Regioselective Oxidation of 3-Substituted Pyridinium Salts
Abstract
:Introduction
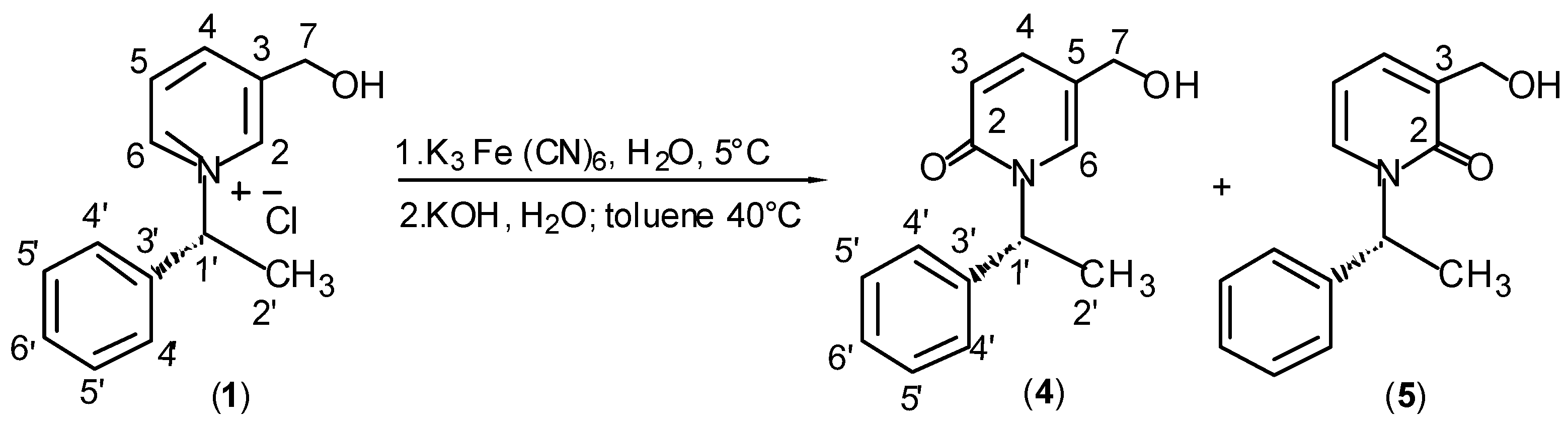
