Anti-Inflammatory and Anti-Oxidative Activities of Phenolic Compounds from Alnus sibirica Stems Fermented by Lactobacillus plantarum subsp. argentoratensis
Abstract
:1. Introduction
2. Results and Discussion
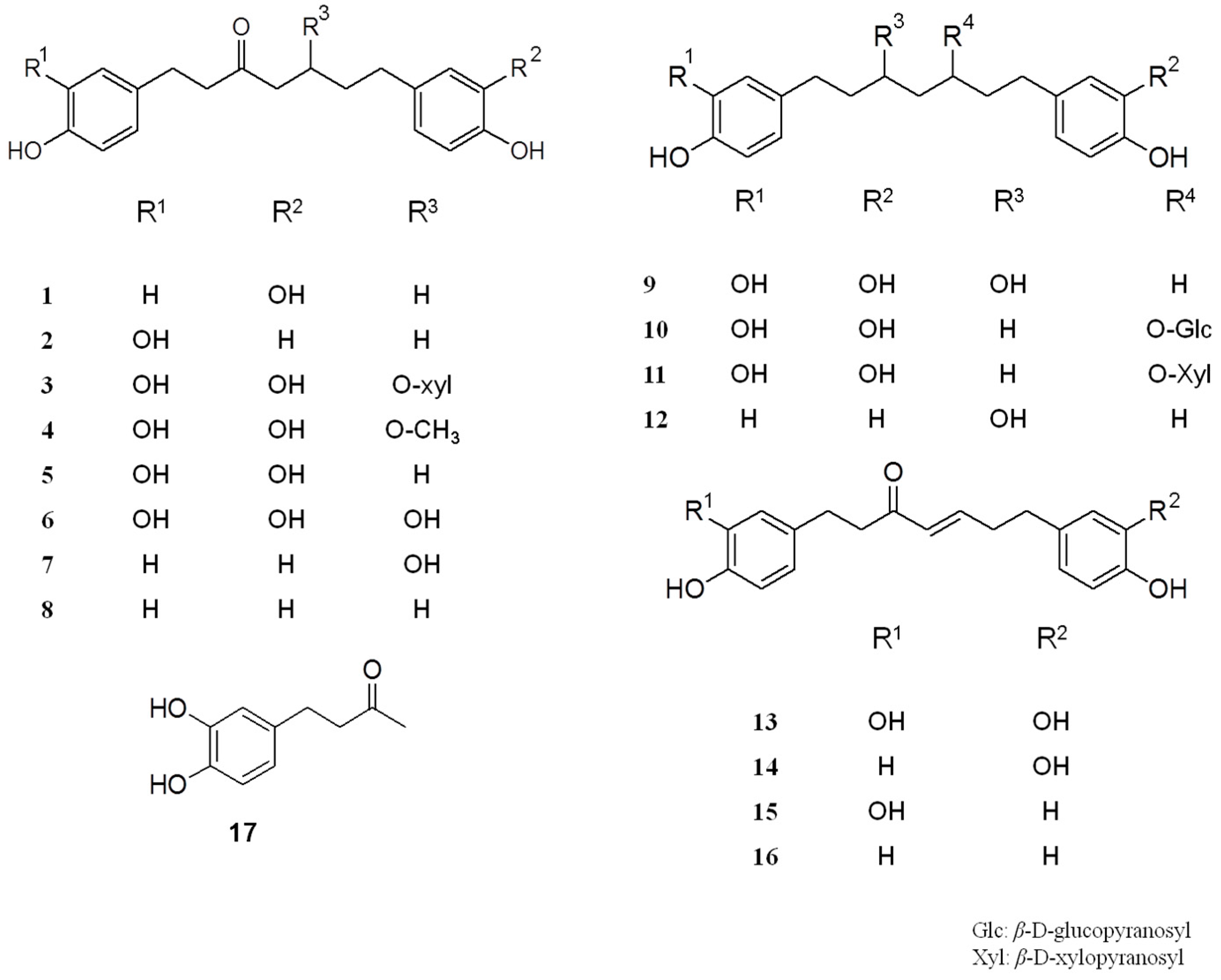
2.1. Isolation and Structural Identification
2.2. Evaluation of Anti-Oxidative and Anti-Inflammatory Activities
3. Experimental Section
3.1. General Experimental Procedure
3.2. Plant Material
3.3. Extraction and Fermentation
3.4. Isolation
3.5. Measurement of DPPH Radical Scavenging Activity
3.6. Cell Culture
3.7. Measurement of Inhibition of NO Production
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kun, C.; Hua, F.; Weiyi, T. Effects of pure breed fermentation on major effective component in Semen sojae praeparatum. J. Gansu Coll. Trad. Chin. Med. 2006, 23, 39–41. [Google Scholar]
- Gao, H.; Jia, T.Z. Comparative research on quality of various Shenqu fermented with different pure inoculation. China J. Chin. Mater. Med. 2008, 33, 2323–2325. [Google Scholar]
- Kamauchi, H.; Kon, T.; Kinoshita, K.; Takahashi, K.; Koyama, K. Three new terpenoids, sterebins O, P1, and P2, isolated from Stevia rebaudiana fermented by Saccharomyces cerevisiae. Tetrahedron Lett. 2014, 55, 7203–7205. [Google Scholar] [CrossRef]
- Kamauchi, H.; Kon, T.; Kinoshita, K. New diterpenoids isolated from Stevia rebaudiana fermented by Saccharomyces cerevisiae. Tetrahedron Lett. 2015, 56, 4377–4382. [Google Scholar] [CrossRef]
- Yu, J.; Xie, J.; Mao, X.J.; Wei, H.; Zhao, S.L.; Ma, Y.-G.; Li, N.; Zhao, R.-H. Comparison of laxative and antioxidant activities of raw, processed and fermented Polygoni Multiflori Radix. Chin. J. Nat. Med. 2012, 10, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J. Korean Folk Medicine; Seoul National University Publishing Center Press: Seoul, Korea, 1966. [Google Scholar]
