Genistein Improves Skin Flap Viability in Rats: A Preliminary In Vivo and In Vitro Investigation
Abstract
:1. Introduction
2. Results
2.1. Macroscopic Analysis of Flap Survival
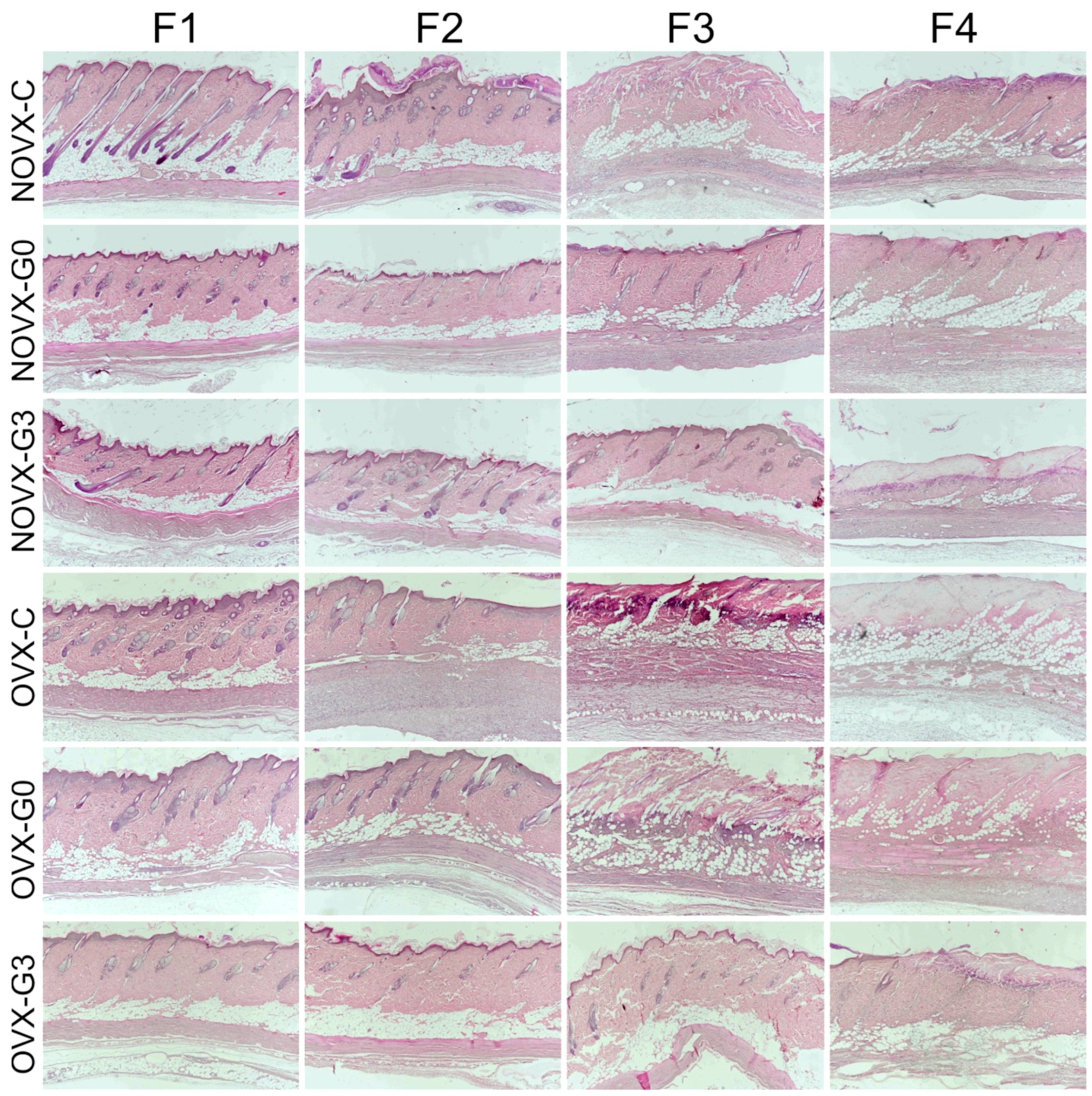
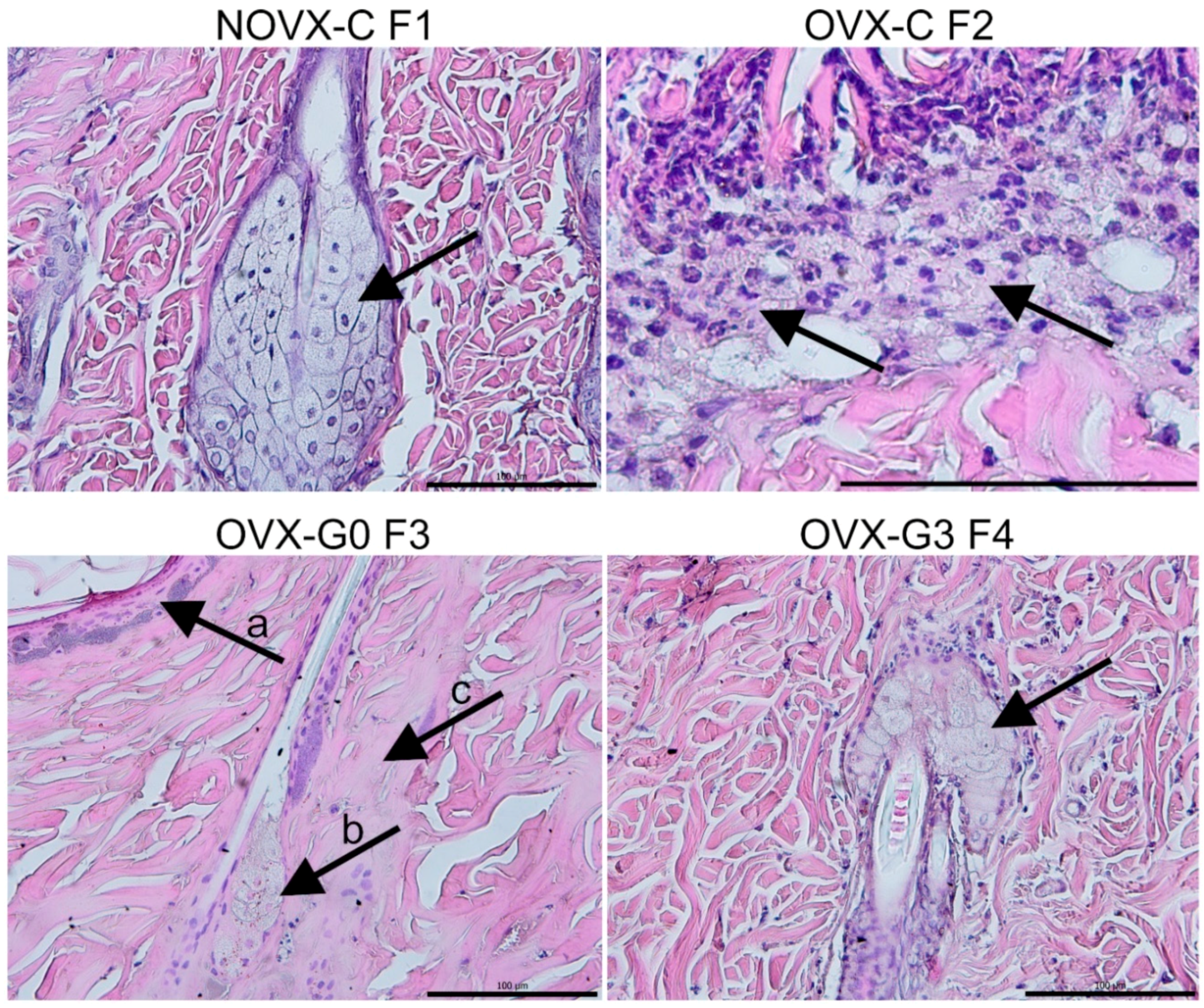
2.2. Flap Histology
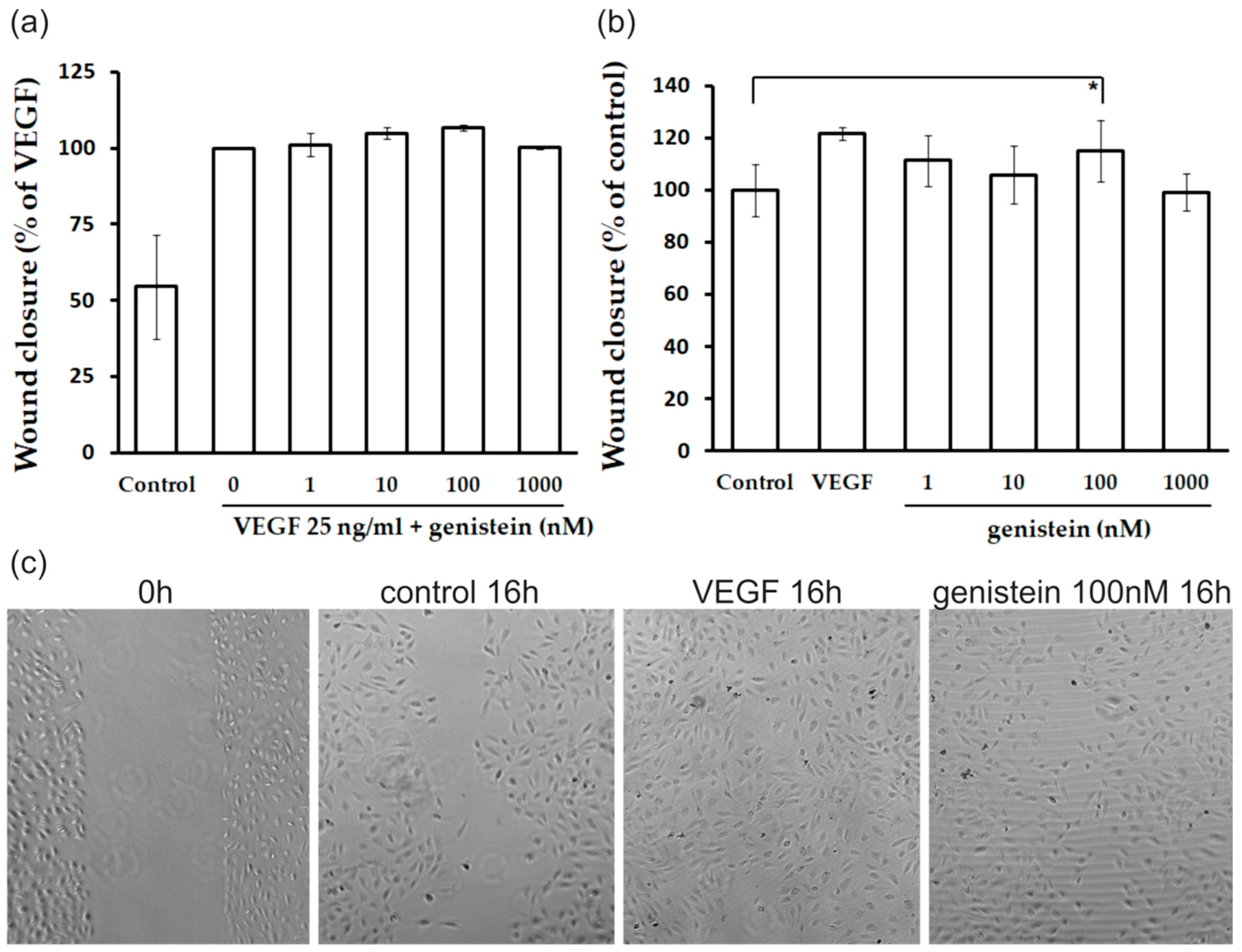
2.3. Proliferation and Migration of Endothelial Cells
2.4. Gene Expression Profile of Endothelial Cells
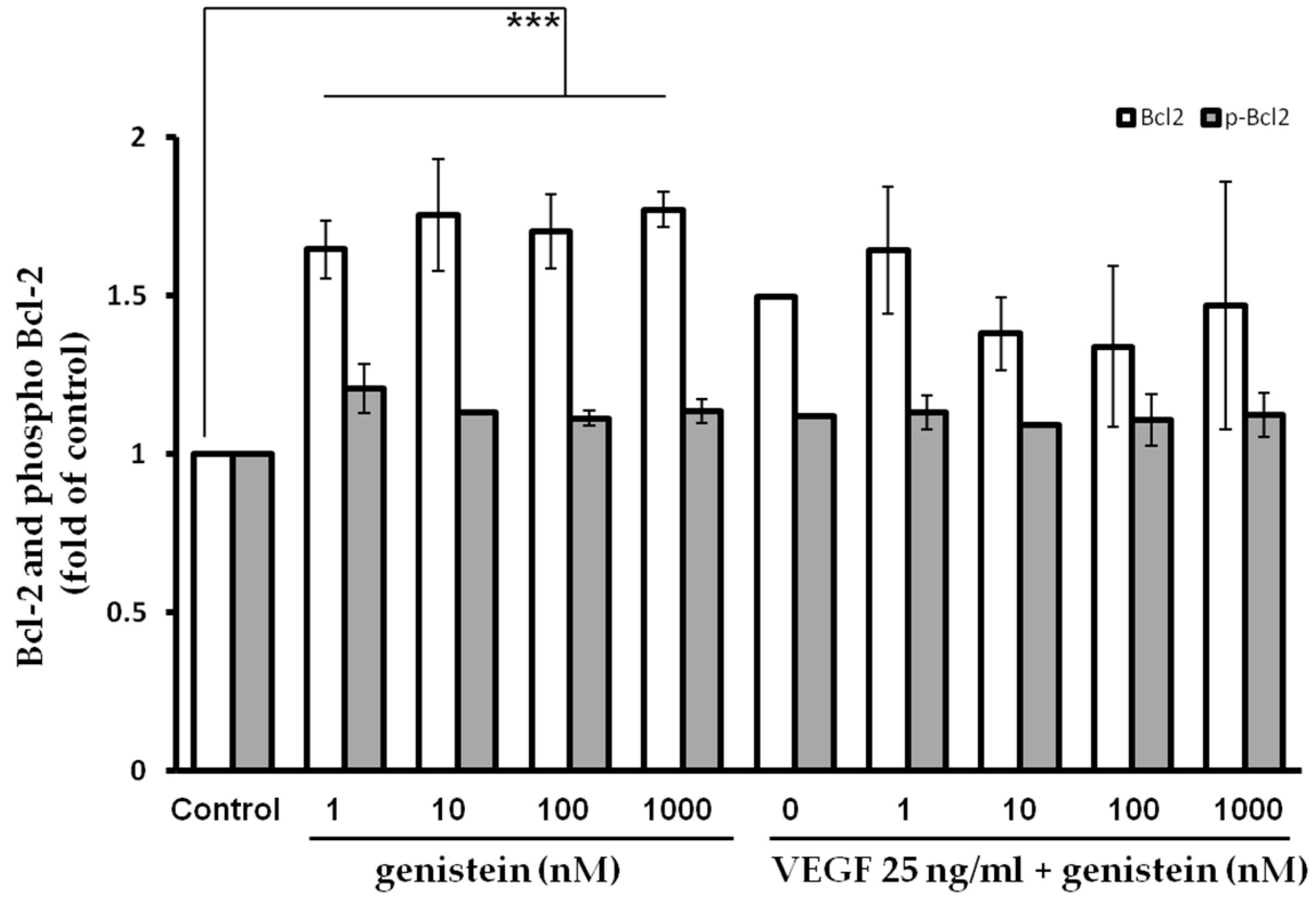
2.5. Bcl-2 Quantification after Genistein Treatment
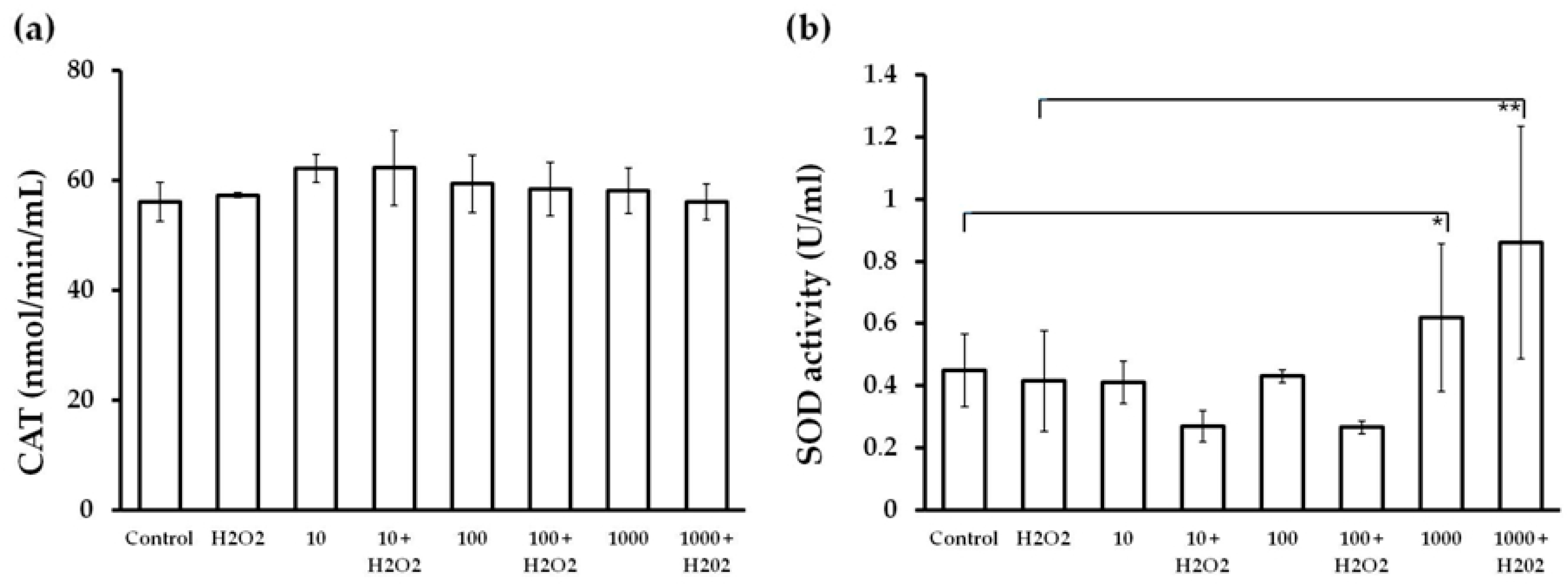
2.6. CAT and SOD Activity in Endothelial Cells
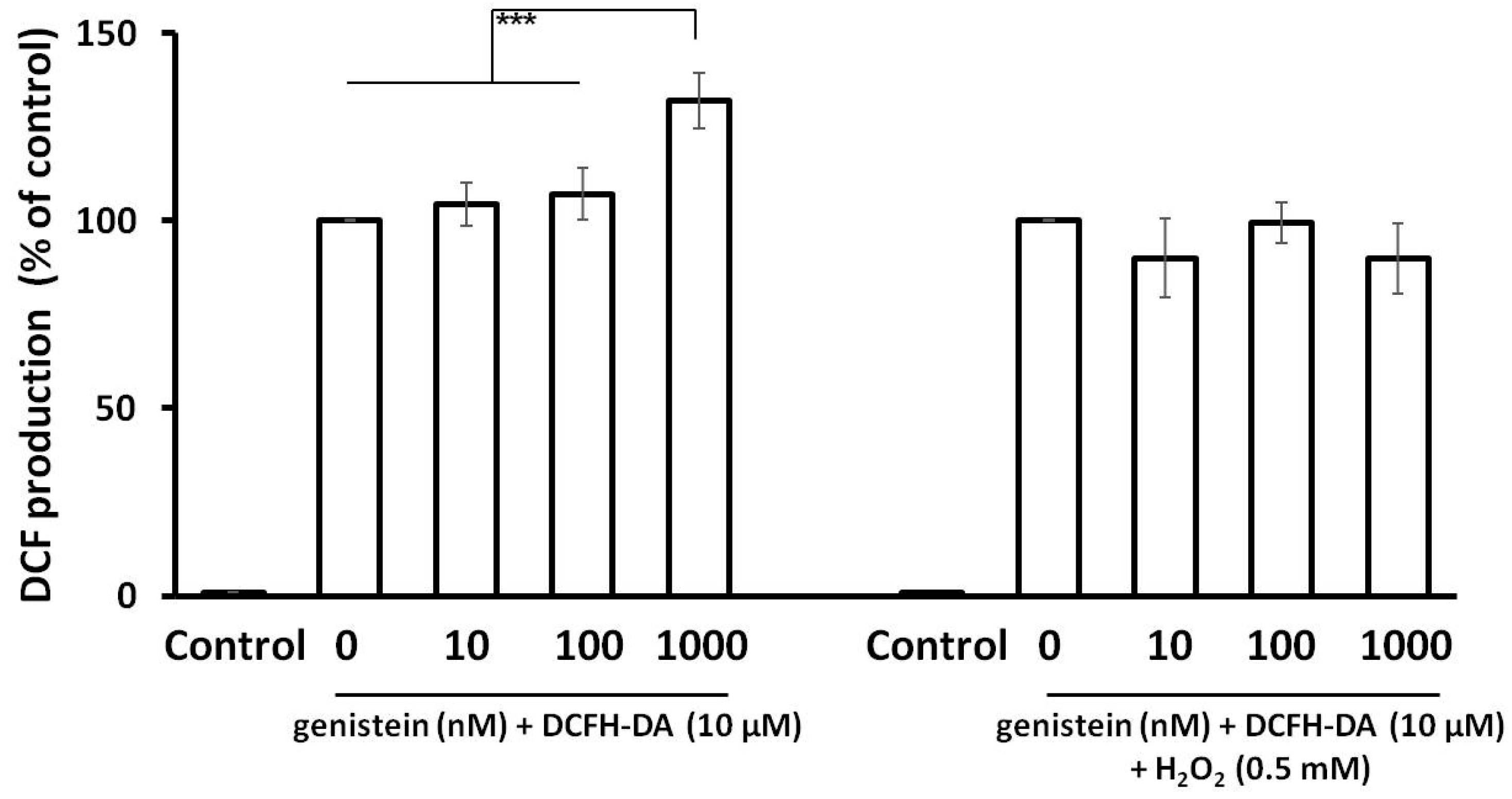
2.7. Genistein and ROS
3. Discussion
4. Materials and Methods
4.1. Animal Model
4.2. Genistein Treatment
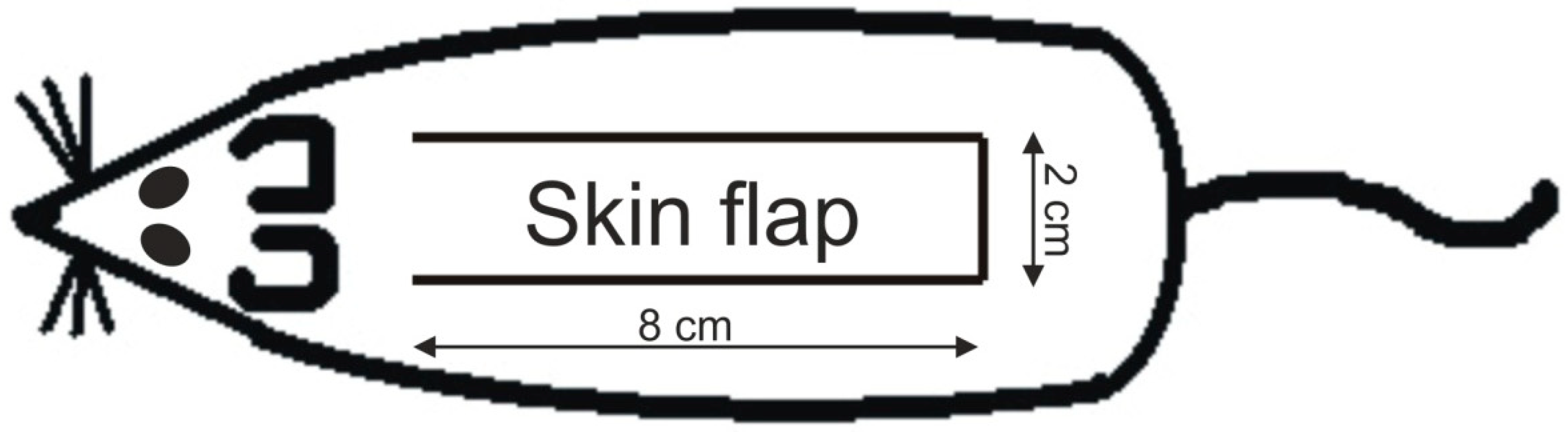
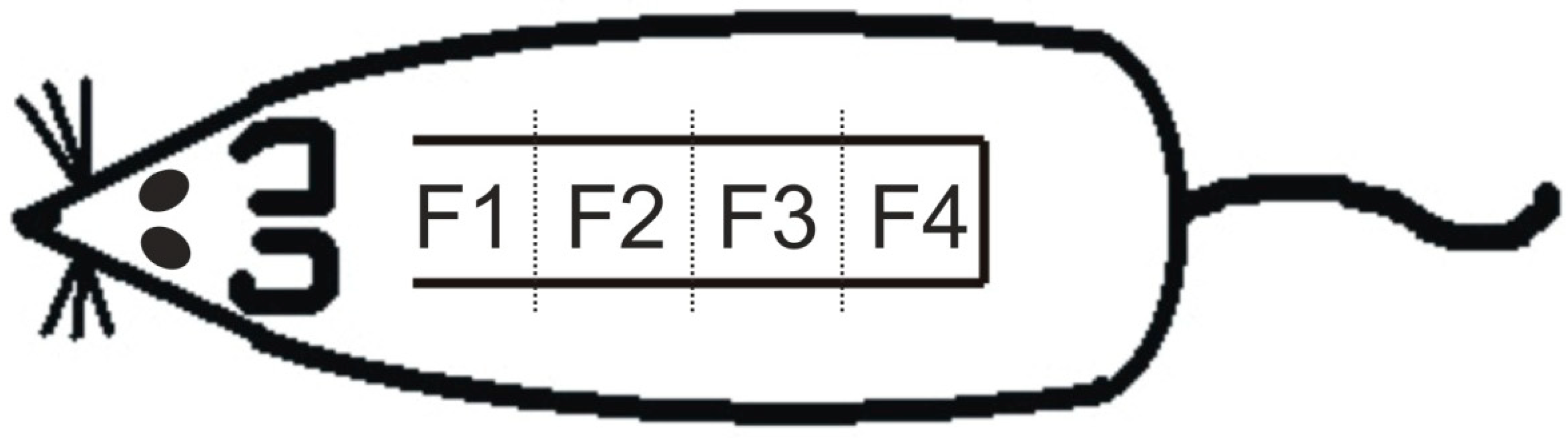
4.3. Measurement of Survived Area on the Skin Flap
4.4. Basic Histology
4.5. Endothelial Cells
4.6. Cell Viability Assay
4.7. In Vitro Migration (Wound Healing) Assay
4.8. RNA Isolation and cDNA Synthesis
4.9. Gene Expression Profiling
4.10. PCR Data Analysis and Statistics
4.11. Flow Cytometric (FCM) Analysis of Genistein Effect on Pro-/Anti-Apoptotic Gene Expressions
4.12. CAT and SOD Activity
4.13. Detection of Intracellular Oxidative Stress
4.14. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Emmerson, E.; Hardman, M.J. The role of estrogen deficiency in skin ageing and wound healing. Biogerontology 2012, 13, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Toutain, C.E.; Brouchet, L.; Raymond-Letron, I.; Vicendo, P.; Berges, H.; Favre, J.; Fouque, M.J.; Krust, A.; Schmitt, A.M.; Chambon, P.; et al. Prevention of skin flap necrosis by estradiol involves reperfusion of a protected vascular network. Circ. Res. 2009, 104, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Pinfildi, C.E.; Liebano, R.E.; Hochman, B.S.; Ferreira, L.M. Helium-neon laser in viability of random skin flap in rats. Lasers Surg. Med. 2005, 37, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Kryger, Z.; Zhang, F.; Dogan, T.; Cheng, C.; Lineaweaver, W.C.; Buncke, H.J. The