Introduction
A high content of chemical pollutants in soils and water may adversely affect plant and animal growth and in turn human health. Contamination of soils with such pollutants could be due to the application of chemical fertilizers, sewage, sludge, herbicides, industrial activities, etc. or irrigation with contaminated water. Although a literature survey reveals that nitrilotriacetic acid (H3NTA), and its metal complexes are extensively used in medical, biological and environmental studies, very little work has been carried out on (H3NTA) and its metal complexes from a purely chemical point of view. The aim of the present investigation is to study such properties in order to understand the way in which such materials are effective.
Nitrilotriacetic acid (H
3NTA) is perhaps historically the most important of the complexones other than H
4EDTA. We were able to find over 100 publications dealing with the use of [NTA]
n- and its metal complexes in water and soil treatment as well as in biological and medical research. [NTA]
n- was also used in detergents to replace sodium tripolyphosphate [
1,
2]. Among other fields in which [NTA]
n- or its metal chelates were used are dying and bleaching [
3].
Rabenstien and Kula [
4] observed that the rate of ligand exchange in Cd, Zn and [Pb: (NTA)] when [H
2NTA]
- is in the mono-protonated from is significantly less than the rate when [NTA]
3- is in the totally ionized form. Maguire [
5] explained the slower rate of [HNTA]
2- reaction with copper ion than [NTA]
3- to be due to the fact that a rate limiting proton transfer step from nitrogen in [HNTA]
2- must occur before complex formation takes place.
Despite the immense number of publications of the use of H
3NTA and its chelates in different applied, medical, biological and industrial fields, we were able to locate only one publication [
6] on the preparation and identification of solid HM
IINTA complexes. The importance of the acid nitrilotriacetates stems from the various uses of NTA complexes in acid media. Although H
3NTA forms 1:1 and 1:2 [metal:(NTA)] complex [
7], the present study will be concerned with the 1:1 complexes only.
Results and discussion
The infrared spectrum of nitrilotriacetic acid over the range 4000-200 cm-1 shows three interesting bands at 1434, 1733 and 3041 cm-1. The bands of interest in the IR spectrum of H2KNTA are those at 1440, 1720 and 3040 cm-1. The bands at 1730 and 3040 cm-1 do not appear in the IR spectrum of K3NTA where there is a broad band at 1650-1600 cm-1 and another one at 1420 cm-1.
The fact that the band at ~ 1430 cm
-1 is present in the spectra of the three compounds and which was previously assigned to the symmetric stretching frequency of the COO- [
9] suggests that H
3NTA is present in the zwitterion form.
Again the presence of the band at ~ 1730 cm
-1 in the spectra of H
3NTA and H
2KNTA justifies its previous assignment to the undissociated –COOH stretching vibration [
10].
Finally the band at ~ 3040 cm
-1 which appears in the spectra of H
3NTA and H
2KNTA is assigned to some sort of N-H stretching frequency. Comparing this band with that for gaseous ammonia (3335-3337 cm
-1) [
10] and those for complexed ammonia in ammonio complexes (3250-3050 cm
-1) [
10], it may be argued that the N-H frequency is lowered via some sort of hydrogen bonding. Tajmir-Riahi [
11] who assigned the broad band at 2700 cm
-1 in some Schiff bases to an intramolecular hydrogen bonding (O-H…N) gave a similar argument. This band shifts to 3000 cm
-1 in the spectra of Schiff base complexes as the hydrogen bond arrangement changes to (O…HN
+).
We sought to establish whether this hydrogen bonding in our ligand (H3NTA) is intramolecular or intermolecular by comparing the melting points of nitrilotriacetic acid (246 oC) with that of citric acid (152 oC) where the two acids are tricarboxylic and the molecular weights are almost the same (191 and 192 respectively). This large difference in the melting points suggests that the hydrogen bond in H3NTA is intermolecular.
