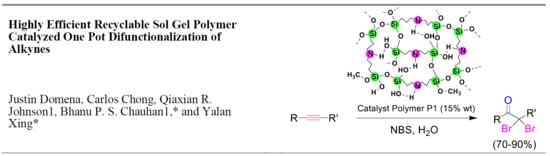
Highly Efficient Recyclable Sol Gel Polymer Catalyzed One Pot Difunctionalization of Alkynes
Abstract
:1. Introduction
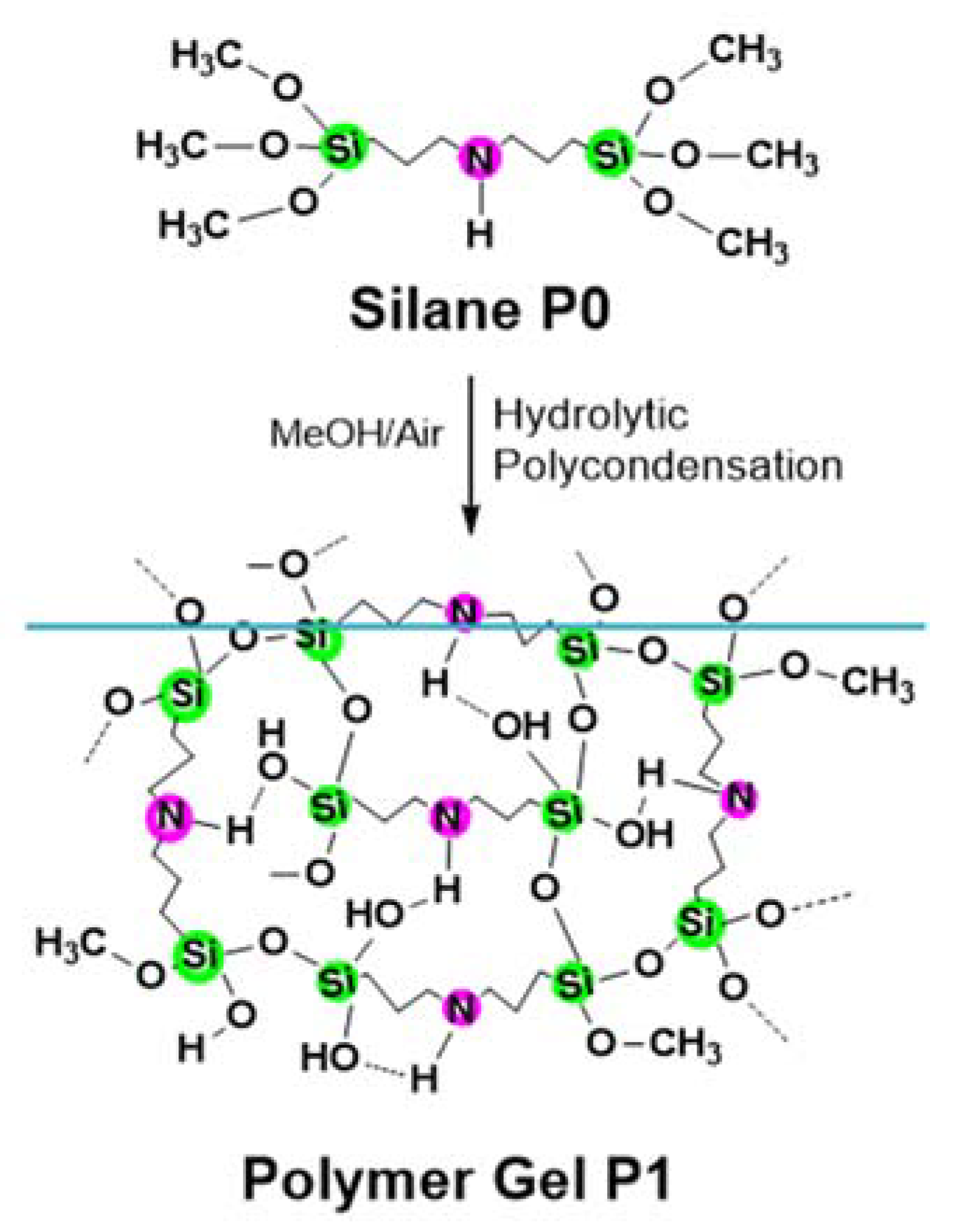
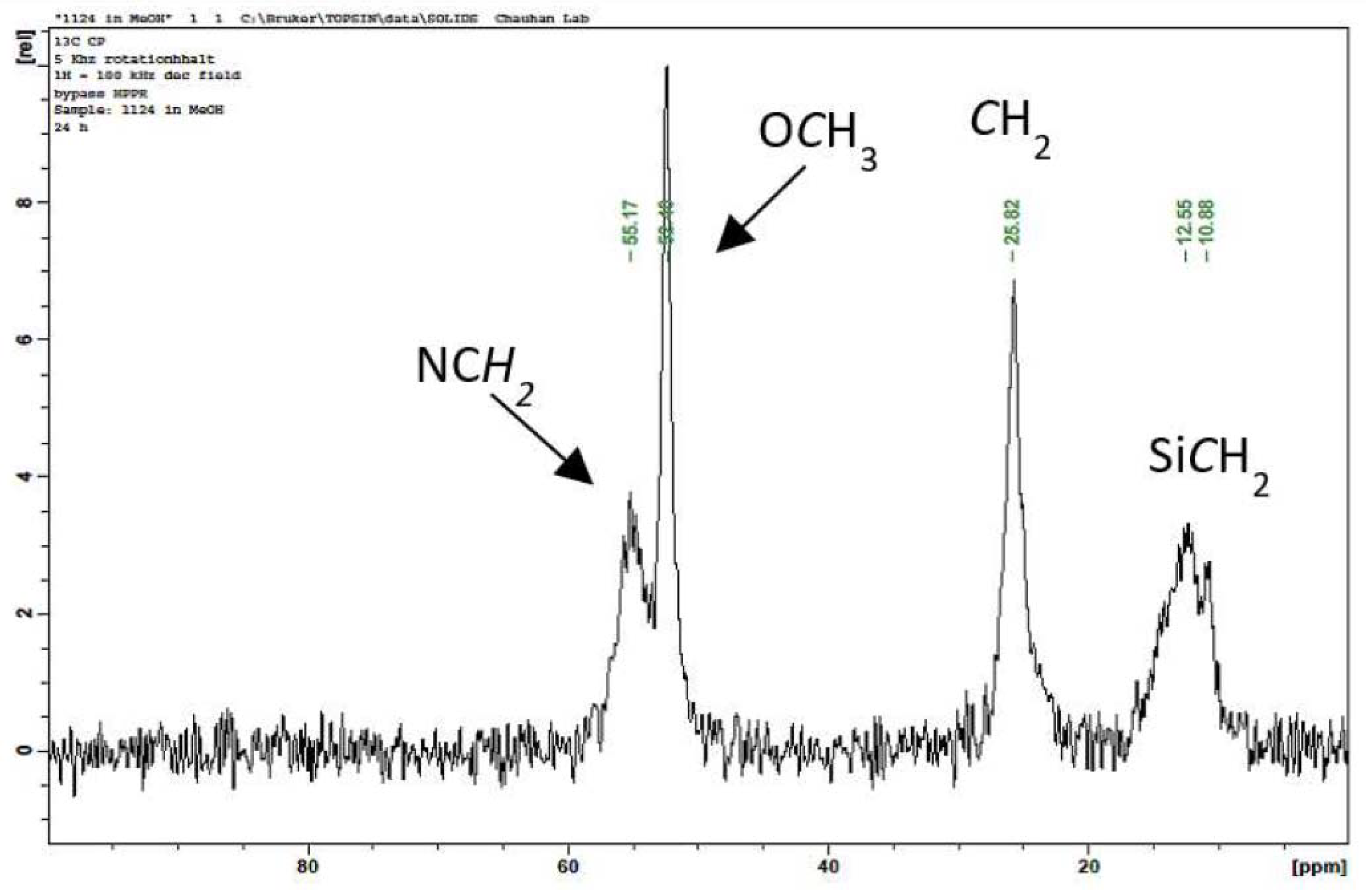
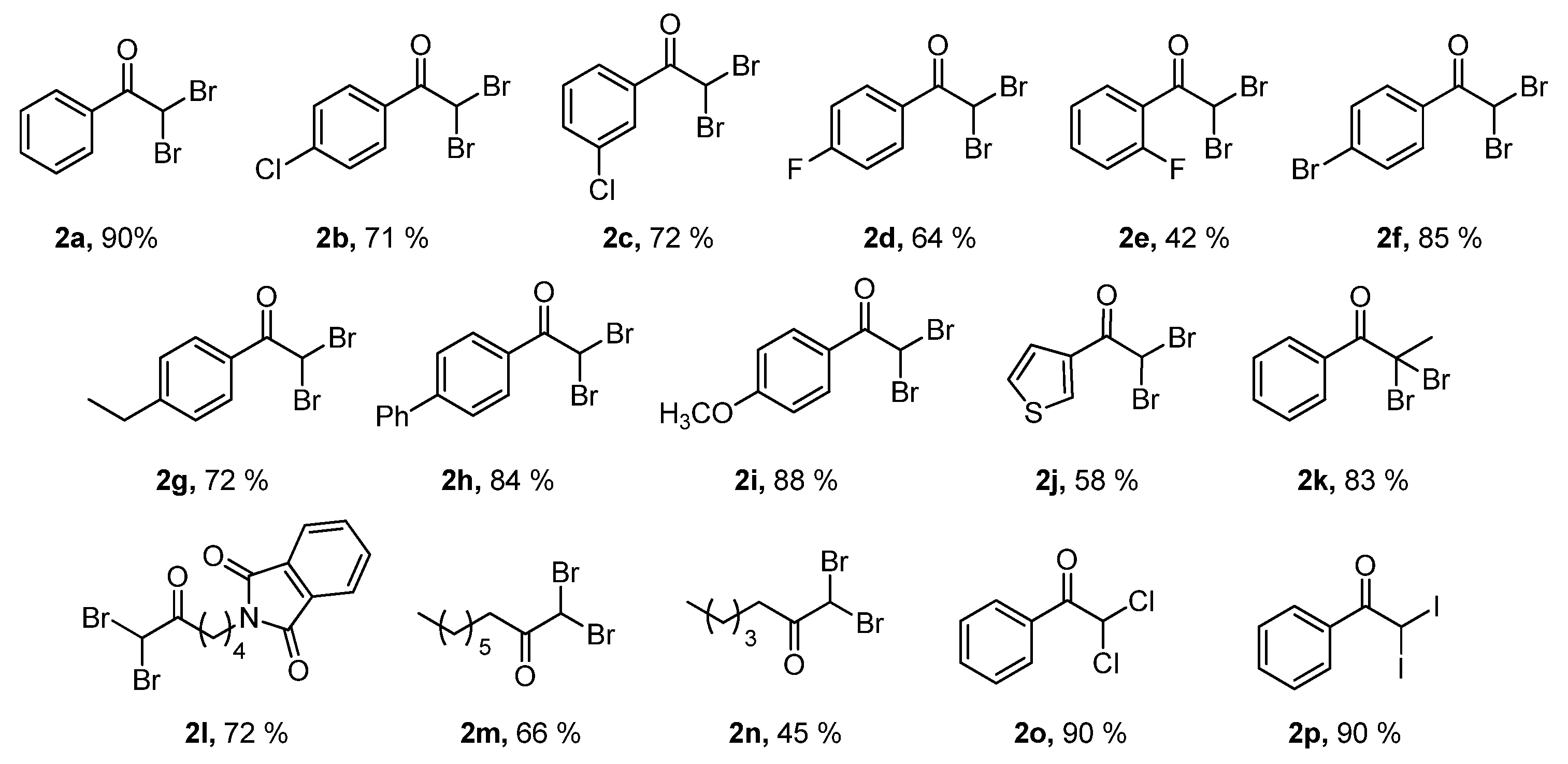
2. Results
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beller, M.; Seayad, J.; Tillack, A.; Jiao, H. Catalytic Markovnikov and anti-Markovnikov Functionalization of Alkenes and Alkynes: Recent Developments and Trends. Angew. Chem. Int. Ed. 2004, 43, 3368–3398. [Google Scholar] [CrossRef] [PubMed]
- Tlahuext-Aca, A.; Hopkinson, M.N.; Garza-Sanchez, A.G.; Glorius, F. Alkyne Difunctionalization by Dual Gold/Photoredox Catalysis. Chem. Eur. J. 2016, 22, 5909–5913. [Google Scholar] [CrossRef] [PubMed]
- Koike, T.; Akita, M. Fine Design of Photoredox Systems for Catalytic Fluoromethylation of Carbon-carbon Multiple Bonds. Acc. Chem. Res. 2016, 49, 1937–1945. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Wen, J.; Yang, D.; Jing, H.; You, J.; Wang, H. Direct Difunctionalization of Alkynes with Sulfinic Acids and Molecular Iodine: A Simple and Convenient Approach to (E)-β-iodovinyl Sulfones. RSC Adv. 2015, 5, 4416–4419. [Google Scholar] [CrossRef]
- Hirner, J.J.; Faizi, D.J.; Blum, S.A. Alkoxyboration: Ring-Closing Addition of B-O σ Bonds Across Alkynes. J. Am. Chem. Soc. 2014, 136, 4740–4745. [Google Scholar] [CrossRef] [PubMed]