effects of VEGF on survival of a random flap in the rat: Examination of various routes of administration. Br. J. Plast. Surg. 2000, 53, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.; Kunar, D.R.; Cummings, C.W. Effect of pentoxyfylline on myocutaneous flap viability in pigs. Otolaryngol. Head Neck Surg. 1993, 109, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Bailet, J.W.; Hoffman, L.F.; Trachy, R.E.; Weymuller, E.A. The effect of nifedipine on skin flap survival in rats. Laryngoscope 1994, 104, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Ulusoy, M.G.; Uysal, A.; Kocer, U.; Karaaslan, O.; Cuzdan, S.S.; Ayyildiz, A.; Ustun, H. Improved flap viability with site-specific delivery of sildenafil citrate using fibrin glue. Ann. Plast. Surg. 2005, 55, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Richards, L.; Lineaweaver, W.C.; Stile, F.; Zhang, J.; Zhang, F. Effect of hyperbaric oxygen therapy on the tubed pedicle flap survival in a rat model. Ann. Plast. Surg. 2003, 50, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Kuczmannova, A.; Gal, P.; Varinska, L.; Treml, J.; Kovac, I.; Novotny, M.; Vasilenko, T.; Dall’Acqua, S.; Nagy, M.; Mucaji, P. Agrimonia eupatoria L. And Cynara cardunculus L. Water infusions: Phenolic profile and comparison of antioxidant activities. Molecules 2015, 20, 20538–20550. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.S.; Pinfildi, C.E.; Gomes, H.C.; Liebano, R.E.; Arias, V.E.; Silveira, T.S.; Ferreira, L.M. Effect of low-level laser therapy with output power of 30 mw and 60 mw in the viability of a random skin flap. Photomed. Laser Surg. 2010, 28, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Kelly, P.M.; Keely, N.O.; Bright, S.A.; Yassin, B.; Ana, G.; Fayne, D.; Zisterer, D.M.; Meegan, M.J. Novel selective estrogen receptor ligand conjugates incorporating endoxifen-combretastatin and cyclofenil-combretastatin hybrid scaffolds: Synthesis and biochemical evaluation. Molecules 2017, 22, 1440. [Google Scholar] [CrossRef] [PubMed]