- Nomura, M.; Tokoroyama, T.; Kubota, T. Diarylheptanoids and other constituents from wood of Alnus japonica. Phytochemistry 1981, 20, 1097–1104. [Google Scholar] [CrossRef]
- Uvarova, N.I.; Oshitok, G.I.; Suprunov, N.I.; Elyakov, G.B. Triterpenoids and other constituents from the Far-Eastern species of Alnus. Phytochemistry 1972, 11, 741–743. [Google Scholar] [CrossRef]
- Lee, M.W.; Tanaka, T.; Nonaka, G.I.; Nishioka, I. Dimeric ellagitannins from Alnus japonica. Phytochemistry 1992, 31, 2835–2839. [Google Scholar] [CrossRef]
- Kim, M.H.; Park, K.H.; Kim, S.R.; Park, K.J.; Lee, M.W. Two new phenolic compounds from the leaves of Alnus Sibirica Fisch. Ex Turcz. Nat. Prod. Res. 2016, 30, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.M.; Kwon, Y.M.; Lee, J.H.; Yon, K.H.; Lee, M.W. Melanogenesis inhibitory activities of diarylheptanoids from Alnus hirsuta Turcz in B16 mouse melanoma cell. Arch. Pharm. Res. 2002, 25, 885–888. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.W.; Kim, J.S.; Cho, S.M.; Lee, Y.A.; Kim, K.H.; Kim, S.; Lee, M. Diarylheptanoids from stem bark of Alnus hirsuta var. Sibirica. Korean J. Pharmacogn. 2000, 31, 28–33. [Google Scholar]
- Lee, M.W.; Tanaka, T.; Nanaka, G.; Nishioka, I. Hirsunin, an ellagitannin with a diarylheptanoid moiety, from Alnus hirsuta var microphylla. Phytochemistry 1992, 31, 967–970. [Google Scholar]
- Lee, M.W.; Jeong, D.W.; Lee, Y.A.; Park, M.S.; Toh, S.H. Flavonoids from the leaves of Alnus hirsute. J. Pharm. Soc. Korea 1999, 43, 547–552. [Google Scholar]
- Lee, M.W.; Kim, N.Y.; Park, M.S.; Ahn, K.H.; Toh, S.H.; Hahn, D.R.; Kim, Y.C.; Chung, H.T. Diarylheptanoids with in vitro inducible nitric oxide synthesis inhibitory activity from Alnus hirsute. Planta Med. 2000, 66, 551–553. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.W.; Kim, J.H.; Jeong, D.W.; Ahn, K.H.; Toh, S.H.; Surh, Y.J. Inhibition of cyclooxigenase-2 expression by diarylheptanoids from the bark of Alnus hirsuta var. Sibirica. Biol. Pharm. Bull. 2000, 23, 517–518. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.W.; Song, J.Y.; Chin, Y.W.; Sung, S.H. Anti-adipogenic diarylheptanoids from Alnus hirsuta f. Sibirica on 3T3-L1 cells. Bioorg. Med. Chem. Lett. 2013, 23, 2069–2073. [Google Scholar] [CrossRef] [PubMed]
- Bae, C.I.; Gong, J.M.; Oh, J.W.; Kim, H.J.; Oh, G.J.; Park, S.K.; Chung, S.G.; Cho, E.H. Studied on the cytotoxic constituent of Alnus hirsuta. J. Pharm. Soc. Korea 1997, 41, 559–564. [Google Scholar]
- Lee, M.W.; Park, M.S.; Jeong, D.W.; Kim, K.H.; Kim, H.H. Diarylheptanoids from the leaves of Alnus hirsuta Turcz. Arch. Pharm. Res. 2000, 23, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Kuroyanagi, M.; Shimomae, M.; Nagashima, Y.; Muto, N.; Okuda, T. New diarylheptanoids from Alnus japonica and their antioxidative activity. Chem. Pharm. Bull. 2005, 53, 1519–1523. [Google Scholar] [CrossRef] [PubMed]
- Giang, P.M.; Son, P.T.; Matsunami, K.; Otsuka, H. New diarylheptanoids from Amomum muricarpum Elmer. Chem. Pharm. Bull. 2006, 54, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Sunnerheim-Sjöberg, K.; Per-Göran, K. Platyphylloside: Metabolism and digestibility reduction in vitro. J. Chem. Ecol. 1995, 21, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Fuchino, H.; Konishi, S.; Satoh, T.; Yagi, A.; Saitsu, K.; Tatsumi, T.; Tanaka, N. Chemical Evaluation of Betula species in Japan. II. Constituents of Betula platyphylla var. japonica. Chem. Pharm. Bull. 1996, 44, 1033–1038. [Google Scholar] [CrossRef]
- Gonzalez-Laredo, R.F.; Chen, J.; Karchesy, Y.M.; Karchesy, J.J. Four new diarylheptanoid glycosides from Alnus rubra bark. Nat. Prod. Res. 1999, 13, 75–80. [Google Scholar]
