Antinociceptive and Antibacterial Properties of Anthocyanins and Flavonols from Fruits of Black and Non-Black Mulberries
Abstract
:1. Introduction
2. Results
2.1. Determination of Anthocyanin and Flavonol Contents
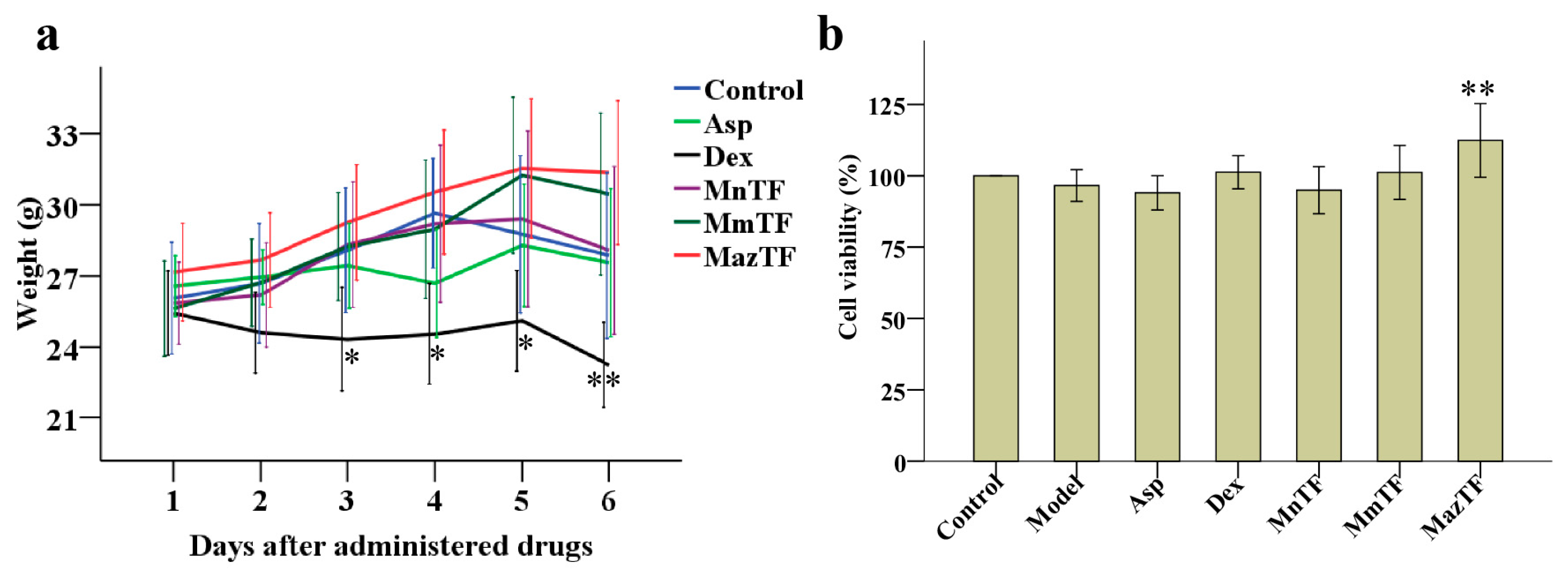
2.2. Toxicity Assessment of TFs
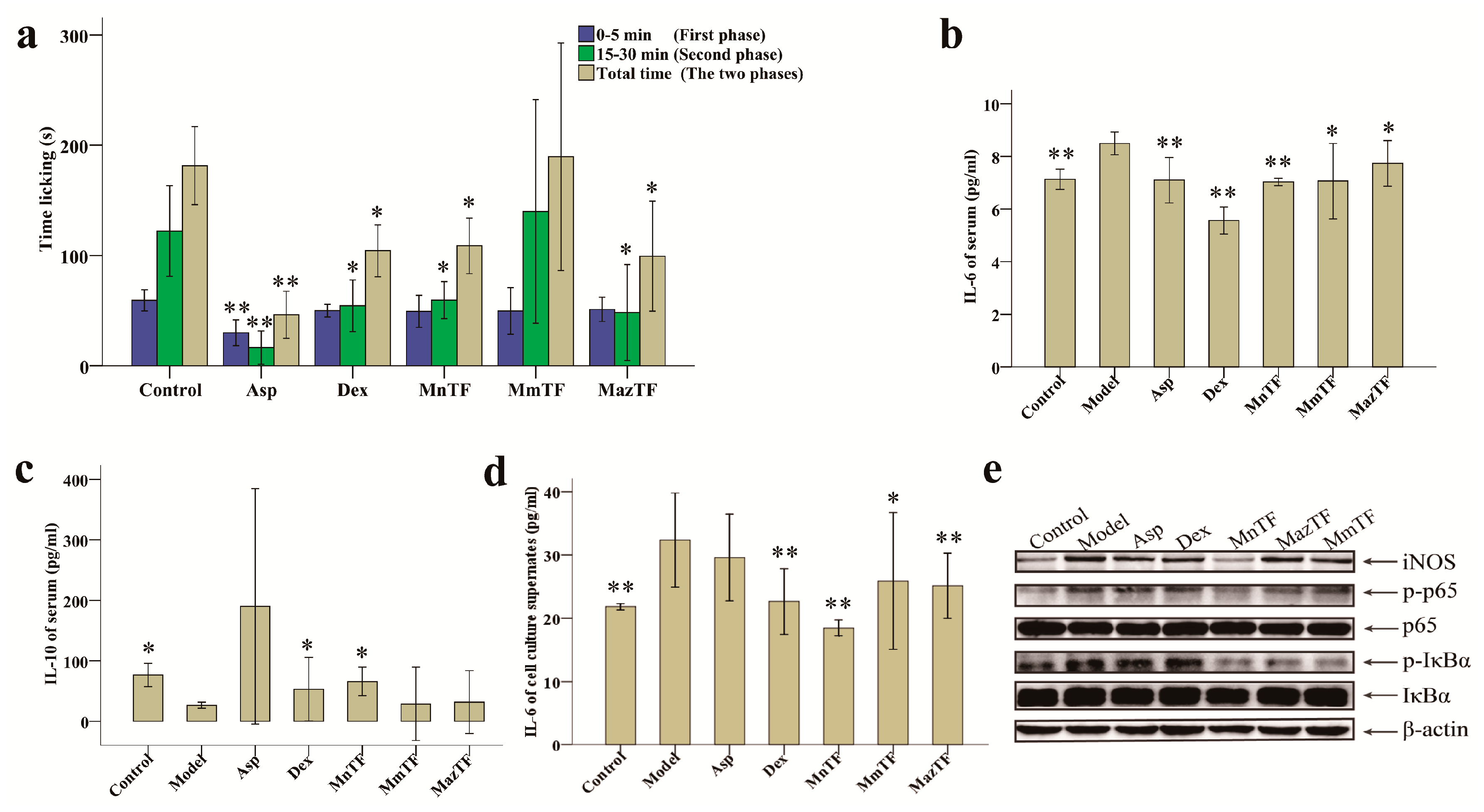
2.3. Antinociceptive Activities of TFs
2.4. Effects of TFs on Cytokines and Pain-Related Proteins
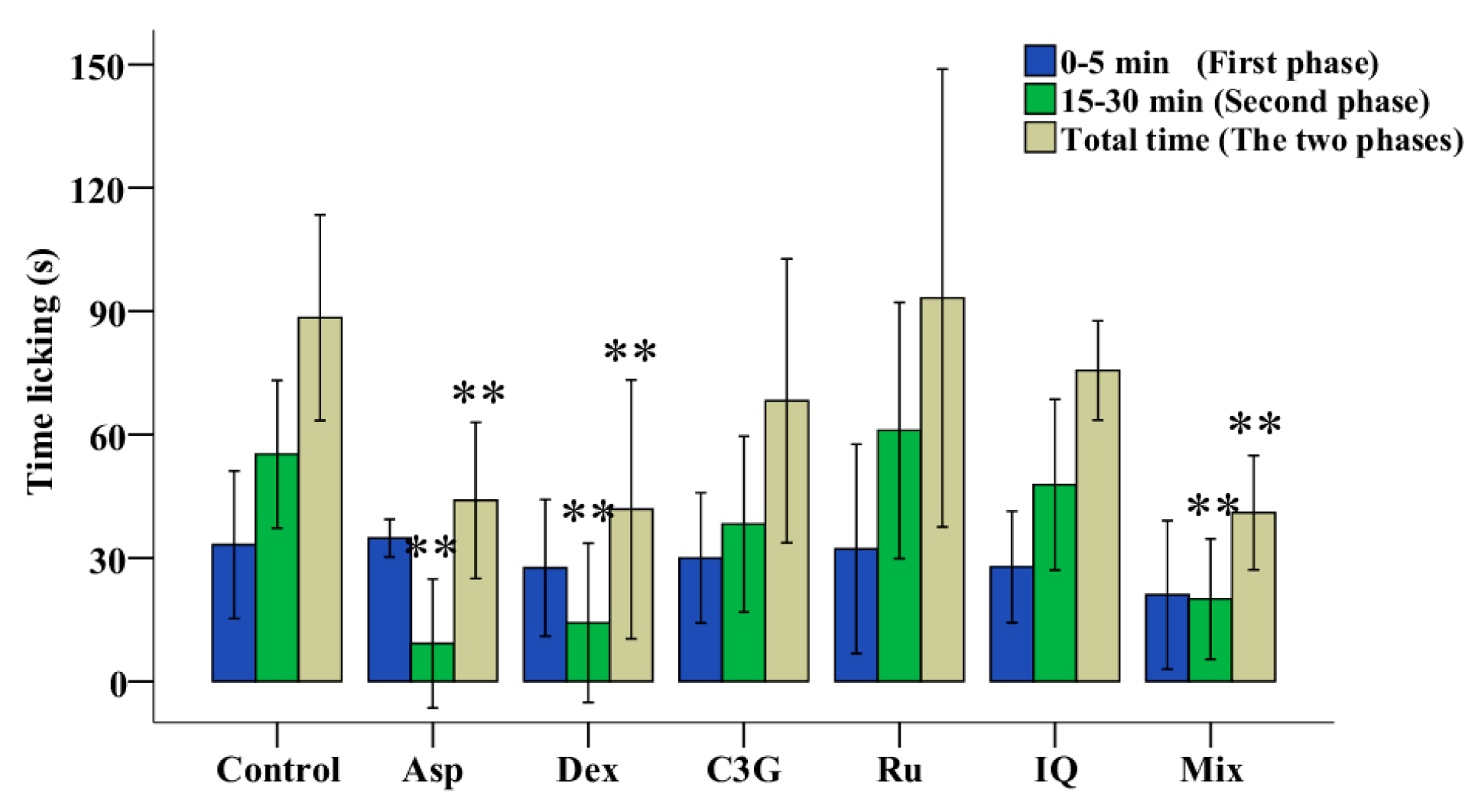
2.5. Antinociceptive Activities of C3G, Ru and IQ
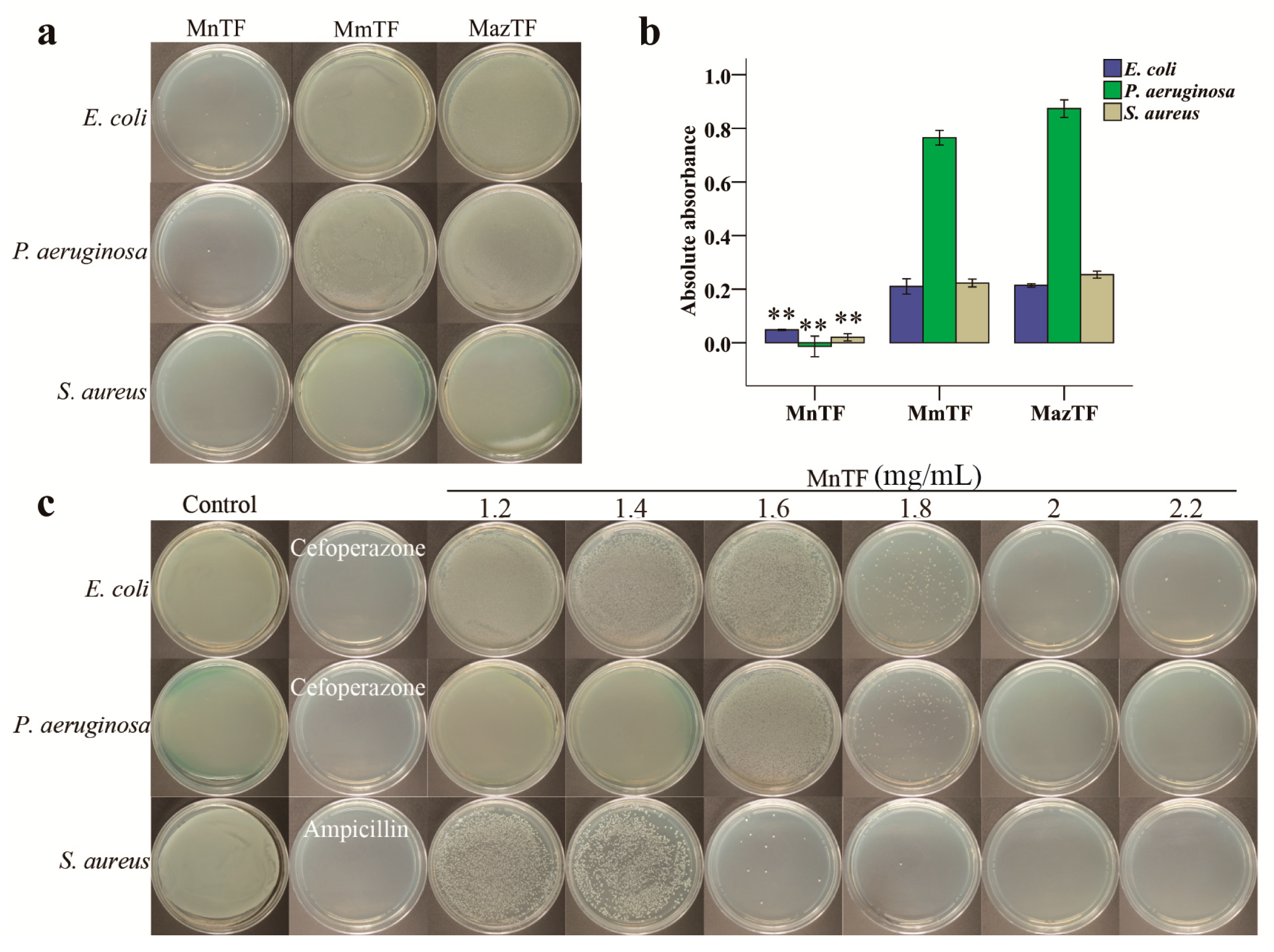
2.6. Antibacterial Activities of TFs
3. Discussion
4. Materials and Methods
4.1. Mulberry Fruits and Animals
4.2. Chemicals and Reagents
4.3. Extraction of TFs
4.4. UPLC–TUV/QDa Conditions and Determination of TFs
4.5. Evaluation of Toxicity and Antinociceptive Activity in Mice
4.6. Immunological Procedures
4.6.1. Blood Collection for Cytokines Analysis
4.6.2. Cell Culture, Cytotoxicity and Western Blot Analysis
4.7. Antibacterial Assays
4.7.1. Comparison of Antibacterial Activities
4.7.2. MBC of MnTF
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACN | acetonitrile |
| Asp | aspirin |
| C3G | cyanidin-3-O-glucoside |
| C3R | cyanidin-3-O-rutinoside |
| Dex | dexamethasone |
| ELISA | enzyme-linked immunosorbent assay |
| HPLC | high-performance liquid chromatography |
| iNOS | inducible nitric oxide synthase |
| IQ | isoquercetin |
| IFN-γ | interfron γ |
| IL-6 | interleukin 6 |
| IL-10 | interleukin 10 |
| Ka | kaempferol |
| LPS | lipopolysaccharide |
| MazTF | total flavonoid of Morus alba ‘Zhenzhubai’ |
| MBC | minimum bactericidal concentration |
| MnTF | total flavonoid of Morus nigra |
| MmTF | total flavonoid of Morus mongolica |
| MeOH | methanol |
| Mh | morin hydrate |
| NF-κB | nuclear factor-κB |
| NO | nitric oxide |
| p-IκBα | phospho-IκBα |
| p-p65 | phospho-p65 |
| Pg3G | pelargonidin-3-O-glucoside |
| Qu | quercetin |
| Ru | quercetin-3-O-rutinlside |
| SD | standard deviation |
| TF | total flavonoids |
| TFs | total flavonoids of M. nigra, M. mongolica, and M. alba ‘Zhenzhubai’ |
| TNF-α | tumor necrosis factor α |
| UPLC | ultra-performance liquid chromatographic |
| UPLC-TUV/QDa | ultra-performance liquid chromatography–tunable ultraviolet/mass single-quadrupole detector |
References