- Uifalean, A.; Schneider, S.; Ionescu, C.; Lalk, M.; Iuga, C.A. Soy isoflavones and breast cancer cell lines: Molecular mechanisms and future perspectives. Molecules 2016, 21, 13. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A. Potential risks and benefits of phytoestrogen-rich diets. Int. J. Vitam. Nutr. Res. 2003, 73, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Mezei, O.; Banz, W.J.; Steger, R.W.; Peluso, M.R.; Winters, T.A.; Shay, N. Soy isoflavones exert antidiabetic and hypolipidemic effects through the PPAR pathways in obese Zucker rats and murine RAW 264.7 cells. J. Nutr. 2003, 133, 1238–1243. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Lee, S.M.; Jung, I.K.; Lim, Y.; Kim, J.H. Effects of genistein on early-stage cutaneous wound healing. Biochem. Biophys. Res. Commun. 2011, 410, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Vasilenko, T.; Slezak, M.; Novotny, M.; Kovac, I.; Durkac, J.; Tomkova, I.; Torma, N.; Vrzgula, A.; Lenhardt, L.; Levkut, M.; et al. Pre-and/or postsurgical administration of estradiol benzoate increases skin flap viability in female rats. Aesthet. Plast. Surg. 2013, 37, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef] [PubMed]
- Morito, K.; Hirose, T.; Kinjo, J.; Hirakawa, T.; Okawa, M.; Nohara, T.; Ogawa, S.; Inoue, S.; Muramatsu, M.; Masamune, Y. Interaction of phytoestrogens with estrogen receptors alpha and beta. Biol. Pharm. Bull. 2001, 24, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Deschamps, A.M.; Murphy, E. Activation of a novel estrogen receptor, GPER, is cardioprotective in male and female rats. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1806–H1813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Etgen, A.M.; Jover-Mengual, T.; Zukin, R.S. Neuroprotective actions of estradiol and novel estrogen analogs in ischemia: Translational implications. Front. Neuroendocrinol. 2011, 32, 336–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, S.; Brown, C.M.; Wise, P.M. Neuroprotective effects of estrogens following ischemic stroke. Front. Neuroendocrinol. 