Exploration of the Inhibitory Potential of Varespladib for Snakebite Envenomation
Abstract
:1. Introduction
2. Results
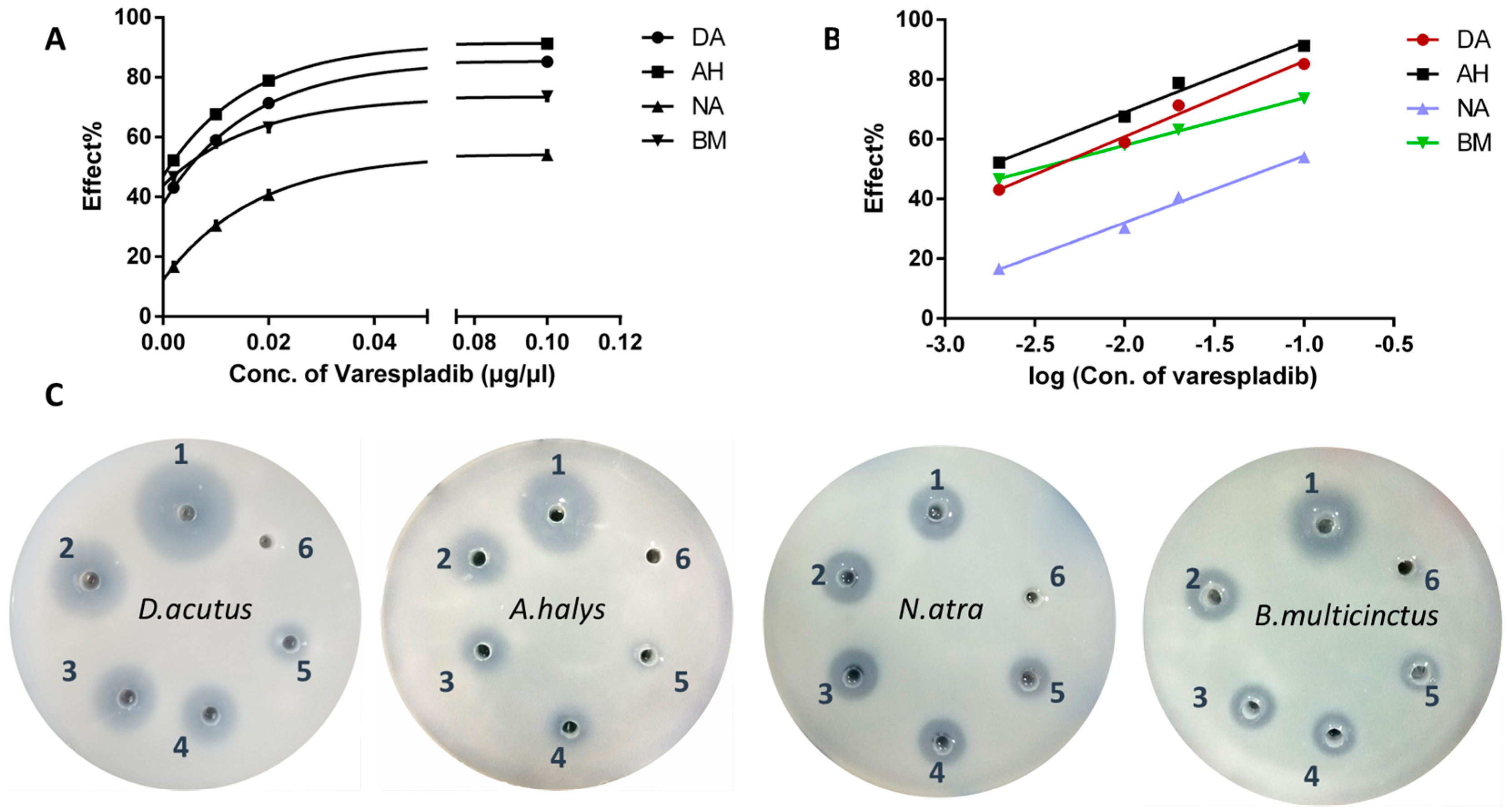
2.1. Inhibition of Phospholipase A2 In Vitro
- D. acutus, Y = 25.28X + 111.4, R2 = 0.9865; IC50 = 0.0037 μg/μL
- A. halys, AH: Y = 23.47X + 115.9, R2 = 0.9867; IC50 = 0.0016 μg/μL
- N. atra, Y = 22.36X + 76.82, R2 = 0.9916; IC50 = 0.063 μg/μL
- B. multicinctus, Y = 15.92X + 89.80, R2 = 0.9991. IC50 = 0.0032 μg/μL
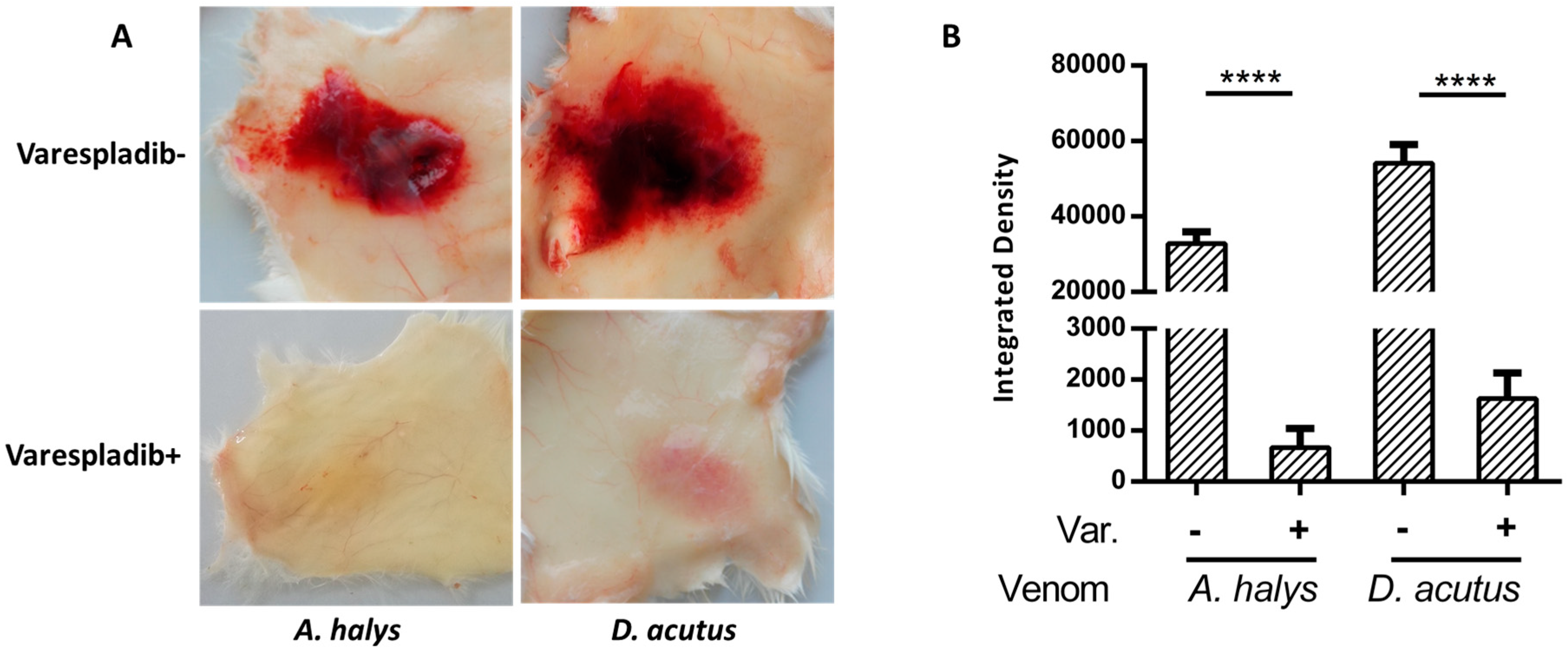
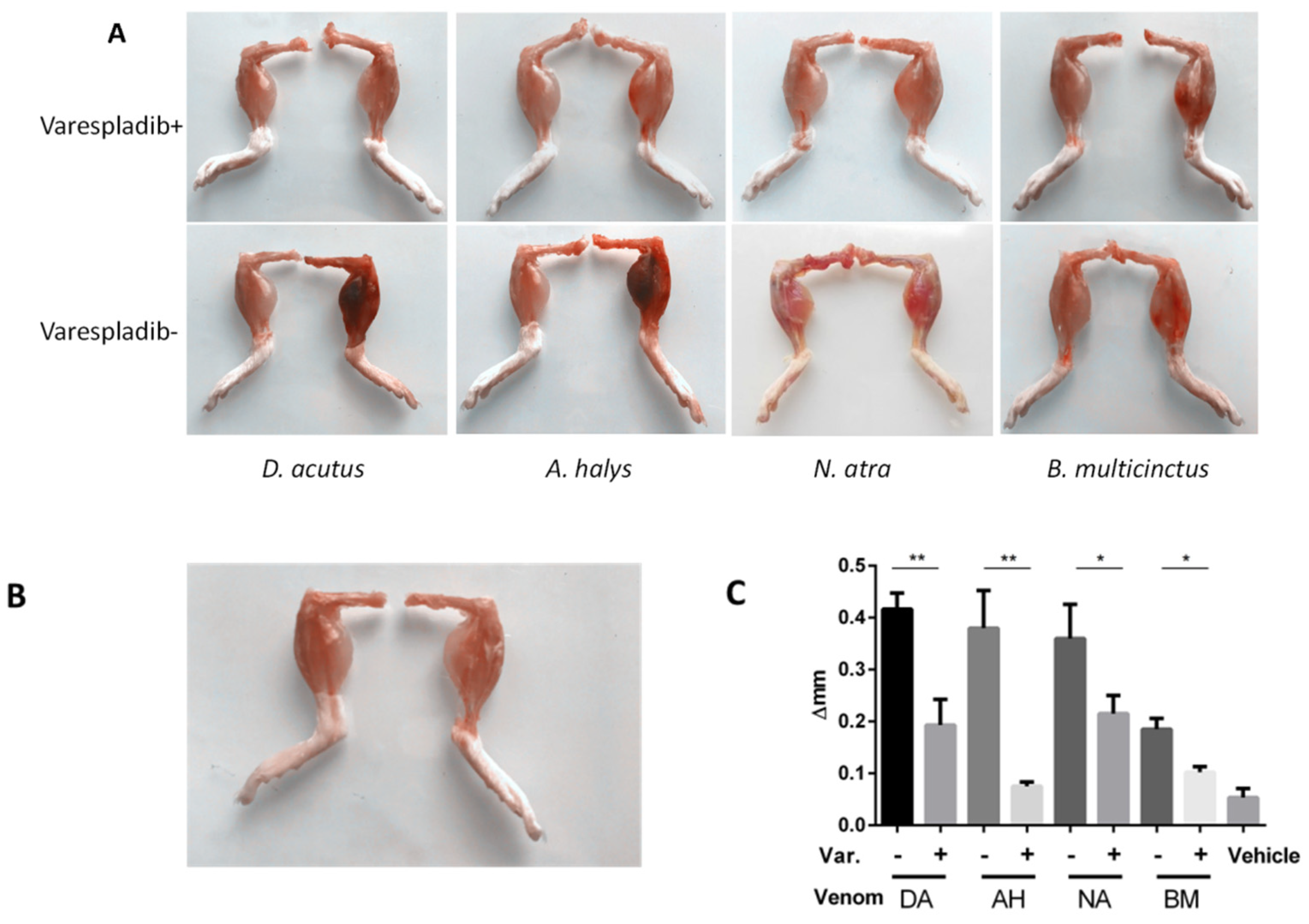
2.2. Anti-Hemorrhage Effect In Vivo
2.3. Physiological and Behavioral Signs
2.4. Local Reaction in the Gastrocnemius Muscle
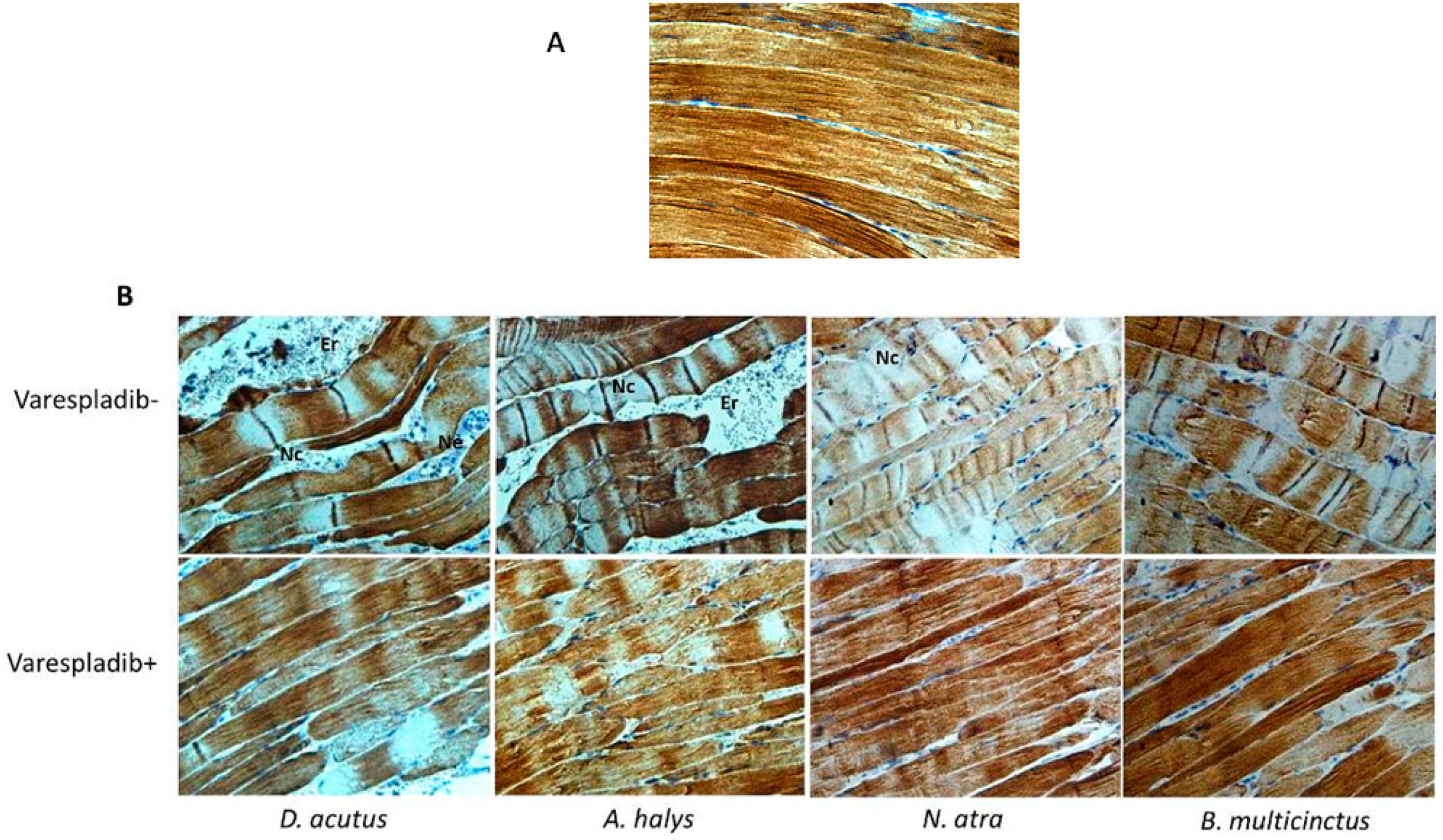
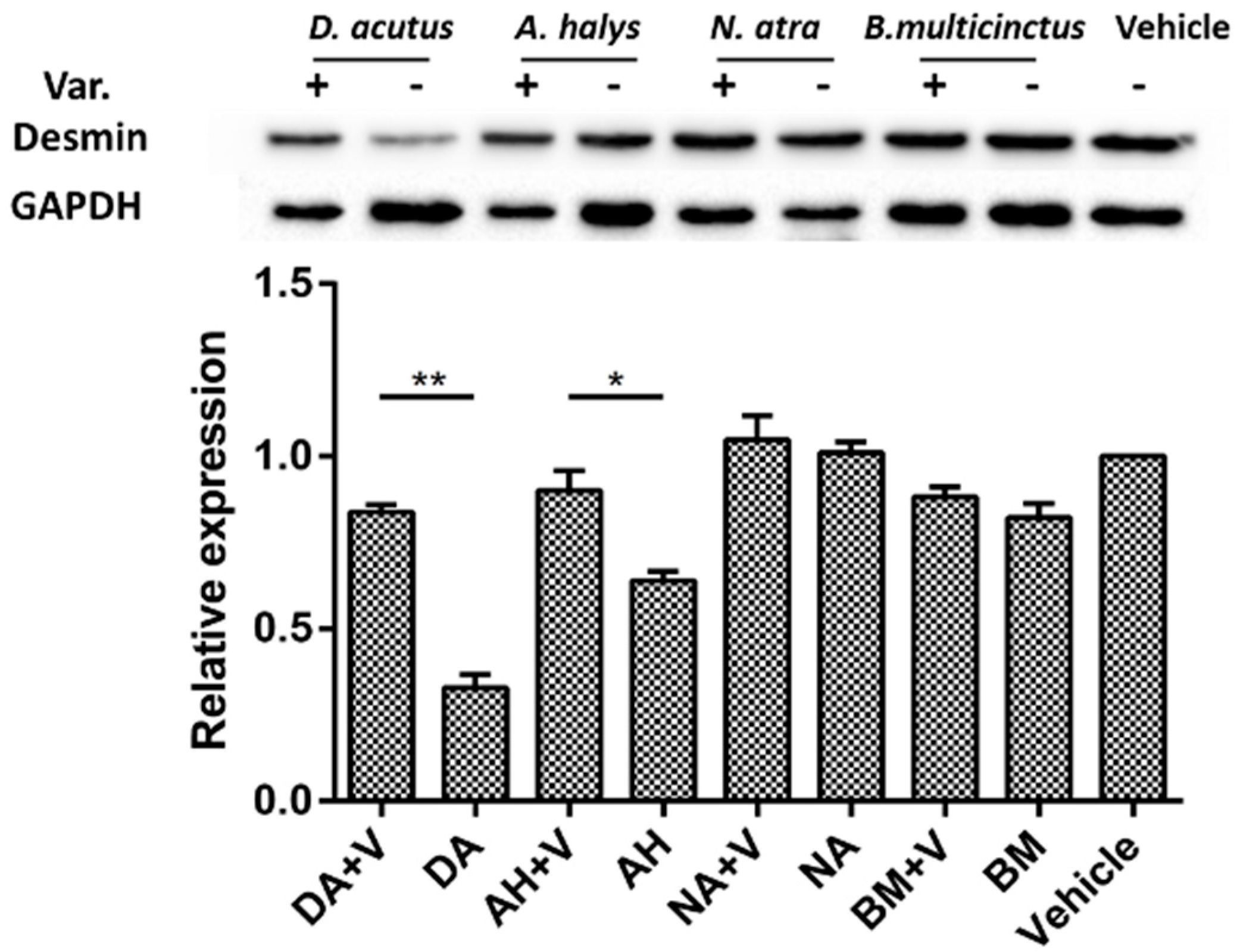
2.5. Immunohistochemistry and Desmin Loss
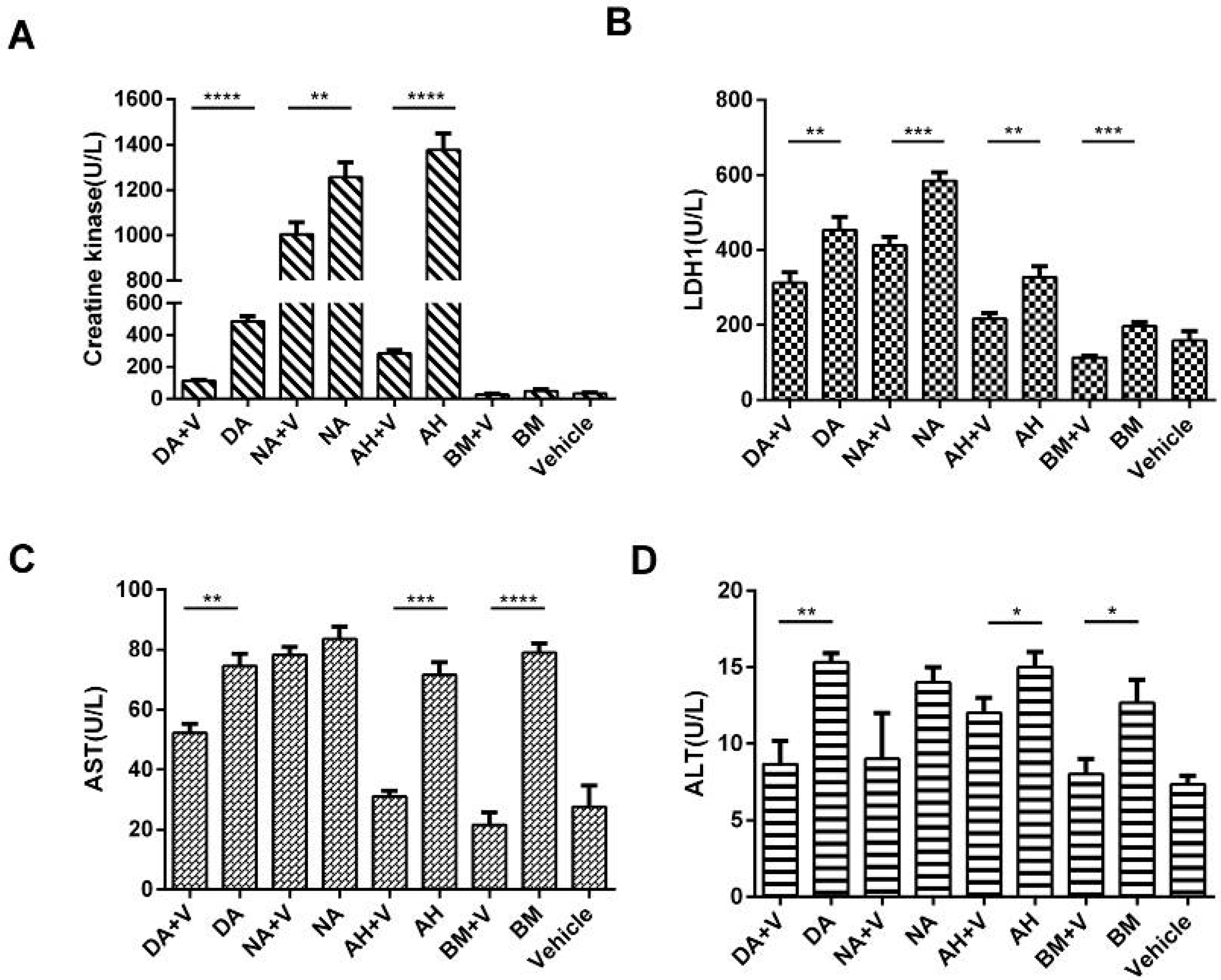
2.6. Biochemical Assays of Serum CK, LDH1, AST, and ALT Levels
2.7. The Estimation of the Median Effective Dose (ED50) of Varespladib
3. Discussion
4. Materials and Methods
4.1. Reagents and Materials
4.2. Inhibition of Varespladib on svPLA2 In Vitro
4.3. Anti-Hemorrhage Effect In Vivo
4.4. Inhibition to Myotoxicity
4.4.1. Desmin Degradation in Envenomed Gastrocnemius Muscle
4.4.2. Immunohistochemical Analysis
4.5. Estimation of the Median Effective Dose (ED50) of Varespladib against Snake Venoms
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CK | Creatine kinase |
| LDH1 | Lactate dehydrogenase isoenzyme 1 |
