Two New β-Dihydroagarofuran Sesquiterpenes from Celastrus orbiculatus Thunb and Their Anti-Proliferative Activity
Abstract
:1. Introduction
2. Results and Discussion
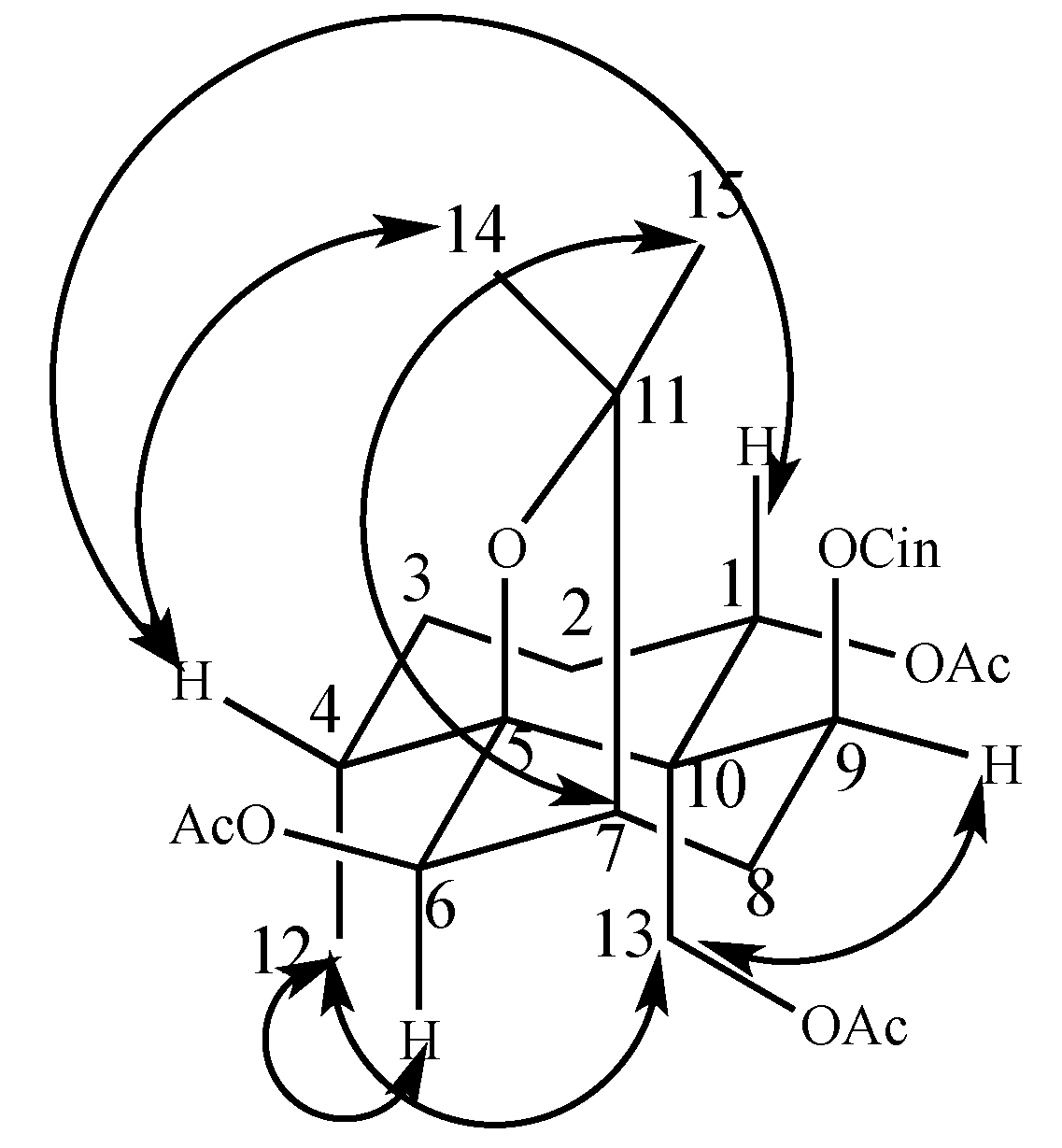
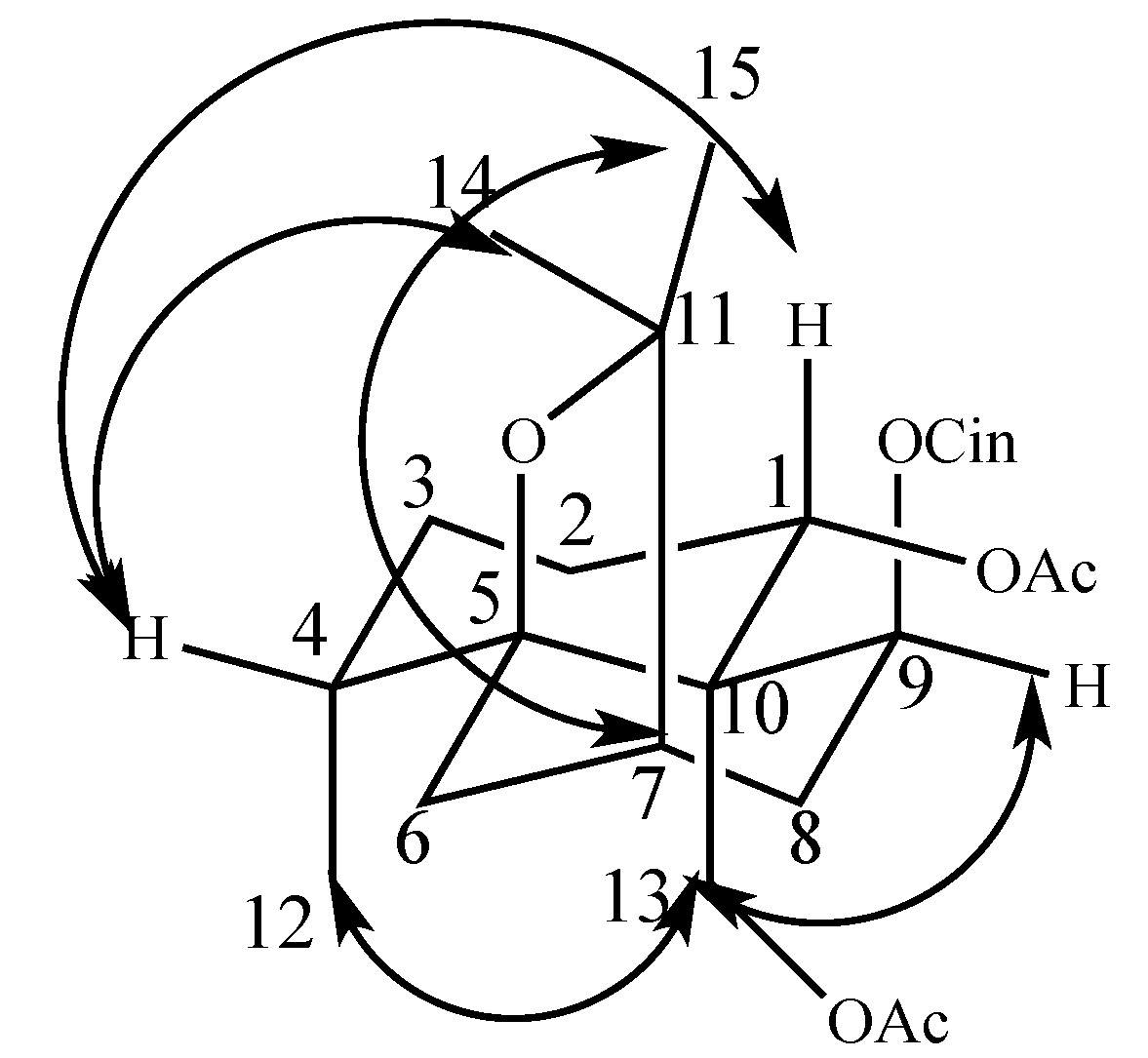
2.1. Two New Identified Compounds (1–2)
2.2. Anti-Proliferative Activity of Compounds 1–19 on HL-60, K562, and HCT116 Cell Lines
3. Experimental
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Seventeen Known Compounds
3.5. Cell Culture
3.6. In Vitro Anti-Proliferative Bioassay
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Guo, Y.Q. Research on the Chemical Constituents and Pharmacological Activities of Celastrus orbiculatus Thunb. Ph.D. Thesis, Shenyang Pharmaceutical University, Shenyang, Liaoning, China, 2004. [Google Scholar]
- State Adiministration of Traditional Chinese Medicine. Editorial Commission of Traditional Chinese Medicine; Traditional Chinese Medicine Shanghai/Shanghai Science & Technology Press: Shanghai, China, 1999; Volume 5, p. 166. [Google Scholar]
- Carroll, A.R.; Davis, R.A.; Addepalli, R.; Fechner, G.A.; Guymer, G.P.; Forster, P.I.; Quinn, R. Cytotoxic agarofurans from the seeds of the Australian rainforest vine Celastrus subspicata. J. Phytochem. Lett. 2009, 72, 163–165. [Google Scholar] [CrossRef]
- Wu, W.J.; Wang, M.G.; Zhu, J.B.; Zhou, W.M.; Hu, Z.N.; Ji, Z.Q. Five new insecticidal sesquiterpenoids from Celastrus angulatus. J. Nat. Prod. 2001, 64, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Takaishi, Y.; Ujita, K.; Tokuda, H.; Nishino, H.; Iwashima, A.; Fujita, T. Errata. Cancer Lett. 1992, 67, 215. [Google Scholar]