2009, 30, 201–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campbell, L.; Emmerson, E.; Davies, F.; Gilliver, S.C.; Krust, A.; Chambon, P.; Ashcroft, G.S.; Hardman, M.J. Estrogen promotes cutaneous wound healing via estrogen receptor beta independent of its antiinflammatory activities. J. Exp. Med. 2010, 207, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Perzelova, V.; Sabol, F.; Vasilenko, T.; Novotny, M.; Kovac, I.; Slezak, M.; Durkac, J.; Holly, M.; Pilatova, M.; Szabo, P.; et al. Pharmacological activation of estrogen receptors-alpha and-beta differentially modulates keratinocyte differentiation with functional impact on wound healing. Int. J. Mol. Med. 2016, 37, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Kanda, N.; Watanabe, S. 17beta-estradiol inhibits oxidative stress-induced apoptosis in keratinocytes by promoting Bcl-2 expression. J. Investig. Dermatol. 2003, 121, 1500–1509. [Google Scholar] [CrossRef] [PubMed]
- Spyridopoulos, I.; Sullivan, A.B.; Kearney, M.; Isner, J.M.; Losordo, D.W. Estrogen-receptor-mediated inhibition of human endothelial cell apoptosis. Estradiol as a survival factor. Circulation 1997, 95, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Jurzak, M.; Adamczyk, K.; Antonczak, P.; Garncarczyk, A.; Kusmierz, D.; Latocha, M. Evaluation of genistein ability to modulate CTGF mrna/protein expression, genes expression of TGFβ isoforms and expression of selected genes regulating cell cycle in keloid fibroblasts in vitro. Acta Pol. Pharm. 2014, 71, 972–986. [Google Scholar] [PubMed]
- Liu, P.Y.; Liu, K.; Wang, X.T.; Badiavas, E.; Rieger-Christ, K.M.; Tang, J.B.; Summerhayes, I.C. Efficacy of combination gene therapy with multiple growth factor cDNAs to enhance skin flap survival in a rat model. DNA Cell Boil. 2005, 24, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Epstein, S.E.; Kornowski, R.; Fuchs, S.; Dvorak, H.F. Angiogenesis therapy: Amidst the hype, the neglected potential for serious side effects. Circulation 2001, 104, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Ge, J.; Lei, Y.; Lu, X. Improvement effect of estrogen on flap reperfusion injury and blood supply. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2009, 23, 964–968. [Google Scholar] [PubMed]