- Craveiro, A.A.; da Costa Prado, A.; Gottlieb, O.R.; De Albuquerque, P.W. Diarylheptanoids of Centrolobium species. Phytochemistry 1970, 9, 1869–1875. [Google Scholar] [CrossRef]
- Ohta, S.; Aoki, T.; Hirata, T.; Suga, T.J. The structures of four diarylheptanoid glycosides from the female flowers of Alnus serrulatorides. J. Chem. Soc. Perkin Trans. 1984, 1, 1635–1642. [Google Scholar] [CrossRef]
- Tung, N.H.; Kim, S.K.; Ra, J.C.; Zhao, Y.Z.; Sohn, D.H.; Kim, Y.H. Antioxidative and hepatoprotective diarylheptanoids from the bark of Alnus japonica. Planta Med. 2010, 76, 626–629. [Google Scholar] [CrossRef] [PubMed]
- Baranovsky, A.; Schmitt, B.; Fowler, D.J. Schneider B, Synthesis of new biosynthetically important diarylheptanoids and their oxa- and fluoro-analogues by three different strategies. Synth. Commun. 2003, 34, 1019–1045. [Google Scholar] [CrossRef]
- Giang, P.M.; Son, P.T.; Matsunami, K.; Otsuka, H. One new and several minor diarylheptanoids from Amomum muricarpum. Nat. Prod. Res. 2012, 26, 1195–1200. [Google Scholar] [CrossRef] [PubMed]
- Monge, P.; Scheline, R.; Solheim, E. The metabolism of zingerone, a pungent principle of ginger. Xenobiotica 1976, 6, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Yoon, K.H.; Yoon, S.H.; Ahn, H.S.; Lee, M.W. Quantitative analysis and validation of hirsutenone and muricarpone B from fermented Alnus sibirica. Nat. Prod. Sci. 2017, 23, 146–150. [Google Scholar] [CrossRef]
- Kim, H.J.; Yeom, S.H.; Kim, M.K.; Shim, J.G.; Paek, I.N.; Lee, M.W. Nitric oxide and prostaglandin E2 synthesis inhibitory activities of diarylheptanoids from the barks of Alnus japonica Steudel. Arch. Pharm. Res. 2005, 28, 177–179. [Google Scholar] [CrossRef] [PubMed]
