1. Introduction
Salvia, the largest genus of the Lamiaceae family, comprises nearly 900 species distributed throughout the world. Some species of
Salvia have been cultivated for their use in folk medicine and for culinary purposes.
Salvia lavandulifolia Vahl. (
Salvia officinalis subsp.
lavandulifolia (Vahl) Gams or Spanish sage) is native to the Iberian Peninsula and grows from south-east of Morocco to the Mediterranean area of France. It is a small woody herbaceous perennial shrub, ranging from 17 to 100 cm high, with mauve-blue flowers that usually grows on the sandy-calcareous soils of mountainous areas (from 300 to more than 1000 m above sea level) [
1].
The essential oil (EO) composition of the genus
Salvia is highly variable, depending on climatic, geographical and seasonal conditions as well as genotypic factors [
2,
3,
4,
5].
S. lavandulifolia EO (SlEO) has been described as a safe memory enhancer, suggesting it could be used in Alzheimer’s disease therapy [
6,
7,
8,
9]. Moreover, SlEO presents other notable activities with medical applications, which include spasmolitic [
10], oestrogenic [
11] and antimicrobial [
12] properties.
The most common technique used for the analysis of volatile components is gas chromatography coupled with a flame ionization detector (GC-FID) or with a mass spectrometry detector (GC-MS), because these methods provide qualitative and quantitative data, even for complex mixtures, with high sensitivity and resolution. Some studies have reported the relative quantitation of SlEO from other Spanish regions [
1,
7,
13,
14,
15,
16] and from other countries (Poland [
17], Germany [
12] and Brazil [
18,
19]). None of them have studied SlEOs from Murcia nor made a chiral characterization of the EOs from this species. The chiral distribution of the principal compounds, as an essential part of quality assurance, helps to determine the genuineness or adulteration of EOs [
20]. Besides, the enantiomeric ratio of a compound can cause significant differences from the point of view of sensorial analysis and bioactivity [
21,
22].
Reactive oxygen species (superoxide radical, hydrogen peroxide, etc.) have been implicated in the pathology of several human diseases (cancer, atherosclerosis, malaria, rheumatoid arthritis and neurodegenerative diseases, and aging effects [
23,
24,
25,
26,
27]), and in the deterioration of food and cosmetic products [
28]. The use of EOs as natural antioxidant is regarded as a safe alternative to synthetic ones [
29,
30].
Lipoxygenase enzyme (LOX) produces hydroperoxyeicosatetraenoic acids (HPETEs) and hydroxyoctadecadienoic acids (HODEs) from arachidonic acid and linoleic acid, respectively [
31]. SlEO can be useful to inhibit this enzyme and, therefore, may represent a valid strategy for prevention and therapy of Alzheimer’s, cancer and inflammatory diseases [
32]. As well as this bioactivity, SlEO shows acetylcholinesterase (AChE) inhibitory activity [
6,
9,
11,
33], so it can be used as memory enhancer in Alzheimer’s disease [
34,
35].
Some studies from other regions or countries reported the antibacterial and antifungal activities of EOs from
Salvia genus [
19,
36,
37,
38]. These activities are highly related with the particular EO composition and microbial strain used in the assays. The antimicrobial capacity of EOs lends weight to their use as natural preservatives.
In this study, we describe the detailed volatile compositions of four SlEOs from the province of Murcia (Spain) and evaluate whether they fulfill the requirements of ISO 3526 [
39]. The enantiomeric ratios of the commercially available compounds are also reported. The in vitro antioxidant and antimicrobial activities of these SlEOs are studied to ascertain their potential use as natural preservatives. The inhibition of LOX and AChE by SlEOs could lead to useful strategies for the treatment of inflammatory and Alzheimer’s diseases and for natural insecticidal purposes.
3. Materials and Methods
3.1. Essential Oils
SlEOs were obtained from 300 g of the aerial part of plants grown in the south-east of Spain by means of hydrodistillation for 3 h in a Clevenger-type apparatus [
59], dried over anhydrous sodium sulfate and stored at 4 °C until use. SlEO-1 and -4 were obtained from plants grown in the Lower Meso-Mediterranean bioclimatic zone, SlEO-2 from plants grown in the Upper Meso-Mediterranean bioclimatic zone and SlEO-3 from plants grown in the Supra-Mediterranean bioclimatic zone [
60]. Plant species were identified in the Plant Biology Department of Murcia University by Dr. Pedro Sanchez-Gomez. The Department of Biochemistry and Molecular Biology-A stores the voucher specimens (BMBA160611, BMBA160612, BMBA160613, BMBA160614 for SlEO-1, -2, -3 and -4, respectively).