- Gopinathan, G.; Milagre, C.; Pearce, O.M.; Reynolds, L.E.; Hodivala-Dilke, K.; Leinster, D.A.; Zhong, H.; Hollingsworth, R.E.; Thompson, R.; Whiteford, J.R.; et al. Interleukin-6 stimulates defective angiogenesis. Cancer Res. 2015, 75, 3098–3107. [Google Scholar] [CrossRef] [PubMed]
- Mukwaya, A.; Peebo, B.; Xeroudaki, M.; Ali, Z.; Lennikov, A.; Jensen, L.; Lagali, N. Factors regulating capillary remodeling in a reversible model of inflammatory corneal angiogenesis. Sci. Rep. 2016, 6, 32137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagy, J.A.; Vasile, E.; Feng, D.; Sundberg, C.; Brown, L.F.; Manseau, E.J.; Dvorak, A.M.; Dvorak, H.F. VEGF-A induces angiogenesis, arteriogenesis, lymphangiogenesis, and vascular malformations. Cold Spring Harb. Symp. Quant. Boil. 2002, 67, 227–237. [Google Scholar] [CrossRef]
- Hayashi, H.; Sano, H.; Seo, S.; Kume, T. The Foxc2 transcription factor regulates angiogenesis via induction of integrin beta3 expression. J. Biol. Chem. 2008, 283, 23791–23800. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.C.; Chuang, S.M.; Hsu, C.J.; Tsai, C.H.; Wang, S.W.; Tang, C.H. CTGF increases vascular endothelial growth factor-dependent angiogenesis in human synovial fibroblasts by increasing miR-210 expression. Cell. Death Dis. 2014, 5, e1485. [Google Scholar] [CrossRef] [PubMed]
- Sertie, A.L.; Sossi, V.; Camargo, A.A.; Zatz, M.; Brahe, C.; Passos-Bueno, M.R. Collagen xviii, containing an endogenous inhibitor of angiogenesis and tumor growth, plays a critical role in the maintenance of retinal structure and in neural tube closure (knobloch syndrome). Hum. Mol. Genet. 2000, 9, 2051–2058. [Google Scholar] [CrossRef] [PubMed]