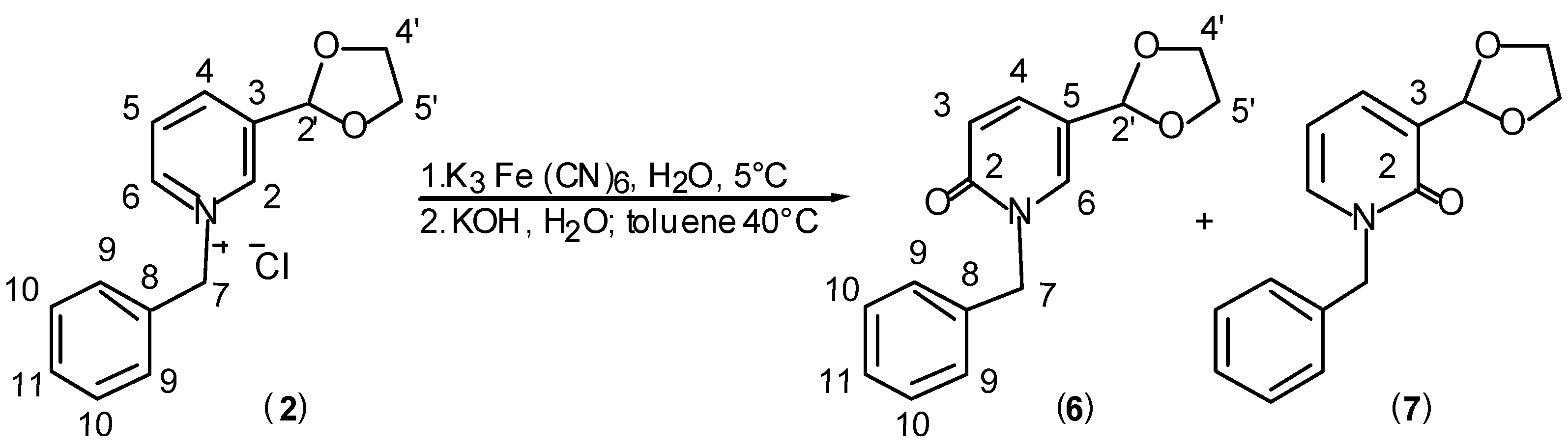
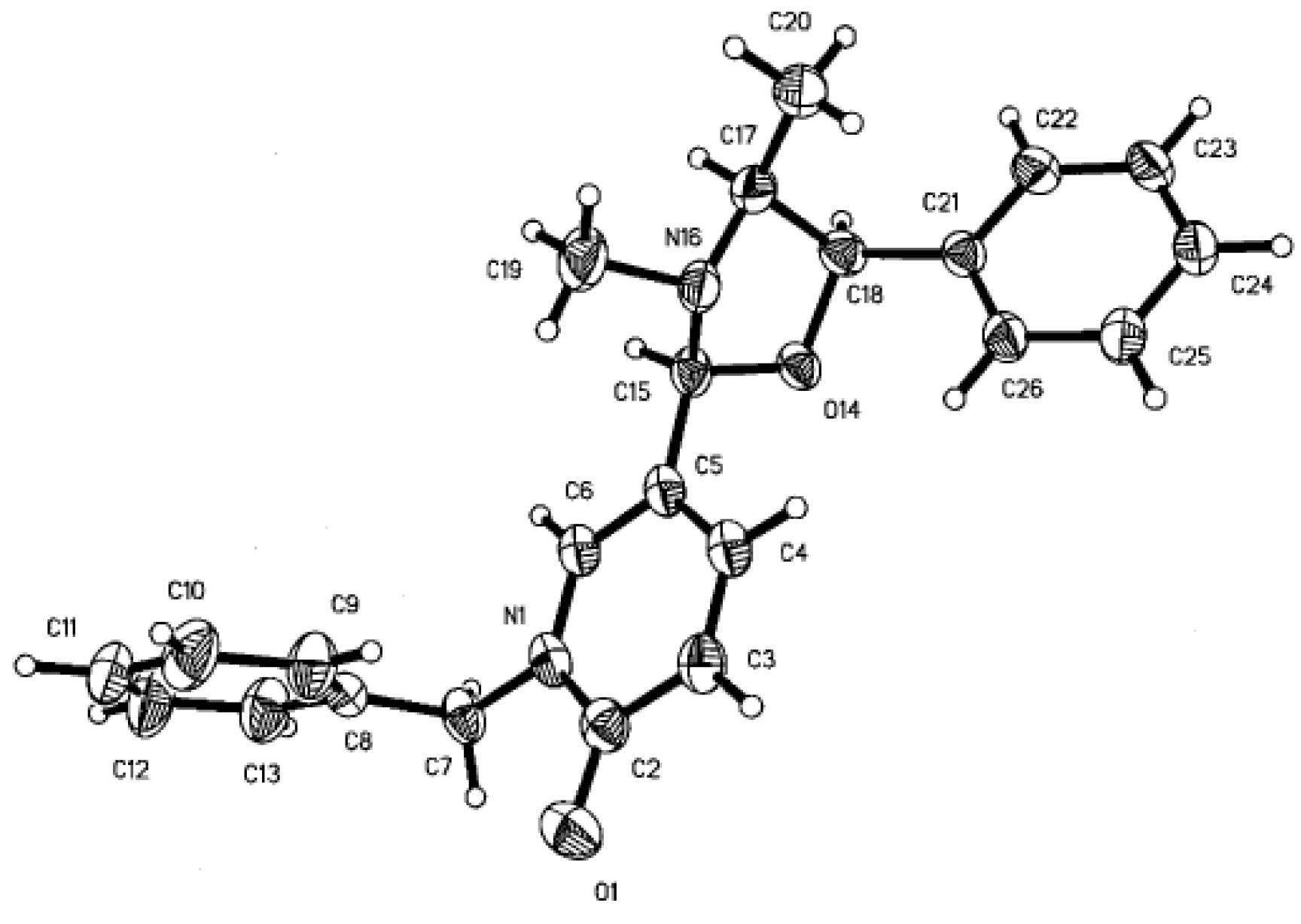
Results and Discussion
Conclusions
Experimental
General.
