Constituents of the Fruits of Citrus medica L. var. sarcodactylis and the Effect of 6,7-Dimethoxy-coumarin on Superoxide Anion Formation and Elastase Release
Abstract
:1. Introduction
2. Results and Discussion
2.1. Purification and Characterization
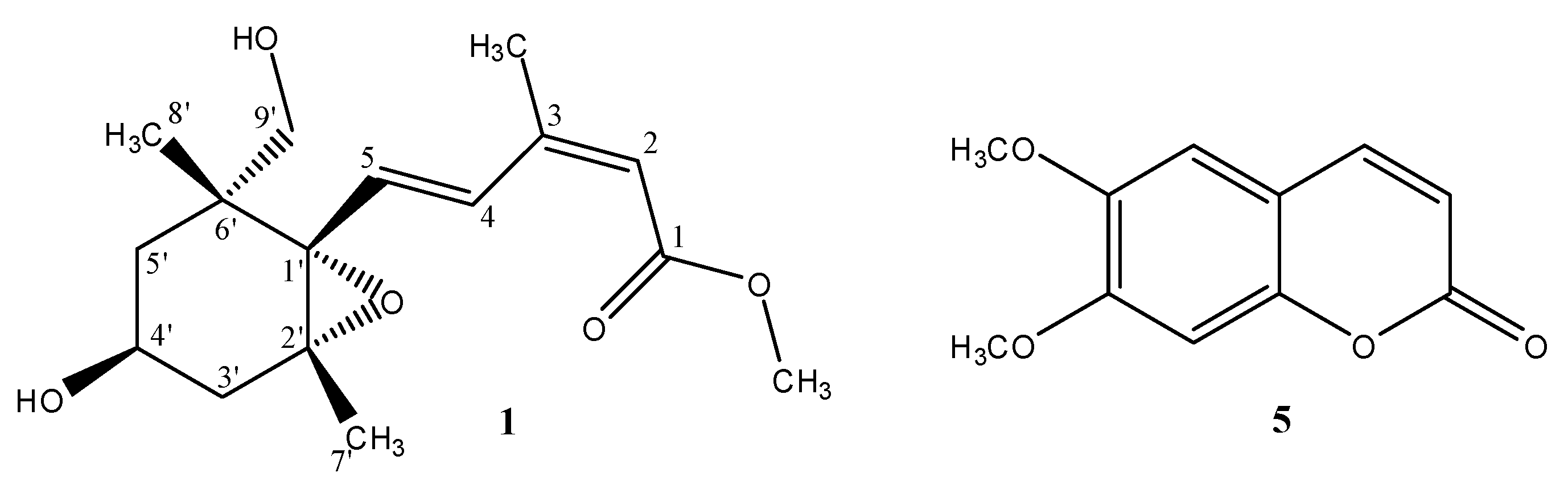
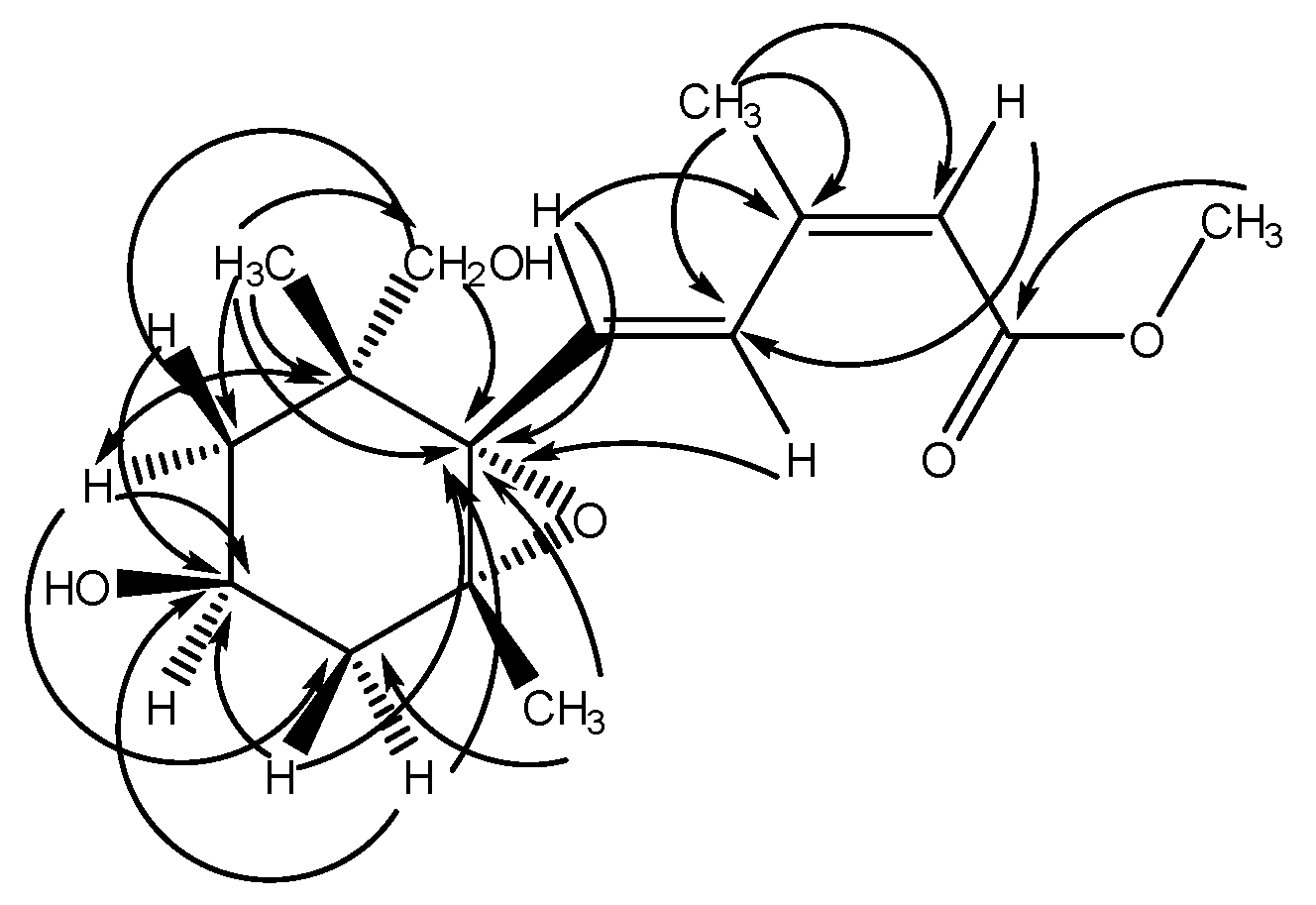
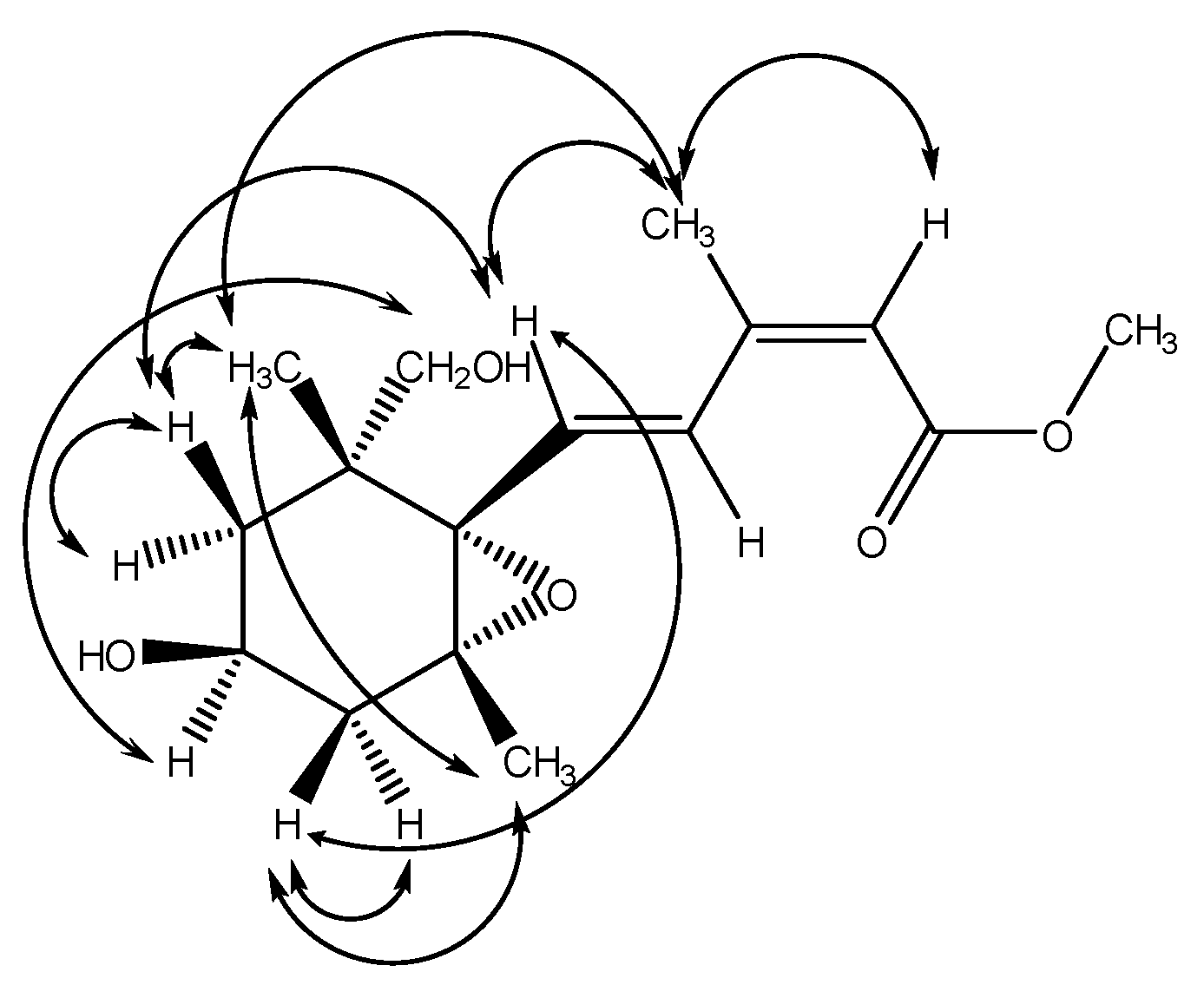
2.2. Structural Elucidation of Compound 1
2.3. The Inhibitory Activity of Superoxide Anion Generation and Elastase Release
3. Materials and Methods
3.1. General Information
3.2. Materials
3.3. Extraction and Isolation
3.4. Bioactivity Examination
3.4.1. Preparation of Human Neutrophils
3.4.2. Inhibition of Superoxide Anion Generation
3.4.3. Inhibition of Elastase Release
3.4.4. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chiu, N.Y.; Chang, K.H. The Illustrated Medicinal Plants of Taiwan, 1st ed.; SMC Publishing: Taipei, Taiwan, 1987; Volume III, p. 121. [Google Scholar]
- Wang, Y.; Qian, J.; Cao, J.; Wang, D.; Liu, C.; Yang, R.; Li, X.; Sun, C. Antioxidant capacity, anticancer ability and flavonoids composition of 35 Citrus (Citrus reticulata Blanco) varieties. Molecules 2017, 22, 1114. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.Y.; Wu, T.S.; Kuo, Y.H. Chemical constituents and cytotoxicity from the stem bark of Citrus medica. Heterocycles 2009, 78, 1309–1316. [Google Scholar]
- Roohbakhsh, A.; Parhiz, H.; Soltani, F.; Rezaee, R.; Iranshahi, M. Molecular mechanisms behind the biological effects of hesperidin and hesperetin for the prevention of cancer and cardiovascular diseases. Life Sci. 2015, 124, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Murthy, K.N.; Jayaprakasha, G.K.; Patil, B.S. Cytotoxicity of obacunone and obacunone glucoside in human prostate cancer cells involves Akt-mediated programmed cell death. Toxicology 2015, 329, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Leclere, L.; Fransolet, M.; Cote, F.; Cambier, P.; Arnould, T.; Van Cutsem, P.; Michiels, C. Heat-modified Citrus pectin induces apoptosis-like cell death and autophagy in HepG2 and A549 cancer cells. PLoS ONE 2015, 10, e0115831. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.Y.; Chen, Y.; Zou, L.; Zhang, Y.; Hu, F.; Feng, J.; Shen, J.; Wei, B. Naringin inhibits growth and induces apoptosis by a mechanism dependent on reduced activation of NF-κB/COX-2 caspase-1 pathway in HeLa cervical cancer cells. Int. J. Oncol. 2014, 45, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.; Stintzing, F.C.; Briemle, D.; Beckmann, C.; Meyer, U.; Gründemann, C. In vitro antiallergic effects of aqueous fermented preparations from Citrus and Cydonia fruits. Planta Med. 2012, 78, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Mokbel, M.S.; Hashinaga, F. Evaluation of the antioxidant activity of extracts from buntan (Citrus grandis Osbeck) fruit tissues. Food Chem. 2006, 94, 529–534. [Google Scholar] [CrossRef]
- Tsai, H.L.; Chang, S.K.; Chang, S.J. Antioxidant content and free radical scavenging ability of fresh red pummelo [Citrus grandis (L.) Osbeck] juice and freeze-dried products. J. Agric. Food Chem. 2007, 55, 2867–2872. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.N.; Shin, J.G.; Jang, H.D. Antioxidant and antidiabetic activity of Dangyuja (Citrus grandis Osbeck) extract treated with Aspergillus saitoi. Food Chem. 2009, 117, 35–41. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and anti-inflammatory properties of the Citrus flavonoids hesperidin and hesperetin: An updated review of their molecular mechanisms and experimental models. Phytother. Res. 2015, 29, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Park, S.W.; Chung, I.M.; Jung, Y.S. Anti-platelet effects of yuzu extract and its component. Food Chem. Toxicol. 2011, 49, 3018–3024. [Google Scholar] [CrossRef] [PubMed]
- Shende, S.; Ingle, A.P.; Gade, A.; Rai, M. Green synthesis of copper nanoparticles by Citrus medica Linn. (Idilimbu) juice and its antimicrobial activity. World J. Microbiol. Biotechnol. 2015, 31, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Chubukov, V.; Mingardon, F.; Schackwitz, W.; Baidoo, E.E.; Alonso-Gutierrez, J.; Hu, Q.; Lee, T.S.; Keasling, J.D.; Mukhopadhyay, A. Acute limonene toxicity in Escherichia coli is caused by limonene hydroperoxide and alleviated by a point mutation in alkyl hydroperoxidase AhpC. Appl. Environ. Microbiol. 