Simultaneous Analysis of 20 Mycotoxins in Grapes and Wines from Hexi Corridor Region (China): Based on a QuEChERS–UHPLC–MS/MS Method
Abstract
1. Introduction
2. Results and Discussions
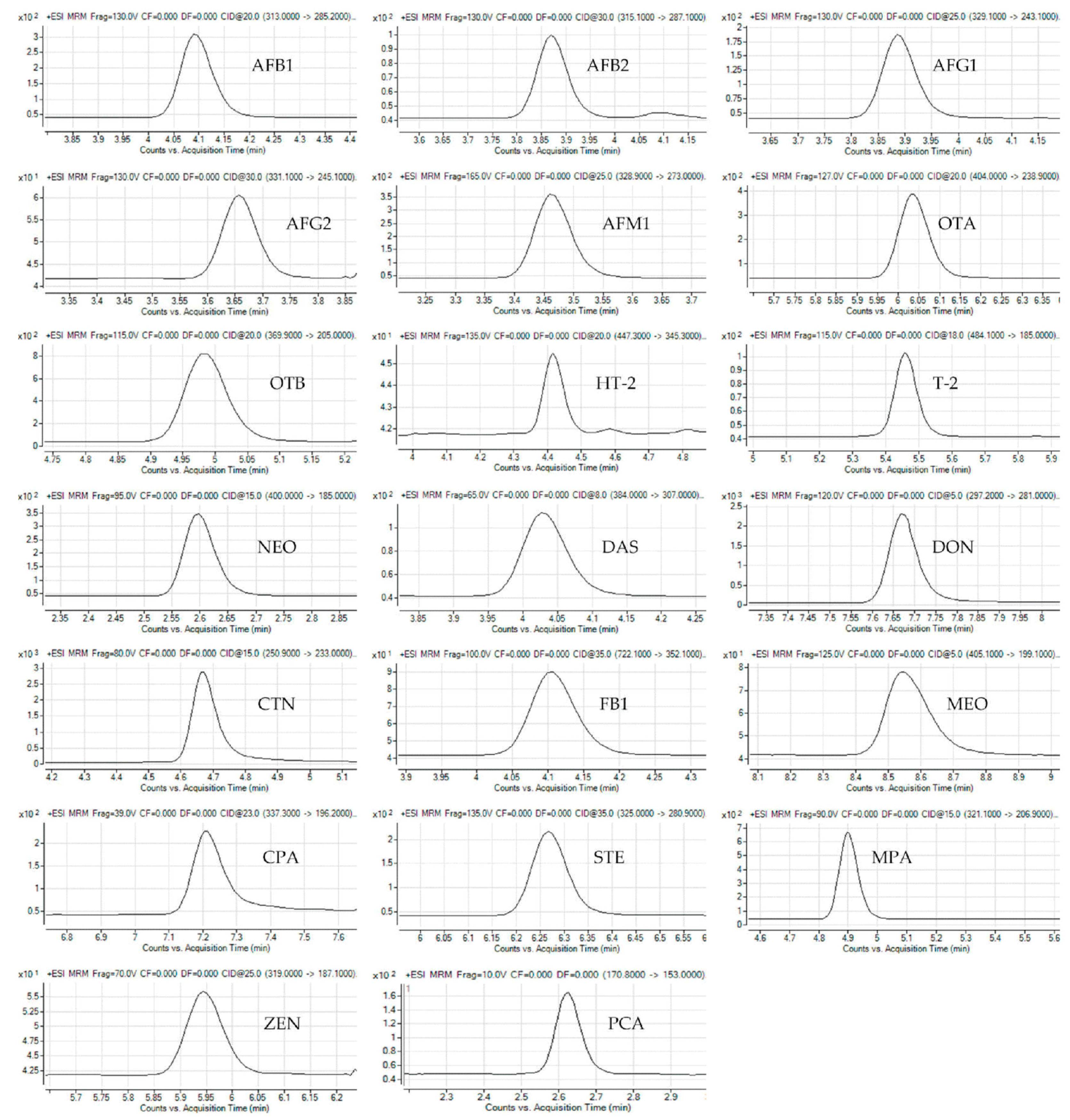
2.1. Optimization of MS/MS Parameters and Chromatographic Separation
2.2. Optimization of the QuEChERS Procedure
2.2.1. Selection of Wine/Water Ratio
2.2.2. Selection of Salt Amount and Type
2.2.3. Selection of Clean-Up
2.3. Method Validation
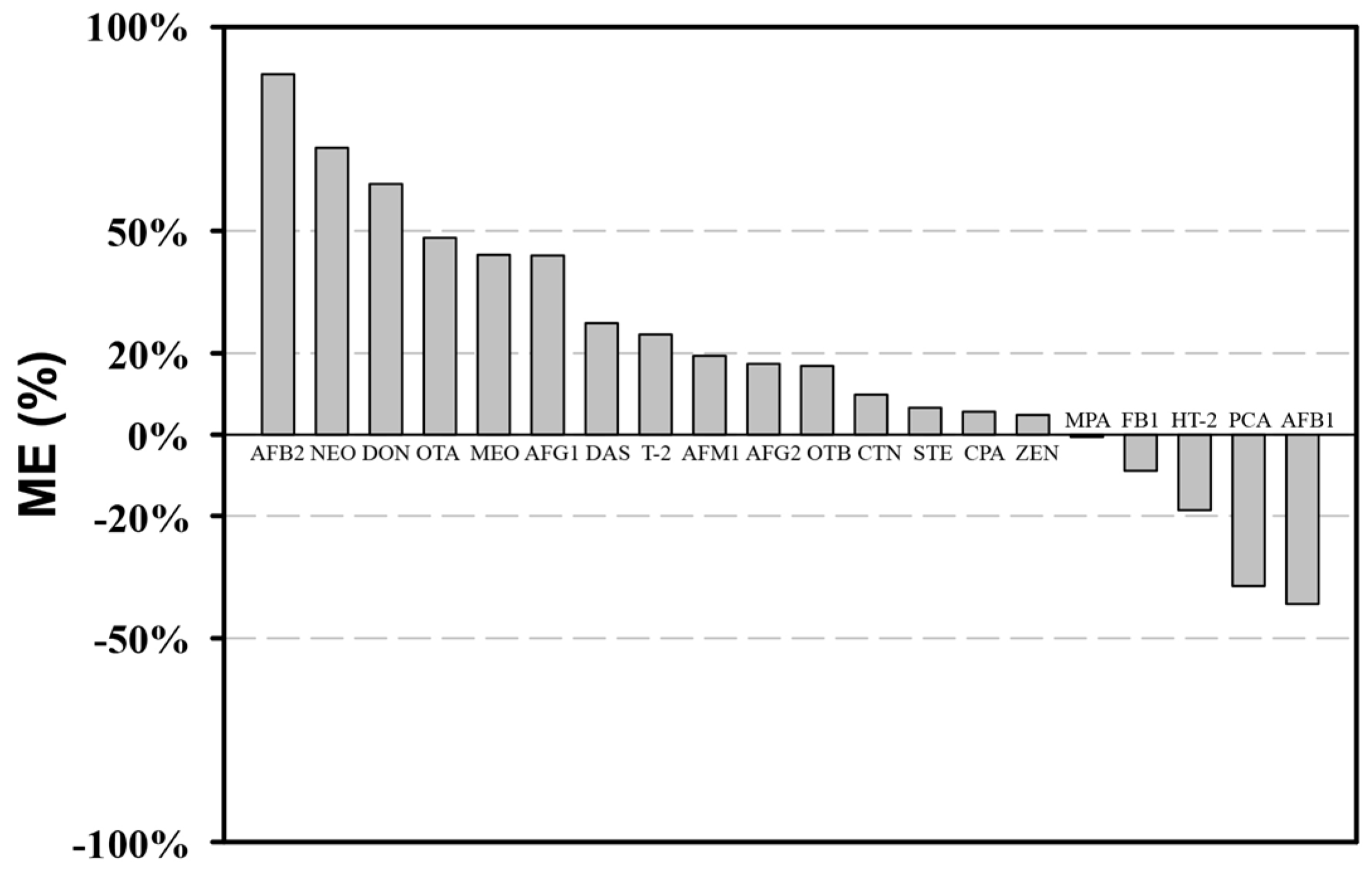
2.3.1. Matrix Effect Evaluation
2.3.2. Calibration Curves, Linearity, LOD and LOQ
2.3.3. Precision and Recovery
2.4. Application to Real Sample
3. Materials and Methods
3.1. Chemicals and Standards
3.2. Optimization of the Extraction Method
3.2.1. The Wine/Water Ratio Optimization
3.2.2. The Amount and Type of Salt Optimization
3.2.3. The Clean-Up Optimization
3.2.4. Optimized QuEChERS Procedure
3.3. Chromatographic and Mass Spectrometric Conditions
3.4. Method Validation Study
3.5. Analysis of Mycotoxins in Grape and Wine Samples
3.6. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Pena, A.; Cerejo, F.; Silva, L.J.G.; Lino, C.M. Ochratoxin A survey in Portuguese wine by LC-FD with direct injection. Talanta 2010, 82, 1556–1561. [Google Scholar] [CrossRef] [PubMed]
- Freire, L.; Passamani, F.R.F.; Thomas, A.B.; Silva, L.M.; Paschoal, F.N.; Pereira, G.E.; Prado, G.; Batista, L.R. Influence of physicathinl and chemical characteristics of wine grapes on the incidence of Penicillium and Aspergillus fungi in grapes and ochratoxin A in wines. Int. J. Food Microbiol. 2017, 241, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Paterson, R.R.M.; Venâncio, A.; Lima, N.; Guilloux-Bénatier, M.; Rousseaux, S. Predominant mycotoxins, mycotoxigenic fungi and climate change related to wine. Food Res. Int. 2018, 103, 478–491. [Google Scholar] [CrossRef] [PubMed]
- Hussein, H.S.; Brasel, J.M. Toxicity, metabolism, and impact of mycotoxins on humans and animals. Toxicology 2001, 167, 101–134. [Google Scholar] [CrossRef]
