Inhibitory Effect of Flavonolignans on the P2Y12 Pathway in Blood Platelets
Abstract
:1. Introduction
2. Results
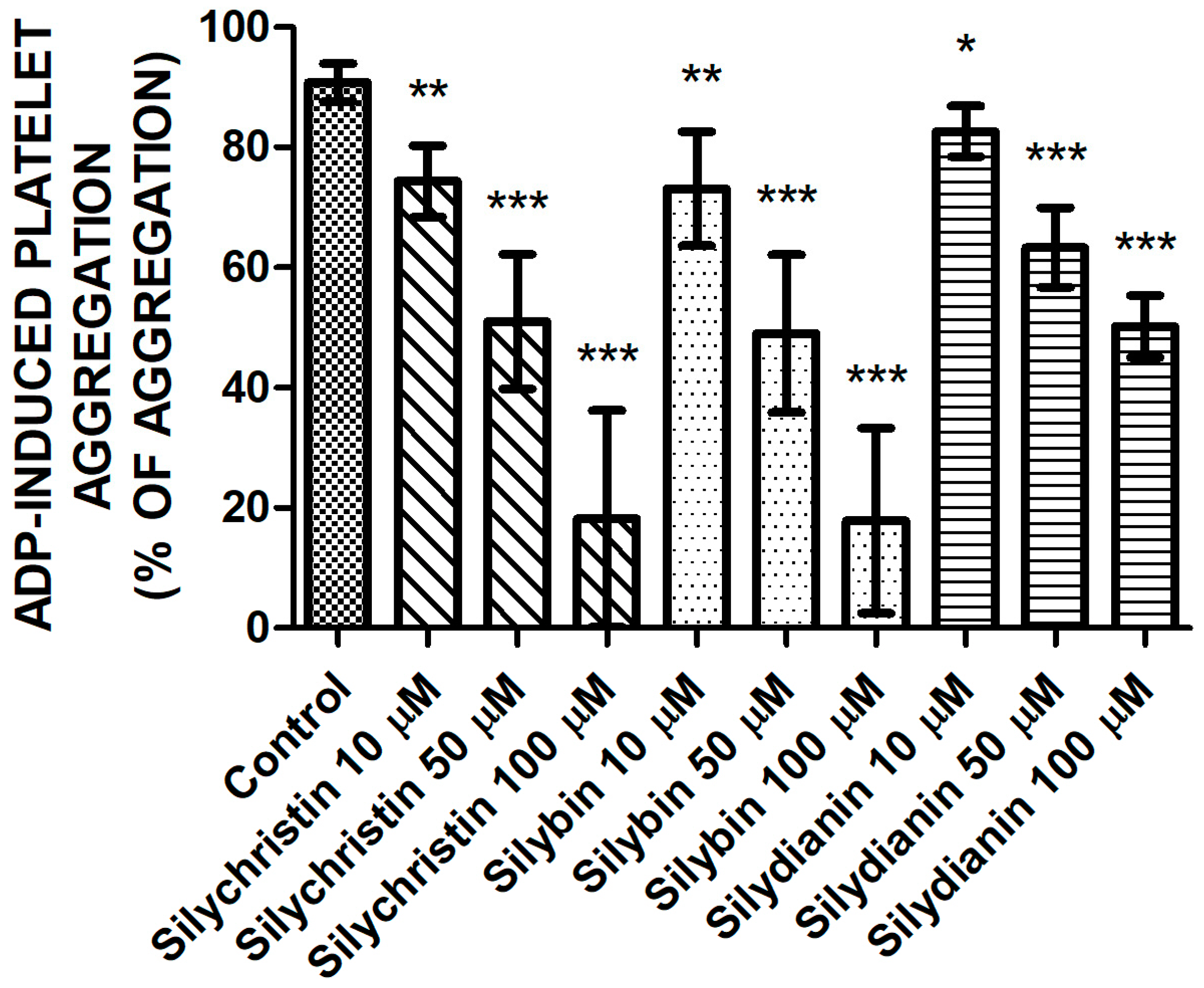
2.1. Effect of Flavonolignans on ADP Induced Blood Platelet Aggregation
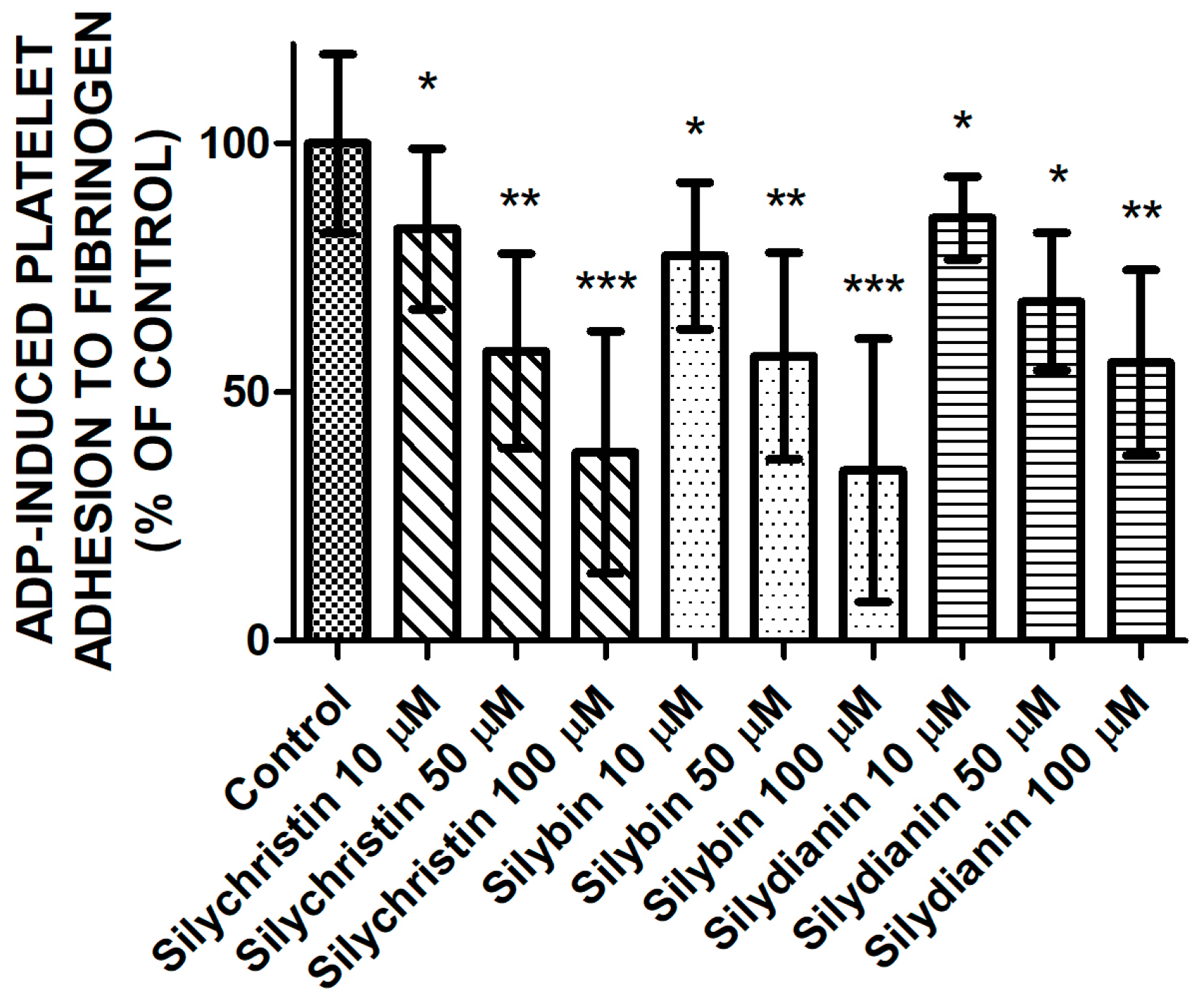
2.2. Effect of Flavonolignans on ADP Induced Blood Platelet Adhesion to Fibrinogen
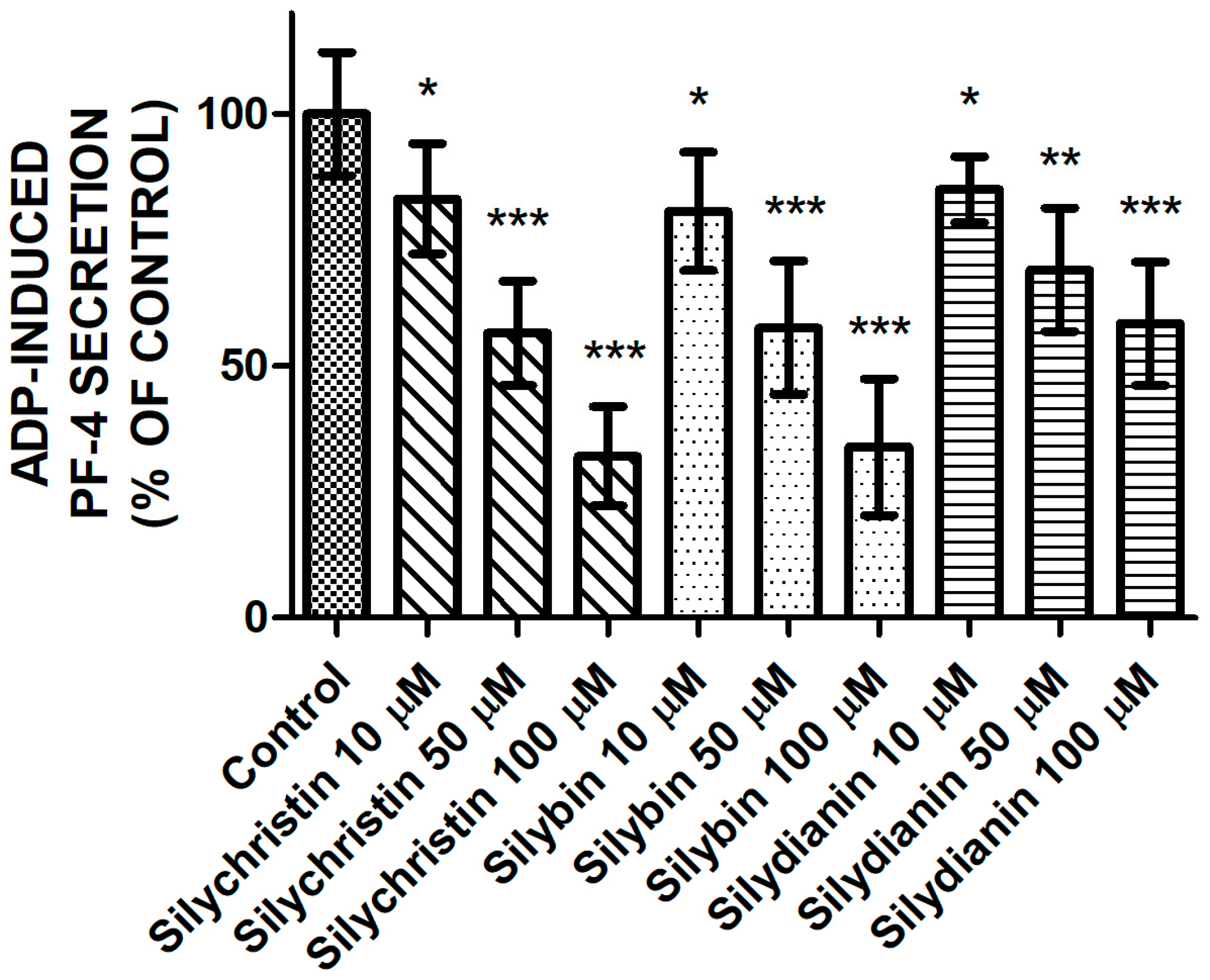
2.3. Effect of Flavonolignans on PF-4 Secretion from Blood Platelets Stimulated by ADP
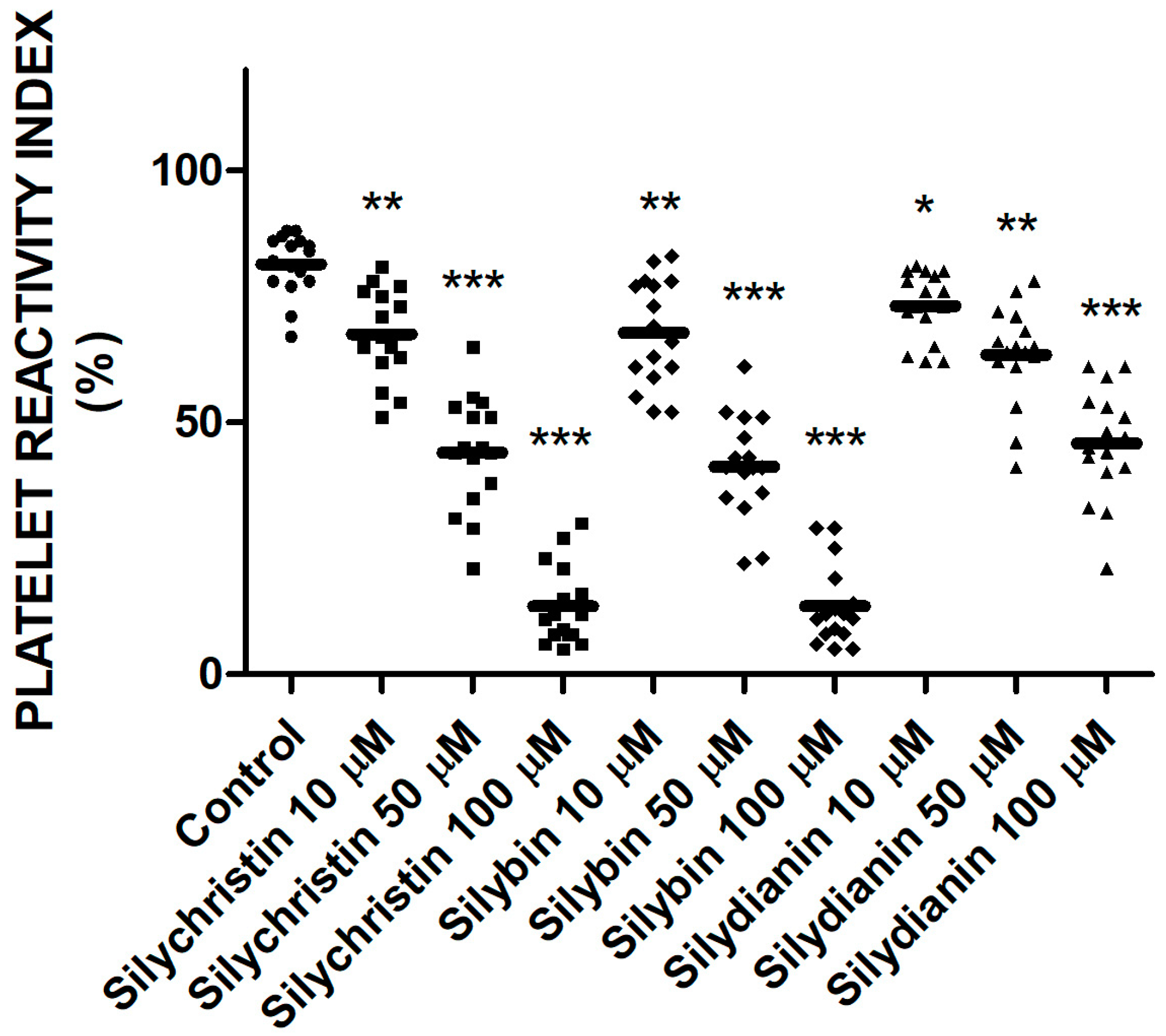
2.4. Effect of Flavonolignans on Blood Platelets VASP Phosphorylation
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Blood Samples
4.3. Isolation of Platelet-Rich Plasma and Blood Platelets
4.4. Sample Preparation Protocol
4.5. Blood Platelet Aggregation Induced by ADP
4.6. ADP Induced Blood Platelet Adhesion to Fibrinogen
4.7. Platelet Factor 4 Secretion
4.8. Estimation of Platelet Vasodilator-Stimulated Phosphoprotein (VASP) Phosphorylation
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Daniel, J.L.; Dangelmaier, C.; Jin, J.; Ashby, B.; Smith, J.B.; Kunapuli, S.P. Molecular basis for adp-induced platelet activation. I. Evidence for three distinct adp receptors on human platelets. J. Biol. Chem. 1998, 273, 2024–2029. [Google Scholar] [CrossRef] [PubMed]
- Savi, P.; Beauverger, P.; Labouret, C.; Delfaud, M.; Salel, V.; Kaghad, M.; Herbert, J.M. Role of p2y1 purinoceptor in adp-induced platelet activation. FEBS Lett. 1998, 422, 291–295. [Google Scholar] [CrossRef]
