Substituent Effects on the Stability of Thallium and Phosphorus Triple Bonds: A Density Functional Study
1. Introduction
2. Methodology
3. General Considerations
4. Results and Discussion
4.1. Small Ligands on Substituted RTl≡PR
 P. It must be noted that the fact that the lone pair of phosphorus has s character and the valence p orbital of phosphorus is much smaller than that of thallium means that both factors can vigorously affect the bonding overlaps between phosphorus and thallium atoms. Therefore, it is anticipated that the triple bond in these RTl≡PR species is very weak. This prediction is confirmed by the three DFT calculations shown in Table 1. All of the values for the Wiberg bond index (WBI) [59,60,61] are a little bit higher than 1.0, rather than 2.0. That is to say, regardless of whether small electropositive or small electronegative groups are attached, the RTl≡PR systems possess a quite weak Tl≡P triple bond.
P. It must be noted that the fact that the lone pair of phosphorus has s character and the valence p orbital of phosphorus is much smaller than that of thallium means that both factors can vigorously affect the bonding overlaps between phosphorus and thallium atoms. Therefore, it is anticipated that the triple bond in these RTl≡PR species is very weak. This prediction is confirmed by the three DFT calculations shown in Table 1. All of the values for the Wiberg bond index (WBI) [59,60,61] are a little bit higher than 1.0, rather than 2.0. That is to say, regardless of whether small electropositive or small electronegative groups are attached, the RTl≡PR systems possess a quite weak Tl≡P triple bond.4.2. Large Ligands on Substituted R′Tl≡PR′
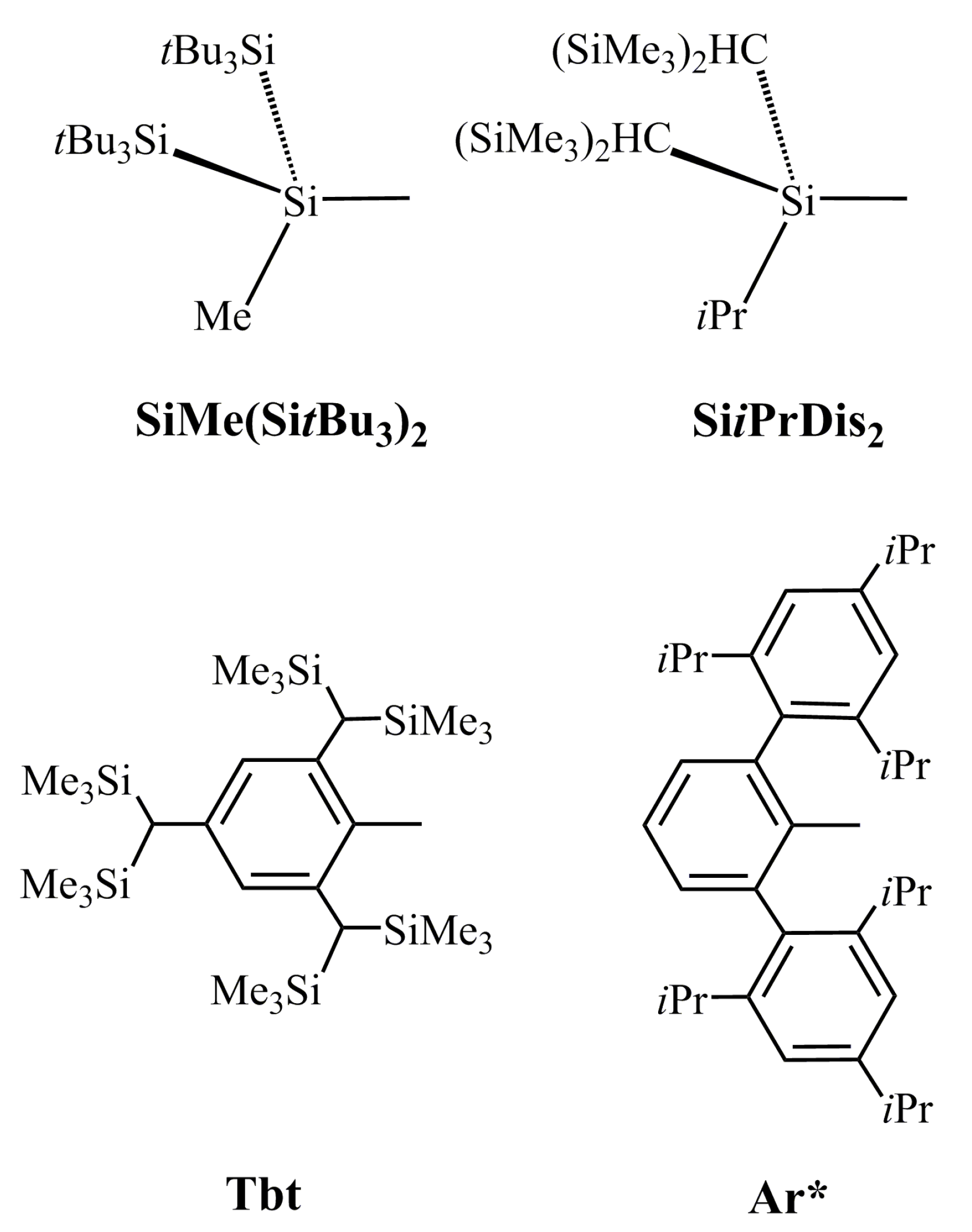
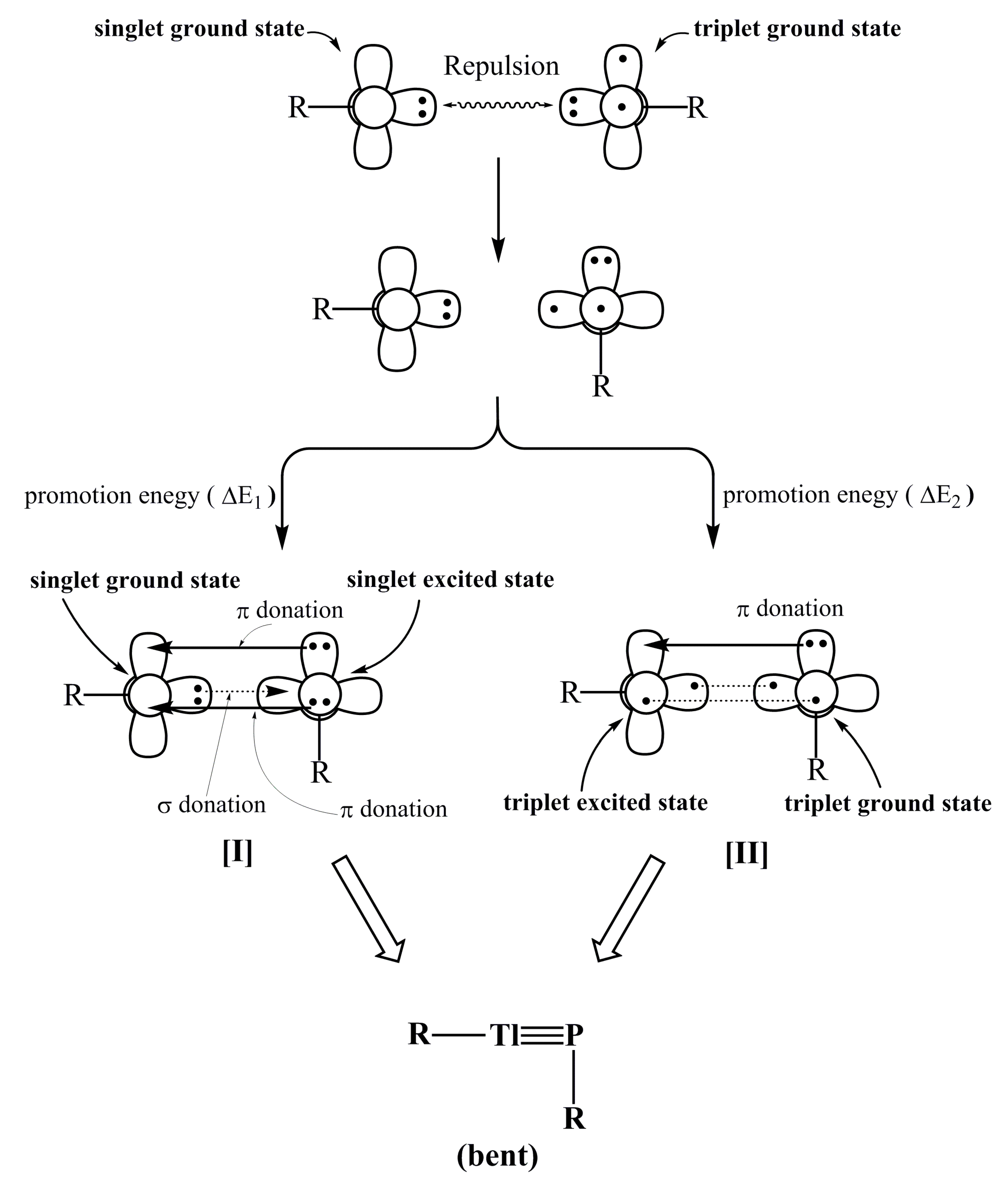
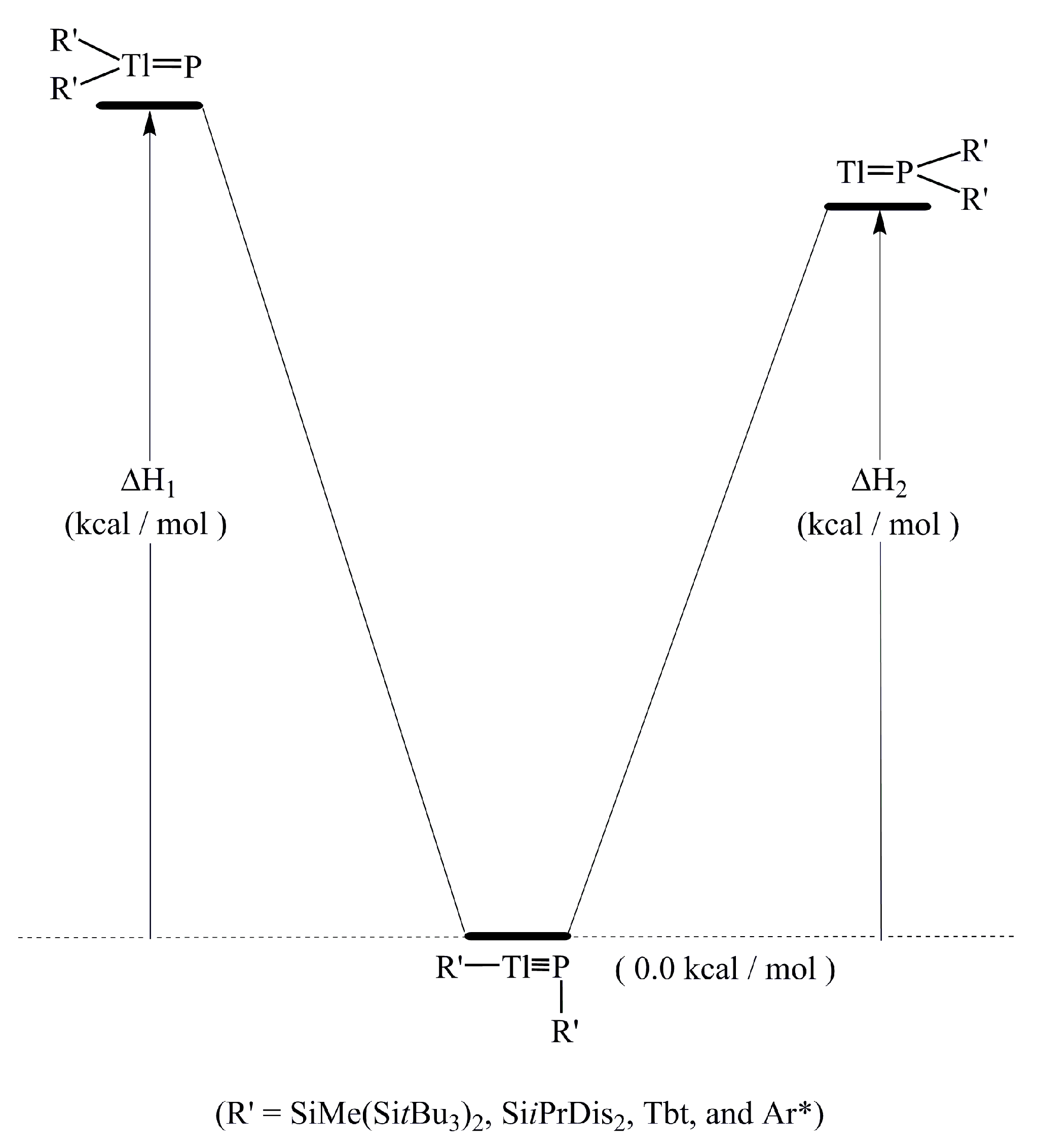
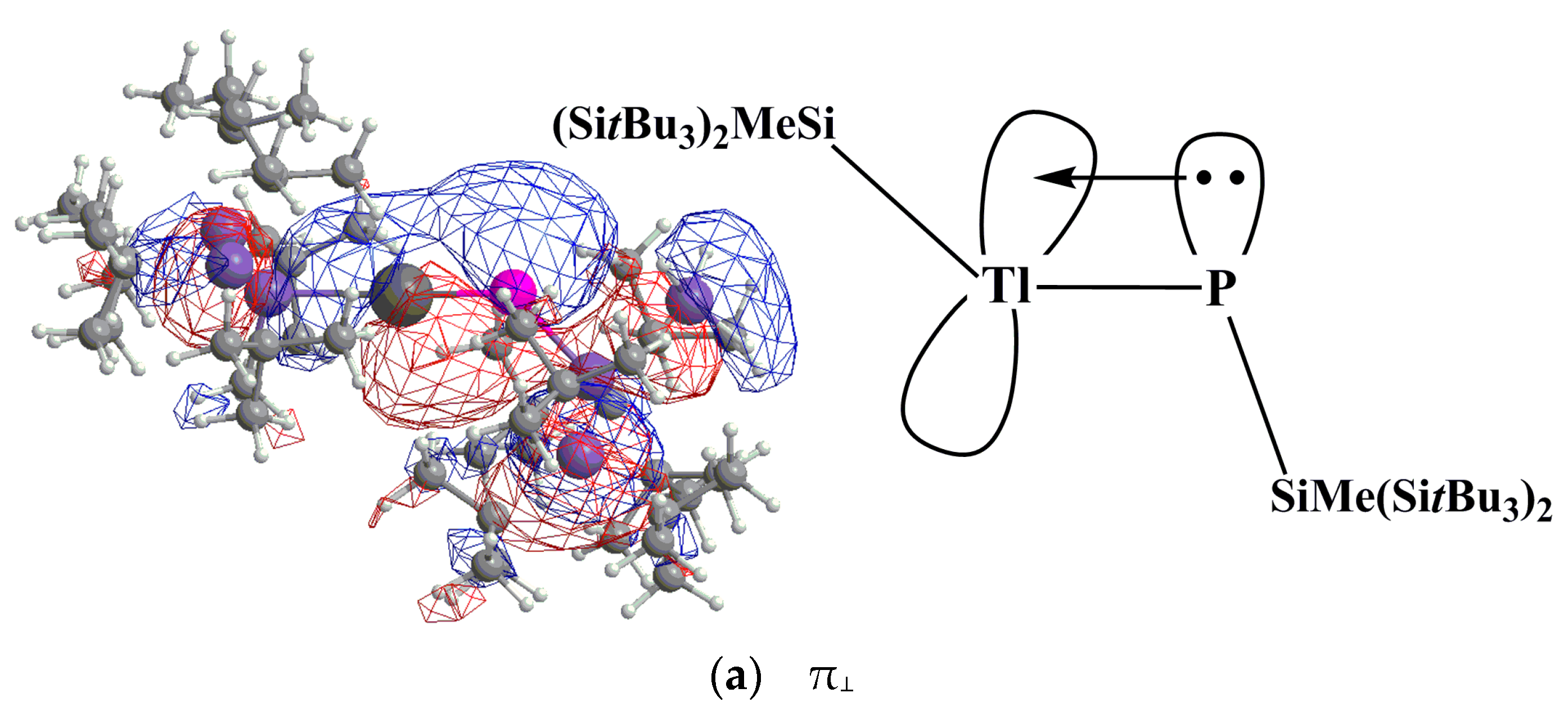
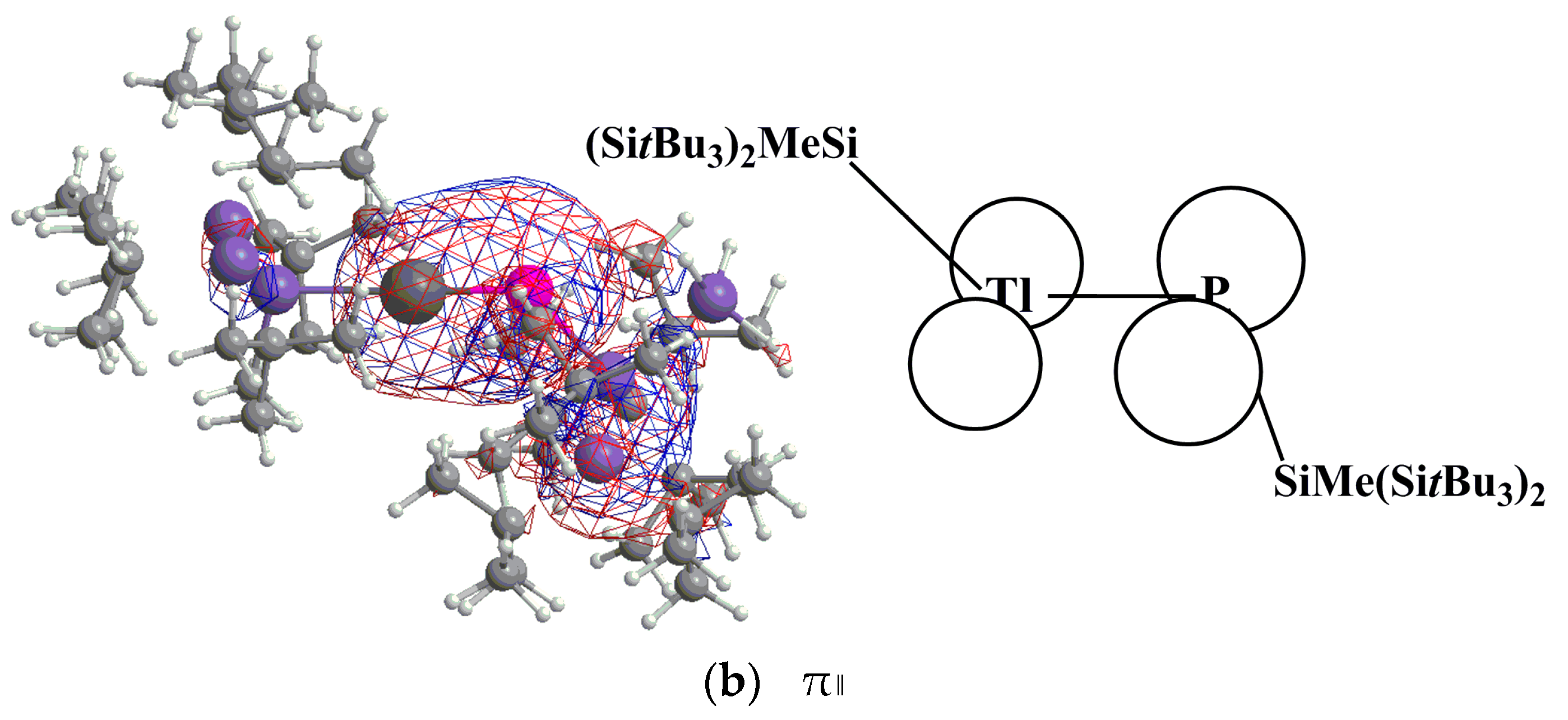
 P. In this model, the electrons that are donated from the lone pair of phosphorus have s character, as shown in Figure 1. Moreover, the size of 2p orbital of P is also much smaller than the 6p orbital of Tl. These two factors combined produce a weak Tl≡P triple bond in the R′Tl≡PR′ species. Supporting theoretical evidence in Table 2 shows that the WBI for R′Tl≡PR′ is 2.21, 2.37, 2.13, and 2.20 for R = SiMe(SitBu3)2, SiiPrDis2, Tbt, and Ar*, respectively. These WBI values are much smaller than the value for acetylene (2.99).
P. In this model, the electrons that are donated from the lone pair of phosphorus have s character, as shown in Figure 1. Moreover, the size of 2p orbital of P is also much smaller than the 6p orbital of Tl. These two factors combined produce a weak Tl≡P triple bond in the R′Tl≡PR′ species. Supporting theoretical evidence in Table 2 shows that the WBI for R′Tl≡PR′ is 2.21, 2.37, 2.13, and 2.20 for R = SiMe(SitBu3)2, SiiPrDis2, Tbt, and Ar*, respectively. These WBI values are much smaller than the value for acetylene (2.99). PR′.
