Screening and Identification of the Metabolites in Rat Plasma and Urine after Oral Administration of Areca catechu L. Nut Extract by Ultra-High-Pressure Liquid Chromatography Coupled with Linear Ion Trap–Orbitrap Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of the Major Components in ACNE by UHPLC-LTQ-Orbitrap
2.1.1. Alkaloids
2.1.2. Tannins
2.1.3. Amino Acid
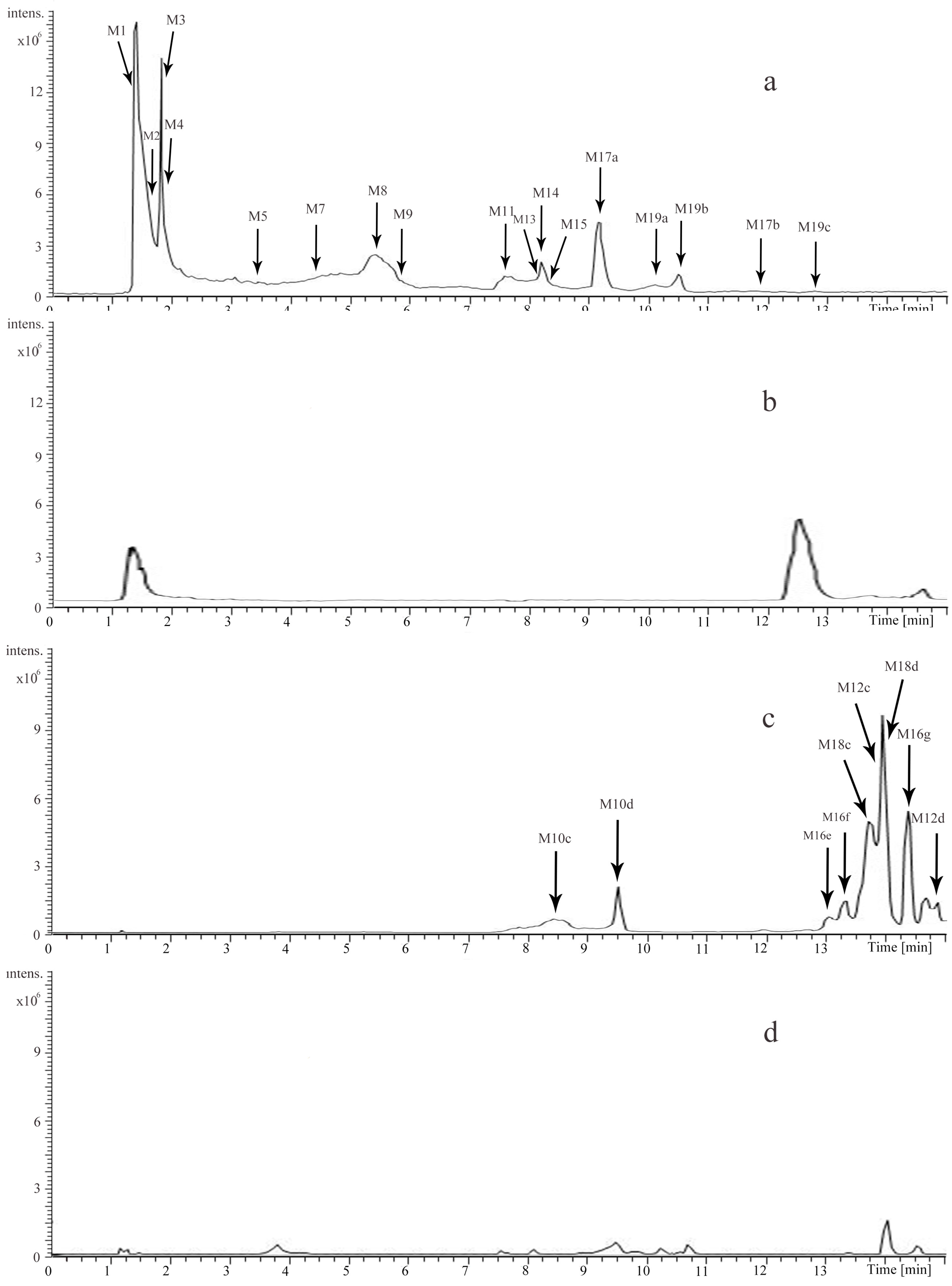
2.2. Identification of the Major Metabolites in ACNE by UHPLC-LTQ-Qrbitrap
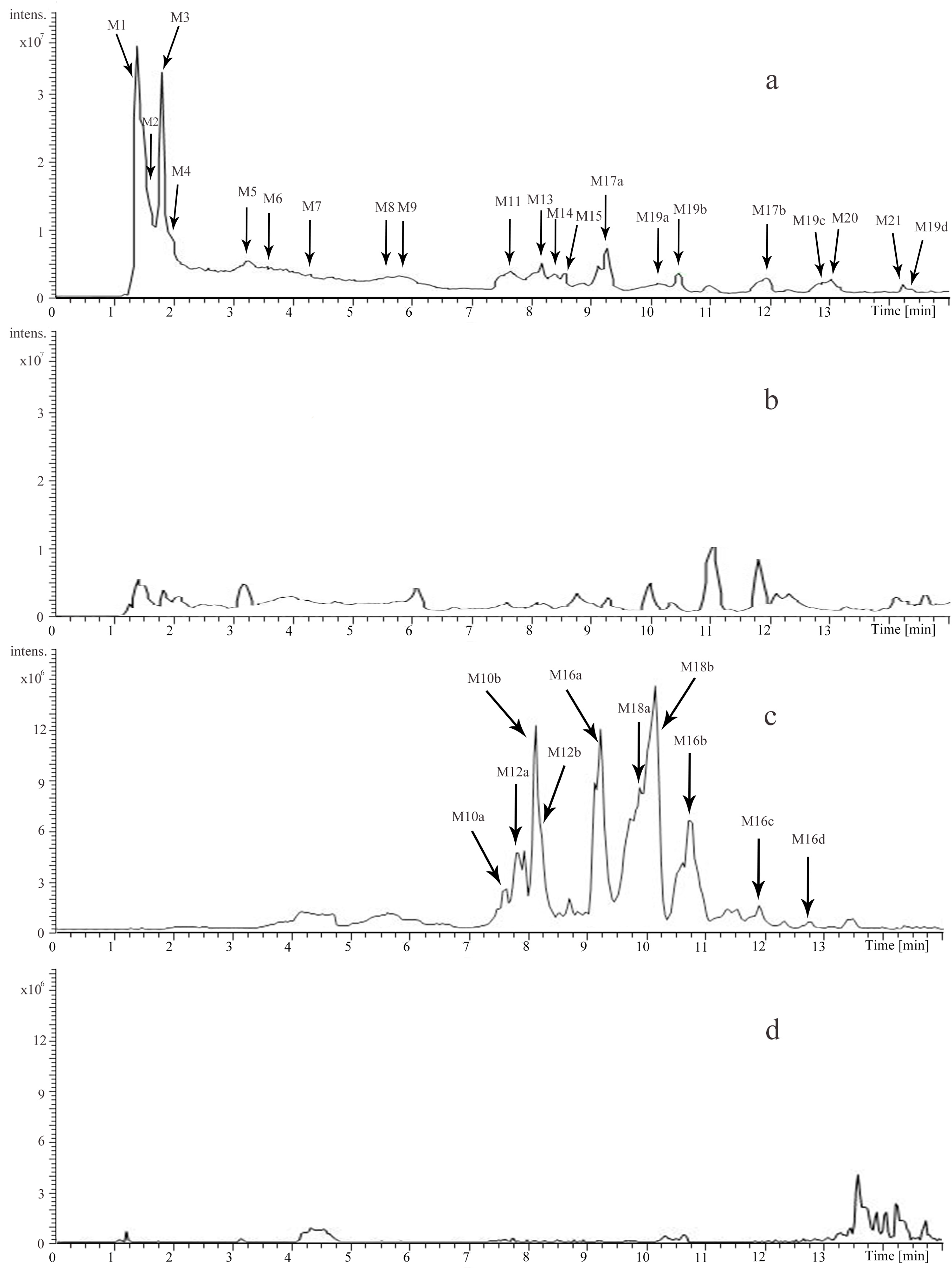
2.2.1. Prototypes (M1, M2, M3, M5, M6, M15, M17)
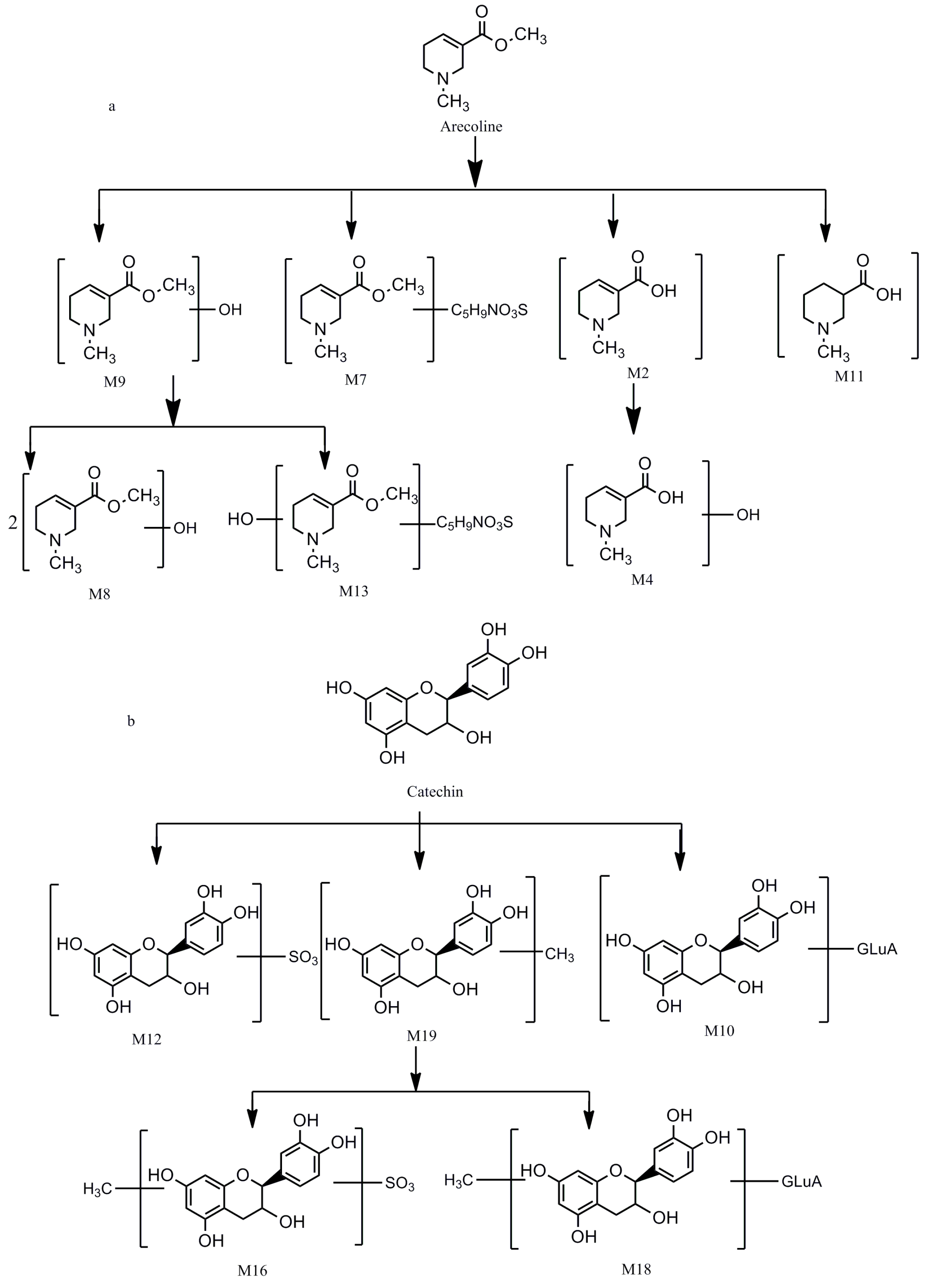
2.2.2. Methylated Metabolites (M19, M21)
2.2.3. Methylated and Sulfated Metabolites (M16)
2.2.4. Methylated and Glucuronidated Metabolites (M18)
2.2.5. Sulfated Metabolites (M12)
2.2.6. Glucuronidated Metabolites (M10)
2.2.7. Oxidative Metabolites (M4, M9)
2.2.8. Mercapturic Acid-Conjugated Metabolites (M7)
2.2.9. Oxidative and Mercapturic Acid Conjugated Metabolites (M13)
2.2.10. Dimer (M8)
2.2.11. Other Metabolites (M11, M14, M20)
3. Materials and Methods
3.1. Chemicals and Materials
3.2. Preparation of the Areca catechu L. Nut Extract
3.3. Animal and Sample Collection
3.4. Sample Preparation
3.5. Instrumentation and Analytical Conditions
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, P.; Lv, H.T.; Zhang, A.H.; Sun, H.; Yan, G.L.; Han, Y.; Wu, X.H.; Wang, X.J. Improved ultra-performance liquid chromatography with electrospray ionization quadrupole-time-of-flight high-definition mass spectrometry method for the rapid analysis of the chemical constituents of a typical medical formula: Liuwei Dihuang Wan. J. Sep. Sci. 2013, 36, 3511–3516. [Google Scholar] [CrossRef] [PubMed]