- Dorsam, R.T.; Kunapuli, S.P. Central role of the p2y12 receptor in platelet activation. J. Clin. Invest. 2004, 113, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Kahner, B.N.; Shankar, H.; Murugappan, S.; Prasad, G.L.; Kunapuli, S.P. Nucleotide receptor signaling in platelets. J. Thromb. Haemost. 2006, 4, 2317–2326. [Google Scholar] [CrossRef] [PubMed]
- Wiviott, S.D.; Braunwald, E.; McCabe, C.H.; Montalescot, G.; Ruzyllo, W.; Gottlieb, S.; Neumann, F.J.; Ardissino, D.; De Servi, S.; Murphy, S.A.; et al. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N. Engl. J. Med. 2007, 357, 2001–2015. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Szelenberger, R.; Saluk, J.; Nowak, P. Flavonolignans inhibit adp induced blood platelets activation and aggregation in whole blood. Int. J. Biol. Macromol. 2017, 95, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Gachet, C.; Hechler, B. The p2y receptors and thrombosis. WIREs Membr. Transp. Signal 2013, 2, 241–253. [Google Scholar] [CrossRef]
- Kim, S.; Kunapuli, S.P. P2y12 receptor in platelet activation. Platelets 2011, 22, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, S.; Ding, Z. Role of p2y12 receptor in thrombosis. Adv. Exp. Med. Biol. 2017, 906, 307–324. [Google Scholar] [PubMed]
- Bijak, M. Silybin, a major bioactive component of milk thistle (silybum marianum l. Gaernt.)-chemistry, bioavailability, and metabolism. Molecules 2017, 22, 1942. [Google Scholar] [CrossRef] [PubMed]
- Aleil, B.; Jacquemin, L.; De Poli, F.; Zaehringer, M.; Collet, J.P.; Montalescot, G.; Cazenave, J.P.; Dickele, M.C.; Monassier, J.P.; Gachet, C. Clopidogrel 150 mg/day to overcome low responsiveness in patients undergoing elective percutaneous coronary intervention: Results from the vasp-02 (vasodilator-stimulated phosphoprotein-02) randomized study. JACC Cardiovasc. Interv. 