2015, 81, 4690–4696. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.C.; Li, H.C.; Wu, P.H.; Huang, P.H.; Wang, Y.T. Assessment of oligogalacturonide from Citrus pectin as a potential antibacterial agent against foodborne pathogens. J. Food Sci. 2014, 79, 1541–1544. [Google Scholar] [CrossRef] [PubMed]
- Vollmerhausen, T.L.; Ramos, N.L.; Dzung, D.T.; Brauner, A. Decoctions from Citrus reticulata Blanco seeds protect the uroepithelium against Escherichia coli invasion. J. Ethnopharmacol. 2013, 150, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.Y.; Li, C.H.; Shen, Y.C.; Wu, T.S. Anti-inflammatory principles from the stem and root barks of Citrus medica. Chem. Pharm. Bull. 2010, 58, 61–65. [Google Scholar] [CrossRef] [PubMed]
- La, V.D.; Zhao, L.; Epifano, F.; Genovese, S.; Grenier, D. Anti-inflammatory and wound healing potential of Citrus auraptene. J. Med. Food 2013, 16, 961–964. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, D.; Bruschetta, G.; Di Paola, R.; Ahmad, A.; Campolo, M.; Cuzzocrea, S.; Esposito, E.; Navarra, M. The anti-inflammatory and antioxidant effects of bergamot juice extract (BJe) in an experimental model of inflammatory bowel disease. Clin. Nutr. 2014, 33, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Mitoshi, M.; Kuriyama, I.; Nakayama, H.; Miyazato, H.; Sugimoto, K.; Kobayashi, Y.; Jippo, T.; Kuramochi, K.; Yoshida, H.; Mizushina, Y. Suppression of allergic and inflammatory responses by essential oils derived from herbal plants and Citrus fruits. Int. J. Mol. Med. 2014, 33, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Noh, H.J.; Hwang, D.; Lee, E.S.; Hyun, J.W.; Yi, P.H.; Kim, G.S.; Lee, S.E.; Pang, C.; Park, Y.J.; Chung, K.H.; et al. Anti-inflammatory activity of a new cyclic peptide, citrusin XI, isolated from the fruits of Citrus unshiu. J. Ethnopharmacol. 2015, 163, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Hattori, S.; Shimokoriyama, M.; Kanao, M. Studies on flavanone glycosides. IV. The glycosides of ripe fruit peel and flower petals of Citrus aurantium L. J. Am. Chem. Soc. 1952, 74, 3614–3615. [Google Scholar] [CrossRef]