- Egmond, H.P.V.; Schothorst, R.C.; Jonker, M.A. Regulations relating to mycotoxins in food. Anal. Bioanal. Chem. 2007, 389, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: Occurrence and risk assessment. Food Addit. Contam. 1996, 13, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Ferracane, R.; Haidukowsky, M.; Cozzi, G.; Visconti, A.; Ritieni, A. Fumonisin B(2) production by Aspergillus niger from grapes and natural occurrence in must. Food Addit. Contam. Part A 2009, 26, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Ferracane, R.; Visconti, A.; Ritieni, A. Natural occurrence of fumonisin B2 in red wine from Italy. Food Addit. Contam. Part A 2010, 27, 1136–1141. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, J.M.; Larsen, T.O.; Nielsen, K.F. Widespread occurrence of the mycotoxin fumonisin B(2) in wine. J. Agric. Food Chem. 2010, 58, 4853–4857. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ortega, P.; Gilbert-López, B.; García-Reyes, J.F.; Ramos-Martos, N.; Molina-Díaz, A. Generic sample treatment method for simultaneous determination of multiclass pesticides and mycotoxins in wines by liquid chromatography-mass spectrometry. J. Chromatogr. A 2012, 1249, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Pizzutti, I.R.; De, K.A.; Scholten, J.; Righi, L.W.; Cardoso, C.D.; Rohers, G.N.; Da, S.R. Development, optimization and validation of a multimethod for the determination of 36 mycotoxins in wines by liquid chromatography-tandem mass spectrometry. Talanta 2014, 129, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.R.; Farhana, N.I.; Salleh, B. Occurrence of Aspergillus spp. and aflatoxin B1 in Malaysian foods used for human consumption. J. Food Sci. 2011, 76, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Cheraghali, A.M.; Yazdanpanah, H.; Doraki, N.; Abouhossain, G.; Hassibi, M.; Aliabadi, S.; Aliakbarpoor, M.; Amirahmadi, M.; Askarian, A.; Fallah, N. Incidence of aflatoxins in Iran pistachio nuts. Food Chem. Toxicol. 2007, 45, 812–816. [Google Scholar] [CrossRef] [PubMed]
- Onji, Y.; Aoxt, Y.; Tani, N. Analysis of fusarium mycotoxins by gas chromatography/mass spectrometry without chemical derivatization. JSM Mycotoxins 2009, 1994, 49–51. [Google Scholar] [CrossRef]