| AST | Aspartate transaminase |
| ALT | Alanine transaminase |
References
- Williams, D.; Gutiérrez, J.M.; Harrison, R.; Warrell, D.A.; White, J.; Winkel, K.D.; Gopalakrishnakone, P. The Global Snake Bite Initiative: An antidote for snake bite. Lancet 2010, 375, 89–91. [Google Scholar] [CrossRef]
- Rägo, L.; Marroquin, A.M.; Nübling, C.M.; Sawyer, J. Treating snake bites—A call for partnership. Lancet 2015, 386, 2252. [Google Scholar] [CrossRef]
- Visser, L.E.; Kyei-Faried, S.; Belcher, D.W.; Geelhoed, D.W.; van Leeuwen, J.S.; van Roosmalen, J. Failure of a new antivenom to treat Echis ocellatus snake bite in rural Ghana: The importance of quality surveillance. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J.; Arias, A.S.; Rodríguez, Y.; Quesada-Bernat, S.; Sánchez, L.V.; Chippaux, J.P.; Pla, D.; Gutiérrez, J.M. Preclinical evaluation of three polyspecific antivenoms against the venom of Echis ocellatus: Neutralization of toxic activities and antivenomics. Toxicon 2016, 119, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.; Engmark, M.; Milbo, C.; Johannesen, J.; Lomonte, B.; Gutiérrez, J.; Lohse, B. From Fangs to Pharmacology: The Future of Snakebite Envenoming Therapy. Curr. Pharm. Des. 2016, 22, 5270–5293. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.; Macedo, J.K.; Feoli, A.; Escalante, T.; Rucavado, A.; Gutierrez, J.M.; Fox, J.W. Muscle Tissue Damage Induced by the Venom of Bothrops asper: Identification of Early and Late Pathological Events through Proteomic Analysis. PLoS Negl. Trop. Dis. 2016, 10, e0004599. [Google Scholar] [CrossRef] [PubMed]