- Horiuch, M.; Murakami, C.; Fukamiya, N.; Yu, D.; Chen, T.H.; Bastow, K.F.; Zhang, D.C.; Takaishi, Y.; Imakura, Y.; Lee, K.H. Tripfordines A-C, Sesquiterpene Pyridine Alkaloids from Tripterygium wilfordii, and Structure Anti-HIV activity relationships of tripterygium alkaloids. J. Nat. Prod. 2006, 69, 1271–1274. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.Z.; Hwang, B.Y.; Kim, H.S.; Lee, J.H.; Kim, Y.H.; Lee, J.J. Anti-inflammatory constituents of Celastrus orbiculatus inhibit the NF-γB activation and No production. J. Nat. Prod. 2002, 65, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Nicolas, F.; Oberti, J.C.; Ravelo, A.G.; Estevez-Braun, A. β-Agarofurans and sesquiterpene pyridine alkaloids from maytenus spinosa. J. Nat. Prod. 2014, 77, 1853–1863. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, M.L.; Cortes-Selva, F.; Perez-Victoria, J.M.; Jimenez, I.A.; Gonzalez, A.G.; Munoz, O.M.; Gamarro, F.; Castanys, S.; Ravelo, A.G. Chemosensitization of a multidrug-resistant leishmania tropica line by new sesquiterpene from maytenus magellanica and maytenus chubutensis. J. Med. Chem. 2001, 44, 4668–4676. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, I.A.; Bazzocchi, I.L.; Nunez, M.J.; Mukainaka, T.; Tokuda, H.; Nishino, H.; Konoshima, T.; Ravelo, A.G. Absolute configuration of sesquiterpenes from Crossopetalum tonduzii and their inhibitory effects on Epstein-Barr virus early antigen activation in Raji cells. J. Nat. Prod. 2003, 66, 1047–1050. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, C.R.; Jimenez, I.A.; Tokuda, H.; Kushida, H.; Bazzocchi, I.L. Antitumor-promoting effects of new sesquiterpenes from Crossopetalum tonduzii. Chem. Biodivers. 