3.2. Reagents and Solvents
The chemical compounds used for the antioxidant methods, the reagents for LOX and AChE inhibition assays and reference antibacterial and antifungal compounds were purchased from Sigma-Aldrich (Madrid, Spain). All compounds were of analytical grade (purity higher than 95%). All culture media were acquired from VWR Chemicals (Barcelona, Spain): Mueller-Hinton agar (MHA), Mueller-Hinton broth (MHB), Roswell Park Memorial Institute medium (RPMI-1640), Sabouraud dextrose agar (SDA), tryptic soy broth (TSB) and yeast peptone dextrose (YPD). Solvents of analytic grade and buffers were purchased from Merck (Madrid, Spain). Type I (18 MΩ·cm) deionized water (MilliQ-Reference, Millipore, Madrid, Spain) was used in this work.
3.3. FGC-FID and FGC-MS
The relative proportions of the SlEO compounds were determined using a GC7890 chromatograph (Agilent, Madrid, Spain) equipped with a FID. The detector temperature was 320 °C. The sandwich injections (0.2 µL air, 0.2 µL isooctane, 0.2 µL air, 0.3 µL sample and 0.2 µL air, described from plunger to needle) were made using a MPS-2XT automatic multi-purpose sampler (Gerstel, Sevilla, Spain).
The chromatography was performed in a low bleed capillary fused-silica column, SLB-5ms from Supelco (Madrid, Spain; 15 m length × 0.1 mm internal diameter × 0.1 µm film thickness) with hydrogen as carrier gas (0.8 mL/min) which generates a head pressure of 46.345 psi. This carrier gas was produced with an electrolytic Parker-Domnik-Hunter (Barcelona, Spain) generator.
The injection conditions were as follows: septum purge 3 mL/min, split ratio 100:1 and injector temperature 300 °C. GC oven temperature was kept at 60 °C and programmed to raise up to 300 °C as follows: to 92 °C at a rate 15 °C/min, to 96 °C at a rate of 1 °C/min, to 108 °C at a rate of 20 °C/min and kept constant for 0.5 min, to 120 °C at a rate of 5 °C/min, to 160 °C at a rate of 20 °C/min, to 170 °C at a rate of 5 °C/min and to 300 °C at a rate of 30 °C/min, kept constant at 300 °C for 0.5 min.
The absolute concentrations were determined using calibration curves of each commercially available component described in the SlEOs (
Table S1), using the gas chromatograph previously described, coupled with an Agilent MS5975 mass spectrometer with electronic impact ionization and single quadrupole. MS was adjusted to the following conditions: electron ionization energy 70 eV, electron-multiplier voltage 1129, acquisition mass range 30–300
m/
z, 21.035 scans·s
−1, transfer line temperature 280 °C, ion source temperature 230 °C, MS quadrupole temperature 150 °C.
Compounds were identified by comparison of their retention times and the mass spectra of commercially available pure standards (
Figures S2 and S3) and the NIST 08 and Wiley 7 data bases. Several dilutions of SlEOs have been made in 2,2,4-trimethylpentane, to obtain their relative and absolute determinations.
3.4. EsGC-MS
A Chiraldex B-DM column (Astec, Madrid, Spain; 30 m length × 0.25 mm internal diameter × 0.12 µm film thickness) from Supelco, made of 2,3-di-O-methyl-6-t-butylsilyl β-cyclodextrin, non-bonded to fused silica column, was installed in the previously described device. The injections were similar to the one previously described but, in this case, 0.5 μL of sample was injected. The injector and transfer line temperatures were 200 °C. Hydrogen was used as carrier gas (constant flow of 2.5 mL/min, 8 psi starting column head pressure). The column temperature was programmed to increase from 35 °C to 170 °C at a rate of 4 °C/min.
To identify both enantiomers, the retention times and the mass spectra of commercially available pure standards were compared with those of the SlEO compounds, and confirmed with the NIST and Wiley spectral data bases (
Figure S4).