- Pinho, F.M.O.; Yu, L.; Burdmann, E.A. Snakebite-Induced Acute Kidney Injury in Latin America. Semin. Nephrol. 2008, 28, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Marsh, N.; Gattullo, D.; Pagliaro, P.; Losano, G. The gaboon viper, Bitis gabonica: Hemorrhagic, metabolic, cardiovascular and clinical affects of the venom. Life Sci. 1997, 61, 763–769. [Google Scholar] [CrossRef]
- Montecucco, C.; Gutiérrez, J.M.; Lomonte, B. Cellular pathology induced by snake venom phospholipase A2 myotoxins and neurotoxins: Common aspects of their mechanisms of action. Cell. Mol. Life Sci. 2008, 65, 2897–2912. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.; Vargas-Vargas, N.; Pla, D.; Sasa, M.; Rey-Suárez, P.; Sanz, L.; Gutiérrez, J.M.; Calvete, J.J.; Lomonte, B. Snake Venomics of Micrurus Alleni and Micrurus Mosquitensis from the Caribbean Region of Costa Rica Reveals Two Divergent Compositional Patterns in New World Elapids. Toxicon 2015, 107, 217–233. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M. Current challenges for confronting the public health problem of snakebite envenoming in Central America. J. Venom. Anim. Toxins Incl. Trop. Dis. 2014, 20. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, T.H.; Khatri, V.; Reifler, L.M.; Lavonas, E.J. Incidence of immediate hypersensitivity reaction and serum sickness following administration of crotalidae polyvalent immune Fab antivenom: A meta-analysis. Acad. Emerg. Med. 2012, 19, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Schiermeier, Q. Africa braced for snakebite crisis. Nature 2015, 525, 299. [Google Scholar] [CrossRef] [PubMed]
- Ranawaka, U.K.; Lalloo, D.G.; de Silva, H.J. Neurotoxicity in Snakebite-The Limits of Our Knowledge. PLoS Negl. Trop. Dis. 2013, 7, e2302. [Google Scholar] [CrossRef] [PubMed]
- Gay, C.; Maruñak, S.; Teibler, P.; Leiva, L.; Acosta, O. Effect of monospecific antibodies against baltergin in myotoxicity induced by bothrops alternatus venom from northeast of argentina. Role of metalloproteinases in muscle damage. Toxicon 2013, 63, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Vaiyapuri, S.; Wagstaff, S.C.; Harrison, R.A.; Gibbins, J.M.; Hutchinson, E.G. Evolutionary analysis of novel serine proteases in the Venom Gland transcriptome of Bitis gabonica rhinoceros. PLoS ONE 2011, 6, e21532. [Google Scholar] [CrossRef] [PubMed]
- Francis, B.R.; Jorge Da Silva, N.; Seebart, C.; Casais E Silva, L.L.; Schmidt, J.J.; Kaiser, I.I. Toxins isolated from the venom of the Brazilian coral snake (Micrurus frontalis frontalis) include hemorrhagic type phospholipases A2 and postsynaptic neurotoxins. Toxicon 1997, 35, 1193–1203. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Ponce-Soto, L.A.; Marangoni, S.L.B. Systemic and local myotoxicity induced by snake venom group II phospholipases A2: Comparison between crotoxin, crotoxin B and a Lys49 PLA2 homologue. Toxicon 2008, 51, 80–92. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, N.D.; Garcia, R.C.; Kleber Ferreira, A.; Rodrigo Batista, D.; Carlos Cassola, A.; Maria, D.; Lebrun, I.; Mendes Carneiro, S.; Castro Afeche, S.; Marcourakis, T.; et al. Neurotoxicity of coral snake phospholipases A2 in cultured rat hippocampal neurons. Brain Res. 2014, 1552, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Rudrammaji, L.M.; Machiah, K.D.; Kantha, T.P.; Gowda, T.V. Role of catalytic function in the antiplatelet activity of phospholipase A2 cobra (Naja naja naja) venom. Mol. Cell. Biochem. 2001, 219, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.; Camargo, E.A.; Ribela, M.T.C.P.; Damico, D.C.; Marangoni, S.; Antunes, E.; De Nucci, G.; Landucci, E.C.T. Inflammatory oedema induced by Lachesis muta muta (Surucucu) venom and LmTX-I in the rat paw and dorsal skin. Toxicon 2009, 53, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Garcia Denegri, M.E.; Maruñak, S.; Todaro, J.S.; Ponce-Soto, L.A.; Acosta, O.; Leiva, L. Neutralisation of the pharmacological activities of Bothrops alternatus venom by anti-PLA2 IgGs. Toxicon 2014, 86, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Snyder, D.W.; Bach, N.J.; Dillard, R.D.; Draheim, S.E.; Carlson, D.G.; Fox, N.; Roehm, N.W.; Armstrong, C.T.; Chang, C.H.; Hartley, L.W.; et al. Pharmacology