- Zhan, C.S.; Xiong, A.Z.; Shen, D.P.; Li, Y.; Wang, Z.T. Characterization of the Principal Constituents of Danning Tablets, a Chinese Formula Consisting of Seven Herbs, by an UPLC-DAD-MS/MS Approach. Molecules 2016, 21, 631. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.M.; Zhang, Q.Y.; Feng, F. Chemical profiling approach to evaluate the influence of traditional and simplified decoction methods on the holistic quality of Da-Huang-Xiao-Shi decoction using high-performance liquid chromatography coupled with diode-array detection and time-of-flight mass spectrometry. J. Sep. Sci. 2016, 39, 1442–1453. [Google Scholar] [PubMed]
- Yan, G.L.; Zou, D.; Zhang, A.H.; Tan, Y.L.; Sun, H.; Wang, X.J. UPLC-Q-TOF-MS/MS fingerprinting for rapid identification of chemical constituents of Ermiao Wan. Anal. Methods 2014, 7, 846–862. [Google Scholar] [CrossRef]
- Luo, K.W.; Shi, Q.S.; Feng, F. Characterization of global metabolic profile of Zhi-Zi-Hou-Po decoction in rat bile, urine and feces after oral administration based on a strategy combining LC-MS and chemometrics. J. Chromatogr. B 2016, 1040, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Xu, F.; Zhang, Y.Z.; Huang, S.; Zang, X.Y.; Zhao, X.; Zhang, L.; Shang, M.Y.; Yang, D.H.; Wang, X. The profiling and identification of the absorbed constituents and metabolites of Paeoniae Radix Rubra decoction in rat plasma and urine by the HPLC-DAD-ESI-IT-TOF-MS(n) technique: A novel strategy for the systematic screening and identification of absorbed. J. Pharm. Biomed. Anal. 2013, 83, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wu, C.S.; Zhang, J.L.; Li, Y.F. A new strategy for the discovery of epimedium metabolites using high-performance liquid chromatography with high resolution mass spectrometry. Anal. Chim. Acta 2013, 768, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.C.; Liu, R.; Yang, P.; Liang, J.Y. Rapid identification of efavirenz metabolites in rats and humans by ultra high performance liquid chromatography combined with quadrupole time-of-flight tandem mass spectrometry. J. Sep. Sci. 2015, 38, 1529–1536. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Lv, Z.H.; Li, G.; Xu, X.L.; Zhang, C.H.; Cao, P.; Huang, J.G.; Si, L.Q. Study of the in vitro metabolism of TJ0711 using ultra high performance liquid chromatography with quadrupole time-of-flight and ultra fast liquid chromatography with quadrupole linear ion trap mass spectrometry. J. Sep. Sci. 2015, 38, 1837–1849. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Zeng, W.L.; He, C.Y.; Bligh, S.; Liu, Q.; Yang, L.; Wang, Z.T. Characterization of metabolites of sweroside in rat urine using ultra-high-performance liquid chromatography combined with electrospray ionization quadrupole time-of-flight tandem mass spectrometry and NMR spectroscopy. J. Mass Spectrom. 2014, 49, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.H.; Guo, C.N.; Chen, Z.L.; Liu, Y.H.; He, L.M.; Zeng, Z.L.; Yan, C.Q.; Pan, G.F.; Li, S.P. Metabolism of nitazoxanide in rats, pigs, and chickens: Application of liquid chromatography coupled to hybrid linear ion trap/Orbitrap mass spectrometer. J. Chromatogr. B 2015, 1000, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.Q.; Ma, X.Y.; Liu, Y.; Lu, L.N.; Yang, R.R.; Yu, G.H.; Sun, M.H.; Xin, S.K.; Tian, S.M.; Chen, X.J. An Approach to Characterizing the Complicated Sequential Metabolism of Salidroside in Rats. Molecules 2016, 21, 706. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Liu, Y.J.; Wu, N.; Sun, T.; He, X.Y.; Gao, Y.X.; Wu, C.J. Areca catechu L. (Arecaceae): A review of its traditional uses, botany, phytochemistry, pharmacology and toxicology. J. Ethnopharmacol. 2015, 164, 340–356. [Google Scholar] [CrossRef] [PubMed]
- Azeez, S.; Amudhan, S.; Adiga, S.; Rao, N.; Udupa, L.A. Wound healing profile of Areca catechu extracts on different wound models in wistar rats. Kuwait Med. J. 2007, 39, 48–52. [Google Scholar]
- Bhandare, A.M.; Vyawahare, N.S.; Kshirsagar, A.D. Anti-migraine effect of Areca Catechu L. nut extract in bradykinin-induced plasma protein extravasation and vocalization in rats. J. Ethnopharmacol. 2015, 171, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Dar, A.; Khatoon, S.; Rahman, G.; Atta, U.R. Anti-depressant activities of Areca catechu fruit extract. Phytomed. Int. J. Phytother. Phytopharmacol. 