Preparation of Chiral Pyridinium Salt (1).
Spectral Data.
General Procedure for Synthesis of Pyridinium Salts (2) and (3).
Spectral Data.
General Procedure for Synthesis of 1H-pyridin-2-ones (4+5), (6+7) and (8).
Spectral Data.
Acknowledgements
References and Notes
- Coffen, D. L.; Hengartner, U.; Katonak, A.; Mulligan, M. E.; Burdick, D. C.; Olson, G. L.; Todaro, L. J. J. Org. Chem. 1984, 49, 5109. Amat, M.; Llor, N.; Hidalgo, J.; Hernández, A.; Bosch, J. Tetrahedron Asymmetry 1996, 7, 4, 977. and references cited therein; Kuethe, J. T.; Padwa, A. Tetrahedron Lett. 1997, 38, 1505. and references cited therein; Mabic, S.; Castagnoli, N., Jr. J. Org. Chem. 1996, 61, 309. Munchhof, M. J.; Meyers, A. I. J. Org. Chem. 1995, 60, 7086. Möhrle, H.; Weber, H. Tetrahedron 1970, 26, 2953. and references cited therein.
- Micouin, L.; Bonin, M.; Cherrier, M.-P.; Mazurier, A.; Tomas, A.; Quirion, J.-Ch.; Husson, H.-P. Tetrahedron 1996, 52(22), 7719. and references therein.
- Prill, E. A. McElvain. Org. Syn. 1943, 2, 419. [Google Scholar]
- Becher, I. Synthesis 1980, 589.
- Fujii, T.; Yoshifuji, S.; Mishishita, K.; Mitsukushi, M.; Yoshida, K. Chem. Pharm. Bull. 1973, 20, 2695. and references cited therein; Gonzalez- Bello, C.; Abell, C.; Leeper, F. J. J. Chem. Soc. Perkin Trans.1. 1997, 1017. Cooksey, C. J.; Johnson, M. D. J. Chem. Soc. B. 1968, 10, 1191. Ruchirawat, S.; Sunkul, S.; Thbtaranonyh, Y. Tetrahedron Letters 1977, 27, 2335.
- Buurman, D. J.; van der Plas, H. C. J. Heterocyclic Chem. 1986, 23, 1015.
- Cotin, R. C.; Morrow, J. C.; Rapoport, H. J. Org. Chem. 1976, 41, 535.
- Uchida, H.; Nishida, A.; Nakagawa, M. Tetrahedron Letters 1999, 40, 113.
- Gnecco, D.; Marazano, C.; Enríquez, R. G.; Terán, J.L.; Sanchez, M. R.; Galindo, A. Tetrahedron Asymmetry 1998, 9, 2027.
- Wong, Y. S.; Marazano, C.; Gnecco, D.; Genisson, Y.; Das, B. C. J. Org. Chem. 1997, 62, 729. [PubMed]Genisson, Y.; Marazano, C.; Mehmandoust, M.; Gnecco, D.; Das, B.C. Synlett. 1992, 431.
- Tlekhusezh, M. A.; Makuilov, R. V.; Badovskaya, L. A. Molecules 2000, 5, M141.
- Sheldrick, G. M. SHELX-97. Program for Refinement of Crystal Structures. University of Göttingen: Germany, 1997. [Google Scholar]
- Sample Availability: Available from the authors.