- Catano, B.; Lee, J.; Kim, C.; Farrell, D.; Petersen, J.L.; Xing, Y. Iron (III) Catalyzed Halo-functionalization of Alkynes. Tetrahedron Lett. 2015, 56, 4124–4127. [Google Scholar] [CrossRef]
- Xing, Y.; Zhang, M.; Ciccarelli, S.; Lee, J.; Catano, B. Au (III)-Catalyzed Formation of α-Halomethyl Ketones from Terminal Alkynes. Eur. J. Org. Chem. 2017, 781–785. [Google Scholar] [CrossRef]
- Kimpe, N.D.; Verhe, R. The Chemistry of α-Haloketones, α-Haloaldehydes, and α-Haloimines; Wiley: New York, NY, USA, 1999. [Google Scholar]
- Yang, Q.; Liu, J.; Zhang, L.; Li, C. Functionalized periodic mesoporous organosilicas for catalysis. J. Mater. Chem. 2009, 19, 1945–1955. [Google Scholar] [CrossRef]
- Alauzun, J.; Besson, E.; Mehdi, A.; Reyé, C.; Corriu, R.J.P. Reversible covalent chemistry of CO2: An opportunity for nanostructured hybrid organic–inorganic materials. Chem. Mater. 2008, 20, 503–513. [Google Scholar] [CrossRef]
- Asefa, T.; MacLachlan, M.J.; Coombs, N.; Ozin, G.A. Periodic mesoporous organosilicas with organic groups inside the channel walls. Nature 1999, 402, 867–871. [Google Scholar] [CrossRef]
- Loy, D.A.; Shea, K.J. Bridged polysilsesquioxanes. Highly porous hybrid organic-inorganic materials. Chem. Rev. 1995, 95, 1431–1442. [Google Scholar] [CrossRef]