- Marini, H.; Polito, F.; Altavilla, D.; Irrera, N.; Minutoli, L.; Calo, M.; Adamo, E.B.; Vaccaro, M.; Squadrito, F.; Bitto, A. Genistein aglycone improves skin repair in an incisional model of wound healing: A comparison with raloxifene and oestradiol in ovariectomized rats. Br. J. Pharmacol. 2010, 160, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Pavese, J.M.; Krishna, S.N.; Bergan, R.C. Genistein inhibits human prostate cancer cell detachment, invasion, and metastasis. Am. J. Clin. Nutr. 2014, 100, 431S–436S. [Google Scholar] [CrossRef] [PubMed]
- Xiong, P.; Wang, R.; Zhang, X.; DeLa Torre, E.; Leon, F.; Zhang, Q.; Zheng, S.; Wang, G.; Chen, Q.H. Design, synthesis, and evaluation of genistein analogues as anti-cancer agents. Anti-Cancer Med. Chem. 2015, 15, 1197–1203. [Google Scholar] [CrossRef]
- Williams, R.J.; Spencer, J.P.; Rice-Evans, C. Flavonoids: Antioxidants or signalling molecules? Free Radic. Biol. Med. 2004, 36, 838–849. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Rimbach, G.; Moini, H.; Weber, S.; Packer, L. ESR and cell culture studies on free radical-scavenging and antioxidant activities of isoflavonoids. Toxicology 2002, 179, 171–180. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, Z.; Pang, X.; Yang, J.; Yu, H.; Zhang, Y.; Zhou, H.; Zhao, J. MiR-34a/sirtuin-1/foxo3a is involved in genistein protecting against ox-LDL-induced oxidative damage in HUVECs. Toxicol. Lett. 2017, 277, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Eo, H.; Lee, H.J.; Lim, Y. Ameliorative effect of dietary genistein on diabetes induced hyper-inflammation and oxidative stress during early stage of wound healing in alloxan induced diabetic mice. Biochem. Biophys. Res. Commun. 2016, 478, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Lin, Y.C.; Ma, X.Y.; Jiang, Z.Y.; Lan, S.P. High concentrations of genistein exhibit pro-oxidant effects in primary muscle cells through mechanisms involving 5-lipoxygenase-mediated production of reactive oxygen species. Food Chem. Toxicol. 2014, 67, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Defilippi, P.; van Hinsbergh, V.; Bertolotto, A.; Rossino, P.; Silengo, L.; Tarone, G. Differential distribution and modulation of expression of alpha 1/beta 1 integrin on human endothelial cells. J. Cell Boil. 1991, 114, 855–863. [Google Scholar] [CrossRef] [Green Version]