of LY315920/S-5920, [[3-(aminooxoacetyl)-2-ethyl-1-(phenylmethyl)-1H-indol-4-yl]oxy] acetate, a potent and selective secretory phospholipase A2 inhibitor: A new class of anti-inflammatory drugs, SPI. J. Pharmacol. Exp. Ther. 1999, 288, 1117–1124. [Google Scholar] [PubMed]
- Lewin, M.; Samuel, S.; Merkel, J.; Bickler, P. Varespladib (LY315920) appears to be a potent, broad-spectrum, inhibitor of snake venom phospholipase A2 and a possible pre-referral treatment for envenomation. Toxins 2016, 8, 248. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Pan, H.; Liao, K.; Yang, M.; Huang, C. Snake Venom PLA 2, a Promising Target for Broad-Spectrum Antivenom Drug Development. BioMed Res. Int. 2017, 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Bustillo, S.; García-Denegri, M.E.; Gay, C.; Van de Velde, A.C.; Acosta, O.; Angulo, Y.; Lomonte, B.; Gutiérrez, J.M.; Leiva, L. Phospholipase A2 enhances the endothelial cell detachment effect of a snake venom metalloproteinase in the absence of catalysis. Chem.-Biol. Interact. 2015, 240, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Bustillo, S.; Gay, C.C.; Denegri, M.E.G.; Ponce-Soto, L.A.; de Kier Joffé, E.B.; Acosta, O.; Leiva, L.C. Synergism between baltergin metalloproteinase and Ba SPII RP4 PLA2 from Bothrops alternatus venom on skeletal muscle (C2C12) cells. Toxicon 2012, 59, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Xiong, S.; Luo, Y.; Zhong, L.; Xiao, H.; Pan, H.; Liao, K.; Yang, M.; Huang, C. Investigation