- He, J.B.; Yan, Y.M.; Ma, X.J.; Lu, Q.; Li, X.S.; Su, J.; Li, Y.; Liu, G.M.; Cheng, Y.X. Sesquiterpenoids and diarylheptanoids from Nidus vespae and their inhibitory effects on nitric oxide production. Chem. Biodivers. 2011, 8, 2270–2276. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds (1–17) are available from the authors. |
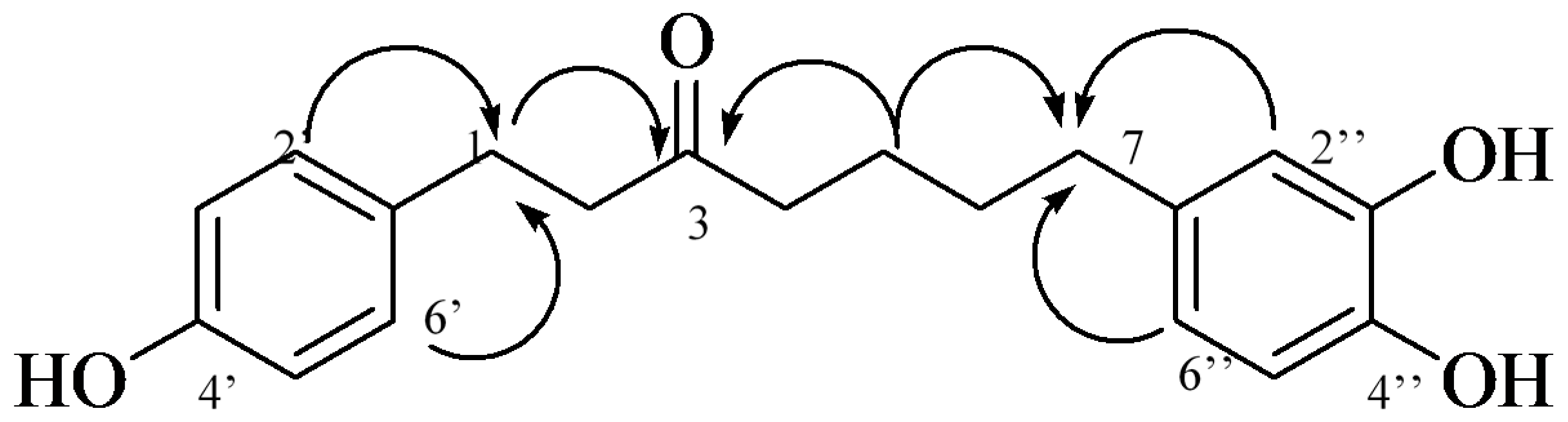
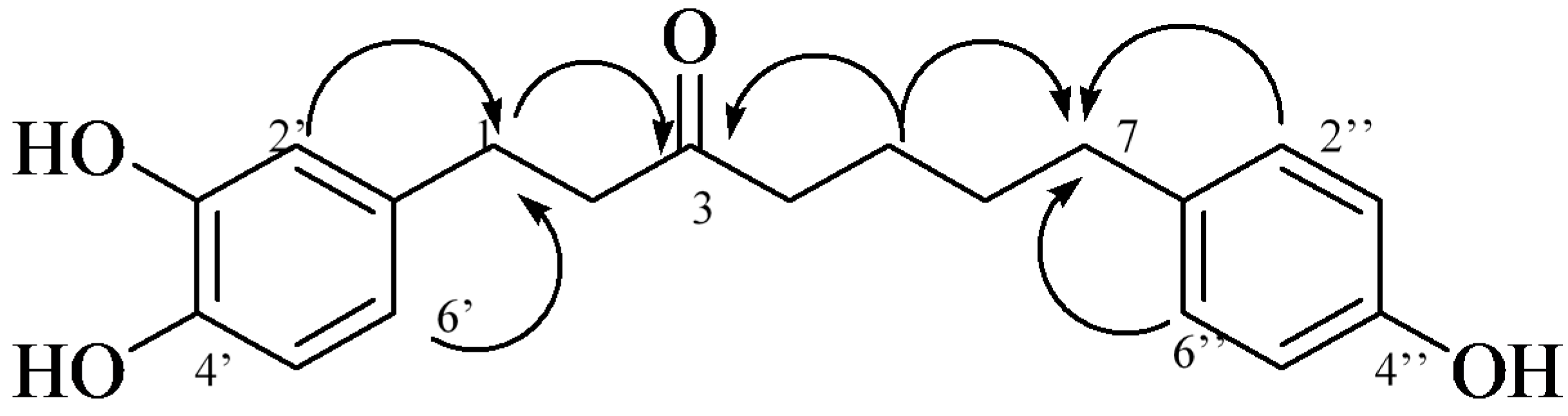
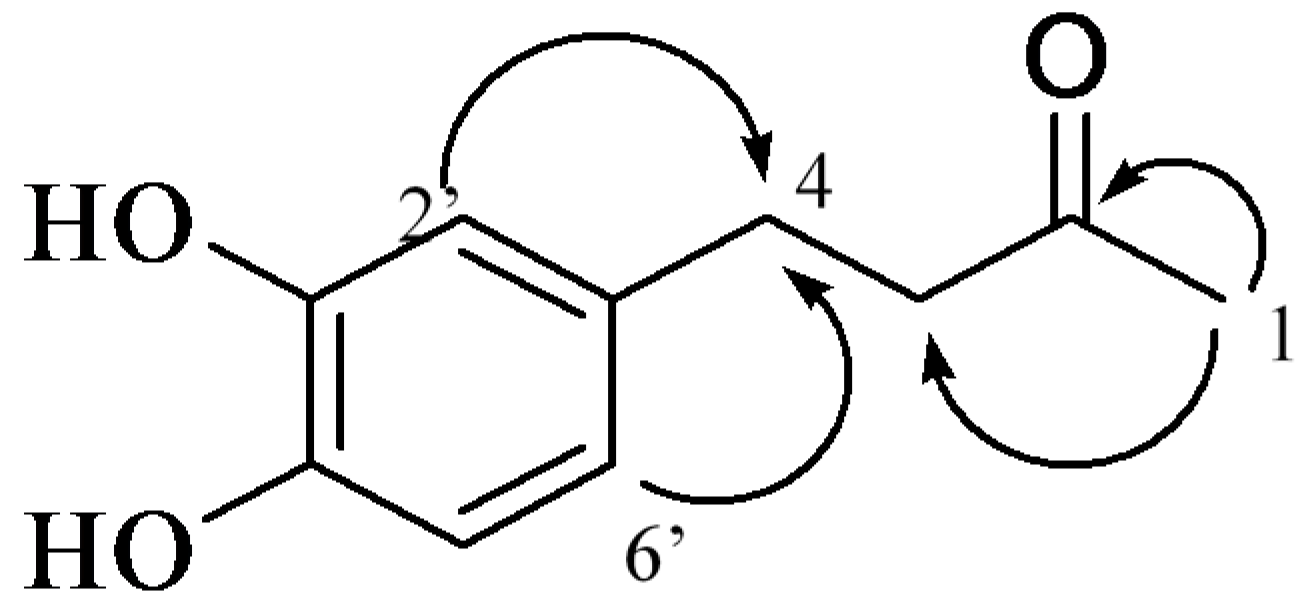
| Position | 1 | Position | 2 | ||
|---|---|---|---|---|---|
| (DMSO-d6, 600 and 150 MHz) | (DMSO-d6, 600 and 150 MHz) | ||||
| δC | δH | δC | δH | ||
| 1 | 28.76 | 2.65–2.75 (m) | 1 | 28.98 | 2.65–2.75 (m) |
| 2 | 44.08 | 2.65–2.75 (m) | 2 | 44.12 | 2.65–2.75 (m) |
| 3 | 210.72 | 3 | 210.72 | ||
| 4 | 42.16 | 2.45–2.55 (m) | 4 | 42.16 | 2.45–2.55 (m) |
| 5 | 23.15 | 1.45–1.55 (m) | 5 | 23.12 | 1.45–1.55 (m) |
| 6 | 31.09 | 1.45–1.55 (m) | 6 | 31.09 | 1.45–1.55 (m) |
| 7 | 34.68 | 2.45–2.55 (m) | 7 | 34.49 | 2.45–2.55 (m) |
| 1′ | 131.71 | 1″ | 132.45 | ||
| 2′, 6′ | 129.67 | 6.98 (2H, d, J = 8.0) | 2″, 6″ | 129.51 | 6.96 (2H, d, J = 8.0) |
| 3′, 5′ | 115.37 | 6.71 (2H, d, J = 8.0) | 3″, 5″ | 115.32 | 6.72 (2H, d, J = 8.0) |
| 4′ | 155.53 | 4″ | 155.37 | ||
| 1″ | 133.39 | 1′ | 132.62 | ||
| 2″ | 115.25 | 6.64 (1H, d, J = 2.0) | 2′ | 115.28 | 6.65 (1H, d, J = 2.0) |
| 3″ | 145.03 | 3′ | 145.14 | ||
| 4″ | 143.22 | 4′ | 143.41 | ||
| 5″ | 115.74 | 6.68 (1H, d, J = 8.0) | 5′ | 115.96 | 6.68 (1H, d, J = 8.0) |
| 6″ | 119.32 | 6.47 (1H, dd, J = 2.0, 8.0) | 6′ | 119.21 | 6.49 (1H, dd, J = 2.0, 8.0) |
| Samples | IC50 (μg/mL) | Compounds | IC50 (μM) |
|---|---|---|---|
| AS | 27.12 ± 3.00 c | 1 + 2 | 47.51 ± 1.00 d |
| FAS | 12.64 ± 3.61 b | 3 | 32.13 ± 0.22 b,c |
| Ascorbic acid | 3.04 ± 0.12 a | 4 | 28.34 ± 0.35 b,c |
| 5 | 20.13 ± 0.13 a | ||
| 6 | 24.98 ± 0.21 a,b | ||
| 7 | 107.79 ± 1.07 h | ||
| 8 | 623.41 ± 13.09 i | ||
| 9 | 34.87 ± 2.98 c | ||
| 10 | 45.11 ± 1.66 d | ||
| 11 | 31.07 ± 0.86 b,c | ||
| 12 | 44.40 ± 0.96 d | ||