- Wu, T.S.; Kuoh, C.S.; Furukawa, H. Acridone alkaloids and a coumarin from Citrus grandis. Phytochemistry 1983, 22, 1493–1497. [Google Scholar]
- McPhail, A.T.; Ju-ichi, M.; Fujitani, Y.; Inoue, M.; Wu, T.S.; Furukawa, H. Isolation and structures of citropone-A and -B from Citrus plants, first examples of naturally-occurring homoacridone alkaloids containing a tropone ring system. Tetrahedron Lett. 1985, 26, 3271–3272. [Google Scholar] [CrossRef]
- Wu, T.S.; Huang, S.C.; Jong, T.T.; Lai, J.S.; Furukawa, H. Honyumine, a new linear pyranoacridone alkaloids from Citrus grandis Osbeck. Heterocycles 1986, 24, 41–43. [Google Scholar] [CrossRef]
- Wu, T.S. Baiyumine-A and -B, two acridone alkaloids from Citrus grandis. Phytochemistry 1987, 26, 871–872. [Google Scholar] [CrossRef]
- Wu, T.S.; Huang, S.C.; Jong, T.T.; Lai, J.S.; Kuoh, C.S. Coumarins, acridone alkaloids and a flavone from Citrus grandis. Phytochemistry 1988, 27, 585–587. [Google Scholar] [CrossRef]
- Wu, T.S. Alkaloids and coumarins of Citrus grandis. Phytochemistry 1988, 27, 3717–3718. [Google Scholar] [CrossRef]
- Huang, S.C.; Chen, M.T.; Wu, T.S. Alkaloids and coumarins from stem bark of Citrus grandis. Phytochemistry 1989, 28, 3574–3576. [Google Scholar] [CrossRef]
- Wu, T.S.; Huang, S.C.; Lai, J.S. Stem bark coumarins of Citrus grandis. Phytochemistry 1994, 36, 217–219. [Google Scholar]
- Takemura, Y.; Ju-ichi, M.; Ito, C.; Furukawa, H.; Tokuda, H. Studies on the inhibitory effects of some acridone alkaloids on Epstein-Barr virus activation. Planta Med. 1995, 61, 366–368. [Google Scholar] [CrossRef] [PubMed]
- Marıín, F.R.; Martinez, M.; Uribesalgo, T.; Castillo, S.; Frutos, M.J. Changes in nutraceutical composition of lemon juices according to different industrial extraction systems. Food Chem. 2002, 78, 319–324. [Google Scholar] [CrossRef]
- Giuffrè, A.M.; Zappia, C.; Capocasale, M. Physico-chemical stability of blood orange juice during frozen storage. Int. J. Food Prop. 2017, in press. [Google Scholar]