3.5. Antioxidant Capacity
Five antioxidant methods were performed with SlEOs and their main individual compounds in triplicate. In the oxygen radical absorbance capacity (ORAC) assay, peroxyl radicals are produced by 2,2′-Azobis(2-methylpropionamidine) dihydrochloride (AAPH), which reacts with fluorescein giving a non-fluorescent compound [
61]. The antioxidants are able to scavenge these peroxyl radicals. Trolox was used as the reference antioxidant. The 2,2-diphenyl-1-picrylhydrazyl (DPPH) [
62] and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) [
63,
64] methods measure the bleaching produced by the reduction of the coloured DPPH and ABTS radicals. Trolox was used as the reference antioxidant. The RdP method measure the ability to reduce ferric ions, according to the method of Oyaizu [
65]. Ascorbic acid was the reference antioxidant used in this method. The ChP method measured the ability to chelate Fe
2+ ion, following the method of Miguel et al. [
66]. EDTA was used as reference antioxidant. The SlEOs were diluted in 100% ethanol in the DPPH, ABTS, RdP and ChP methods, whereas in the ORAC method, SlEOs were diluted in 15% ethanol to obtain a final concentration of 5% ethanol in the reaction medium. All measurements were made at the end-point of the reaction, except in the ORAC method (
Figure S5).
3.6. Inhibition Activity
A LOX preparation from
Glycine max (soybean) was acquired from Sigma-Aldrich. LOX inhibitory activity was determined as previously reported [
67], based on the absorption at 234 nm of the hydroperoxyde conjugated dienes (ε = 25,000 M
−1 cm
−1), which are generated from the oxidation of linoleic acid in the presence of oxygen and LOX. This assay was carried out on a double beam Lambda 35 spectrophotometer (PerkinElmer, Madrid, Spain) with the UV-Winlab software, at 25 °C. SlEOs were diluted in 10% acetonitrile (supergradient quality, A
210 < 0.018) and nordihydroguaiaretic acid (NDGA) was used as standard LOX inhibitor.
AChE VI-S from
Electrophorus electricus was purchased from Sigma-Aldrich. AChE inhibitory activity was measured in triplicate using a 96-well microplate reader at 25 °C, according to Ellman’s method [
68]. This enzyme hydrolyzes acetylthiocholine to acetate and thiocholine, which reacts with 5,5-dithio-bis-(2-nitrobenzoic acid) (DTNB) producing a coloured compound with absorbance at 412 nm. The reaction was measured for 10 min at 25 °C, using a 96-well microplate reader. SlEOs were diluted in 15% ethanol, to obtain a final concentration of 5% ethanol in the reaction medium. Galantamine hydrobromide was used as reference AChE inhibitor.
The DI in both antienzymatic assays were calculated using Equation (1):
where
and
are the steady state rates without and with inhibitor, respectively. The inhibitions of LOX were reported as DI at 150 µg/mL of each SlEO. Higher SlEO concentrations could not be assayed due to their solubility limits. However, AChE inhibition could be expressed as IC
50. To calculate the IC
50 values, data of DI (%) of seven different concentrations of each SlEO were plotted and fitted by non-linear regression according to Equation (2) using Sigma Plot software (systatsoftware.com) (
Figure S6). The inhibitions of LOX and AChE by individual compounds were reported as IC
50 or DI, depending on their inhibition capacities and solubilities.
3.7. Antimicrobial Activity
3.7.1. Microorganisms and Culture Conditions
The following test microorganisms used in this work were acquired from Sigma-Aldrich: Pseudomonas aeruginosa ATCC 9027, Escherichia coli ATCC 8739, Staphylococcus aureus ATCC 6538, and Candida albicans ATCC 10231. The stock cultures were preserved in TSB or YPD with 15% glycerol, for bacteria and yeast cells, respectively, at −80 °C. Isolated colonies from an 18- to 24-h agar plate were transferred to MHB for bacteria and RPMI-1640 for C. albicans.