In order to gain more insight into the structure of H3NTA, the 1H-NMR spectrum of the acid dissolved in DMSO was measured. The spectrum shows the following signals: a singlet signal at δ 3.55-3.79 p.p.m, for the six protons of the three-methylene groups, a broad singlet signal at δ 11.20-11.45 p.p.m with integration values (14.529 / 7.5) = 1.93: 6.11 ~ 2: 6, i.e. two carboxylic, and six methylene protons.
The results of elemental analysis for the studied 1:1 acid complexes, together with some of their physicochemical properties along with the proposed formulae are shown in
Table (1).
The only papers that dealt with the IR spectra of solid acid divalent metal complexes of NTA give the formulae for Mg, Ca, Sr and Ba complexes as (H[Mg(NTA)]·3H
2O, H[Ca(NTA)]·2H
2O, H[Sr(NTA)]·4H
2O and H[Ba(NTA)]·3H
2O respectively) [
6,
12]. In their preparation technique, Tomita and Uneno [
6] recrystallised the solid acid complexes from water. Furthermore, they did not oven-dry the products. The metal, carbon, hydrogen and nitrogen contents of all the eleven complexes described in the present study suggests that they are all anhydrous, thus having the unified formula H[M
II(NTA)]. Their molecular weights were calculated on this basis.
Since the structure of H
3NTA does not allow the formation of square planar complexes, then the 1:1 complexes of the present study should have the tetrahedral alternative. This is confirmed by calculating the magnetic moments of the complexes of the paramagnetic metal ions. The values in B.M. are as follows: H[Cu(NTA)] = 1.83, H[Ni(NTA)] = 3.17, H[Co(NTA)] = 4.30 and H[Mn(NTA)] = 5.81. It is well known that tetrahedral Co(II) complexes have magnetic moments ranging from 4.40-4.88 B.M. while octahedral Co(II) complexes have values around 5.1 B.M [
13,