PR′.5. Conclusions
 P, better interprets the triple bond in RTl≡PR species that feature small substituents. Model [II], whose bonding property is typified as Tl
P, better interprets the triple bond in RTl≡PR species that feature small substituents. Model [II], whose bonding property is typified as Tl  P, better describes the triple bond in R′Tl≡PR′ molecules that feature bulky ligands (Figure 6). However, regardless of whether the substituents in triply bonded RTl≡PR compound are large or small, their Tl≡P triple bonds are quite weak. Two effects can explain these phenomena. The different sizes of the p orbitals in the Tl and P elements mean that their overlapping populations are pretty small and the lone pair of the phosphorus atom has significant amount of s character, which results in poor overlaps between thallium and phosphorus. It is hoped that the results of experimental synthesis and structural characterization will confirm these predictions.
P, better describes the triple bond in R′Tl≡PR′ molecules that feature bulky ligands (Figure 6). However, regardless of whether the substituents in triply bonded RTl≡PR compound are large or small, their Tl≡P triple bonds are quite weak. Two effects can explain these phenomena. The different sizes of the p orbitals in the Tl and P elements mean that their overlapping populations are pretty small and the lone pair of the phosphorus atom has significant amount of s character, which results in poor overlaps between thallium and phosphorus. It is hoped that the results of experimental synthesis and structural characterization will confirm these predictions.Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bino, A.; Ardon, M.; Shirman, E. Formation of a carbon-carbon triple bond by coupling reactions in aqueous solution. Science 2005, 308, 234–235. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Wu, J.; Gu, J.; Wu, W.; Shaik, S.; Hiberty, P.C. Bonding conundrums in the C2 molecule: A valence bond study. J. Chem. Theory Comput. 2011, 7, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Ploshnik, E.; Danovich, D.; Hiberty, P.C.; Shaik, S. The nature of the idealized triple bonds between principal elements and the σ origins of trans-bent geometries—A valence bond study. J. Chem. Theory Comput. 2011, 7, 955–968. [Google Scholar] [CrossRef] [PubMed]
- Danovich, D.; Bino, A.; Shaik, S. Formation of carbon–carbon triply bonded molecules from two free carbyne radicals via a conical intersection. J. Phys. Chem. Lett. 2013, 4, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Power, P.P. π-Bonding and the lone pair effect in multiple bonds between heavier main group elements. Chem. Rev. 1999, 99, 3463–3504. [Google Scholar] [CrossRef] [PubMed]
- Jutzi, P. Stable systems with a triple bond to silicon or its homologues: Another challenge. Angew. Chem. Int. Ed. 2000, 39, 3797–3800. [Google Scholar] [CrossRef]
- Weidenbruch, M. Some recent advances in the chemistry of silicon and its homologues in low coordination states. J. Organomet. Chem. 2002, 646, 39–52. [Google Scholar] [CrossRef]
- Power, P.P. Silicon, germanium, tin and lead analogues of acetylenes. Chem. Commun. 2003, 2091–2101. [Google Scholar] [CrossRef]
- Weidenbruch, M. Molecules with a genuine Si-Si triple bond and a stable derivative of [SiH]+. Angew. Chem. Int. Ed. 2005, 44, 514–516. [Google Scholar] [CrossRef] [PubMed]
- Power, P.P. Synthesis and some reactivity studies of germanium, tin and lead analogues of alkynes. Appl. Organomet. Chem. 2005, 19, 488–493. [Google Scholar] [CrossRef]
- Lein, M.; Krapp, A.; Frenking, G. Why do the heavy-atom analogues of acetylene E2H2 (E = Si−Pb) exhibit unusual structures? J. Am. Chem. Soc. 2005, 127, 6290–6299. [Google Scholar] [CrossRef] [PubMed]
- Sekiguchi, A.; Ichinohe, M.; Kinjo, R. The chemistry of disilyne with a genuine Si–Si triple bond: Synthesis, structure, and reactivity. Bull. Chem. Soc. Jpn. 2006, 79, 825–828. [Google Scholar] [CrossRef]
- Power, P.P. Bonding and reactivity of heavier group 14 element alkyne analogues. Organometallics 2007, 26, 4362–4366. [Google Scholar] [CrossRef]
- Sekiguchi, A. Disilyne with a silicon-silicon triple bond: A new entry to multiple bond chemistry. Pure Appl. Chem. 2008, 80, 447–451. [Google Scholar] [CrossRef]
- Sekiguchi, A.; Kinjo, R.; Ichinohe, M. Interaction of π-bonds of the silicon–silicon triple bond with alkali metals: An isolable anion radical upon reduction of a disilyne. Synt. Met. 2009, 159, 773–780. [Google Scholar] [CrossRef]
- Fischer, R.C.; Power, P.P. π-Bonding and the lone pair effect in multiple bonds involving heavier main group elements: Developments in the new millennium. Chem. Rev. 2010, 110, 3877–3882. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Fischer, R.C.; Merrill, W.A.; Fischer, J.; Pu, L.; Ellis, B.D.; Fettinger, J.C.; Herber, R.H.; Power, P.P. Substituent effects in ditetrel alkyne analogues: Multiple vs. single bonded isomers. Chem. Sci. 2010, 1, 461–478. [Google Scholar] [CrossRef]
- Sasamori, T.; Han, J.S.; Hironaka, K.; Takagi, N.; Nagase, S.; Tokitoh, N. Synthesis and structure of stable 1,2-diaryldisilyne. Pure Appl. Chem. 2010, 82, 603–611. [Google Scholar] [CrossRef]
- Sekiguchi, A.; Kinjo, R.; Ichinohe, M. A stable compound containing a silicon–silicon triple bond. Science 2004, 305, 1755–1758. [Google Scholar] [CrossRef] [PubMed]
- Wiberg, N.; Vasisht, S.K.; Fischer, G.; Mayer, P.Z. Disilynes. III [1] a relatively stable disilyne RSi≡SiR (R = SiMe(SitBu3)2). Anorg. Allg. Chem. 2004, 630, 1823–1831. [Google Scholar] [CrossRef]
- Sasamori, T.; Hironaka, K.; Sugiyama, T.; Takagi, N.; Nagase, S.; Hosoi, Y.; Furukawa, Y.; Tokitoh, N. Synthesis and reactions of a stable 1,2-diaryl-1,2-dibromodisilene: A precursor for substituted disilenes and a 1,2-diaryldisilyne. J. Am. Chem. Soc. 2008, 130, 13856–13868. [Google Scholar] [CrossRef] [PubMed]
- Stender, M.; Phillips, A.D.; Wright, R.J.; Power, P.P. Synthesis and characterization of a digermanium analogue of an alkyne. Angew. Chem. Int. Ed. 2002, 41, 1785–1789. [Google Scholar] [CrossRef]
- Stender, M.; Phillips, A.D.; Power, P.P. Formation of [Ar*Ge{CH2C(Me)C(Me)CH2}CH2C(Me)=]2 (Ar* = C6H3-2,6-Trip2; Trip = C6H2-2,4,6-i-Pr3) via reaction of Ar*GeGeAr* with 2,3-dimethyl-1,3-butadiene: Evidence for the existence of a germanium analogue of an alkyne. Chem. Commun. 2002, 1312–1313. [Google Scholar] [CrossRef]