- Gundogdu, M.; Muradoglu, F.; Sensoy, R.I.G.; Yilmaz, H. Determination of fruit chemical properties of Morus nigra L.; Morus alba L. and Morus rubra L. by HPLC. Sci. Hortic. 2011, 132, 37–41. [Google Scholar] [CrossRef]
- He, N.; Zhang, C.; Qi, X.; Zhao, S.; Tao, Y.; Yang, G.; Lee, T.H.; Wang, X.; Cai, Q.; Li, D.; et al. Draft genome sequence of the mulberry tree Morus notabilis. Nat. Commun. 2013, 4, 2445. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Pu, J.; Liu, D.; Yu, W.; Shao, Y.; Yang, G.; Xiang, Z.; He, N. Anti-inflammatory and antinociceptive properties of flavonoids from the fruits of black mulberry (Morus nigra L.). PLoS ONE 2016, 11, e0153080. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.J.; Jin, H.G.; Woo, E.R.; Lee, S.K.; Kim, H.P. The root barks of Morus alba and the flavonoid constituents inhibit airway inflammation. J. Ethnopharmacol. 2013, 149, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Ozgen, M.; Serce, S.; Kaya, C. Phytochemical and antioxidant properties of anthocyanin-rich Morus nigra and Morus rubra fruits. Sci. Hortic. 2009, 119, 275–279. [Google Scholar] [CrossRef]
- Queiroz, G.T.; Santos, T.R.; Macedo, R.; Peters, V.M.; Leite, M.N.; de Cassia da Silveira e Sa, R.; de Oliveira Guerra, M. Efficacy of Morus nigra L. on reproduction in female Wistar rats. Food Chem. Toxicol. 2012, 50, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Chen, H.; Zhang, C.; Han, M.; Li, T.; Qi, X.; Xiang, Z.; He, N. Definition of eight mulberry species in the genus Morus by internal transcribed spacer-based phylogeny. PLoS ONE 2015, 10, e0135411. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, W.; Zhao, T.; Li, F.; Zhang, M.; Li, J.; Zou, Y.; Wang, W.; Cobbina, S.J.; Wu, X.; et al. Adsorption properties of macroporous adsorbent resins for separation of anthocyanins from mulberry. Food Chem. 2016, 194, 712–722. [Google Scholar] [CrossRef] [PubMed]
- Ercisli, S.; Orhan, E. Chemical composition of white (Morus alba), red (Morus rubra) and black (Morus nigra) mulberry fruits. Food Chem. 2007, 103, 1380–1384. [Google Scholar] [CrossRef]
- Kim, S.B.; Chang, B.Y.; Hwang, B.Y.; Kim, S.Y.; Lee, M.K. Pyrrole alkaloids from the fruits of Morus alba. Bioorg. Med. Chem. Lett. 2014, 24, 5656–5659. [Google Scholar] [CrossRef] [PubMed]
- Levyadun, S.; Gould, K.S. Role of anthocyanins in plant defence. Anthocyanins 2008, 22–28. [Google Scholar] [CrossRef]