- Baquião, A.C.; Zorzete, P.; Reis, T.A.; Assunção, E.; Vergueiro, S.; Correa, B. Mycoflora and mycotoxins in field samples of Brazil nuts. Food Control 2012, 28, 224–229. [Google Scholar] [CrossRef]
- Song, S.; Ediage, E.N.; Wu, A.; De, S.S. Development and application of salting-out assisted liquid/liquid extraction for multi-mycotoxin biomarkers analysis in pig urine with high performance liquid chromatography/tandem mass spectrometry. J. Chromatogr. A 2013, 1292, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.K.; Krystin Kee, S.Y.; Ng, W.; Gopalakrishnakone, P. Determination of trichothecene toxin (T2 mycotoxin) in aqueous sample with solid phase microextraction technique followed by gas chromatography with flame ionization detection. J. Sep. Sci. 2015, 22, 424–426. [Google Scholar] [CrossRef]
- Prieto, A.; Basauri, O.; Rodil, R.; Usobiaga, A.; Fernández, L.A.; Etxebarria, N.; Zuloaga, O. Stir-bar sorptive extraction: A view on method optimisation, novel applications, limitations and potential solutions. J. Chromatogr. A 2010, 1217, 2642–2666. [Google Scholar] [CrossRef] [PubMed]
- Alsharif, A.M.A.; Huat, T.G.; Mun, C.Y.; Lawal, A. Liquid phase microextraction for analysis of mycotoxins in food samples: Review. Res. J. Chem. Environ. Sci. 2015, 3, 5–21. [Google Scholar]
- Liu, Y.; Han, S.; Lu, M.; Wang, P.; Han, J.; Wang, J. Modified QuEChERS method combined with ultra-high performance liquid chromatography tandem mass spectrometry for the simultaneous determination of 26 mycotoxins in sesame butter. J. Chromatogr. B 2014, 970, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Yang, X.; Zhao, X.; Bai, B.; Yao, C.; Liu, N.; Wang, J.; Zhou, C. Vortex-assisted dispersive liquid-liquid microextraction for the analysis of major Aspergillus and Penicillium mycotoxins in rice wine by liquid chromatography-tandem mass spectrometry. Food Control 2017, 73, 862–868. [Google Scholar] [CrossRef]