1997, 4, 41–45. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Maiti, K.; Mukherjee, K.; Houghton, P.J. Leads from Indian medicinal plants with hypoglycemic potentials. J. Ethnopharmacol. 2006, 106, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Hamsar, M.N.; Ismail, S.; Mordi, M.N.; Ramanathan, S.; Mansor, S.M. Antioxidant activity and the effect of different parts of Areca catechu extracts on Glutathione-S-Transferase activity in vitro. Free Radic. Antioxid. 2011, 1, 28–33. [Google Scholar] [CrossRef]
- Wu, P.F.; Chiang, T.A.; Chen, M.T.; Lee, C.P.; Chen, P.H.; Ko, A.M.; Yang, K.J.; Chang, P.Y.; Ke, D.S.; Ko, Y.C. A characterization of the antioxidant enzyme activity and reproductive toxicity in male rats following sub-chronic exposure to areca nut extracts. J. Hazard. Mater. 2010, 178, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.X.; Zang, X.B.; Qiu, F.; Nie, Y.; Hu, Y.M.; Hu, S.M.; Pan, C.H.; Zhou, Y.C. The effect of Areca nut on the sperms of mice. Teratog. Carcinog. Mutagen. 1999, 11, 39–41. [Google Scholar]
- Sun, M.H.; Luo, Z.Q.; Liu, Y.; Yang, R.R.; Lu, L.N.; Yu, G.H.; Ma, X.Y.; Liu, A.X.; Guo, Y.F.; Zhao, H.Y. Identification of the Major Components of Buddleja officinalis Extract and Their Metabolites in Rat Urine by UHPLC-LTQ-Orbitrap. J. Food Sci. 2016, 81, H2587–H2596. [Google Scholar] [CrossRef] [PubMed]
- Zuo, A.H.; Wang, L.; Xiao, H.B.; Li, L.M.; Liu, Y.H.; Yi, J.H. Identification of the absorbed components and metabolites in rat plasma after oral administration of Rhizoma Chuanxiong decoction by HPLC-ESI-MS/MS. J. Pharm. Biomed. Anal. 2011, 56, 1046–1056. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.; Han, Z.; Mei, W.L.; Dai, H.F. Antioxidant and Cytotoxic Phenolic Compounds of Areca Nut (Areca catechu). Chem. Res. Chin. Univ. 2010, 26, 161–164. [Google Scholar]
- Jahns, E. Ueber die Alkaloïde der Arecanuss. Berichte Der Deutschen Chemischen Gesellschaft 2006, 21, 3404–3409. [Google Scholar] [CrossRef]
