2.1. Characterization of PCN and Co@PCN
As shown in
Figure 1a, Fourier-transform infrared (FTIR) spectroscopy was used to define the functional groups and chemical species in the prepared particles. In general, the original CN contains three typical vibration bands, included N–H and C–N bonds, located around 3200 cm
−1 and 1200–1650 cm
−1, respectively. The third peak was detected at 814 cm
−1, which was caused by the out-of-plane bending vibration of heptanazine rings [
35]. The vibrational bands of P–N functional groups of PCN and Co@PCN were not detected, which may be due to the overlap of these vibrational bands with strong C–N vibrational bands [
36,
37,
38]. Nevertheless, a small wavenumber shift occurred in the sharp band around 814 cm
−1 (
Figure 1b), indicating a change in the electron cloud in C–N bonds and C=N bonds induced by phosphorus doping [
39]. A similar phenomenon was also observed in Co@PCN. There was also a new band at 495 cm
−1 in PCN and Co@PCN, which originated from the P–O–P bending mode [
40]. The crystal structure and phase purity of PCN and Co@PCN were assessed by X-ray diffraction (XRD) using CN as a reference sample. The results are shown in
Figure 1c. The pattern of CN had two characteristic peaks. The peak at 12.97° was caused by in-plane repeating units at, and the other one at 27.73° originated from the interlayer stacking of conjugated aromatic units [
35]. The PCN particles showed a similar XRD pattern to the original polyheptanazine, which indicates that polyheptanazine retained its framework well during the phosphorization process. However, there were some small sharp peaks in the P-doped samples, indicating that the samples may contain crystalline phosphate impurities [
23,
40,
41]. In addition, the weakening of the PCN (002) diffraction peak may be caused by the inclusion of hetero-element P during thermal phosphorization, which enlarged the interlayer spacing and reduced the thickness of the graphitic structure. The further decrease in the diffraction intensity of metal-containing samples (Co@PCN) may be attributed to chemical coupling between PCN and metal phosphide clusters [
35,
42].
Figure 1d shows the TG-DSC curve of a mixture of urea and diammonium hydrogen phosphate (DHP). The endothermic peak of the mixture at 134 °C was the melting of urea. The melting point of DHP is about 155 °C.
Figure 1d shows that urea and DHP began to react at 150–230 °C. The corresponding peak at 237 °C was due to the formation of a triazine structure. The polymerization of the triazine compound occurred at 364 °C, which was earlier than the similar peak temperature of CN prepared using pristine urea [
3]. The residues of the mixture of urea and DHP were 3.8% at 550 °C from the curves of TG-DSC (Simultaneous thermogravimetric analysis coupled with differential scanning calorimeter) The obtained product yield (PCN) was about 5.2% from calcination in muffle furnace. The reason for the differences may be due to the different environments in which mixture were located.
As shown in
Figure 2, similar to that of CN and PCN, Co@PCN still presented a distinct nanosheet structure. The composition of chemical species in Co@PCN particles was further investigated using scanning transmission electron microscope (STEM) and energy-dispersive spectrum mapping (EDS) analysis. Significantly, P and Co showed analogous distribution patterns as N (
Figure 2h,i), which demonstrated effective doping of P into the triazine rings of CN. We did not observe nanoclusters on the surface of Co@PCN. It should be noted that the low-density distribution of Co suggested that there were other forms of CoP or cobalt oxides besides the nanoclusters. A considerable part of these may be only sub-nanometer or even atomic in size [
35].