- Pu, L.; Phillips, A.D.; Richards, A.F.; Stender, M.; Simons, R.S.; Olmstead, M.M.; Power, P.P. Germanium and tin analogues of alkynes and their reduction products. J. Am. Chem. Soc. 2003, 125, 11626–11632. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, Y.; Sasamori, T.; Hosoi, Y.; Furukawa, Y.; Takagi, N.; Nagase, S.; Tokitoh, N. Synthesis and properties of a new kinetically stabilized digermyne: New insights for a germanium analogue of an alkyne. J. Am. Chem. Soc. 2006, 128, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Spikes, G.H.; Power, P.P. Lewis base induced tuning of the Ge–Ge bond order in a “digermyne”. Chem. Commun. 2007, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.D.; Wright, R.J.; Olmstead, M.M.; Power, P.P. Synthesis and characterization of 2,6-Dipp2-H3C6SnSnC6H3-2,6-Dipp2 (Dipp = C6H3-2,6-Pri2): A tin analogue of an alkyne. J. Am. Chem. Soc. 2002, 124, 5930–5931. [Google Scholar] [CrossRef] [PubMed]
- Pu, L.; Twamley, B.; Power, P.P. Synthesis and characterization of 2,6-Trip2H3C6PbPbC6H3-2,6-Trip2 (Trip = C6H2-2,4,6-i-Pr3): A stable heavier group 14 element analogue of an alkyne. J. Am. Chem. Soc. 2000, 122, 3524–3525. [Google Scholar] [CrossRef]
- Karni, M.; Apeloig, Y.; Schröder, D.; Zummack, W.; Rabezzana, R.; Schwarz, H. Nachweis von HCSiF und HCSiCl als die ersten Moleküle mit formalen C≡Si-Bindungen. Angew. Chem. Int. Ed. 1999, 38, 332–335. [Google Scholar]
- Danovich, D.; Ogliaro, F.; Karni, M.; Apeloig, Y.; Cooper, D.L.; Shaik, S. Silynes (RC identical with SiR′) and disilynes (RSi identical with SiR′): Why are less bonds worth energetically more? Angew. Chem. Int. Ed. 2001, 40, 4023–4027. [Google Scholar] [CrossRef]
- Gau, D.; Kato, T.; Saffon-Merceron, N.; Cozar, A.D.; Cossio, F.P.; Baceiredo, A. Synthesis and structure of a base-stabilized C-phosphino-Si-amino silyne. Angew. Chem. Int. Ed. 2010, 49, 6585–6589. [Google Scholar] [CrossRef] [PubMed]
- Lühmann, N.; Müller, T. A compound with a Si-C triple bond. Angew. Chem. Int. Ed. 2010, 49, 10042–10046. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.-Y.; Su, M.-D.; Chu, S.-Y. A stable species with a formal Ge≡C triple bond–a theoretical study. Chem. Phys. Lett. 2001, 341, 122–131. [Google Scholar] [CrossRef]
- Wu, P.-C.; Su, M.-D. The effect of substituents on the stability of triply bonded galliumuantimony molecules: A new target for synthesis. Dalton. Trans. 2011, 40, 4253–4260. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-C.; Su, M.-D. Triply bonded stannaacetylene (RC≡SnR): Theoretical designs and characterization. Inorg. Chem. 2011, 50, 6814–6822. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-C.; Su, M.-D. A new target for synthesis of triply bonded plumbacetylene (RC≡PbR): A theoretical design. Organometallics 2011, 30, 3293–3303. [Google Scholar] [CrossRef]
- Paetzold, P. Iminoboranes. Adv. Inorg. Chem. 1987, 31, 123–132. [Google Scholar]
- Paetzold, P. Boron Chemistry. In Proceedings of the 6th International Meeting on Born Chemistry, Bechyne, Slovakia, 22–26 June 1987; Hermanek, S., Ed.; World Scientific: Singapore, 1987; p. 446. [Google Scholar]
- Paetzold, P. New perspectives in boron-nitrogen chemistry-I. Pure Appl. Chem. 1991, 63, 345–363. [Google Scholar] [CrossRef]
- Paetzold, P. Boron-Nitrogen analogues of cyclobutadiene, benzene and cyclooctatetraene: Interconversions. Phosphorus Sulfur Silicon Relat. Elem. 1994, 93, 39–50. [Google Scholar] [CrossRef]
- Wright, R.J.; Phillips, A.D.; Allen, T.L.; Fink, W.H.; Power, P.P. Synthesis and characterization of the monomeric imides Ar′MNAr″ (M = Ga or In; Ar′ or Ar″ = terphenyl ligands) with two-coordinate gallium and indium. J. Am. Chem. Soc. 2003, 125, 1694–1696. [Google Scholar] [CrossRef] [PubMed]
- Dillon, K.B.; Mathey, F.; Nixon, J.F. Phosphorus: The Carbon Copy: From Organophosphorus to Phospha-Organic Chemistry; Wiley: New York, NY, USA, 1998; p. 2. [Google Scholar]
- Mosha, D.M.S. The monomeric pentacyanocobaltate (II) anion: Preparation and properties of thallium (I) pentacyanocobaltate (II). J. Chem. Educ. 1982, 59, 1057–1068. [Google Scholar] [CrossRef]
- Baldwin, R.A.; Wells, R.L.; White, P.S. Synthesis and characterization of an organothallium-phosphorus adduct: Crystal structure of (Me3SiCH2)3Tl·P(SiMe3)3. Main Group Chem. 1997, 2, 67–75. [Google Scholar] [CrossRef]
- Francis, M.D.; Jones, C.; Deacon, G.B.; Delbridge, E.E. Synthesis and structural characterization of a novel polyheterocyclopentadienyl thallium (I) complex. Organometallics 1998, 17, 3826–3832. [Google Scholar] [CrossRef]
- Fox, A.R.; Wright, R.J.; Rivard, E.; Power, P.P. Tl2[Aryl2P4]: A thallium complexed diaryltetraphosphabutadienediide and its two-electron oxidation to a diaryltetraphosphabicyclobutane, Aryl2P4. Angew. Chem. Int. Ed. 2005, 44, 7729–7733. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, version Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2013. [Google Scholar]
- Zhao, Y.; Truhlar, D.G. Density functionals with broad applicability in chemistry. Acc. Chem. Res. 2008, 41, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Becke, A.D. Density-Functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Becke, A.D. Density-Functional thermochemistry. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Perdew, J.P.; Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. 1992, B45, 13244–13249. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Dunning, T.H., Jr.; Hay, P.J. Modern Theoretical Chemistry; Schaefer, H.F., III, Ed.; Plenum: New York, NY, USA, 1976; pp. 1–28. [Google Scholar]
- Hay, P.J.; Wadt, W.R. Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J. Chem. Phys. 1985, 82, 270–283. [Google Scholar] [CrossRef]
- Hay, P.J.; Wadt, W.R. Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi. J. Chem. Phys. 1985, 82, 284–298. [Google Scholar]
- Hay, P.J.; Wadt, W.R. Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J. Chem. Phys. 1985, 82, 299–310. [Google Scholar] [CrossRef]
- Check, C.E.; Faust, T.O.; Bailey, J.M.; Wright, B.J.; Gilbert, T.M.; Sunderlin, L.S. Addition of polarization and diffuse functions to the LANL2DZ basis set for p-block elements. J. Phys. Chem. A 2001, 105, 8111–8116. [Google Scholar] [CrossRef]
- The Wiberg Bond Index, Which is Used to Screen Atom Pairs for the Possible Bonding in the Natural Bonding Orbital (NBO) Search, are Performed with the NBO Program (version NBO 6.0; University of Wisconsin System: Madison, WI, USA, 2013). Available online: http://www.chem.wisc.edu/~nbo5 (accessed on 4 July 2017).