- Baruah, H.K. Arecanut, Indias popular masticatory Botany, chemistry and utilization. Econ. Bot. 1958, 12, 315–345. [Google Scholar]
- Huang, J.L.; Mcleish, M.J. High-performance liquid chromatographic determination of the alkaloids in betel nut. J. Chromatogr. A 1989, 475, 447–450. [Google Scholar] [CrossRef]
- Yang, W.Q.; Wang, H.C.; Wang, W.J.; Wang, Y.; Zhang, X.Q.; Ye, W.C. Chemical constituents from the fruits of Areca catechu. J. Chin. Med. Mater. 2012, 35, 400–403. [Google Scholar]
- Shoji, T.; Mutsuga, M.; Nakamura, T.; Kanda, T.; Akiyama, H.; Goda, Y. Isolation and structural elucidation of some procyanidins from apple by low-temperature nuclear magnetic resonance. J. Agric. Food Chem. 2003, 51, 3806–3813. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Troufflard, S.; Serafini, M.; Crozier, A. Absorption, metabolism and excretion of Choladi green tea flavan-3-ols by humans. Mol. Nutr. Food Res. 2009, 53, S44–S53. [Google Scholar] [CrossRef] [PubMed]
- Giri, S.; Idle, J.R.; Chen, C.; Zabriskie, T.M.; Krausz, K.W.; Gonzalez, F.J. A metabolomic approach to the metabolism of the areca nut alkaloids arecoline and arecaidine in the mouse. Chem. Res. Toxicol. 2006, 19, 818–827. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
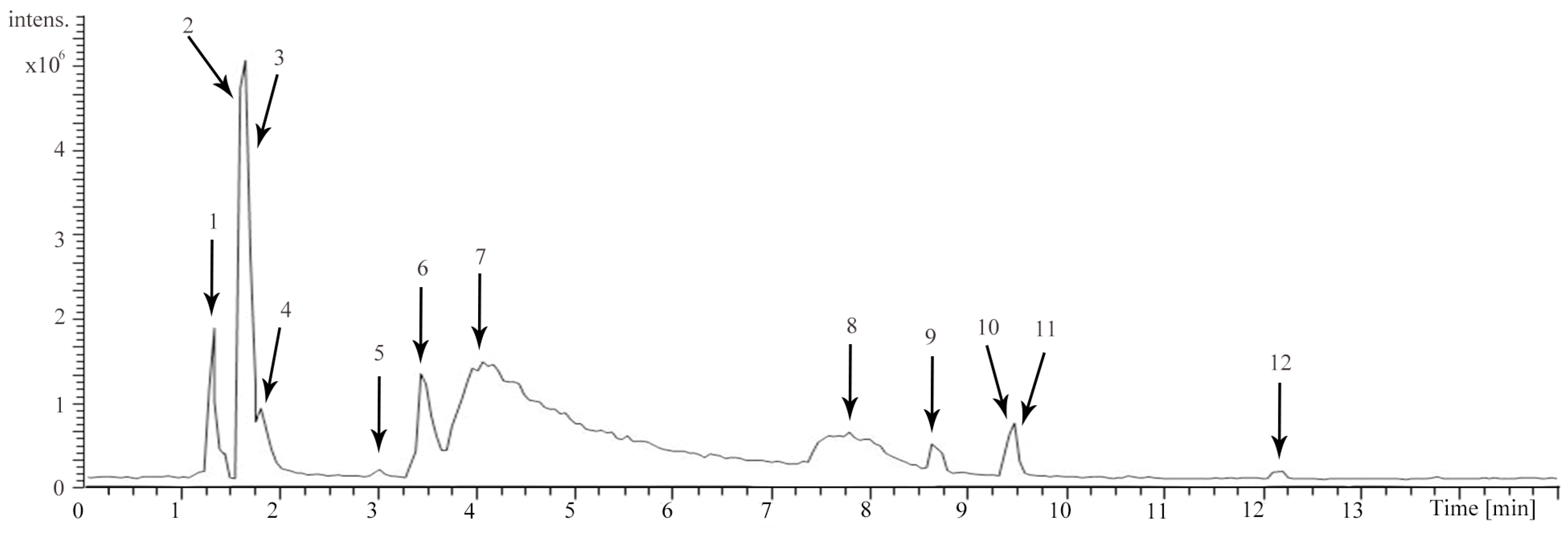
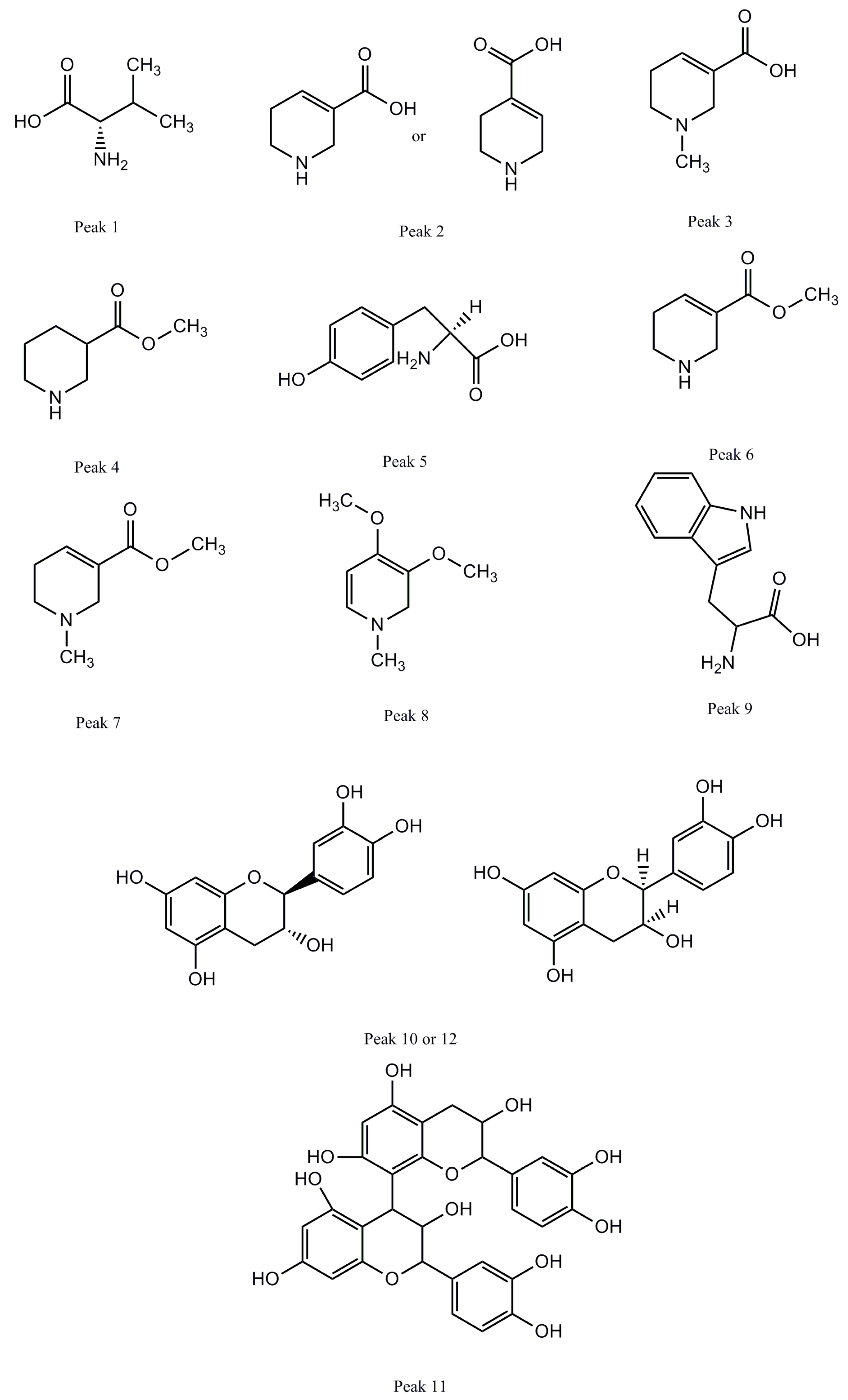
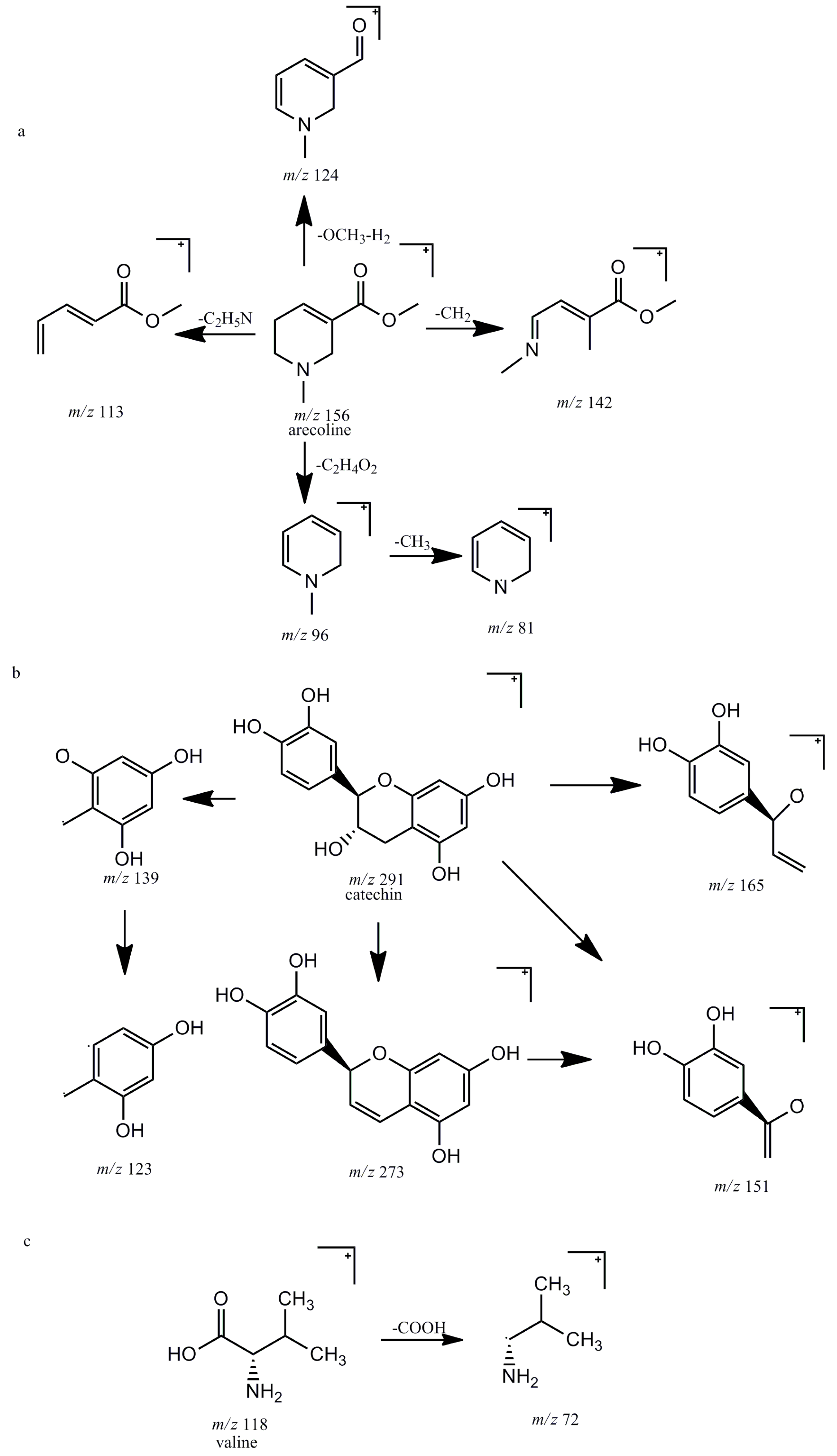
| Component | tR (Min) | [M + H]+ Ion (m/z) | Error (ppm) | Formula | Fragment Ions (m/z) | Identification |
|---|---|---|---|---|---|---|
| 1 | 1.36 | 118.0861 | −1.568 | C5H11NO2 | 72.0806 | Valine |
| 2 | 1.70 | 128.0705 | −0.743 | C6H9NO2 | 110.0601, 81.0334 | Guvacine/Isoguvacine |
| 3 | 1.74 | 142.0862 | −0.085 | C7H11NO2 | 128.0745, 124.0758, 96.0807, 81.0333 | Arecaidine |
| 4 | 1.87 | 144.1018 | −0.869 | C7H13NO2 | 126.0915, 114.0913, 84.0806 | Methyl piperidine-3-carboxylate |
| 5 | 3.05 | 182.0810 | −0.822 | C9H11NO3 | 165.0548, 136.0760 | Tyrosine |