The X-ray photoelectron spectroscopy (XPS) analysis images of CN, PCN, and Co@PCN are given in
Figure 3, and the atomic percentages of these particles are listed in
Table 1. In the C 1s spectrum (
Figure 3b) of CN, the peaks at 285.08 and 288.28 eV were attributed to the sp
2 graphitic carbon (C–C=C) and sp
2-bonded carbon in the triazine rings (N–C=N), respectively [
43]. CN has a lower area ratio of graphitic carbon than PCN, and C–O (289.48 eV) appeared. The C 1s XPS spectra of Co@PCN can be divided into four peaks at 284.93, 287.23, 288.13, and 288.78 eV, corresponding to sp
2 graphitic C, C–N, sp
2-bonded carbon in N–C=N, and sp
2-hybridized C bonded to –NH
2 on the aromatic ring, respectively [
22]. The XPS N 1s spectra of CN (
Figure 3c) at 398.68, 399.28, and 400.68 eV were attributed to sp
2-hybridized nitrogen C–N=C, tertiary nitrogen N–C3, and C–N–H (positive charge localization in heterocycles), respectively [
35]. The last peak positioned at 401.78 eV was identified as C–NH
2 in PCN [
44,
45]. Two additional peaks for Co@PCN located at 398.98 and 400.48 eV corresponded to N–Co and C–NH
2, respectively [
34,
46]. The C 1s XPS spectra of PCN and Co@PCN (
Figure 3d) can be divided into two peaks at 531.43 eV and 532.88 eV, corresponding to adsorbed O and oxygen and H
2O on the surface [
22]. The P 2p XPS spectra of PCN and Co@PCN contained peaks at 133.13, 133.93, and 134.83 eV, which correspond to P–N, P=N, and P–O bonds, respectively (
Figure 3e) [
36,
39,
47]. This implied that P likely replaced C in the
s-triazine units to form P–N and P=N bonds because the binding energies of P–C coordination bonds (131.2–132.2 eV) were lower than that of P–N and P=N bonds [
22]. P–O bonds were ascribed to oxidized P on the surface [
39]. The Co 2p spectrum is displayed in
Figure 3f, and the peaks positioned at 781.43 and 797.63 eV were associated with Co
3+. The peaks at 783.03 and 801.48 eV were related to Co
2+ [
48,
49]. The peaks at 803.98 and 786.48 eV were assigned to the satellite peaks of Co 2p
1/2 and 2p
3/2 [
49]. The peaks at 779.6 and 795.0 eV were related to the Co−N
x structure of Co@PCN, which formed due to van der Waals forces [
50,
51].
The thermogravimetric analysis (TGA) curves of CN, PCN, and Co@PCN are shown in
Figure 4 (in nitrogen), and the corresponding thermal decomposition data are given in
Table 2. The initial decomposition temperature, the temperature at 50% weight loss, and the temperature at the maximal weight were recorded as T
−5, T
−50, and T
−max, respectively. CN presented a one-step decomposition process over the whole temperature range. Unlike CN, PCN exhibited three different thermal decomposition processes (
Figure 4b). The decomposition of PCN at 200–550 °C may be attributed to the decomposition of a small amount of oxidized P and oligomers. The T
−50 of PCN and Co@PCN were higher than that of CN, indicating that the formation of a graphitic polyheptanazine framework was not affected by phosphorus doping or cobalt loading. The residues of PCN and Co@PCN at 750 °C were 26.3% and 47.1%, respectively, which were much higher than that of CN, typically 0% at 750 °C. This indicated that phosphorus doping greatly increased the residues of CN, possibly due to the formation of P−N bonds, which encouraged the retention of P in the condensed phase during thermal decomposition.
2.3. Thermal Stability and Fire Resistance of TPU and Its Composites
The thermal decomposition of the TPU composites in nitrogen is shown in
Figure 6, and the corresponding parameters are listed in
Table 3. All nanocomposites exhibited similar degradation behavior to that of pure TPU (
Figure 6). T
−5 of CN-TPU decreased slightly after the introduction of CN, while T
−5 of PCN-TPU was 19 °C lower than that of pure TPU, which may be attributed to the presence of a small amount of oxidized P in PCN. The initial decomposition temperature of Co@PCN-TPU was 4 °C higher than that of pure TPU, which was due to the higher thermal stability of Co@PCN. T
−50 of CN-TPU and PCN-TPU were basically the same as that of pure TPU, while T
−50 of Co@PCN-TPU was 10 °C higher than that of pure TPU. The residues of CN-TPU, PCN-TPU, and Co@PCN-TPU at 700 °C were 5.2, 9.5, and 10.7%, respectively, which were 4.4% higher than pure TPU. This phenomenon demonstrated that the introduction of phosphorus increased the residues, which was attributed to the effect of P–N bonds [
12]. The overall influence of PCN and Co@PCN was that the incorporated additives did not influence the decomposition of polyurethane bonds but slightly increased the residue percentage formed during the decomposition of polyols (the second decomposition step in
Figure 5b).