- Inagaki, S.; Guan, S.; Fukushima, Y.; Ohsuna, T.; Terasaki, O. Novel mesoporous materials with a uniform distribution of organic groups and inorganic oxide in their frameworks. J. Am. Chem. Soc. 1999, 121, 9611–9614. [Google Scholar] [CrossRef]
- Shea, K.J.; Loy, D.A. Bridged polysilsesquioxanes. Molecular-engineered hybrid organic–inorganic materials. Chem. Mater. 2001, 13, 3306–3319. [Google Scholar] [CrossRef]
- Zhang, Z.; Dai, S. Preparation and characterization of novel inorganic–organic mesoscopic ordered composites with bridges formed by coordination compounds. J. Am. Chem. Soc. 2001, 123, 9204–9205. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Han, S.; Che, H.; Hua, L. Fluorescent hybrid with electron acceptor methylene viologen units inside the pore walls of mesoporous MCM-48 silica. Langmuir 2010, 26, 3555–3561. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Zhang, D.; Zhai, Y.; Feng, C.; Chen, J.; Li, H. Periodic mesoporous organosilicas: A type of hybrid support for water mediated reactions. Chem. Asian J. 2007, 2, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Watkins, E.; Ilavsky, J.; Metroke, T.L.; Wang, P.; Lee, B.; Schaefer, DW. Water-barrier properties of mixed bis[trimethoxysilylpropyl]amine and vinyltriacetoxysilane films. J. Phys. Chem. B 2007, 111, 7041–7051. [Google Scholar] [CrossRef] [PubMed]
