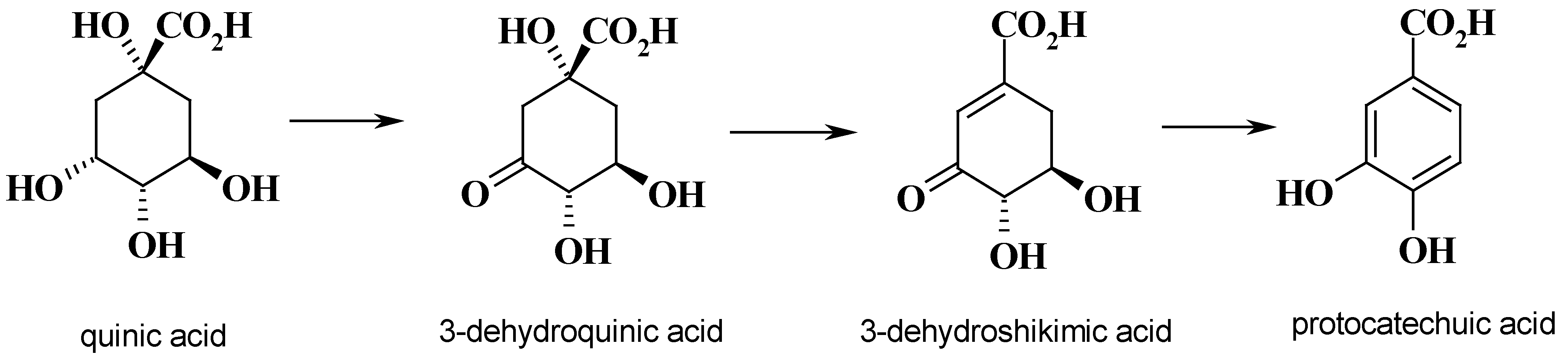
Syntheses
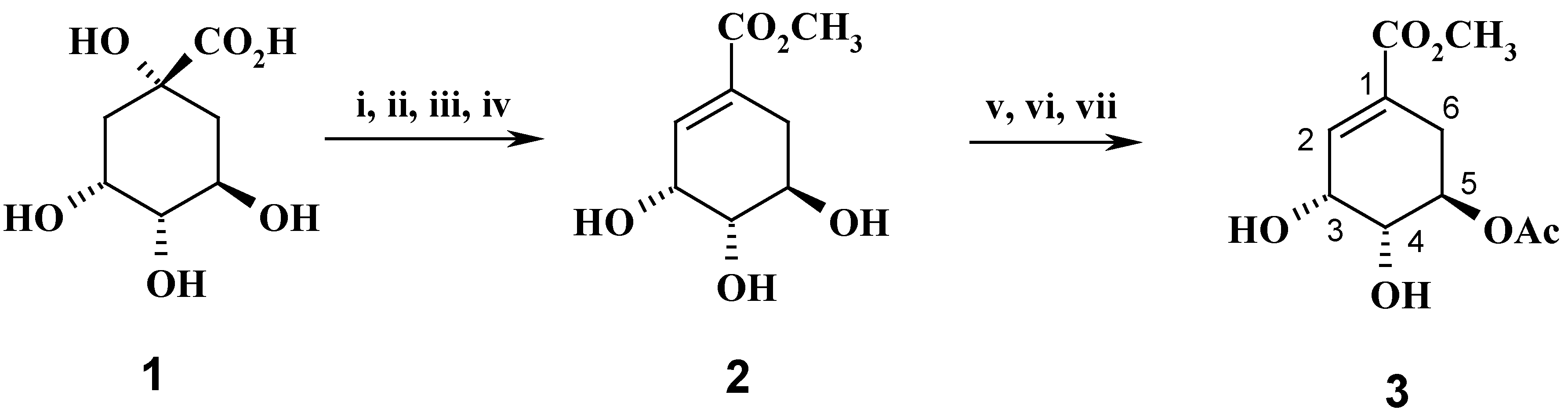
Methyl 5-acetyl shikimate (3): Synthesis of compound 3 was described in ref. 11. 1H-NMR (200 MHz, CDCl3), δ 2.09 (s, 3H, AcO), 2.25-2.35 (dd, 1H, H-6ax), 2.81-2.91 (dd, 1H, H-6eq), 3.73 (s, 3H, CO2CH3), 3.85 (dd, 1H, H-4), 4.42 (m, 1H, H-3), 5.18 (m, 1H, H-5), 6.85 (m, 1H, H-2).