- Wiberg, K.B. Application of the pople-santry-segal CNDO method to the cyclopropylcarbinyl and cyclobutyl cation and to bicyclobutane. Tetrahedron 1968, 24, 1083–1096. [Google Scholar] [CrossRef]
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 1998, 88, 899–993. [Google Scholar] [CrossRef]
- Emsley, J. The Elements, 2nd ed.; Clarendon Press: Oxford, UK, 1991; p. 285. [Google Scholar]
- Pyykko, P.; Desclaux, J.-P. Relativity and the periodic system of elements. Acc. Chem. Res. 1979, 12, 276–282. [Google Scholar] [CrossRef]
- Kutzelnigg, W. Chemical bonding in higher main group elements. Angew. Chem. Int. Ed. Engl. 1984, 23, 272–276. [Google Scholar] [CrossRef]
- Pyykko, P. Relativistic effects in structural chemistry. Chem. Rev. 1988, 88, 563–623. [Google Scholar] [CrossRef]
- Pyykko, P. Strong closed-shell interactions in inorganic chemistry. Chem. Rev. 1997, 97, 597–636. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Nagase, S. Silicon−Silicon triple bonds: Do substituents make disilynes synthetically accessible? Organometallics 1997, 16, 2489–2495. [Google Scholar] [CrossRef]
- Kobayashi, K.; Takagi, N.; Nagase, S. Do bulky aryl groups make stable silicon−silicon triple bonds synthetically accessible? Organometallics 2001, 20, 234–236. [Google Scholar] [CrossRef]
- Dapprich, S.; Frenking, G. Investigation of donor-acceptor interactions: A charge decomposition analysis using fragment molecular orbitals. J. Phys. Chem. 1995, 99, 9352–9367. [Google Scholar] [CrossRef]
- Glendening, E.D.; Weinhold, F. Natural resonance theory: I. General formalism. J. Comput. Chem. 1998, 19, 593–606. [Google Scholar] [CrossRef]
- Glendening, E.D.; Weinhold, F. Natural resonance theory: II. Natural bond order and valency. J. Comput. Chem. 1998, 19, 610–623. [Google Scholar] [CrossRef]
- Glendening, E.D.; Weinhold, F.; Badenhoop, J.K. Natural resonance theory. III. Chemical applications. J. Comput. Chem. 1998, 19, 628–632. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
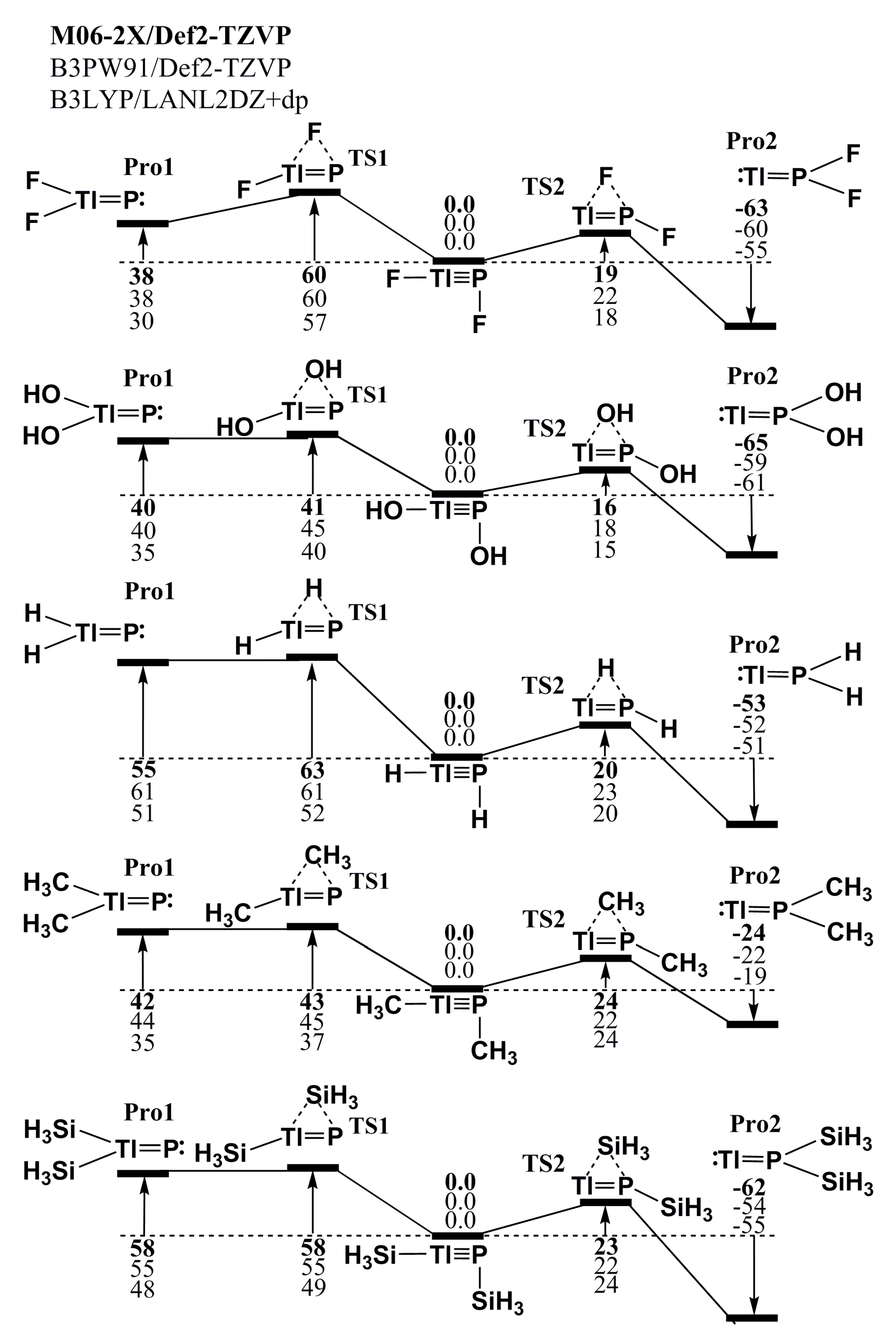
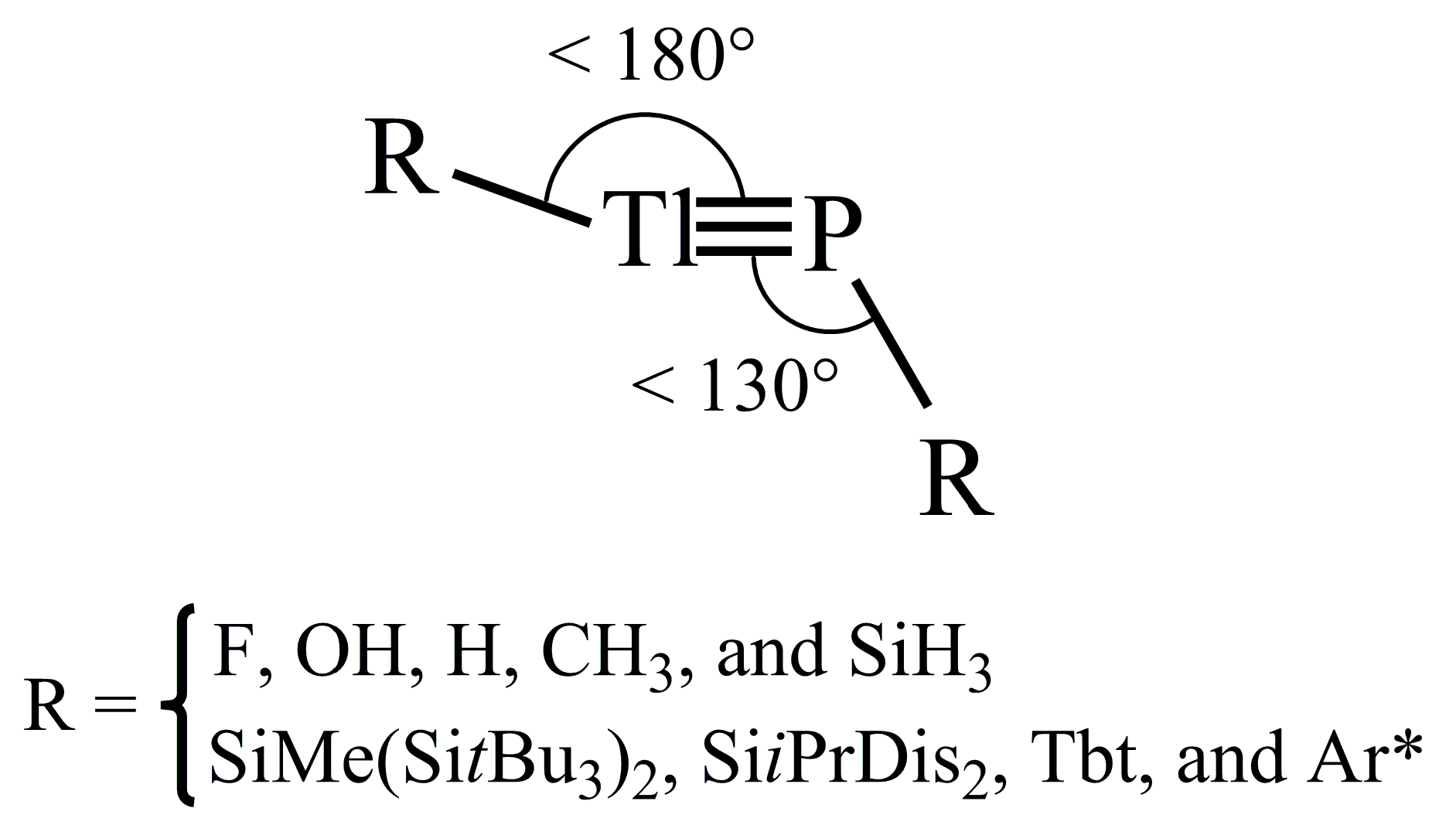
| R | F | OH | H | CH3 | SiH3 |
|---|---|---|---|---|---|
| Tl≡P (Å) | 2.422 | 2.437 | 2.320 | 2.339 | 2.313 |
| (2.425) | (2.443) | (2.327) | (2.349) | (2.336) | |
| [2.455] | [2.480] | [2.331] | [2.360] | [2.337] | |
| R-P-Tl (°) | 179.7 | 179.1 | 179.1 | 175.2 | 174.6 |
| (179.7) | (176.5) | (178.5) | (174.5) | (175.7) | |
| [178.5] | [177.9] | [178.2] | [171.3] | [179.1] | |
| P-Tl-R (°) | 94.63 | 98.92 | 86.51 | 100.4 | 94.76 |
| (96.59) | (101.5) | (86.82) | (102.2) | (92.71) | |
| [94.22] | [100.1] | [86.36] | [102.6] | [90.78] | |
| R-P-Tl-R (°) | 180.0 | 179.4 | 179.1 | 178.0 | 177.0 |