- Hunks, W.J.; Ozin, G.A. Challenges and advances in the chemistry of periodic mesoporous organosilicas (PMOs). J. Mater. Chem. 2005, 15, 3716–3724. [Google Scholar] [CrossRef]
- Hartmann, S.; Brandhuber, D.; Hüsing, N. Glycol-modified silanes: Novel possibilities for the synthesis of hierarchically organized (hybrid) porous materials. Acc. Chem. Res. 2017, 40, 885–894. [Google Scholar] [CrossRef] [PubMed]
- Mehdi, A.; Reye, C.; Corriu, R. From molecular chemistry to hybrid nanomaterials. Design and functionalization. Chem. Soc. Rev. 2011, 40, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Park, S.S.; Shin, J.H.; Zhao, D.; Ha, C.-S. Free-standing and bridged amine-functionalized periodic mesoporous organosilica films. J. Mater. Chem. 2010, 20, 7854–7858. [Google Scholar] [CrossRef]
- Nguyen, T.P.; Hesemann, P.; Tran, T.M.L.; Moreau, J.J.E. Nanostructured polysilsesquioxanes bearing amine and ammonium groups by micelle templating using anionic surfactants. J. Mater. Chem. 2010, 20, 3910–3917. [Google Scholar] [CrossRef]
- Corriu, R.J.P.; Mehdi, A.; Reyé, C.; Thieuleux, C. Control of coordination chemistry in both the framework and the pore channels of mesoporous hybrid materials. New J. Chem. 2003, 27, 905–908. [Google Scholar] [CrossRef]