- Wei, J.; Cao, J.; Tian, K.; Hu, Y.; Su, H.; Wan, J.; Li, P. Trace determination of five organophosphorus pesticides by using QuEChERS coupled with dispersive liquid–liquid microextraction and stacking before micellar electrokinetic chromatography. Anal. Methods 2015, 7, 5801–5807. [Google Scholar] [CrossRef]
- Soisungnoen, P.; Burakham, R.; Srijaranai, S. Determination of organophosphorus pesticides using dispersive liquid-liquid microextraction combined with reversed electrode polarity stacking mode-micellar electrokinetic chromatography. Talanta 2012, 98, 62–68. [Google Scholar] [CrossRef] [PubMed]
- García-López, M.; Canosa, P.; Rodríguez, I. Trends and recent applications of matrix solid-phase dispersion. Anal. Bioanal. Chem. 2008, 391, 963–974. [Google Scholar] [CrossRef] [PubMed]
- Matuszewski, B.K.; Constanzer, M.L.; Chavezeng, C.M. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal. Chem. 2003, 75, 3019–3030. [Google Scholar] [CrossRef] [PubMed]
- Lozano, A.; Kiedrowska, B.; Scholten, J.; de Kroon, M.; de Kok, A.; Fernandez-Alba, A.R. Miniaturisation and optimisation of the Dutch mini-Luke extraction method for implementation in the routine multi-residue analysis of pesticides in fruits and vegetables. Food Chem. 2016, 192, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Al-Taher, F.; Cappozzo, J.; Zweigenbaum, J.; Lee, H.J.; Jackson, L.; Ryu, D. Detection and quantitation of mycotoxins in infant cereals in the U.S. market by LC-MS/MS using a stable isotope dilution assay. Food Control 2017, 72, 27–35. [Google Scholar] [CrossRef]
- Koesukwiwat, U.; Sanguankaew, K.; Leepipatpiboon, N. Evaluation of a modified QuEChERS method for analysis of mycotoxins in rice. Food Chem. 2014, 153, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Soleimany, F.; Jinap, S.; Faridah, A.; Khatib, A. A UPLC–MS/MS for simultaneous determination of aflatoxins, ochratoxin A, zearalenone, DON, fumonisins, T-2 toxin and HT-2 toxin, in cereals. Food Control 2012, 25, 647–653. [Google Scholar] [CrossRef]