- Harkar, S.; Razdan, T.K.; Waight, E.S. Steroids, chromone and coumarins from Angelica officinalis. Phytochemistry 1984, 23, 419–426. [Google Scholar] [CrossRef]
- Wu, T.S.; Chang, F.C.; Wu, P.L.; Kuoh, C.S.; Chen, I.S. Constituents of Leaves of Tetradium Glabrifolium. J. Chin. Chem. Soc. 1995, 42, 929–934. [Google Scholar] [CrossRef]
- Sendker, J.; Böker, I.; Lengers, I.; Brandt, S.; Jose, J.; Stark, T.; Hofmann, T.; Fink, C.; Abdel-Aziz, H.; Hensel, A. Phytochemical characterization of low molecular weight constituents from marshmallow roots (Althaea officinalis) and inhibiting effects of the aqueous extract on human hyaluronidase-1. J. Nat. Prod. 2017, 80, 290–297. [Google Scholar] [CrossRef] [PubMed]
- He, H.Y.; Ling, L.; Zhou, M. Isolation and structure elucidation of two dimeric limettins from fingered citron. Youji Huaxue 1987, 3, 193–196. [Google Scholar]
- Sugimoto, T.; Miyase, T.; Kuroyanagi, M.; Ueno, A. Limonoids and quinolone alkaloids from Evodia rutaecarpa bentham. Chem. Pharm. Bull. 1988, 36, 4453–4461. [Google Scholar] [CrossRef]
- Aquino, R.; Cáceres, A.; Morelli, S.; Rastrelli, L. An extract of Tagetes lucida and its phenolic constituents as antioxidants. J. Nat. Prod. 2002, 65, 1773–1776. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.Y.; Juang, S.H.; Huang, G.J.; Liao, Y.R.; Chen, Y.F.; Wu, C.C.; Chang, H.T.; Wu, T.S. The Constituents of Michelia compressa var. formosana and their bioactivities. Int. J. Mol. Sci. 2014, 15, 10926–10935. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.R.; Sharma, N.D.; Harrison, J.S.; Malone, J.F.; McRoberts, W.C.; Hamilton, J.T.G.; Harper, D.B. Enzyme-catalysed synthesis and reactions of benzene oxide/oxepine derivatives of methyl benzoates. Org. Biomol. Chem. 2008, 6, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Chan, Y.Y.; Kuo, P.C.; Chen, Y.F.; Chang, R.J.; Chen, I.S.; Wu, S.J.; Wu, T.S. The constituents of roots and stems of Illigera luzonensis and their anti-platelet aggregation effects. Int. J. Mol. Sci. 2014, 15, 13424–13436. [Google Scholar] [CrossRef] [PubMed]