2008, 1, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Capasso, R.; Milic, N.; Capasso, F. Milk thistle in liver diseases: Past, present, future. Phytother. Res. 2010, 24, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M. [flavonolignans - compounds not only for liver treatment]. Pol. Merkur. Lekarski 2017, 42, 34–37. [Google Scholar] [PubMed]
- Bijak, M.; Ponczek, M.B.; Nowak, P. Polyphenol compounds belonging to flavonoids inhibit activity of coagulation factor x. Int. J. Biol. Macromol. 2014, 65, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Ziewiecki, R.; Saluk, J.; Ponczek, M.; Pawlaczyk, I.; Krotkiewski, H.; Wachowicz, B.; Nowak, P. Thrombin inhibitory activity of some polyphenolic compounds. Med. Chem. Res. 2014, 23, 2324–2337. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Saluk-Bijak, J. Flavonolignans inhibit the arachidonic acid pathway in blood platelets. BMC Complement. Altern. Med. 2017, 17, 396. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Dziedzic, A.; Saluk-Bijak, J. Flavonolignans reduce the response of blood platelet to collagen. Int. J. Biol. Macromol. 2018, 106, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Dziedzic, A.; Synowiec, E.; Sliwinski, T.; Saluk-Bijak, J. Flavonolignans inhibit il1-β-induced cross-talk between blood platelets and leukocytes. Nutrients 2017, 9, 1022. [Google Scholar] [CrossRef] [PubMed]