- Chatterjee, N.S.; Utture, S.; Banerjee, K.; Ahammed Shabeer, T.P.; Kamble, N.; Mathew, S.; Ashok Kumar, K. Multiresidue analysis of multiclass pesticides and polyaromatic hydrocarbons in fatty fish by gas chromatography tandem mass spectrometry and evaluation of matrix effect. Food Chem. 2016, 196, 1–8. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the some wines are available from the authors. |
| Mycotoxin | Structure | Adduct ion | Precursor Ion (m/z) | Product ion a (m/z) | Fragmentor (V) | Collision Energy (V) |
|---|---|---|---|---|---|---|
| AFB1 (aflatoxin B1) |  | [M + H]+ | 313.0 | 285.2/241.1 | 130 | 20/20 |
| AFB2 (aflatoxin B2) |  | [M + H]+ | 315.1 | 287.1/269.1 | 130 | 30/30 |
| AFG1 (aflatoxin G1) |  | [M + H]+ | 329.1 | 243.1/311.1 | 130 | 25/20 |
| AFG2 (aflatoxin G2) |  | [M + H]+ | 331.1 | 245.1/285.1 | 130 | 30/25 |
| AFM1 (aflatoxin M1) |  | [M + H]+ | 328.9 | 273.0/228.9 | 165 | 25/50 |
| CPA (cyclopiazonic acid) |  | [M + H]+ | 337.3 | 196.2/182.1 | 39 | 23/20 |
| CTN (citrinin) |  | [M + H]+ | 250.9 | 233.0/205.0 | 80 | 15/25 |
| DAS (diacetoxyscirpenol) |  | [M + NH4]+ | 384.0 | 307.0/247.0 | 65 | 8/10 |
| DON (deoxynivalenol) |  | [M + H]+ | 297.2 | 281.0/265.0 | 120 | 5/25 |
| FB1 (fumonisin B1) |  | [M + H]+ | 722.1 | 334.0/352.1 | 100 | 40/35 |
| HT-2 (HT-2 toxin) |  | [M + Na]+ | 447.3 | 284.8/345.3 | 135 | 20/20 |
| MEO (mevinolin) |  | [M + H]+ | 405.1 | 199.1/285.1 | 125 | 5/5 |
| MPA (mycophenolic acid) |  | [M + H]+ | 321.1 | 206.9/274.7 | 90 | 15/15 |
| NEO (neosolaniol) |  | [M + NH4]+ | 400.0 | 305.0/185.0 | 95 | 10/15 |
| OTA (ochratoxin A) |  | [M + H]+ | 404.0 | 238.9/357.9 | 127 | 20/10 |
| OTB (ochratoxin B) |  | [M + H]+ | 369.9 | 205.0/324.0 | 115 | 20/10 |
| PCA (penicillic acid) |  | [M + H]+ | 170.8 | 153.0/125.0 | 10 | 0/15 |
| STE (sterigmatocystine) |  | [M + H]+ | 325.0 | 280.9/252.9 | 135 | 35/55 |
| T-2 (T-2 toxin) |  | [M + NH4]+ | 484.1 | 305.0/215.0 | 115 | 10/18 |
| ZEN (zearalenone) |  | [M + H]+ | 319.0 | 187.1/184.9 | 70 | 25/40 |
| Mycotoxin | Matrix Effect (%)/Recovery (%) (n = 3) | |||||
|---|---|---|---|---|---|---|
| 1:9 | 2:8 | 3:7 | 5:5 | 7:3 | 8:2 | |
| AFB1 | −26/94 | 31/116 | 33/101 | 68/84 | 51/69 | 68/64 |
| AFB2 | −32/183 | 15/96 | 9/98 | 34/85 | 15/68 | 32/66 |