- Zhou, G.; Simerly, T.; Golovko, L.; Tychinin, I.; Trachevsky, V.; Gomza, Y.; Vasiliev, A. Highly functionalized bridged silsesquioxanes. J. Sol-Gel Sci. Technol. 2012, 62, 470–482. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |



| Entry [a] | Catalyst | H2O Equiv. | Solvent (1.25 mL) | Yield [b] | |
|---|---|---|---|---|---|
| 2a | 3 | ||||
| 1 | Gel P1 | 5 | 1,4 Dioxane | 0 | 16 |
| 2 | Gel P1 | 5 | Acetonitrile | 6 | 23 |
| 3 | Gel P1 | 5 | THF | 0 | 24 |
| 4 | Gel P1 | 5 | Acetone | 45 | 5 |
| 5 | Gel P1 | 10 | Acetone | 53 | 6 |
| 6 | Gel P1 | 50 | Acetone | 90 | 8 |
| 7 | Silica gel [c] | 50 | Acetone | 68 | 10 |
| 8 | No | 50 | Acetone | 78 | 10 |
| 9 | No | 0 | Acetone | 0 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domena, J.; Chong, C.; Johnson, Q.R.; Chauhan, B.P.S.; Xing, Y. Highly Efficient Recyclable Sol Gel Polymer Catalyzed One Pot Difunctionalization of Alkynes. Molecules 2018, 23, 1879. https://doi.org/10.3390/molecules23081879
Domena J, Chong C, Johnson QR, Chauhan BPS, Xing Y. Highly Efficient Recyclable Sol Gel Polymer Catalyzed One Pot Difunctionalization of Alkynes. Molecules. 2018; 23(8):1879. https://doi.org/10.3390/molecules23081879
Chicago/Turabian StyleDomena, Justin, Carlos Chong, Qiaxian R. Johnson, Bhanu P. S. Chauhan, and Yalan Xing. 2018. "Highly Efficient Recyclable Sol Gel Polymer Catalyzed One Pot Difunctionalization of Alkynes" Molecules 23, no. 8: 1879. https://doi.org/10.3390/molecules23081879