- Ravindra, P.V.; Narayan, M.S. Antioxidant activity of the anthocyanin from carrot (Daucus carota) callus culture. Int. J. Food Sci. Nutr. 2003, 54, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk, J.; Saluk-Juszczak, J.; Posmyk, M.; Janas, K.; Wachowicz, B. Red cabbage anthocyanins may protect blood plasma proteins and lipids. Open Life Sci. 2011, 6, 565–574. [Google Scholar] [CrossRef]
- Chen, T.; Hu, S.; Zhang, H.; Guan, Q.; Yang, Y.; Wang, X. Anti-inflammatory effects of Dioscorea alata L. anthocyanins in a TNBS-induced colitis model. Food Funct. 2017, 8, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Cremonini, E.; Mastaloudis, A.; Hester, S.N.; Verstraeten, S.V.; Anderson, M.; Wood, S.M.; Waterhouse, A.L.; Fraga, C.G.; Oteiza, P.I. Anthocyanins inhibit tumor necrosis alpha-induced loss of Caco-2 cell barrier integrity. Food Funct. 2017, 8, 2915–2923. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.W.; Synytsya, A.; Capek, P.; Bleha, R.; Pohl, R.; Park, Y.I. Structural analysis and anti-obesity effect of a pectic polysaccharide isolated from Korean mulberry fruit Oddi (Morus alba L.). Carbohydr. Polym. 2016, 146, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Yimam, M.; Lee, Y.C.; Moore, B.; Jiao, P.; Hong, M.; Nam, J.B.; Kim, M.R.; Hyun, E.J.; Chu, M.; Brownell, L.; et al. Analgesic and anti-inflammatory effects of UP1304, a botanical composite containing standardized extracts of Curcuma longa and Morus alba. J. Integr. Med. 2016, 14, 60–68. [Google Scholar] [CrossRef]
- Chen, C.; You, L.J.; Abbasi, A.M.; Fu, X.; Liu, R.H. Optimization for ultrasound extraction of polysaccharides from mulberry fruits with antioxidant and hyperglycemic activity in vitro. Carbohydr. Polym. 2015, 130, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J.; Lin, J.Y. Anti-inflammatory effects of phenolic extracts from strawberry and mulberry fruits on cytokine secretion profiles using mouse primary splenocytes and peritoneal macrophages. Int. Immunopharmacol. 2013, 16, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Synytsya, A.; Kim, H.B.; Choi, D.J.; Lee, S.; Lee, J.; Kim, W.J.; Jang, S.; Park, Y.I. Purification, characterization and immunomodulating activity of a pectic polysaccharide isolated from Korean mulberry fruit Oddi (Morus alba L.). Int. Immunopharmacol. 