| 13 | 28.99 ± 1.07 b,c | ||
| 14 + 15 | 60.31 ± 0.39 e | ||
| 16 | 101.21 ± 1.03 g | ||
| 17 | 68.89 ± 2.00 f | ||
| Ascorbic acid | 46.03 ± 0.45 d | ||
| Samples | IC50 (μg/mL) | Compounds | IC50 (μM) |
|---|---|---|---|
| AS | 12.34 ± 0.44 b | 1 + 2 | 15.57 ± 3.24 b,c |
| FAS | 4.22 ± 0.39 a | 3 | 9.69 ± 2.34 a,b |
| L-NMMA | 35.53 ± 3.45 c | 4 | 11.45 ± 2.86 a,b |
| 5 | 6.57 ± 1.18 a | ||
| 6 | 21.28 ± 2.93 c,d | ||
| 7 | 30.00 ± 2.86 e | ||
| 8 | 32.12 ± 3.00 e | ||
| 9 | 10.25 ± 2.51 a,b | ||
| 10 | 81.91 ± 5.98 g | ||
| 11 | 38.90 ± 4.01 f | ||
| 12 | 27.77 ± 1.30 d,e | ||
| 13 | 5.77 ± 2.35 a | ||
| 14 + 15 | 11.94 ± 4.03 a,b | ||
| 16 | 15.85 ± 2.06 b,c | ||
| 17 | 22.15 ± 2.91 c,d | ||
| L-NMMA | 41.95 ± 6.48 f | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, T.T.; Yin, J.; Lee, M. Anti-Inflammatory and Anti-Oxidative Activities of Phenolic Compounds from Alnus sibirica Stems Fermented by Lactobacillus plantarum subsp. argentoratensis. Molecules 2017, 22, 1566. https://doi.org/10.3390/molecules22091566
Le TT, Yin J, Lee M. Anti-Inflammatory and Anti-Oxidative Activities of Phenolic Compounds from Alnus sibirica Stems Fermented by Lactobacillus plantarum subsp. argentoratensis. Molecules. 2017; 22(9):1566. https://doi.org/10.3390/molecules22091566
Chicago/Turabian StyleLe, Thi Tam, Jun Yin, and MinWon Lee. 2017. "Anti-Inflammatory and Anti-Oxidative Activities of Phenolic Compounds from Alnus sibirica Stems Fermented by Lactobacillus plantarum subsp. argentoratensis" Molecules 22, no. 9: 1566. https://doi.org/10.3390/molecules22091566
APA StyleLe, T. T., Yin, J., & Lee, M. (2017). Anti-Inflammatory and Anti-Oxidative Activities of Phenolic Compounds from Alnus sibirica Stems Fermented by Lactobacillus plantarum subsp. argentoratensis. Molecules, 22(9), 1566. https://doi.org/10.3390/molecules22091566