| (180.0) | (178.8) | (179.2) | (178.8) | (179.1) | |
| [180.0] | [179.2] | [179.8] | [179.9] | [179.4] | |
| QP (1) | 0.16 | 0.076 | −0.63 | −0.37 | −0.83 |
| (0.17) | (0.13) | (−0.60) | (−0.33) | (−0.72) | |
| [0.096] | [0.021] | [−0.62] | [−0.39] | [−0.76] | |
| QTl (2) | 1.19 | 1.14 | 1.12 | 1.07 | 0.82 |
| (1.11) | (1.03) | (0.87) | (0.99) | (0.75) | |
| [1.25] | [1.17] | [0.99] | [1.13] | [0.89] | |
| ΔEST for Tl⎼R (kcal/mol) (3) | 102.1 | 83.57 | 84.85 | 66.82 | 75.96 |
| (103.7) | (80.69) | (85.69) | (67.38) | (77.63) | |
| [102.2] | [83.15] | [83.05] | [67.94] | [74.40] | |
| ΔEST for P⎼R (kcal/mol) (4) | −28.91 | −17.53 | −30.75 | −26.43 | −15.84 |
| (−33.35) | (−21.29) | (−35.49) | (−30.26) | (−18.68) | |
| [−31.76] | [−20.24] | [−33.16] | [−29.21] | [−14.46] | |
| HOMO—LUMO (kcal/mol) | 184.1 | 167.6 | 210.6 | 151.2 | 142.1 |
| (131.6) | (118.1) | (212.0) | (149.3) | (145.1) | |
| [182.5] | [169.1] | [215.4] | [146.5] | [148.5] | |
| BE (kcal/mol) (5) | 95.58 | 83.57 | 84.85 | 66.82 | 75.96 |
| (95.74) | (82.10) | (85.69) | (67.38) | (77.63) | |
| [93.43] | [83.15] | [83.05] | [67.94] | [74.40] | |
| WBI (6) | 1.159 | 1.162 | 1.456 | 1.382 | 1.404 |
| (1.194) | (1.197) | (1.491) | (1.415) | (1.417) | |
| [1.191] | [1.178] | [1.475] | [1.403] | [1.372] |
| R′ | SiMe(SitBu3)2 | SiiPrDis2 | Tbt | Ar* |
|---|---|---|---|---|
| Tl≡P (Å) | 2.386 | 2.384 | 2.385 | 2.336 |
| ∠R′–Tl–P (°) | 166.9 | 166.4 | 168.9 | 161.2 |
| ∠Tl–P–R′ (°) | 122.3 | 113.7 | 116.2 | 115.6 |
| ∠R′–Tl–P–R′ (°) | 171.4 | 179.5 | 173.9 | 174.4 |
| QTl (1) | 0.975 | 0.739 | 1.166 | 1.218 |
| QP (2) | −0.860 | −0.826 | −0.344 | −0.257 |
| ΔEST for Tl—R′ (kcal/mol) (3) | 35.91 | 35.52 | 31.27 | 30.24 |
| ΔEST for P—R′ (kcal/mol) (4) | −43.10 | −37.47 | −39.74 | −40.52 |
| HOMO—LUMO (kcal/mol) | 71.27 | 27.21 | 58.05 | 39.34 |
| BE (kcal/mol) (5) | 80.24 | 85.43 | 62.51 | 67.89 |
| ΔH1 (kcal/mol) (6) | 91.34 | 90.49 | 89.22 | 87.11 |
| ΔH2 (kcal/mol) (6) | 73.98 | 72.83 | 71.27 | 74.01 |
| WBI (7) | 2.116 | 2.273 | 2.127 | 2.201 |
| Orbital | Occupancy | X | Y | X – Y | Q | |
|---|---|---|---|---|---|---|
| 218 | 2.000000 | 0.000757 | 0.000586 | 0.000171 | −0.002462 | |
| 219 | 2.000000 | 0.001036 | 0.000522 | 0.000513 | −0.004450 | |
| 220 | 2.000000 | 0.000932 | 0.000539 | 0.000394 | −0.006342 | |
| 221 | 2.000000 | 0.000026 | 0.004350 | −0.004325 | −0.002504 | |
| 222 | 2.000000 | 0.001151 | −0.000164 | 0.001315 | −0.001354 | |
| 223 | 2.000000 | 0.000081 | 0.003145 | −0.003064 | −0.001960 | |
| 224 | 2.000000 | 0.000037 | 0.002403 | −0.002366 | −0.000054 | |
| 225 | 2.000000 | 0.001777 | 0.029263 | −0.027486 | −0.030329 | |
| 226 | 2.000000 | 0.000477 | 0.013735 | −0.013259 | −0.007124 | |