| AFG1 | −27/193 | 13/86 | 16/69 | 47/81 | 31/76 | 44/76 |
| AFG2 | −12/182 | 26/92 | 25/81 | 42/89 | 13/50 | 26/49 |
| AFM1 | −58/63 | −60/72 | −57/71 | −64/84 | −68/71 | −69/76 |
| CPA | −13/124 | −27/88 | −24/81 | −37/88 | −33/79 | −30/63 |
| CTN | 53/84 | 37/88 | 50/89 | 54/94 | 61/88 | 68/85 |
| DAS | 312/84 | 289/90 | 325/90 | 357/96 | 413/88 | 435/89 |
| DON | 177/73 | 6/36 | 9/72 | −21/115 | −33/148 | −18/240 |
| FB1 | −93/109 | −92/94 | −94/108 | −93/101 | −93/91 | −92/74 |
| HT-2 | 30/95 | 7/110 | 19/102 | 48/88 | 28/81 | 22/116 |
| MEO | 113/62 | 97/61 | 67/73 | 89/84 | 0/85 | 7/77 |
| MPA | −23/116 | −7/90 | −24/107 | 8/81 | −11/88 | 19/68 |
| NEO | −8/71 | −43/90 | −41/74 | −51/86 | −57/80 | −56/75 |
| OTA | −14/120 | 1/106 | −31/126 | −1/106 | −16/111 | 29/74 |
| OTB | 76/87 | 64/90 | 84/85 | 81/91 | 85/87 | 92/84 |
| PCA | −48/105 | −45/90 | −59/115 | −43/83 | −61/102 | −46/70 |
| STE | −33/196 | −7/145 | −16/175 | 31/115 | 11/116 | 51/92 |
| T-2 | 556/78 | 455/100 | 508/95 | 496/105 | 532/94 | 541/94 |
| ZEN | −25/104 | −14/98 | −27/115 | 3/89 | −11/79 | 12/67 |
| Mycotoxin | Matrix Effect (%)/Recovery (%) (n = 3) | |||||||
|---|---|---|---|---|---|---|---|---|
| A a | B | C | D | E | F | G | H | |
| AFB1 | −26/100 | 31/118 | 33/89 | 68/100 | 51/89 | 68/87 | 53/107 | 55/89 |
| AFB2 | −32/120 | 15/103 | 9/113 | 34/104 | 15/124 | 32/74 | 11/99 | 31/87 |
| AFG1 | −27/113 | 13/109 | 16/108 | 47/101 | 31/102 | 44/84 | 33/104 | 37/88 |
| AFG2 | −12/117 | 26/115 | 25/117 | 42/117 | 13/112 | 26/100 | 23/108 | 22/85 |
| AFM1 | −71/46 | −72/56 | −73/68 | −72/65 | −73/83 | −73/79 | −71/80 | −71/88 |
| CPA | −26/52 | −26/78 | −33/92 | −32/94 | −21/98 | −32/116 | −30/109 | −17/94 |
| CTN | 46/55 | 52/64 | 53/69 | 47/78 | 48/93 | 46/93 | 47/87 | 40/93 |
| DAS | 118/68 | 176/72 | 187/82 | 198/85 | 221/93 | 209/92 | 209/89 | 198/92 |
| DON | −59/158 | −74/257 | −59/120 | −62/134 | −67/95 | −75/80 | −39/77 | −46/84 |
| FB1 | −94/100 | −93/99 | −93/98 | −92/86 | −94/85 | −94/134 | −94/74 | −95/101 |
| HT-2 | 31/98 | 15/102 | 44/96 | 33/94 | 33/103 | 36/90 | 46/89 | 21/100 |