2005, 2, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.G.; Tincusi, B.M.; Bazzocchi, I.L.; Tokuda, H.; Nishino, H.; Konoshima, T.; Jiménez, I.A.; Ravelo, A.G. Anti-tumor rromoting effects of sesquiterpenes from Maytenus cuzcoina (Celastraceae). Bioorg. Med. Chem. 2000, 8, 1773–1778. [Google Scholar] [CrossRef]
- Petrestelo, N.R.; Jimenez, I.A.; Tokuda, H.; Hayashi, H.; Bazzocchi, I.L. Sesquiterpenes from Maytenus jelskii as potential cancer chemopreventive agents. J. Nat. Prod. 2010, 73, 127–132. [Google Scholar]
- Núñez, M.J.; Jiménez, I.A.; Mendoza, C.R.; Chavez-Sifontes, M.; Martinez, M.L.; Ichiishi, E.; Tokuda, R.; Tokuda, H.; Bazzocchi, I.L. Dihydro-β-agarofuran sesquiterpenoids from celastraceae species as anti-tumor-promoting agents: Structure-activity relationship. Eur. J. Med. Chem. 2016, 111, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.Q.; Huang, G.S.; Ma, Y.X.; Wu, X.L.; Song, Q.B. Alkaloids from Celastrus angulatus. J. Nat. Prod. 1992, 55, 1320–1322. [Google Scholar] [CrossRef]
- Tu, Y.Q.; Hu, Y.J. Structure of sesquiterpenoids from Celastrus angulatus. J. Nat. Prod. 1993, 56, 126–129. [Google Scholar] [CrossRef]
- Wibowo, M.; Levrier, C.; Sadowski, M.C.; Nelson, C.C.; Wang, Q.; Holst, J.; healy, P.C.; Hofmann, A.; Davis, R.A. Bioactive dihydro-β-agarofuran sesquiterpenoids from the Australian rainforest plant Maytenus bilocularis. J. Nat. Prod. 2016, 79, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Ning, R.; Lei, Y.; Liu, S.; Wang, H.; Zhang, R.; Wang, W.; Zhu, Y.; Zhang, H.; Zhao, W. Natural β-dihydroagarofuran-Type Sesquiterpenoids as Cognition-Enhancing and Neuroprotective Agents from Medicinal Plants of the Genus Celastrus. J. Nat. Prod. 2015, 78, 2175–2186. [Google Scholar] [CrossRef] [PubMed]