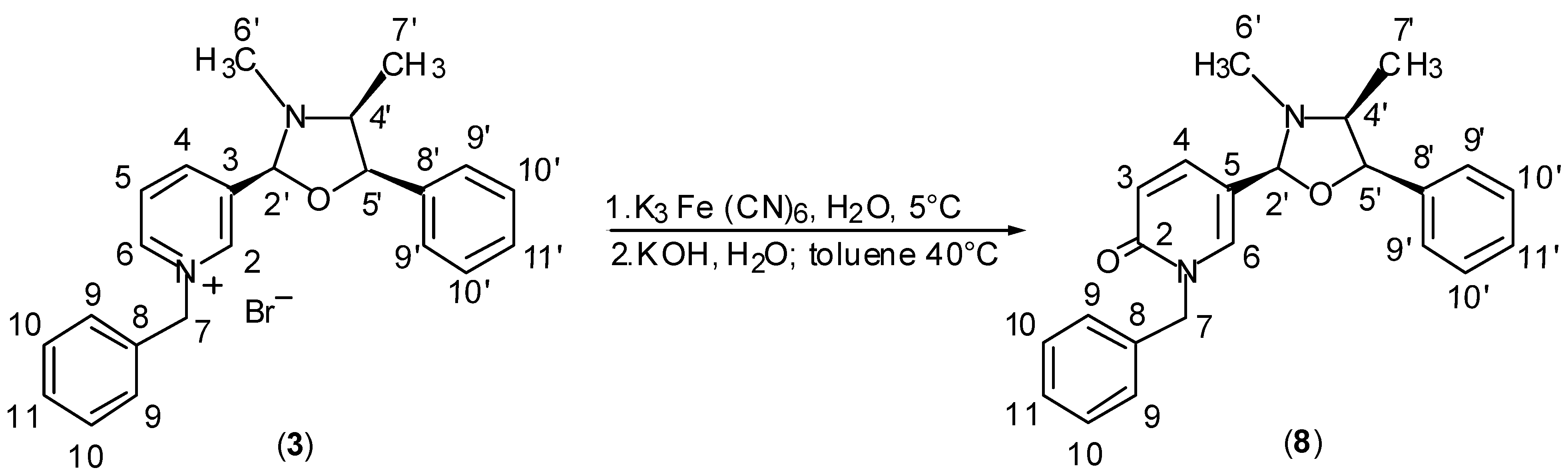
© 2000 by Molecular Diversity Preservation International (MDPI). Reproduction permitted for noncommercial purposes.
Share and Cite
Terán, J.L.; Gnecco, D.; Galindo, A.; Juárez, J.R.; Enríquez, R.G.; Soriano, M.; Reynolds, W.F. Regioselective Oxidation of 3-Substituted Pyridinium Salts. Molecules 2000, 5, 1175-1181. https://doi.org/10.3390/51001175
Terán JL, Gnecco D, Galindo A, Juárez JR, Enríquez RG, Soriano M, Reynolds WF. Regioselective Oxidation of 3-Substituted Pyridinium Salts. Molecules. 2000; 5(10):1175-1181. https://doi.org/10.3390/51001175
Chicago/Turabian StyleTerán, Joel L., Dino Gnecco, Alberto Galindo, Jorge R. Juárez, Raúl G. Enríquez, Manuel Soriano, and W. F. Reynolds. 2000. "Regioselective Oxidation of 3-Substituted Pyridinium Salts" Molecules 5, no. 10: 1175-1181. https://doi.org/10.3390/51001175
APA StyleTerán, J. L., Gnecco, D., Galindo, A., Juárez, J. R., Enríquez, R. G., Soriano, M., & Reynolds, W. F. (2000). Regioselective Oxidation of 3-Substituted Pyridinium Salts. Molecules, 5(10), 1175-1181. https://doi.org/10.3390/51001175