| MEO | −15/89 | −33/93 | −6/102 | −26/117 | −20/123 | −21/113 | −32/88 | −21/90 |
| MPA | −35/66 | −31/66 | −39/73 | −40/86 | −32/92 | −39/97 | −35/105 | −24/94 |
| NEO | −53/54 | −53/63 | −53/73 | −51/72 | −49/85 | −58/89 | −51/85 | −53/91 |
| OTA | −29/67 | −20/57 | −28/65 | −33/77 | −27/80 | −35/87 | −5/97 | 3/92 |
| OTB | 74/57 | 80/66 | 79/73 | 75/79 | 76/86 | 78/83 | 76/90 | 77/90 |
| PCA | −52/76 | −58/80 | −64/77 | −69/127 | −68/103 | −72/96 | −14/110 | 6/89 |
| STE | −33/59 | −31/59 | −29/58 | −26/65 | −15/68 | −21/70 | −50/112 | −44/102 |
| T-2 | 370/43 | 375/57 | 359/69 | 307/85 | 297/104 | 283/103 | 278/95 | 291/93 |
| ZEN | −31/62 | −33/64 | −39/69 | −38/79 | −35/88 | −38/89 | −46/100 | −40/95 |
| Mycotoxin | Calibration | R2 | Linear Range (μg/L) | LOD (μg/L) | LOQ (μg/L) |
|---|---|---|---|---|---|
| AFB1 | y = 5.6980x + 0.1230 | 0.9990 | 5–200 | 0.25 | 0.75 |
| AFB2 | y = 33.0713x + 116.2999 | 0.9993 | 1.25–50 | 0.13 | 0.39 |
| AFG1 | y = 71.1585x + 249.3916 | 0.9988 | 5–200 | 0.25 | 0.75 |
| AFG2 | y = 10.0093x + 4.5423 | 0.9991 | 1.25–50 | 0.06 | 0.18 |
| AFM1 | y = 4.4951x + 3.7299 | 0.9995 | 5–200 | 0.1 | 0.3 |
| CPA | y = 3.1361x − 13.8214 | 0.9987 | 5–200 | 0.25 | 0.75 |
| CTN | y = 923.0092x + 1433.5419 | 0.9995 | 5–200 | 0.1 | 0.3 |
| DAS | y = 0.8656x − 1.8377 | 0.9980 | 5–200 | 0.1 | 0.3 |
| DON | y = 0.8969x + 3045.7580 | 0.9971 | 5–200 | 5 | 15 |
| FB1 | y = 0.4647x − 0.9568 | 0.9994 | 5–200 | 1 | 3 |
| HT-2 | y = 0.0326x + 0.1858 | 0.9963 | 5–200 | 10 | 30 |
| MEO | y = 1.6624x − 4.5656 | 0.9992 | 5–200 | 0.1 | 0.3 |
| MPA | y = 6.9560x − 16.4431 | 0.9993 | 5–200 | 0.1 | 0.3 |
| NEO | y = 42.1866x + 72.9181 | 0.9994 | 5–200 | 0.1 | 0.3 |
| OTA | y = 3.6812x − 6.8163 | 0.9998 | 5–200 | 0.1 | 0.3 |
| OTB | y = 195.8790x + 507.8370 | 0.9991 | 5–200 | 0.1 | 0.3 |
| PCA | y = 10.5717x + 34.0765 | 0.9993 | 5–200 | 1 | 3 |
| STE | y = 5.9077x − 0.9029 | 0.9999 | 5–200 | 0.1 | 0.3 |
| T-2 | y = 1.0423x − 2.7770 | 0.9983 | 5–200 | 0.25 | 0.75 |
| ZEN | y = 0.5345x − 0.5351 | 0.9992 | 5–200 | 1 | 1 |
| Area | Total Number of Samples | Number of Samples with Mycotoxins Detected | ||||