- Van Hinsbergh, V.W.; Binnema, D.; Scheffer, M.A.; Sprengers, E.D.; Kooistra, T.; Rijken, D.C. Production of plasminogen activators and inhibitor by serially propagated endothelial cells from adult human blood vessels. Arterioscler. Thromb. Vasc. Biol. 1987, 7, 389–400. [Google Scholar] [CrossRef]
- Martinez-Poveda, B.; Quesada, A.R.; Medina, M.A. Hyperforin, a bio-active compound of St. John’s wort, is a new inhibitor of angiogenesis targeting several key steps of the process. Int. J. Cancer 2005, 117, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Chatterton, R.T.; Michel, N.; Bryk, M.; Lee, O.; Ivancic, D.; Heinz, R.; Zalles, C.M.; Helenowski, I.B.; Jovanovic, B.D.; et al. Soy isoflavone supplementation for breast cancer risk reduction: A randomized phase ii trial. Cancer Prev. Res. 2012, 5, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Lohr, J.M.; Karimi, M.; Omazic, B.; Kartalis, N.; Verbeke, C.S.; Berkenstam, A.; Frodin, J.E. A phase I dose escalation trial of AXP107-11, a novel multi-component crystalline form of genistein, in combination with gemcitabine in chemotherapy-naive patients with unresectable pancreatic cancer. Pancreatology 2016, 16, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Crisafulli, A.; Altavilla, D.; Squadrito, G.; Romeo, A.; Adamo, E.B.; Marini, R.; Inferrera, M.A.; Marini, H.; Bitto, A.; D’Anna, R.; et al. Effects of the phytoestrogen genistein on the circulating soluble receptor activator of nuclear factor kappab ligand-osteoprotegerin system in early postmenopausal women. J. Clin. Endocrinol. Metab. 2004, 89, 188–192. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |

| Group/Subgroup | No. of Rats | Ovariectomy (O)/ Sham Surgery (S) | Treatment Started | Dose mg/kg | |
|---|---|---|---|---|---|
| NOVX | -C | 8 | S—3M prior flap surgery | from the day of flap surgery | 0 |
| -G0 | 8 | S—3M prior flap surgery | from the day of flap surgery | 1 | |
| -G3 | 8 | S—3M prior flap surgery | 3 days prior flap surgery | 1 | |
| OVX | -C | 8 | O—3M prior flap surgery | from the day of flap surgery | 0 |