of the inhibitory potential of phospholipase A2inhibitor gamma from Sinonatrix annularis to snake envenomation. Toxicon 2017, 137, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, A.; Gil, R.J.; Kulawik, T.; Pronicki, M.; Karkucińska-Wieogonekckowska, A.; Szymańska-Deogonekbińska, T.; Gil, K.; Lagwinski, N.; Czarnowska, E. Type of desmin expression in cardiomyocytes—A good marker of heart failure development in idiopathic dilated cardiomyopathy. J. Intern. Med. 2012, 272, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, A.; Gil, R.J.; Grajkowska, W.; Nasierowska-Guttmejer, A.M.; Rzezak, J.; Kulawik, T. Significance of low desmin expression in cardiomyocytes in patients with idiopathic dilated cardiomyopathy. Am. J. Cardiol. 2013, 111, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Rosenson, R.S.; Hislop, C.; Elliott, M.; Stasiv, Y.; Goulder, M.; Waters, D. Effects of varespladib methyl on biomarkers and major cardiovascular events in acute coronary syndrome patients. J. Am. Coll. Cardiol. 2010, 56, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Le, Z.; Li, X.; Yuan, P.; Liu, P.; Huang, C. Orthogonal optimization of prokaryotic expression of a natural snake venom phospholipase A2 inhibitor from Sinonatrix annularis. Toxicon 2015, 108, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Jiang, K.; Shu, P. Snakes and snake toxins. In Biotoxin Development and Utilization; Chemical Industry Press: Beijing, China, 2007; pp. 44–45. ISBN 9787122007773. [Google Scholar]