- Offermanns, S. Activation of platelet function through g protein-coupled receptors. Circ. Res. 2006, 99, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Wentworth, J.K.; Pula, G.; Poole, A.W. Vasodilator-stimulated phosphoprotein (vasp) is phosphorylated on ser157 by protein kinase c-dependent and -independent mechanisms in thrombin-stimulated human platelets. Biochem. J. 2006, 393, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Gurbel, P.A.; Bliden, K.P.; Hiatt, B.L.; O’Connor, C.M. Clopidogrel for coronary stenting: Response variability, drug resistance, and the effect of pretreatment platelet reactivity. Circulation 2003, 107, 2908–2913. [Google Scholar] [CrossRef] [PubMed]
- Müller, I.; Besta, F.; Schulz, C.; Massberg, S.; Schönig, A.; Gawaz, M. Prevalence of clopidogrel non-responders among patients with stable angina pectoris scheduled for elective coronary stent placement. Thromb. Haemost. 2003, 89, 783–787. [Google Scholar] [PubMed]
- Bagoly, Z.; Sarkady, F.; Magyar, T.; Kappelmayer, J.; Pongrácz, E.; Csiba, L.; Muszbek, L. Comparison of a new p2y12 receptor specific platelet aggregation test with other laboratory methods in stroke patients on clopidogrel monotherapy. PLoS ONE 2013, 8, e69417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pampuch, A.; Cerletti, C.; de Gaetano, G. Comparison of vasp-phosphorylation assay to light-transmission aggregometry in assessing inhibition of the platelet adp p2y12 receptor. Thromb. Haemost. 2006, 96, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Gorog, D.A.; Fuster, V. Platelet function tests in clinical cardiology: Unfulfilled expectations. J. Am. Coll. Cardiol. 2013, 61, 2115–2129. [Google Scholar] [CrossRef] [PubMed]