2013, 17, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Ley, J.P.; Schwerzler, J.; Lieder, B.; Beltran, L.; Ziemba, P.M.; Hatt, H.; Hans, J.; Widder, S.; Krammer, G.E.; et al. Nonivamide, a capsaicin analogue, exhibits anti-inflammatory properties in peripheral blood mononuclear cells and U-937 macrophages. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- DeVon, H.A.; Piano, M.R.; Rosenfeld, A.G.; Hoppensteadt, D.A. The association of pain with protein inflammatory biomarkers: A review of the literature. Nurs. Res. 2014, 63, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Lopes-de-Araujo, J.; Neves, A.R.; Gouveia, V.M.; Moura, C.C.; Nunes, C.; Reis, S. Oxaprozin-loaded lipid nanoparticles towards overcoming NSAIDs side-effects. Pharm. Res. 2016, 33, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Roth, S.H. Coming to terms with nonsteroidal anti-inflammatory drug gastropathy. Drugs 2012, 72, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.N.; Badyal, D.K. Biological studies of turmeric oil, part 3: Anti-inflammatory and analgesic properties of turmeric oil and fish oil in comparison with aspirin. Nat. Prod. Commun. 2014, 9, 225–228. [Google Scholar] [PubMed]
- McQuay, H.J.; Moore, R.A. Dose-response in direct comparisons of different doses of aspirin, ibuprofen and paracetamol (acetaminophen) in analgesic studies. Br. J. Clin. Pharmacol. 2007, 63, 271–278. [Google Scholar] [CrossRef] [PubMed]
- White, A.C., Jr.; Kang, G. Antibiotics, microbiota and health: Are there dangers hiding in plain sight? Curr. Opin. Infect. Dis. 2015, 28, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Dellit, T.H.; Owens, R.C.; McGowan, J.E., Jr.; Gerding, D.N.; Weinstein, R.A.; Burke, J.P.; Huskins, W.C.; Paterson, D.L.; Fishman, N.O.; Carpenter, C.F.; et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin. Infect. Dis. 2007, 44, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Shuai, Q.; Chen, H.; Fan, L.; Zeng, Q.; He, N. Cloning and expression analyses of the anthocyanin biosynthetic genes in mulberry plants. Mol. Genet. Genom. 2014, 289, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Pu, J.S.; Xiang, Z.H.; He, N.J. Extraction and antioxidant activity of total flavonoids from black mulberry fruit. Food Sci. 2014, 35, 7–12. [Google Scholar]