- Hamilton, M.; Russo, R.; Thurston, R. Trimmed Spearman-Karber Method for Estimating Median Lethal Concentrations in Toxicity Bioassays. Environ. Sci. Technol. 1977, 11, 714–719. [Google Scholar] [CrossRef]
Sample Availability: Samples of the snake venom lyophilized powder of D. acutus, A. halys, B. multicinctus, and N. atra are available from the authors. |
| Venom | Challenge Dose (μg) | ED50 (μg/g) (95% CI) | Standard Deviation |
|---|---|---|---|
| D. acutus | 240 | 1.14 (0.86–1.50) | 0.0608 |
| A. halys | 120 | 0.45 (0.33–0.62) | 0.0699 |
| B. multicinctus | 12 | 15.23 (12.08–19.23) | 0.0515 |
| N. atra | 28 | 22.09(17.31–28.19) | 0.0540 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhang, J.; Zhang, D.; Xiao, H.; Xiong, S.; Huang, C. Exploration of the Inhibitory Potential of Varespladib for Snakebite Envenomation. Molecules 2018, 23, 391. https://doi.org/10.3390/molecules23020391
Wang Y, Zhang J, Zhang D, Xiao H, Xiong S, Huang C. Exploration of the Inhibitory Potential of Varespladib for Snakebite Envenomation. Molecules. 2018; 23(2):391. https://doi.org/10.3390/molecules23020391
Chicago/Turabian StyleWang, Yiding, Jing Zhang, Denghong Zhang, Huixiang Xiao, Shengwei Xiong, and Chunhong Huang. 2018. "Exploration of the Inhibitory Potential of Varespladib for Snakebite Envenomation" Molecules 23, no. 2: 391. https://doi.org/10.3390/molecules23020391