| -G0 | 8 | O—3M prior flap surgery | from the day of flap surgery | 1 | |
| -G3 | 8 | O—3M prior flap surgery | 3 days prior flap surgery | 1 | |
| Gene Symbol | Gene Name | Genistein + VEGF/VEGF |
|---|---|---|
| COL18A1 | Collagen Type XVIII Alpha 1 Chain | 2.2 |
| CTGF | Connective Tissue Growth Factor | 2.4 |
| CXCL5 | C-X-C Motif Chemokine Ligand 5 | 2.8 |
| IL-6 | Interleukin 6 | 2.1 |
| ITGB3 | Integrin Subunit Beta 3 | 2.6 |
| MMP-14 | Matrix Metallopeptidase 14 | 2.4 |
| TIMP-2 | TIMP Metallopeptidase Inhibitor 2 | 2.2 |
| TIMP-3 | TIMP Metallopeptidase Inhibitor 3 | 2.2 |
| VEGF-A | Vascular Endothelial Growth Factor A | 2.4 |
| Gene Symbol | Gene Name | Genistein/Control |
|---|---|---|
| CXCL10 | C-X-C Motif Chemokine Ligand 10 | −2.7 |
| SERPINF1 | Serpin Family F Member 1 | −2.1 |
| Analysis | Staining Solution | Manufacturer |
|---|---|---|
| Protein analysis | Bcl-2 (124) Mouse mAb (PE Conjugate) 1:50 | Cell Signaling Technology, Danvers, MA, USA |
| Phospho-Bcl-2 (Ser70) Rabbit mAb (Alexa Fluor® 488 Conjugate) 1:50 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fáber, L.; Kováč, I.; Mitrengová, P.; Novotný, M.; Varinská, L.; Vasilenko, T.; Kello, M.; Čoma, M.; Kuruc, T.; Petrová, K.; et al. Genistein Improves Skin Flap Viability in Rats: A Preliminary In Vivo and In Vitro Investigation. Molecules 2018, 23, 1637. https://doi.org/10.3390/molecules23071637
Fáber L, Kováč I, Mitrengová P, Novotný M, Varinská L, Vasilenko T, Kello M, Čoma M, Kuruc T, Petrová K, et al. Genistein Improves Skin Flap Viability in Rats: A Preliminary In Vivo and In Vitro Investigation. Molecules. 2018; 23(7):1637. https://doi.org/10.3390/molecules23071637
Chicago/Turabian StyleFáber, Lenka, Ivan Kováč, Petra Mitrengová, Martin Novotný, Lenka Varinská, Tomáš Vasilenko, Martin Kello, Matúš Čoma, Tomáš Kuruc, Klaudia Petrová, and et al. 2018. "Genistein Improves Skin Flap Viability in Rats: A Preliminary In Vivo and In Vitro Investigation" Molecules 23, no. 7: 1637. https://doi.org/10.3390/molecules23071637