3.7.2. MIC and MBC or MFC
MIC were determined using the broth microdilution method in 96-well microplates, according to the M07-A10 [
69] standard for bacteria and the M27-A3 [
70] for
Candida. Briefly, two-fold dilutions of a 40 µL/mL SlEOs were prepared to obtain a final concentration range of 0.08–20 µL/mL with 0.5% Tween
®80 and 2.5% DMSO. The antimicrobial activities of main compounds were also tested in the concentration range of 0.12–15 mM with 0.5% Tween
®80 and 2.5% DMSO as solvent. The final strain concentration was 5 × 10
5 CFU/mL in MHB for the bacteria and 0.5−2.5 × 10
3 in RPMI-1640 for the yeast. These plates were incubated for 24 h for bacteria and 48 h for
Candida, both at 35 ± 1 °C, under aerobic conditions on a plate shaker at 100 rpm. Streptomycin (0.06–8.00 µg/mL) and fluconazole (0.13–16 µg/mL) were used as reference antibacterial and antifungal compound, respectively. The negative and positive controls were made to test that all solutions were sterile and that 0.5% tween and 2.5% DMSO, used for emulsifying the SlEOs, did not show any antimicrobial activity. MIC is defined as the lowest concentration of EO or individual compound with no visible growth of microorganisms, at the end of the incubation period. Then, 100 µL of each well without growth in the MIC assay were spread on MHA (bacteria) or SDA (yeast) and incubated for 24 h at 35 ± 1 °C to determinate the MBC or MFC. The MBC or MFC was defined as the lowest EO concentration in which microorganisms failed to grow in broth and on agar. All determinations were carried out in triplicate.
3.8. Statistical Analysis
The statistical analyses of data were made using both univariate and multivariate methods [
71] Data were recorded as mean ± standard deviation of at least triplicate determinations. Data values of 0.0 in the tables means values lower than 0.05 units. Data quality was analyzed by ANOVA, and means were confronted using Tukey’s (HSD) test, considering differences to be significant at
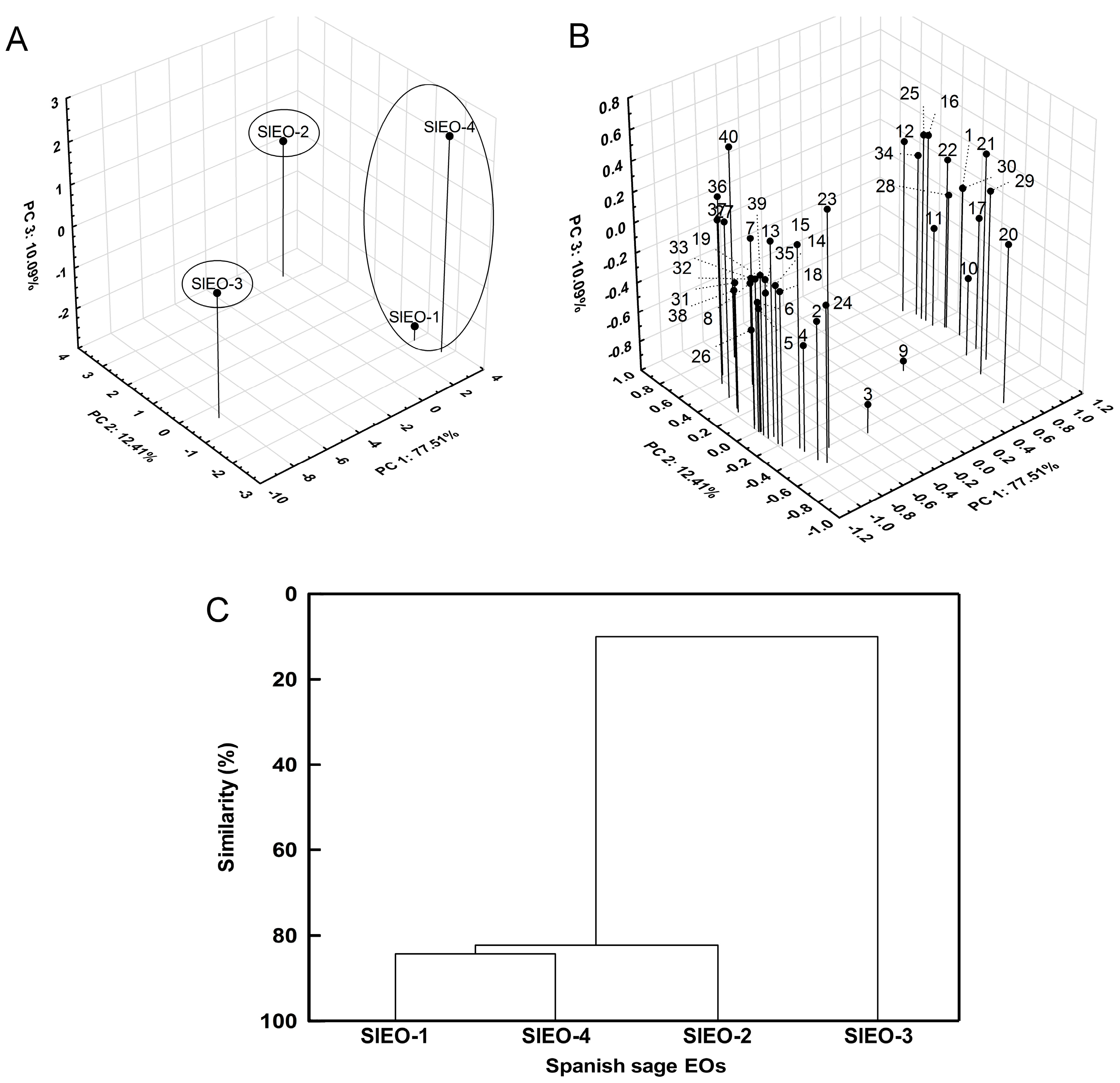
p < 0.05, represented by different letters next to numerical values in the text and tables. To determine similarity between SlEOs, PCA and AHC based on Euclidean distance were performed. Statistica software (software.dell.com) was used to conduct all statistical analyses.
4. Conclusions
S. lavandulifolia has been widely used for its medicinal and flavoring properties [
7]. The composition of its EO can vary depending on several factors [