- Mena, P.; Sánchez-Salcedo, E.M.; Tassotti, M.; Martínez, J.J.; Hernández, F.; Rio, D.D. Phytochemical evaluation of eight white (Morus alba L.) and black (Morus nigra L.) mulberry clones grown in Spain based on UHPLC-ESI-MSn metabolomic profiles. Food Res. Int. 2016, 89, 1116–1122. [Google Scholar] [CrossRef]
- Rainsford, K.D. Profile and mechanisms of gastrointestinal and other side effects of nonsteroidal anti-inflammatory drugs (NSAIDs). Am. J. Med. 1999, 107, 27S–35S. [Google Scholar] [CrossRef]
- Hunskaar, S.; Hole, K. The formalin test in mice: Dissociation between inflammatory and non-inflammatory pain. Pain 1987, 30, 103–114. [Google Scholar] [CrossRef]
- Alvarez Perez Gil, A.L.; Barbosa Navarro, L.; Patipo Vera, M.; Petricevich, V.L. Anti-inflammatory and antinociceptive activities of the ethanolic extract of Bougainvillea xbuttiana. J. Ethnopharmacol. 2012, 144, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, P.; Tang, C.; Wang, Y.; Li, Y.; Zhang, H. Antinociceptive and anti-inflammatory activities of extract and two isolated flavonoids of Carthamus tinctorius L. J. Ethnopharmacol. 2014, 151, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, M.I.; Pereira, A.F.; Nogueira, R.B.; Rolim, F.E.; Brito, G.A.; Wong, D.V.; Lima-Junior, R.C.; de Albuquerque Ribeiro, R.; Vale, M.L. The antioxidant effects of the flavonoids rutin and quercetin inhibit oxaliplatin-induced chronic painful peripheral neuropathy. Mol. Pain 2013, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Lapa Fda, R.; Gadotti, V.M.; Missau, F.C.; Pizzolatti, M.G.; Marques, M.C.; Dafre, A.L.; Farina, M.; Rodrigues, A.L.; Santos, A.R. Antinociceptive properties of the hydroalcoholic extract and the flavonoid rutin obtained from Polygala paniculata L. in mice. Basic Clin. Pharmacol. 2009, 104, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xu, M.L.; Liu, J.; Wang, Y.; Hu, J.H.; Wang, M.H. Sonchus asper extract inhibits LPS-induced oxidative stress and pro-inflammatory cytokine production in RAW264.7 macrophages. Nutr. Res. Pract. 2015, 9, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.H.; Baek, S.J. Molecular targets of dietary polyphenols with anti-inflammatory properties. Yonsei Med. J. 2005, 46, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Meng, H.; McClendon, C.L.; Dai, Z.; Li, K.; Zhang, X.; He, S.; Shang, E.; Liu, Y.; Lai, L. Discovery of novel 15-lipoxygenase activators to shift the human arachidonic acid metabolic network toward inflammation resolution. J. Med. Chem. 2016, 59, 4202–4209. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.J.; Lin, J.T.; Chen, Y.C.; Liu, S.C.; Lu, F.J.; Chang, T.J.; Wang, M.; Lin, H.W.; Chang, Y.Y. Suppressive effect of carotenoid extract of Dunaliella salina alga on production of LPS-stimulated pro-inflammatory mediators in RAW264.7 cells via NF-κB and JNK inactivation. J. Funct. Foods 2013, 5, 607–615. [Google Scholar] [CrossRef]
- Chan, K.C.; Huang, H.P.; Ho, H.H.; Huang, C.N.; Lin, M.C.; Wang, C.J. Mulberry polyphenols induce cell cycle arrest of vascular smooth muscle cells by inducing NO production and activating AMPK and p53. J. Funct. Foods 2015, 15, 604–613. [Google Scholar] [CrossRef]
- Matsui, H.; Ihara, Y.; Fujio, Y.; Kunisada, K.; Akira, S.; Kishimoto, T.; Yamauchitakihara, K. Induction of interleukin (IL)-6 by hypoxia is mediated by nuclear factor (NF)-κB and NF-IL6 in cardiac myocytes. Cardiovasc. Res. 1999, 270, 11463–11471. [Google Scholar] [CrossRef]
- Vallabhapurapu, S.; Karin, M. Regulation and function of NF-κB transcription factors in the immune system. Annu. Rev. Immunol. 2009, 27, 693–733. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Cao, Y.; Greten, F.R.; Li, Z.W. NF-κB in cancer: From innocent bystander to major culprit. Nat. Rev. Cancer 2002, 2, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Menghini, L.; Ferrante, C.; Leporini, L.; Recinella, L.; Chiavaroli, A.; Leone, S.; Pintore, G.; Vacca, M.; Orlando, G.; Brunetti, L. An hydroalcoholic chamomile extract modulates inflammatory and immune response in HT29 cells and isolated rat colonic inflammation. Phytother. Res. 2016, 30, 1513–1518. [Google Scholar] [CrossRef] [PubMed]
- Bian, A.L.; Hu, H.Y.; Rong, Y.D.; Wang, J.; Wang, J.X.; Zhou, X.Z. A study on relationship between elderly sarcopenia and inflammatory factors IL-6 and TNF-α. Eur. J. Med. Res. 2017, 22, 25. [Google Scholar] [CrossRef] [PubMed]
- Menghini, L.; Leporini, L.; Pintore, G.; Ferrante, C.; Recinella, L.; Orlando, G.; Vacca, M.; Brunetti, L. A natural formulation (imoviral) increases macrophage resistance to LPS-induced oxidative and inflammatory stress in vitro. J. Biol. Regul. Homeost. Agents 2014, 28, 775–782. [Google Scholar] [PubMed]