| 227 | 2.000000 | 0.008445 | 0.068258 | −0.059813 | −0.018272 | |
| HOMO | 228 | 2.000000 | −0.005339 | 0.003033 | −0.008432 | −0.004437 |
| LUMO | 229 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |
| 230 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | |
| sum | 456.000000 | 0.028853 | 0.241774 | −0.212922 | −0.107250 |
| R′Tl≡PR′ | WBI | NBO Analysis | NRT Analysis | |||
|---|---|---|---|---|---|---|
| Occupancy | Hybridization | Polarization | Total/Covalent/Ionic | Resonance Weight | ||
| R′ = SiMe(SitBu3)2 | 2.11 | σ = 2.21 | σ : 0.5116 Tl (sp1.27) + 0.8592 P (sp2.07) | 26.18% (Tl) | 2.22/1.55/0.67 | Tl⎼P: 23.17% Tl=P: 66.87% Tl≡P: 9.94% |
| 73.82% (P) | ||||||
| π⊥ = 1.84 | π⊥: 0.3114 Tl (sp4.77) + 0.9503 P (sp1.42) | 9.70% (Tl) | ||||
| 90.30% (P) | ||||||
| π‖ = 1.92 | π‖: 0.6833 Tl (sp99.87) + 0.7556 P (sp99.99) | 5.69% (Tl) | ||||
| 94.31% (P) | ||||||
| R′ = SiiPrDis2 | 2.37 | σ = 1.83 | σ : 0.6422 Tl (sp0.86) + 0.7665 P (sp20.18) | 41.24% (Tl) | 2.59/0.83/1.76 | Tl⎼P: 17.35% Tl=P: 71.14% Tl≡P: 11.51% |
| 58.76% (P) | ||||||
| π⊥ = 1.92 | π⊥: 0.4064 Tl (sp99.99) + 0.9137 P (sp44.72) | 16.51% (Tl) | ||||
| 83.49% (P) | ||||||
| π‖ = 1.93 | π‖: 0.4551 Tl (sp99.99) + 0.8997 P (sp94.99) | 14.79% (Tl) | ||||
| 85.21% (P) | ||||||
| R′ = Tbt | 2.13 | σ = 1.77 | σ : 0.6888 Tl (sp0.94) + 0.7249 P (sp38.46) | 47.45% (Tl) | 2.08/1.59/0.49 | Tl⎼P: 27.42% Tl=P: 63.76% Tl≡P: 8.82% |
| 52.55% (P) | ||||||
| π⊥ = 1.94 | π⊥: 0.4133 Tl (sp35.51) + 0.9244 P (sp87.83) | 23.43% (Tl) | ||||
| 82.74% (P) | ||||||
| π‖ = 1.90 | π‖: 0.4118 Tl (sp99.89) + 0.9077 P (sp99.99) | 17.28% (Tl) | ||||
| 82.72% (P) | ||||||
| R′ = Ar* | 2.20 | σ = 1.96 | σ: 0.7362 Tl (sp0.04) + 0.6767 P (sp64.96) | 54.20% (Tl) | 2.17/1.66/0.51 | Tl⎼P: 19.82% Tl=P: 71.69% Tl≡P: 8.49% |
| 45.80% (P) | ||||||
| π⊥ = 1.77 | π⊥: 0.3177 Tl (sp99.99) + 0.9482 P (sp99.99) | 10.09% (Tl) | ||||
| 89.91% (P) | ||||||
| π‖ = 1.92 | π‖: 0.4083 Tl (sp99.99) + 0.9128 P (sp99.99) | 16.67% (Tl) | ||||
| 83.33% (P) | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, J.-S.; Yang, M.-C.; Su, M.-D. Substituent Effects on the Stability of Thallium and Phosphorus Triple Bonds: A Density Functional Study. Molecules 2017, 22, 1111. https://doi.org/10.3390/molecules22071111
Lu J-S, Yang M-C, Su M-D. Substituent Effects on the Stability of Thallium and Phosphorus Triple Bonds: A Density Functional Study. Molecules. 2017; 22(7):1111. https://doi.org/10.3390/molecules22071111
Chicago/Turabian StyleLu, Jia-Syun, Ming-Chung Yang, and Ming-Der Su. 2017. "Substituent Effects on the Stability of Thallium and Phosphorus Triple Bonds: A Density Functional Study" Molecules 22, no. 7: 1111. https://doi.org/10.3390/molecules22071111