14]. This tetrahedral structure is supported by the IR spectra, (
Table 2), of the solid complexes where all these spectra contained the υ
(COOM) (str.) band at around 1600 cm
-1.
Table 2 also shows an interesting observation where the υ
OH (str.) for group A metal complexes ranges from 3375-3458 cm
-1 while that for group B metal complexes ranges from 3502-3628 cm
-1. This may be explained in the light of the M-O bond strength which is expected to be stronger in group A complexes with smaller ionic radii than in group B metal complexes with larger ionic radii. This is at once reflected on the O-H bond strength being weaker in-group complexes.
The assignment of the M-N and M-O stretching frequencies (
Table 2) is supported by the work of Condrate and Nakamoto [
11] who located the M-N stretching frequencies for some divalent metal glycine complexes in the range 550-450 cm
-1 and M-O stretching bands between 420 and 280 cm
-1. The two principal bands assigned as υ
C=O stretching (υ
1) and υ
M-O (υ
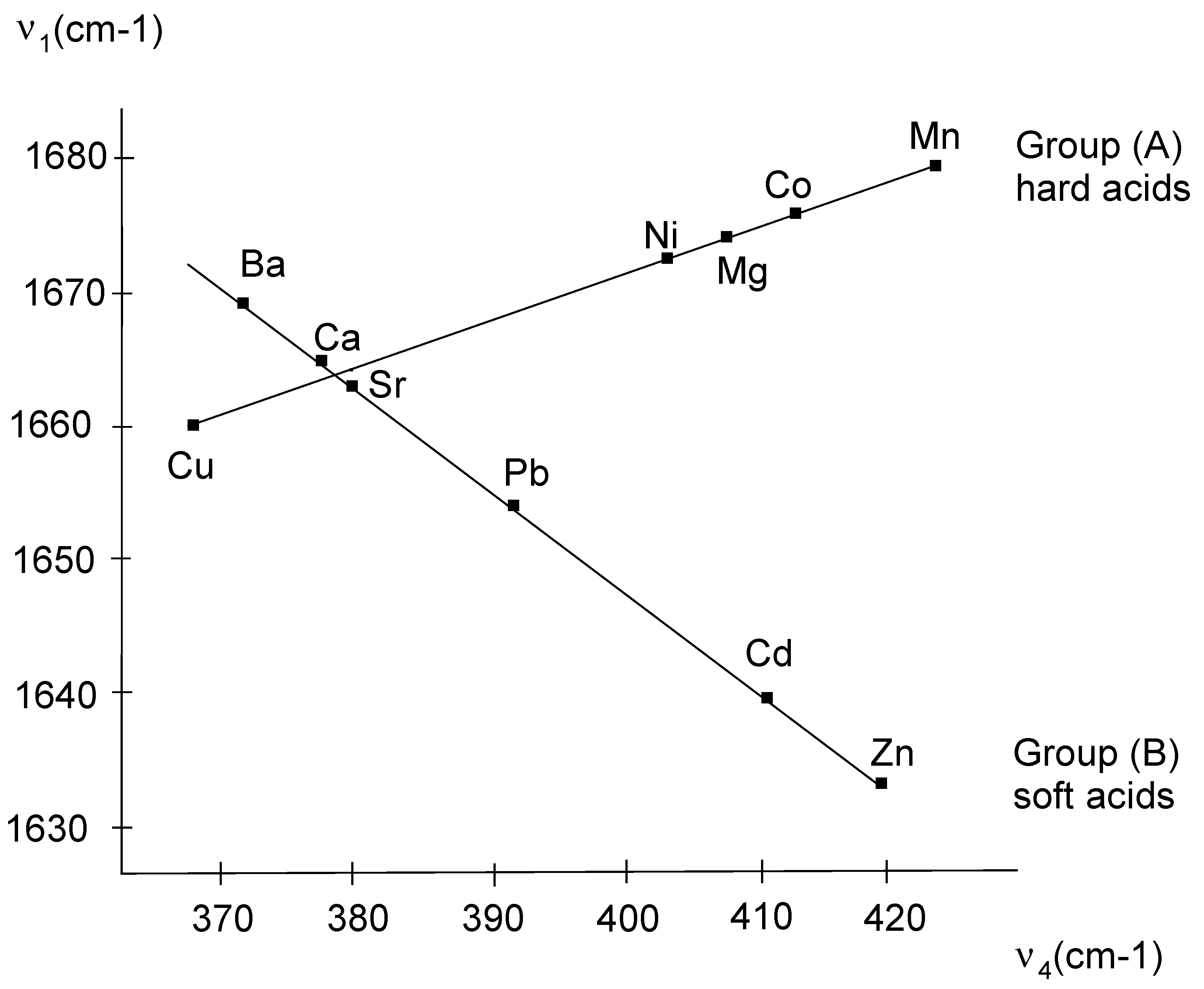
4) are metal sensitive, since these bands shifts progressively to higher frequencies as the ionic radius of the metals change. This is exhibited by the linear relationship between υ
1 and υ
4 as illustrated in
Fig. (1).
Before discussing the UV-visible spectra of the colored complexes of divalent Cu, Ni & Co it should be noted that upon dissolution in water the tetrahedral complexes add two water molecules in cis- position to give an octahedral structure [
4].
The UV-visible spectrum of the Cu(II) complex showed a broad band with λ
max at 784 nm (1.27 x 10
4 cm
-1) which is assigned to
2E →
2T2 transition. 10 Dq equals 12700 cm
-1. This value is very close to that calculated by Dreisch for the aqueous Cu(II) [
15], which is equal to 12200 cm
-1.