- Kyrkanides, S.; Olschowka, J.A.; Williams, J.P.; Hansen, J.T.; O’Banion, M.K. TNFα and IL-1β mediate intercellular adhesion molecule-1 induction via microglia–astrocyte interaction in CNS radiation injury. J. Neuroimmunol. 1999, 95, 95–106. [Google Scholar] [CrossRef]
- Starkie, R.; Ostrowski, S.R.; Jauffred, S.; Febbraio, M.; Pedersen, B.K. Exercise and IL-6 infusion inhibit endotoxin-induced TNF-α production in humans. FASEB J. 2003, 17, 884–886. [Google Scholar] [CrossRef] [PubMed]
- Khanapure, S.P.; Garvey, D.S.; Janero, D.R.; Letts, L.G. Eicosanoids in inflammation: Biosynthesis, pharmacology, and therapeutic frontiers. Curr. Top. Med. Chem. 2007, 7, 311–340. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Jianrong, L.I.; Jiang, Y. Polysaccharides from mulberry leaf in relation to their antioxidant activity and antibacterial ability. J. Food Process Eng. 2010, 33, 39–50. [Google Scholar] [CrossRef]
- Thabti, I.; Elfalleh, W.; Tlili, N.; Ziadi, M.; Campos, M.G.; Ferchichi, A. Phenols, flavonoids, and antioxidant and antibacterial activity of leaves and stem bark of Morus species. Int. J. Food Prop. 2014, 17, 842–854. [Google Scholar] [CrossRef]
- Vazquezguillamet, M.C.; Vazquez, R.; Micek, S.T.; Kollef, M.H. Predicting resistance to Piperacillin-Tazobactam, Cefepime and Meropenem in Septic Patients with bloodstream infection due to Gram-negative bacteria. Clin. Infect. Dis. 2017, 65, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Doernberg, S.B.; Lodise, T.P.; Thaden, J.T.; Munita, J.M.; Cosgrove, S.E.; Arias, C.A.; Boucher, H.W.; Corey, G.R.; Lowy, F.D.; Murray, B.; et al. Gram-positive bacterial infections: Research priorities, accomplishments, and future directions of the antibacterial resistance leadership group. Clin. Infect. Dis. 2017, 64 (Suppl. 1), S24–S29. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.H.; Lee, A.R.; Yang, C.H. Antioxidative and anti-Inflammatory activities of polyhydroxy flavonoids of GEORGI. Biosci. Biotechnol. Biochem. 2014, 70, 2371–2380. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.H.; Kim, M.S.; Le, M.Q.; Song, Y.S.; Bak, Y.; Ryu, H.W.; Oh, S.R.; Yoon, D.Y. Fargesin exerts anti-inflammatory effects in THP-1 monocytes by suppressing PKC-dependent AP-1 and NF-ĸB signaling. Phytomed. Int. J. Phytother. Phytopharmacol. 