The flame retardancy of TPU and its nanocomposites under nitrogen atmosphere was evaluated by microcalorimetry (MCC). As shown in
Figure 7 and
Table 4, the peak heat release rate (pHRR) and total heat release (THR) of pure TPU during combustion were 407.8 kW/m
2 and 86.34 MJ/m
2, respectively. The pHRR for PCN-TPU was 33.8% lower while the pHRR of Co@PCN-TPU decreased by 16.4% compared with pure TPU. It can be seen that CN has a good thermal inhibition effect in a nitrogen atmosphere.
Figure 7b shows that the incorporation of PCN and Co@PCN further reduced the THR of the nanocomposites. It demonstrated that the introduction of phosphorus or cobalt is beneficial to further reduce the THR of nanocomposites under nitrogen atmosphere.
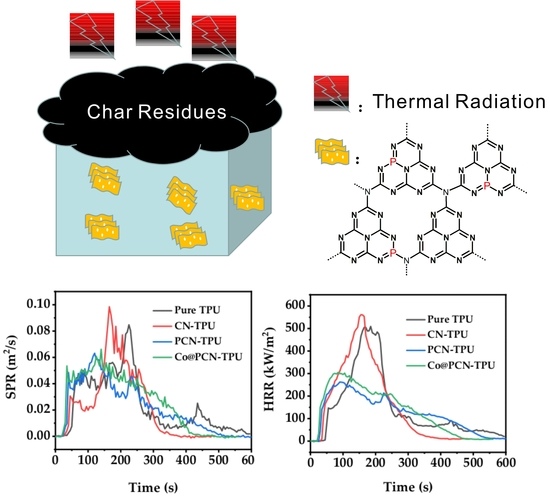
The HRR and THR curves obtained from cone calorimetry tests (CCT) of pure TPU and its nanocomposites are shown in
Figure 8. The pHRR of pure TPU was 509.4 kW/m
2 while that of CN-TPU was 561.4 kW/m
2, which was the highest of all samples (
Figure 8a). It demonstrated that the addition of untreated CN alone did not help reduce the pHRR of thermoplastic polyurethane. After the introduction of PCN and Co@PCN into the polymer, the pHRR decreased by 48.5% and 40.1% for PCN-TPU and Co@PCN-TPU, respectively, rivaled by pure TPU. PCN and Co@PCN changed the pyrolysis process of TPU, forming more char layers, which was mainly attributed to the formation of P–N bonds and P=N bonds in triazine rings, which promoted the retention of P in the condensed phase and produced more high-quality residues.
The results in
Figure 8b show that PCN-TPU and Co@PCN-TPU degraded slightly earlier than pure TPU samples. The THR of pure TPU, CN-TPU, PCN-TPU, and Co@PCN-TPU were 86.34 MJ/m
2, 82.31 MJ/m
2, 74.01 MJ/m
2, and 76.68 MJ/m
2, respectively (
Table 4). PCN and Co@PCN encouraged the production of more residues and reduced the mass loss, thus reducing the value of total heat release. The CCT results showed that the flame-retardant efficiency of PCN was better than that of Co@PCN.
The smoke-production rate (SPR) curves of pure TPU and its composites are shown in
Figure 8c. The pSPR was greatly reduced after the introduction of PCN and Co@PCN, indicating that smoke toxicity of TPU decreased. Under the same filling contents, PCN-TPU presented the lowest pSPR and showed the best smoke-suppression performance of all samples. This was attributed to the charring ability of the condensed phase of modified polyheptanazine, which reduced heat release and smoke production.
Figure 8d gives the mass loss curves of nanocomposites as a function of time. The lower mass loss was ascribed to the formation of a high-quality char on the nanocomposite surface and its structure improvement [
52]. Only 10.18% of pure TPU remained after the CCT test. The PCN-TPU plot shows a high residue value of 17.04%, which indicates that P-doped polyheptanazine was cross-linked. Moreover, the char residues formed on the surface can prevent heat and mass transfer, further reducing mass loss and improving fire safety.
It has been reported that the char residues of cyclo-P–N bond composites include P–O–P bonds and P–O–C bonds [
53,
54]. Combined with the TG data, it can be inferred that the increase of residues during cone calorimetry tests may be attributed to the production of phosphoric acid or polyphosphate during pyrolysis, which promotes the formation of heat-resistant carbonaceous compounds.