|---|---|---|---|---|---|---|
| PCA a | MPA | CPA | FB1 | ZEN | ||
| Jiayuguan | 3 | - | 1 (9.27) | 3 (2.94–7.44) | - | - |
| Zhangye | 9 | 9.04 | 2 (0.10–0.68) | - | - | - |
| Wuwei | 24 | 11.71 | 2 (0.10–81.26) | 5 (0.11–0.17) | 1 (0.84) | 2 (0.29–0.36) |
| Year | Total Number of Samples | Number of Samples with Mycotoxins Detected | ||||
|---|---|---|---|---|---|---|
| OTA a | MPA | CPA | AFB2 | ZEN | ||
| 2007 | 1 | - | 1 (1.37) | - | - | - |
| 2008 | 1 | - | - | - | - | - |
| 2009 | 2 | - | 2 (1.37–1.53) | - | - | - |
| 2010 | 2 | - | 1 (1.53) | - | 1 (0.10) | - |
| 2011 | 2 | - | 1 (1.45) | - | - | - |
| 2012 | 2 | - | 2 (1.60) | - | - | - |
| 2013 | 3 | - | 2 (1.45–1.60) | 1 (3.72) | - | - |
| 2014 | 9 | 1 (1.27) | 6 (1.45–2.31) | 1 (3.90) | 1 (0.10) | 2 (1.82–1.85) |
| 2015 | 14 | 1 (1.27) | 5 (1.37–2.31) | 3 (2.71–4.62) | - | 1 (1.82) |
| 2016 | 6 | - | - | - | - | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, B.; Chen, X.; Han, S.-Y.; Li, M.; Ma, T.-Z.; Sheng, W.-J.; Zhu, X. Simultaneous Analysis of 20 Mycotoxins in Grapes and Wines from Hexi Corridor Region (China): Based on a QuEChERS–UHPLC–MS/MS Method. Molecules 2018, 23, 1926. https://doi.org/10.3390/molecules23081926
Zhang B, Chen X, Han S-Y, Li M, Ma T-Z, Sheng W-J, Zhu X. Simultaneous Analysis of 20 Mycotoxins in Grapes and Wines from Hexi Corridor Region (China): Based on a QuEChERS–UHPLC–MS/MS Method. Molecules. 2018; 23(8):1926. https://doi.org/10.3390/molecules23081926
Chicago/Turabian StyleZhang, Bo, Xia Chen, Shun-Yu Han, Min Li, Teng-Zhen Ma, Wen-Jun Sheng, and Xia Zhu. 2018. "Simultaneous Analysis of 20 Mycotoxins in Grapes and Wines from Hexi Corridor Region (China): Based on a QuEChERS–UHPLC–MS/MS Method" Molecules 23, no. 8: 1926. https://doi.org/10.3390/molecules23081926
APA StyleZhang, B., Chen, X., Han, S.-Y., Li, M., Ma, T.-Z., Sheng, W.-J., & Zhu, X. (2018). Simultaneous Analysis of 20 Mycotoxins in Grapes and Wines from Hexi Corridor Region (China): Based on a QuEChERS–UHPLC–MS/MS Method. Molecules, 23(8), 1926. https://doi.org/10.3390/molecules23081926