| 6 | 3.47 | 142.0862 | −0.115 | C7H11NO2 | 113.9638, 110.0602, 81.0334 | Guvacoline |
| 7 | 4.09 | 156.1018 | −0.115 | C8H13NO2 | 124.0757, 113.0597, 96.0807, 81.0333 | Arecoline |
| 8 | 7.81 | 156.1017 | −0.255 | C8H13NO2 | 128.1069, 113.0597, 81.0333 | Arecolidine |
| 9 | 8.68 | 579.1489 | −1.334 | C30H26O12 | 453.1197, 427.1039, 409.0937, 291.0874 | Procyanidin B2 |
| 10 | 9.48 | 291.0861 | −0.669 | C15H14O6 | 273.0762, 165.0548, 151.0392, 139.0392, 123.0443 | Catechin (C) |
| 11 | 9.53 | 205.0970 | −0.996 | C11H12N2O2 | 188.0917, 159.9331 | Tryptophan |
| 12 | 12.20 | 291.0860 | −0.978 | C15H14O6 | 273.0764, 165.0549, 151.0393, 139.0393, 123.0444 | Epicatechin (EC) |
| Metabolite | tR (Min) | Selected Ion | Calculated Mass (m/z) | Error (ppm) | Formula | Fragmentation | Identification | Source |
|---|---|---|---|---|---|---|---|---|
| M1 | 1.75 | [M + H]+ | 128.0706 | −0.352 | C6H9NO2 | 110.0601, 81.0332 | Guvacine/Isoguvacine | Urine; Plasma |
| M2 | 1.76 | [M + H]+ | 142.0862 | −0.740 | C7H11NO2 | 124.0759, 96.0809, 81.0334 | Arecaidine | Urine; Plasma |
| M3 | 1.78 | [M + H]+ | 144.1018 | −0.661 | C7H13NO2 | 126.0915, 114.0915, 84.0806 | Methyl piperidine-3-carboxylate | Urine; Plasma |
| M4 | 1.96 | [M + H]+ | 158.0811 | −0.315 | C7H11NO3 | 140.0529, 112.0870 | Arecaidine N-oxide | Urine; Plasma |
| M5 | 3.28 | [M + H]+ | 142.0862 | −0.388 | C7H11NO2 | 113.0597, 110.0601, 81.0333 | Guvacoline | Urine; Plasma |
| M6 | 3.58 | [M + H]+ | 156.1018 | −0.418 | C8H13NO2 | 124.0758, 113.0598, 96.0807, 81.0333 | Arecoline | Urine |
| M7 | 4.31 | [M + H]+ | 319.1319 | −1.000 | C13H22N2O5S | 287.1068, 277.1223, 190.0901, 156.1022 | Arecoline mercapturic acid | Urine; Plasma |
| M8 | 5.58 | [M + H]+ | 343.1862 | −0.417 | C16H26N2O6 | 172.0974 | Arecoline N-oxide dimer | Urine; Plasma |
| M9 | 5.79 | [M + H]+ | 172.0966 | −1.103 | C8H13NO3 | 154.0866, 140.0710, 112.0759 | Arecoline N-oxide | Urine; Plasma |
| M10a | 7.60 | [M − H]− | 465.1025 | −0.586 | C21H22O12 | 289.0719 | Glucuronidation of C/EC | Urine |