- Takaishi, Y.; Ohshima, S.; Nakano, K.; Tomimatsu, T.; Tokuda, H.; Nishino, H.; Iwashima, A. Structures of Sesquiterpene Polyol Esters from Celastrus stephanotiifolius with Potential Tumor-Promotion Inhibitory Activity. J. Nat. Prod. 1993, 56, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Yoshihisa, T.; Shouji, T.; Kimiko, N.; Koutarou, M.; Toshiaki, T. Structures of sesquiterpene polyol esters from Tripterygium wilfordii var. Regelii. Phytochemistry 1991, 30, 3027–3031. [Google Scholar]
- Borrelli, F.; Borbone, N.; Capasso, R.; Montesano, D.; Angelo, A.I.; Simona, D. M.; Francesco, C.; Lydia, F.; Rocco, L.; Franco, Z. New Sesquiterpenes with Intestinal Relaxant Effect from Celastrus paniculatus. Planta Med. 2004, 70, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Mingan, W.; Fuheng, C. The study the insects antifeeding constituents of Celastrus orbiculatus. Chem. J. Chin. Univ. 1995, 16, 1248–1250. [Google Scholar]
- Zhu, Y.; Miao, Z.J.; Zhao, W. Cytotoxic Dihydroagarofuranoid Sesquiterpenes from the Seeds of Celastrus orbiculatus. J. Nat. Prod. 2008, 71, 1005–1010. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.Q.; Li, X.; Xu, J.; Meng, D.; Wang, J. Sesquiterpene Esters from the Fruits Celastrus orbiculatus. Chem. Lett. 2005, 34, 764–765. [Google Scholar] [CrossRef]