- Claydon, N.; Frederick, J.; Pople, G.M. Elm bark beetle boring and feeding deterrents from Phomopsis oblonga. Phytochemistry 1985, 24, 937–943. [Google Scholar] [CrossRef]
- Steevesa, V.; Försterb, H.; Pommerb, U.; Savidgea, R. Coniferyl alcohol metabolism in conifers—I. Glucosidic turnover of cinnamyl aldehydes by UDPG: Coniferyl alcohol glucosyltransferase from pine cambium. Phytochemistry 2001, 57, 1085–1093. [Google Scholar] [CrossRef]
- Marco, J.A.; Sanz-Cervera, J.F.; Sancenon, F.; Jakupovic, J.; Rustaiyant, A.; Mohamadit, F. Oplopanone derivatives monoterpene glycosides from Artemisia sieberi. Phytochemistry 1993, 34, 1061–1067. [Google Scholar] [CrossRef]
- Chan, Y.Y.; Leu, Y.L.; Wu, T.S. The constituents of the leaves of Aristolochia heterophylla Hemsl. Chem. Pharm. Bull. 1999, 47, 887–889. [Google Scholar] [CrossRef]
- Gattuso, G.; Barreca, D.; Caristi, C.; Gargiulli, C.; Leuzzi, U. Distribution of flavonoids and furocoumarins in juices from cultivars of Citrus bergamia Risso. J. Agric. Food Chem. 2007, 55, 9921–9927. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Win Aung, K.K.; Ran, X.K.; Wang, X.T.; Dou, D.Q.; Dong, F. A new sesquiterpene lactone from yacon leaves. Nat. Prod. Res. 2017, 31, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wang, L.; Guo, X.J.; Lou, H.X.; Ren, D.G. A new flavonoid glycoside and other constituents from Dracocephalum moldavica. Nat. Prod. Res. 2013, 27, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Takahashi, K.; Nukano, T. Convenient synthesis of optically active abscisic acid and xanthoxin. Tetrahedron 1992, 48, 8229–8238. [Google Scholar] [CrossRef]
- Yang, S.C.; Chung, P.J.; Ho, C.M.; Kuo, C.Y.; Hung, M.F.; Huang, Y.T.; Chang, W.Y.; Chang, Y.W.; Chan, K.H.; Hwang, T.L. Propofol inhibits superoxide production, elastase release, and chemotaxis in formyl peptide-activated human neutrophils by blocking formyl peptide receptor 1. J. Immunol. 2013, 190, 6511–6519. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.P.; Hsieh, P.W.; Chang, Y.J.; Chung, P.J.; Kuo, L.M.; Hwang, T.L. 2-(2-Fluorobenzamido) benzoate ethyl ester (EFB-1) inhibits superoxide production by human neutrophils and attenuates hemorrhagic shock-induced organ dysfunction in rats. Free Radic. Biol. Med. 2011, 50, 1737–1748. [Google Scholar] [CrossRef] [PubMed]