| M11 | 7.67 | [M + H]+ | 144.1019 | −0.175 | C7H13NO2 | 126.0916, 98.0965, 84.0807 | N-methylnipecotic acid | Urine; Plasma |
| M12a | 7.90 | [M − H]− | 369.0282 | 1.872 | C15H14O9S | 289.0723 | Sulfation of C/EC | Urine |
| M10b | 8.12 | [M − H]− | 465.1037 | 1.973 | C21H22O12 | 289.0721 | Glucuronidation of C/EC | Urine |
| M12b | 8.22 | [M − H]− | 369.0282 | 2.279 | C15H14O9S | 289.0723 | Sulfation of C/EC | Urine |
| M13 | 8.33 | [M + H]+ | 335.1270 | −0.369 | C13H22N2O6S | 317.1172, 303.1019, 293.1173, 206.0851, 172.0973 | Arecoline N-oxide mercapturic acid | Urine; Plasma |
| M14 | 8.41 | [M + H]+ | 170.0812 | −0.057 | C8H11NO3 | 152.0711 | Norepinephrine | Urine; Plasma |
| M10c | 8.47 | [M − H]− | 465.1024 | −0.715 | C21H22O12 | 289.0716 | Glucuronidation of C/EC | Plasma |
| M15 | 8.60 | [M + H]+ | 579.1491 | −1.023 | C30H26O12 | 453.1199, 427.1041, 409.0938, 291.0875 | Procyanidin B2 | Urine; Plasma |
| M16a | 9.20 | [M − H]− | 383.0435 | 1.020 | C16H16O9S | 303.0879, 137.0249 | Sulfation + methylation of C/EC | Urine |
| M17a | 9.31 | [M + H]+ | 291.0865 | 0.602 | C15H14O6 | 273.0766, 165.0550, 151.0394, 139.0394, 123.0444 | Catechin(C) | Urine; Plasma |
| M10d | 9.53 | [M − H]− | 465.1031 | 0.790 | C21H22O12 | 289.0721 | Glucuronidation of C/EC | Plasma |
| M18a | 9.86 | [M − H]− | 479.1187 | 0.579 | C22H24O12 | 303.0877, 289.0716, 175.0249 | Methylation + glucuronidation of C/EC | Urine |
| M18b | 10.12 | [M − H]− | 479.1179 | −1.132 | C22H24O12 | 303.0873, 289.0715, 175.0247 | Methylation + glucuronidation of C/EC | Urine |
| M19a | 10.16 | [M + H]+ | 305.1020 | 0.083 | C16H16O6 | 287.0921, 179.0707 | Methylation of C/EC | Urine; Plasma |
| M19b | 10.50 | [M + H]+ | 305.1021 | 0.280 | C16H16O6 | 287.0923, 179.0708 | Methylation of C/EC | Urine; Plasma |
| M16b | 10.73 | [M − H]− | 383.0430 | −0.259 | C16H16O9S | 303.0876, 137.0249 | Sulfation + methylation of C/EC | Urine |