The curves of CO and CO
2 evolution as a function of time obtained from CCT are shown in
Figure 8e,f, and the total amount of CO and CO
2, and the corresponding CO
2/CO ratio of TPU and its nanocomposites after complete combustion are listed in
Table 5. CO and CO
2 are the main components of fire gas. A high concentration of CO will lead to carbon monoxide poisoning due to obstruction of escape in a fire. The lower values of CO
2/CO ratio indicated that the low efficiency of combustion and the conversion of CO to CO
2 are inhibited [
55]. As shown in
Table 5, the yields of CO of PCN-TPU and Co@PCN-TPU were higher than that of pure TPU, but PCN-TPU and Co@PCN-TPU had lower yields of CO
2 compared to pure TPU, leading to a decrease in CO
2/CO ratios (29.7 and 27.8 vs. 44.9). This may be due to the reaction between a small amount of oxidized P with the polymer matrix, which prevents further oxidation of CO release from PCN-TPU and Co@PCN-TPU in the first combustion process into CO
2, whereas that reaction could not occur in the combustion process of pure TPU.
TPU and its nanocomposites passed the UL-94 V-2 rating with melt dripping in the vertical burning test. Furthermore, the dripping object ignited the absorbent cotton. t1 + t2 (Total burning time is used to evaluate the UL-94 rating.) in the UL-94 test decreased as the flame retardants were incorporated. t1 + t2 of pure TPU is 3 s while those of PCN-TPU and Co@PCN-TPU are shorter.
To highlight the improved fire performance of phosphorus-doped polyheptanazine in the TPU matrix, the performance of PCN-TPU was compared with the reported results of other flame-retardant TPU nanocomposites (
Table 6). It can be inferred that PCN encourages the formation of char residues, resulting in decreased pHRR values.
Table 6 also shows that PCN has a definite smoke-suppression effect. In this work, the introduction of PCN to TPU remarkably reduced the PHRR (48.5%), which was better than most reported results. Yang et al. [
56] fabricated cetyltrimethylammonium bromide (CTAB)-modified Ti
3C
2 (MXene) ultra-thin nanosheets (CTAB-Ti
3C
2). Despite excellent flame retardance properties (−51.2% in pHRR), the use of MXene flame retardants has been greatly limited because their preparation methods are expensive and difficult to industrialize. The raw materials used in this paper are urea and diammonium hydrogen phosphate, which are produced industrially, making them easy to acquire. In addition, the preparation method of PCN is simple.
2.4. Evolution of Pyrolysis Gas
Simultaneous thermal analysis coupled with Fourier-transform infrared spectrometry (TG-IR) was used to detect the effect of those particles on the pyrolysis behavior of pure TPU, CN-TPU, PCN-TPU, and Co@PCN-TPU (
Figure 9). TPU nanocomposites showed similar main spectra bands to that of pure TPU, and those bands were attributed to functional groups or components with characteristic band positions (
Figure 10), including CO
2 (2358 cm
−1), aromatic compounds (1508 and 1460 cm
−1), esters (1145 cm
−1), hydrocarbons (2977 and 2880 cm
−1), carbonyl compounds (1751 cm
−1), and HCN (714 cm
−1) [
57,
62,
63]. The results showed that pure TPU and CN-TPU exhibited similar thermal decomposition processes while PCN-TPU decomposed slightly earlier. This demonstrated that incorporating these flame-retardant particles into TPU may not change the primary decomposition process.
Figure 10a shows that the decomposition of PCN-TPU released abundant CO
2 during the first stage, which was related to the slightly earlier decomposition due to the addition of PCN during TGA (
Figure 6) and MCC (
Figure 7). This can enhance the gas-phase flame-retardant mechanism of the nanocomposites. The inhibition of PCN-TPU was better than that of Co@ PCN-TPU in the remaining characteristic band analysis. This can be attributed to the better char-formation ability of PCN in the matrix.
Figure 10b shows the evolution trend of aromatic compounds. The addition of three fillers increased the intensity of the first stage and decreased the intensity of the second stage, which did not prolong the ignition time of the nanocomposite during CCT. This is different from the previous literature that stated Co compounds can inhibit the formation of aromatic compounds [
31]. In addition, PCN-TPU showed a different phenomenon from other samples in the hydrocarbon curve in
Figure 10d. A few hydrocarbons were released during the first stage, which may be attributed to the reaction of a small amount of oxidized P with the polymer matrix, corresponding to premature decomposition of PCN-TPU in TGA.
Figure 10f shows that PCN changed the decomposition pathway, thus avoiding HCN produced by polyheptanazine decomposition. This stage corresponds to the 500–600 °C decomposition stage in TGA.