The UV-visible spectrum of the Ni(II) complex showed two bands: one at 384nm (26042 cm
-1) for the
3A
2g →
3T
1g (F) transition and another band at 609nm (16420 cm
-1) for the
3A
2g →
3T
2g (F) transition, while that for the Co(II) complex showed a shoulder at 456nm (21929.8 cm
-1) for the
4A1
g →
4A
2g (F) transition and a band at 511nm (19569.5 cm
-1) for the
4T
1g (F) →
4T
1g(P) transition. The 10 Dq values for Ni(II) and Co(II) complexes were calculated using:
- (a)
The equations on the energy terms for the triplet state for Ni(II) and the quartet states for Co(II) [
16].
- (b)
The Tanabe and Sugano diagrams for the d8 and d7 systems respectively.
Applying method (a) and knowing that B for Ni(II) = 1080 and for Co(II) = 970, the 10Dq values for the Ni(II) and Co(II) complexes were found to be 17394 cm-1 and 15500 cm-1 respectively. Applying method (b) the 10 Dq values for the Ni(II) and Co(II) complexes were found to be 17312 cm-1 and 15520 cm-1.
The pKa values for the eleven acid complexes calculated from the results of titration of 0.01molar solution of K[M(NTA)] with 0.1molar solution of hydrochloric acid at 25
oC according to the method of calculations described by Albert and Serjeant [
8] are given in
Table (3) together with the pK
a values of the [M(H
2O)
n]
2+ ions [
8], also with the log β
1 values of the [M(NTA)]
n- complexes.
The expected proportionality between the two series of pKa values is clear where the pKa value of any [M(HNTA).(H2O)3] is derived from that of the free [M(H2O)n]2+ ion and that of H3NTA. The relation between pKa of [M(HNTA).(H2O)3] and log β1 of the corresponding [M(NTA).(H2O)2]- complex may be explained according to the fact that the more stable the complex the higher the dissociation of the corresponding acid complex.
X-ray patterns of the prepared solid NTA complexes were taken over the range of 2θθ = 6 – 60
o. The principal “d” lines and its intensity are represented in
Table 4. All complexes are crystalline solids, where Mg, Ca and Sr complexes have a similar structure (isostructural). The Ni, Cu and Cd display a different structure and a very high degree of crystallinity while Co and Zn complexes are characterized by a low degree of crystallinity, but the strongest lines in the radiograms assume almost the same position as in the radiograms of other complexes. Low symmetry, large size of unit cells and various degrees of crystallinity are characteristics of all crystalline complexes.
Preparation of the acid nitrilotriacetates complexes, [HMNTA]
The calculated amounts of the metal carbonates of Mn, Cd, Ca, Sr, Ba and Pb or the basic carbonate of Cu, Ni, Co, Zn and Mg were added to the aqueous suspension of the equivalent amount of nitrilotriacetic acid in water to give 0.10 molar solution. The mixture was heated gently nearly to boiling for about one hour. After complete reaction, the reaction mixture was concentrated by heating, then 96% ethanol was added and the reaction mixture cooled under tap water or in an ice bath. A dense crystalline precipitate was obtained, filtered, washed with 96% ethanol, dried at 105 oC in an oven, then placed in a vacuum dessiccator, over CaCl2. The same procedure was followed for the preparation of H2KNTA and K3NTA where smaller volumes of water and larger volumes of 96% ethanol were used.
The potassium salts of the acid nitrilotriacetates were prepared by the following reaction:
After cooling, the solution was transferred quantitatively to a volumetric flask, then made up to the mark with distilled water. Suitable aliquots of these solutions were titrated potentiometrically against standard hydrochloric acid solution. No more than 0.06 in a pKa value scatter was allowed for a set of readings in any given estimation.
X-ray diffraction patterns (XRD) of the prepared solid complexes were recorded using a Philips Xray diffractometer model PW 1140/90 and Fe-filtered CoK◻ radiation. The X-ray patterns of powdered samples were recorded automatically with a scanning speed of 2o per minute, and the scanning angular range (2θ) was from 6o-60o. All the diffraction patterns were measured at room temperature. The “d” (10-1 nm)-spacing was calculated and compared with data on they’re relative intensities given in the ASTM cards.