- Liu, J.K.; Becker, H.; Zapp, J.; Wu, D. Four sesquiterpenes from the insecticidal plant Celastrus angulatus. Phytochemistry 1995, 40, 841–846. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, R.; Lan, J.; Ning, R.; Wu, D.; Chen, D.; &Zhao, W. β-Dihydroagarofuran-Type Sesquiterpenes from the Seeds of Celastrus monospermus and Their Lifespan-Extending Effects on the Nematode Caenorhabditis elegans. J. Nat. Prod. 2016, 79, 3039–3046. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Chen, Y.; Wu, D.; Zhang, X.; Hao, X. Sesquiterpenoids from Celastrus paniculatus. J. Nat. Prod. 1993, 56, 122–125. [Google Scholar] [CrossRef]
- Rubinstein, L.V.; Shoemaker, R.H.; Paull, K.D.; Simon, R.M.; Tosini, S.; Skehan, P.; Scudiero, D.A.; Monks, A.; Boyd, M.R. Comparison of in vitro anticancer-drug-screening data generated with a tetrazolium assay versus a protein assay against a diverse panel of human tumor cell lines. J. Natl. Cancer Inst. 1990, 82, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
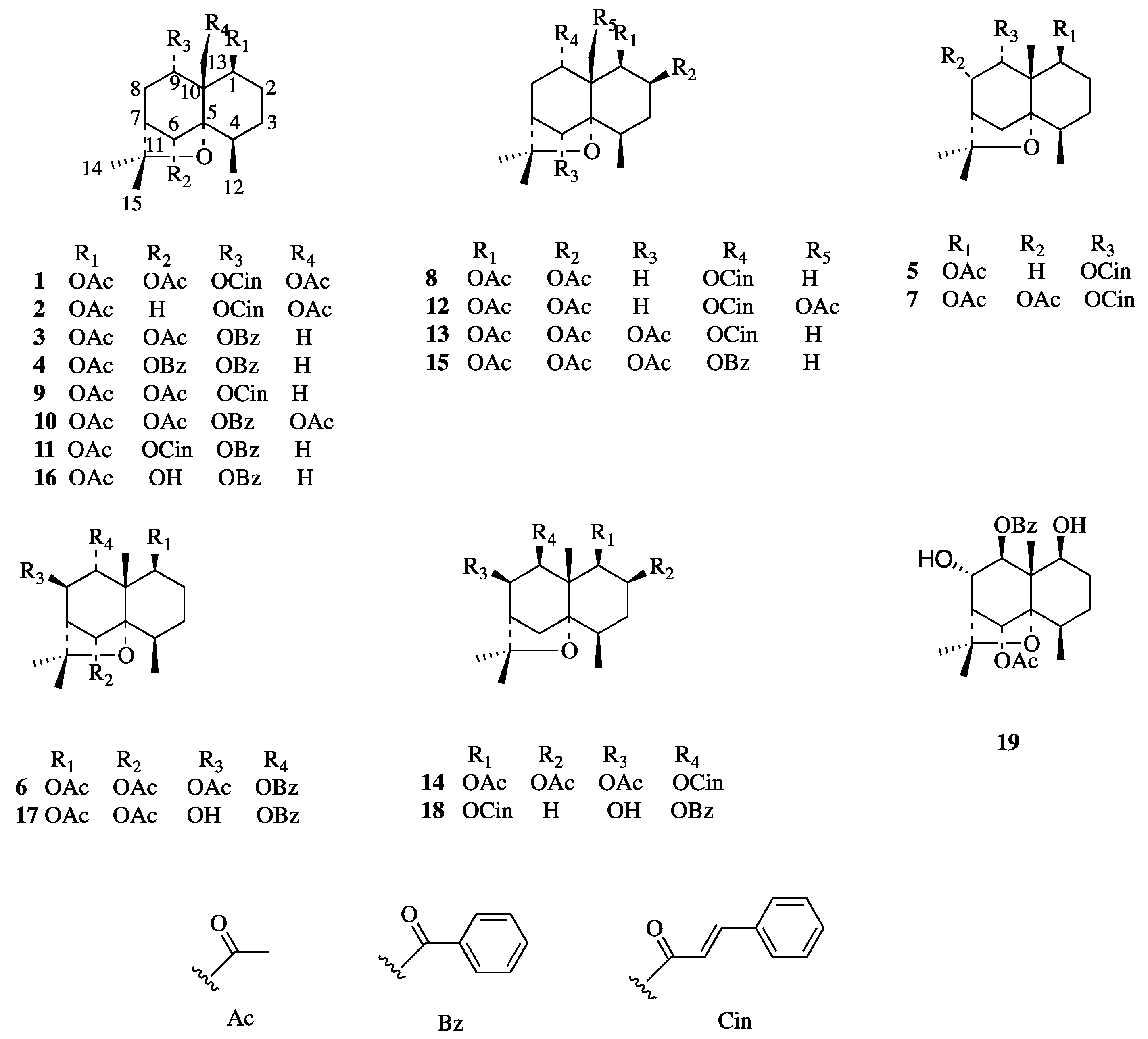


| Position | 1 | 2 |
|---|---|---|
| 1 | 5.58 (1H, dd, J = 12.4 Hz, 4.5 Hz) | 5.53 (1H, dd, J = 12.4 Hz, 4.3 Hz) |
| 2 | 1.86 (1H, m), 1.55 (1H, m) | 1.85 (1H, m), 1.55 (1H, m) |
| 3 | 2.25 (1H, m), 1.46 (1H, m) | 2.26 (1H, m), 1.45 (1H, m) |
| 4 | 2.29 (1H, m) | 1.86 (1H, m) |
| 5 | ||
| 6 | 5.92 (1H, s) | 2.21 (1H, m), 2.01 (1H, m) |
| 7 | 2.19 (1H, m) | 2.04 (1H, m) |
| 8 | 2.43 (1H, m), 2.15 (1H, m) | 2.19 (1H, m), 2.07 (1H, m) |
| 9 | 5.16 (1H, d, J = 7.3 Hz) | 5.20 (1H, d, J = 6.8 Hz) |
| 10 | ||
| 11 | ||
| 12 | 0.97 (3H, d, J = 7.4 Hz) | 1.08 (3H, d, J = 7.8 Hz) |
| 13 | 4.66 (1H, d, J = 12.2 Hz) 4.41 (1H, d, J = 12.3 Hz) | 4.52 (1H, d, J = 12.2 Hz) 4.47 (1H, d, J = 12.1 Hz) |
| 14 | 1.43 (3H, s) | 1.39 (3H, s) |
| 15 | 1.39 (3H, s) | 1.19 (3H, s) |
| Position | 1 | 2 |
|---|---|---|
| 1 | 73.1 | 73.4 |
| 2 | 22.5 | 22.8 |