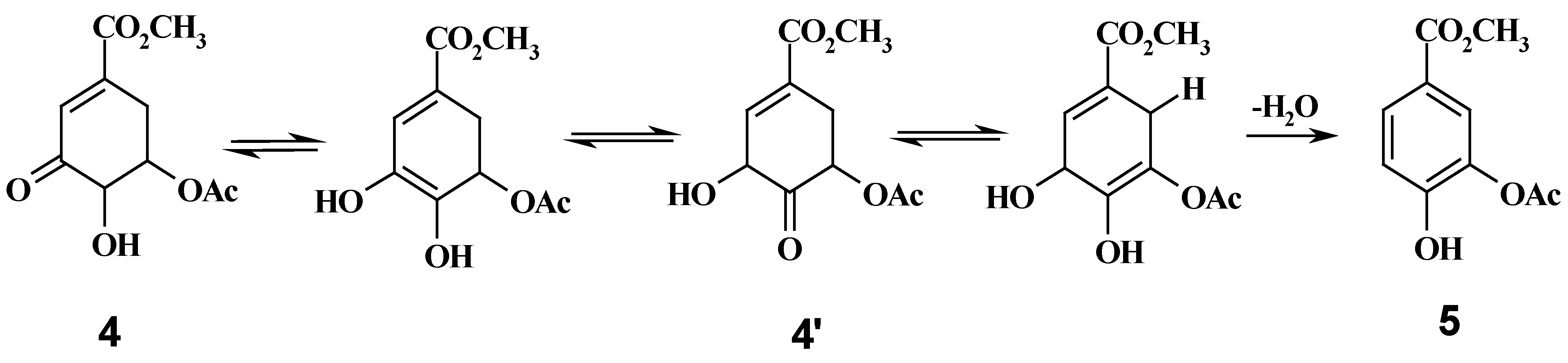
Methyl 5-acetyl-3-keto shikimate (4) and methyl 3-acetyl-protocatechuicate (5): compound 3 (200 mg) was dissolved in acetone (10 mL) under stirring and, Jones’ reagent was added dropwise. When the solution became persistent orange-brown coloured, the reaction was complete as the evidence by TLC. Sodium tiosulphate was added until a green colour was observed, then the reaction mixture was neutralised with barium carbonate and filtered through Celite. Volatile materials were evaporated in vacuo and the residue purified by Si gel chromatography, eluting with ethyl acetate / chloroform (6:4) to give 4 (15 mg, 8% yield) and compound 5 (166 mg, 92% yield), both as colourless oils. 1H-NMR compound 4 (200 MHz, CDCl3): δ 2.15 (s, 3H, AcO), 2.60 (m, 1H, H-6ax), 3.30 (dd, 1H, H-6eq), 3.85 (s, 3H, CO2CH3), 4.30 (d, 1H, H-4), 5.10 (m, 1H, H-5), 6.90 (d, 1H, H-2). 1H-NMR compound 5 (200 MHz, CDCl3): δ 2.33 (s, 3H, AcO), 3.62 (s, 3H, CO2CH3), 6.3 (s, 1H, OH), 7.69 (d, 1H, J= .8 Hz), 7.82 (dd, 1H, J=1.8, 7.9 Hz), 8.12 (d, 1H, J=7.9 Hz).
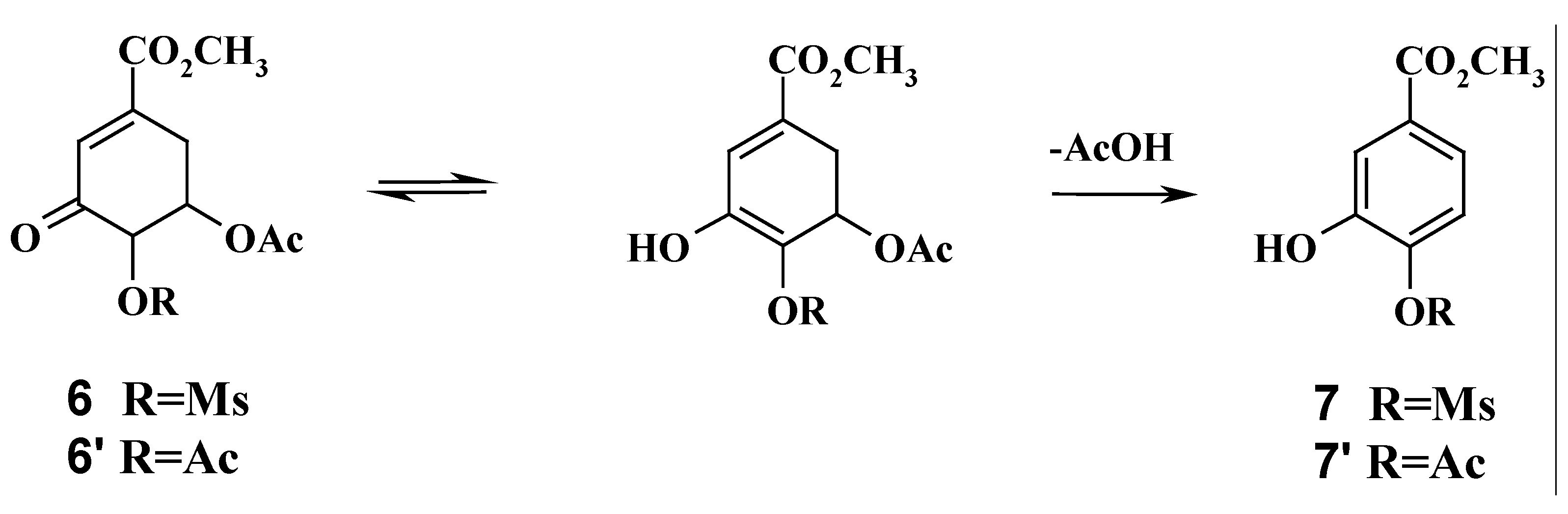
Methyl 5-acetyl-3-keto-4-mesyl shikimate (6): Compound 4 (50 mg) was dissolved in dichloromethane (2.5 mL) and the solution cooled at 0 °C. triethylamine (0.1 mL) was added, followed by dropwise addition of methanesulfonyl chloride (0.05 ml). The reaction mixture was stirred at 0 °C for 2 h. The mixture was then diluted with dichloromethane, the organic phase washed with water and dried over anhydrous sodium sulphate. The volatile materials were removed in vacuo and the residue was purified by Si gel chromatography, eluting with chloroform / ethyl acetate (7:3), to give 6 (58 mg, 86% yield) as a colourless oil. 1H-NMR (200 MHz, CDCl3): δ 2.12 (s, 3H, AcO), 2.60 (m, 1H, H-6ax), 3.11 (s, 3H, SO2CH3), 3.31 (m, 1H, H-6eq), 3.82 (s, 3H, CO2CH3), 4.81 (m, 1H, H-4), 5.05 (m, 1H, H-5), 6.91 (d, 1H, H-2).