14]. In this study, four SlEOs obtained from plants grown in Murcia were soundly analyzed. They were rich in α-pinene, camphene, 1,8-cineole and camphor. Moreover, other compounds were found in significant amounts, such as β-pinene, sabinene, myrcene, limonene and borneol. PCA and AHC analyses showed that SlEO-1, -2 and -4 are very similar, but different from SlEO-3. SlEO-1, -2 and -4 have similar concentrations of linalool (
16),
cis-sabinol (
17), linalyl acetate (
25) and sabinyl acetate (
28), whereas SlEO-3 has higher concentration of camphor (
18), borneol (
19), (−)-myrtenol (
23), (
Z)-citral (
24) and others.
The enantiomeric distribution for sabinene hydrate, camphor, bornyl acetate, borneol and β-caryophyllene were similar in all SlEOs, so they can be used as biomolecular markers for these EOs. The enantiomeric distribution is important to prove the authenticity of EOs.
As regards the antioxidant results, SlEO-3 was the most antioxidant in ORAC, DPPH, ABTS and RdP methods. These differences between methods can be explained by the different antioxidant activities of their individual compounds in each method; for example, linalool has a high ChP, but it is less active than others against ABTS radical.
All the SlEOs showed LOX inhibition at 150 µg/mL. Although bornyl acetate and limonene were the most active against LOX, the two main compounds (camphor and 1,8-cineole) were also able to reduce the rate of the LOX reaction. Moreover, SlEOs are effective against AChE activity. 1,8-Cineole, a major compound in these EOs, is able to reach 50% inhibition at a concentration of 35 µg/mL. These SlEOs could be used for memory enhancement due to their anti-AChE activities and their low proportion of α-thujone [
6,
9,
33]. In fact, there is negligible quantities of α-thujone in SlEOs from Murcia, that prevent their undesirable effects on brain, muscle, kidney and liver cells [
58].
The SlEOs of this study also showed antimicrobial activity against E. coli, S. aureus and C. albicans, but not against P. aeruginosa, which is a highly resistant Gram-negative bacterium. S. aureus and C. albicans showed greater susceptibility to the SlEOs than E. coli, probably because of some individual components, such as limonene and borneol, which are more effective against these two microorganisms.
The extensive knowledge acquired about the composition and the antioxidant, antienzymatic and antimicrobial activities of these SlEOs is useful for confirming their potential applications in agronomic, food, cosmetic and pharmaceutical industries.