| 3 | 26.5 | 26.8 |
| 4 | 33.5 | 40.1 |
| 5 | 89.7 | 87.0 |
| 6 | 78.2 | 36.7 |
| 7 | 48.8 | 43.6 |
| 8 | 34.7 | 33.9 |
| 9 | 69.8 | 69.9 |
| 10 | 53.0 | 50.6 |
| 11 | 82.5 | 82.0 |
| 12 | 16.6 | 17.4 |
| 13 | 65.5 | 65.0 |
| 14 | 25.9 | 24.4 |
| 15 | 30.5 | 30.3 |
| Compounds | IC50 (μΜ) | ||
|---|---|---|---|
| HL-60 | K562 | HCT-116 | |
| 1 | - | - | - |
| 2 | 23.11 | 35.00 | 50.64 |
| 3 | 26.34 | 38.75 | 46.61 |
| 4 | 3.61 | 17.13 | 10.15 |
| 5 | 33.29 | 42.85 | 34.25 |
| 6 | 19.32 | 37.27 | 37.46 |
| 7 | 30.85 | 30.20 | - |
| 8 | - | - | - |
| 9 | - | - | 36.03 |
| 10 | 20.32 | 35.84 | 41.64 |
| 11 | - | - | - |
| 12 | - | - | - |
| 13 | 40.50 | 42.67 | - |
| 14 | - | - | 30.98 |
| 15 | 31.51 | 40.86 | 50.01 |
| 16 | 33.51 | 42.56 | - |
| 17 | 18.87 | 35.22 | - |
| 18 | - | - | - |
| 19 | 23.55 | 47.31 | 31.05 |
| 5-FU | 11.06 | 27.55 | 29.13 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, J.; Han, N.; Lv, G.; Jia, L.; Liu, Z.; Yin, J. Two New β-Dihydroagarofuran Sesquiterpenes from Celastrus orbiculatus Thunb and Their Anti-Proliferative Activity. Molecules 2017, 22, 948. https://doi.org/10.3390/molecules22060948
Zhou J, Han N, Lv G, Jia L, Liu Z, Yin J. Two New β-Dihydroagarofuran Sesquiterpenes from Celastrus orbiculatus Thunb and Their Anti-Proliferative Activity. Molecules. 2017; 22(6):948. https://doi.org/10.3390/molecules22060948
Chicago/Turabian StyleZhou, Jingjing, Na Han, Guanghui Lv, Lina Jia, Zhihui Liu, and Jun Yin. 2017. "Two New β-Dihydroagarofuran Sesquiterpenes from Celastrus orbiculatus Thunb and Their Anti-Proliferative Activity" Molecules 22, no. 6: 948. https://doi.org/10.3390/molecules22060948