2017, 24, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Omara, S.T. MIC and MBC of Honey and Gold Nanoparticles against methicillin-resistant (MRSA) and vancomycin-resistant (VRSA) coagulase-positive S. aureus isolated from contagious Bovine clinical Mastitis. J. Genetic Eng. Biotechnol. 2017, 15, 219–230. [Google Scholar] [CrossRef]
Sample Availability: Samples of MnTF, MmTF, and MazTF are available from the authors. |
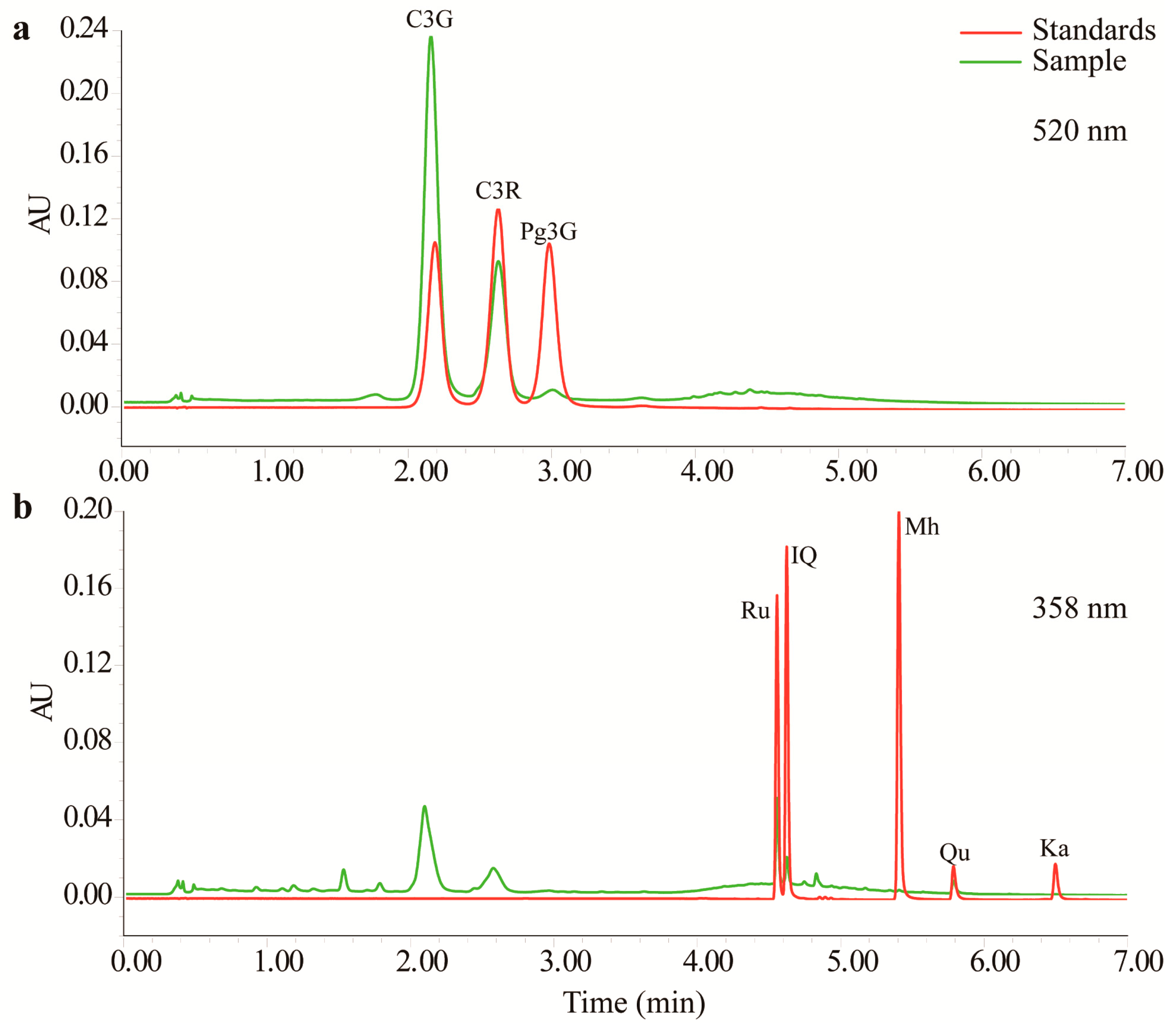
| Compounds | RT a (min) | Regression Equation b | r2 | Content (mg/g) c | Selected Ions by QDa (m/z) | ||
|---|---|---|---|---|---|---|---|
| MnTF | MmTF | MazTF | |||||
| C3G | 2.186 | y = (12.499x + 1.239) × 103 | 0.9999 | 8.2168 ± 0.0238 | 0.2220 ± 0.0024 | ND | 449.18 |
| C3R | 2.627 | y = (8.765x + 1.550) × 103 | 0.9999 | 2.8578 ± 0.0146 | 0.0610 ± 0.0013 | ND | 595.33 |
| P3G | 2.983 | y = (5.230x + 0.770) × 103 | 0.9999 | 0.2539 ± 0.0047 | 0.0057 ± 0.0003 | ND | 433.24 |
| Ru | 4.556 | y = (6.065x + 2.362) × 103 | 0.9999 | 0.4498 ± 0.0075 | 0.2723 ± 0.0013 | 0.0816 ± 0.0015 | 302.93 |
| IQ | 4.624 | y = (2.560x + 0.080) × 103 | 0.9999 | 0.1639 ± 0.0006 | 0.2459 ± 0.0059 | 0.0631 ± 0.0033 | 303.06 |
| Mh | 5.405 | y = (6.880x + 0.226) × 103 | 0.9999 | 0.0002 ± 0.0001 | <0.0001 | <0.0001 | 303.04 |
| Qu | 5.786 | y = (4.870x − 0.074) × 103 | 0.9993 | 0.0716 ± 0.0045 | 0.0029 ± 0.0002 | 0.0036 ± 0.0004 | 303.11 |
| Ka | 6.497 | y = (2.710x + 0.487) × 103 | 0.9986 | <0.0001 | <0.0001 | ND | 287.03 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Yu, W.; Chen, G.; Meng, S.; Xiang, Z.; He, N. Antinociceptive and Antibacterial Properties of Anthocyanins and Flavonols from Fruits of Black and Non-Black Mulberries. Molecules 2018, 23, 4. https://doi.org/10.3390/molecules23010004
Chen H, Yu W, Chen G, Meng S, Xiang Z, He N. Antinociceptive and Antibacterial Properties of Anthocyanins and Flavonols from Fruits of Black and Non-Black Mulberries. Molecules. 2018; 23(1):4. https://doi.org/10.3390/molecules23010004
Chicago/Turabian StyleChen, Hu, Wansha Yu, Guo Chen, Shuai Meng, Zhonghuai Xiang, and Ningjia He. 2018. "Antinociceptive and Antibacterial Properties of Anthocyanins and Flavonols from Fruits of Black and Non-Black Mulberries" Molecules 23, no. 1: 4. https://doi.org/10.3390/molecules23010004