- Strehmel, N.; Böttcher, C.; Schmidt, S.; Scheel, D. Profiling of secondary metabolites in root exudates of Arabidopsis thaliana. Phytochemistry 2014, 108, 35–46. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the purified compounds are available from the authors. |
| Position | δH (mult., J in Hz) | δC |
|---|---|---|
| 1 | 168.2 | |
| 2 | 5.79 (1H, s) | 118.3 |
| 3 | 152.0 | |
| 4 | 8.00 (1H, d, 15.7) | 131.7 |
| 5 | 6.55 (1H, d, 15.7) | 135.7 |
| 1′ | 83.3 | |
| 2′ | 87.8 | |
| 3′ | 2.03 (1H, dd, 13.7, 7.0) 1.73 (1H, dd, 13.7, 10.4) | 46.0 |
| 4′ | 4.12 (1H, m) | 66.0 |
| 5′ | 1.86 (1H, dd, 13.5, 6.9) 1.68 (1H, dd, 13.5, 13.5) | 44.6 |
| 6′ | 49.2 | |
| 7′ | 1.16 (3H, s) | 19.6 |
| 8′ | 0.94 (3H, s) | 16.4 |
| 9′ | 3.82 (1H, d, 7.5) 3.72 (1H, d, 7.5) | 77.3 |
| CH3-3 | 2.10 (3H, s,) | 21.2 |
| OCH3 | 3.70 (3H, s) | 51.6 |
| Compound | IC50 (μM) a | |
|---|---|---|
| Superoxide Anion Generation | Elastase Release | |
| 5 | 3.8 ± 1.4 *** | >10 |
| LY294002 b | 0.4 ± 0.02 *** | 1.5 ± 0.3 *** |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, Y.-Y.; Hwang, T.-L.; Kuo, P.-C.; Hung, H.-Y.; Wu, T.-S. Constituents of the Fruits of Citrus medica L. var. sarcodactylis and the Effect of 6,7-Dimethoxy-coumarin on Superoxide Anion Formation and Elastase Release. Molecules 2017, 22, 1454. https://doi.org/10.3390/molecules22091454
Chan Y-Y, Hwang T-L, Kuo P-C, Hung H-Y, Wu T-S. Constituents of the Fruits of Citrus medica L. var. sarcodactylis and the Effect of 6,7-Dimethoxy-coumarin on Superoxide Anion Formation and Elastase Release. Molecules. 2017; 22(9):1454. https://doi.org/10.3390/molecules22091454
Chicago/Turabian StyleChan, Yu-Yi, Tsong-Long Hwang, Ping-Chung Kuo, Hsin-Yi Hung, and Tian-Shung Wu. 2017. "Constituents of the Fruits of Citrus medica L. var. sarcodactylis and the Effect of 6,7-Dimethoxy-coumarin on Superoxide Anion Formation and Elastase Release" Molecules 22, no. 9: 1454. https://doi.org/10.3390/molecules22091454