- Hwang, d.H.; Kim, Y.I.; Cho, K.H.; Poudel, B.K.; Choi, J.Y.; Kim, D.W.; Shin, Y.J.; Bae, O.N.; Yousaf, A.M.; Yong, C.S.; et al. A novel solid dispersion system for natural product-loaded medicine: Silymarin-loaded solid dispersion with enhanced oral bioavailability and hepatoprotective activity. J. Microencapsul. 2014, 31, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Synowiec, E.; Sitarek, P.; Sliwiński, T.; Saluk-Bijak, J. Evaluation of the cytotoxicity and genotoxicity of flavonolignans in different cellular models. Nutrients 2017, 9, 1356. [Google Scholar] [CrossRef] [PubMed]
- Walkowiak, B.; Michalak, E.; Koziolkiewicz, W.; Cierniewski, C.S. Rapid photometric method for estimation of platelet count in blood plasma or platelet suspension. Thromb. Res. 1989, 56, 763–766. [Google Scholar] [CrossRef]
- Doolittle, R.F.; Schubert, D.; Schwartz, S.A. Amino acid sequence studies on artiodactyl fibrinopeptides. I. Dromedary camel, mule deer, and cape buffalo. Arch. Biochem. Biophys. 1967, 118, 456–467. [Google Scholar] [CrossRef]
- Tuszynski, G.P.; Murphy, A. Spectrophotometric quantitation of anchorage-dependent cell numbers using the bicinchoninic acid protein assay reagent. Anal. Biochem. 1990, 184, 189–191. [Google Scholar] [CrossRef]
- Bijak, M.; Kolodziejczyk-Czepas, J.; Ponczek, M.B.; Saluk, J.; Nowak, P. Protective effects of grape seed extract against oxidative and nitrative damage of plasma proteins. Int. J. Biol. Macromol. 2012, 51, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Nowak, P.; Borowiecka, M.; Ponczek, M.B.; Zbikowska, H.M.; Wachowicz, B. Protective effects of (-)-epicatechin against nitrative modifications of fibrinogen. Thromb. Res. 2012, 130, e123–e128. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Saluk, J.; Antosik, A.; Ponczek, M.B.; Zbikowska, H.M.; Borowiecka, M.; Nowak, P. Aronia melanocarpa as a protector against nitration of fibrinogen. Int. J. Biol. Macromol. 2013, 55, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Saluk, J.; Tsirigotis-Maniecka, M.; Komorowska, H.; Wachowicz, B.; Zaczynska, E.; Czarny, A.; Czechowski, F.; Nowak, P.; Pawlaczyk, I. The influence of conjugates isolated from matricaria chamomilla l. On platelets activity and cytotoxicity. Int. J. Biol. Macromol. 2013, 61, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Zbikowska, H.M.; Antosik, A.; Szejk, M.; Bijak, M.; Olejnik, A.K.; Saluk, J.; Nowak, P. Does quercetin protect human red blood cell membranes against γ-irradiation? Redox Rep. 2014, 19, 65–71. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bijak, M.; Szelenberger, R.; Dziedzic, A.; Saluk-Bijak, J. Inhibitory Effect of Flavonolignans on the P2Y12 Pathway in Blood Platelets. Molecules 2018, 23, 374. https://doi.org/10.3390/molecules23020374
Bijak M, Szelenberger R, Dziedzic A, Saluk-Bijak J. Inhibitory Effect of Flavonolignans on the P2Y12 Pathway in Blood Platelets. Molecules. 2018; 23(2):374. https://doi.org/10.3390/molecules23020374
Chicago/Turabian StyleBijak, Michal, Rafal Szelenberger, Angela Dziedzic, and Joanna Saluk-Bijak. 2018. "Inhibitory Effect of Flavonolignans on the P2Y12 Pathway in Blood Platelets" Molecules 23, no. 2: 374. https://doi.org/10.3390/molecules23020374