Methyl 4,5-diacetyl-3-keto shikimate (6’): Compound 4 (50 mg) was dissolved in pyridine (0.1 mL) and acetic anhydride (0.2 mL) was added. The reaction mixture was allowed to stand at room temperature. After 1 h compound 6’ was formed, as shown by TLC using chloroform / ethyl acetate (8:2) as eluent. Methanol was added to the reaction mixture and the volatile materials were removed in vacuo. The residue was purified by chromatography eluting with chloroform / ethyl acetate (8:2) to give 6’ (51 mg, 86% yield). 1H-NMR (200 MHz, CDCl3): δ 2.12, 2.14 (s, 2x3H, 2xAcO), 2.62 (m, 1H, H-6ax), 3.28 (m, 1H, H-6eq), 3.78 (s, 3H, CO2CH3), 4.80 (m, 1H, H-4), 5.03 (m, 1H, H-5), 6.94 (d, 1H, H-2).
Methyl 4-mesyl-protocatechuicate (7): compound 6 (100 mg) was dissolved in anhydrous DMF (4 mL) and the reaction mixture was heated at 80°C under stirring for 2 h. The reaction mixture was then diluted with ethyl acetate, successively washed with saturated aqueous ammonium chloride, water and brine, and dried over anhydrous sodium sulphate. The solvents were evaporated in vacuo and the residue purified by Si gel chromatography, eluting with chloroform / ethyl acetate (6:4) to give compound 7 (80 mg, 93% yield) as a yellow oil. 1H-NMR (200 MHz, CDCl3): δ 3.13 (s, 3H, SO2CH3) 3.70 (s, 3H, CO2CH3), 6.1 (s, 1H, OH), 7.67 (d, 1H, J=1.8 Hz), 7.91 (dd, 1H, J=1.8, 7.4 Hz), 8.10 (d, 1H, J=7.4 Hz).
Methyl 4-acetyl-protocatechuicate (7’): Compound 6’, under the same reaction conditions as described for compound 6, afforded compound 7’, as a colourless viscous oil, in 94% yield. 1H-NMR (200 MHz, CDCl3): δ 2.25 (s, 3H, AcO), 3.70 (s, 3H, CO2CH3), 6.1 (s, 1H, OH), 7.67 (d, 1H, J=1.8 Hz), 7.91 (dd, 1H, J=1.8, 7.4 Hz), 8.10 (d, 1H, J=7.4 Hz).