| M16c | 11.73 | [M − H]− | 383.0427 | −1.225 | C16H16O9S | 303.0882, 137.0250 | Sulfation + methylation of C/EC | Urine |
| M17b | 11.99 | [M + H]+ | 291.0864 | 0.293 | C15H14O6 | 273.0767, 165.0551, 151.0394, 139.0394, 123.0445 | Epicatechin(EC) | Urine; Plasma |
| M16d | 12.72 | [M − H]− | 383.0429 | −0.729 | C16H16O9S | 303.0882, 137.0250 | Sulfation + methylation of C/EC | Urine |
| M19c | 12.94 | [M + H]+ | 305.1020 | −0.015 | C16H16O6 | 287.0923, 179.0708 | Methylation of C/EC | Urine; Plasma |
| M16e | 13.01 | [M − H]− | 383.0436 | 1.334 | C16H16O9S | 303.0876, 137.0250 | Sulfation + methylation of C/EC | Plasma |
| M20 | 13.06 | [M + H]+ | 184.0968 | −0.271 | C9H13NO3 | 166.0865, 152.0710 | Epinephrine(EP) | Urine |
| M16f | 13.29 | [M − H]− | 383.0432 | 0.289 | C16H16O9S | 303.0875, 137.0249 | Sulfation + methylation of C/EC | Plasma |
| M18c | 13.70 | [M − H]− | 479.1183 | −0.297 | C22H24O12 | 303.0876, 289.0718, 175.0249 | Methylation + glucuronidation of C/EC | Plasma |
| M12c | 13.89 | [M − H]− | 369.0271 | −0.946 | C15H14O9S | 289.0722 | Sulfation of C/EC | Plasma |
| M18d | 13.93 | [M − H]− | 479.1179 | −1.007 | C22H24O12 | 303.0876, 289.0717, 175.0249 | Methylation + glucuronidation of C/EC | Plasma |
| M21 | 14.27 | [M + H]+ | 321.0943 | −2.549 | C16H16O7 | 303.1331, 289.0689 | Methylation of epigallocatechin(EGC) | Urine |
| M16g | 14.34 | [M − H]− | 383.0432 | 2.378 | C16H16O9S | 303.0883, 137.0250 | Sulfation + methylation of C/EC | Plasma |
| M19d | 14.35 | [M + H]+ | 305.1018 | −0.507 | C16H16O6 | 287.0920, 179.0706 | Methylation of C/EC | Urine |
| M12d | 14.68 | [M − H]− | 369.0270 | −1.352 | C15H14O9S | 289.0722 | Sulfation of C/EC | Plasma |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Luo, Z.; Liu, Y.; Wang, H.; Liu, A.; Yu, G.; Li, M.; Yang, R.; Chen, X.; Zhu, J.; et al. Screening and Identification of the Metabolites in Rat Plasma and Urine after Oral Administration of Areca catechu L. Nut Extract by Ultra-High-Pressure Liquid Chromatography Coupled with Linear Ion Trap–Orbitrap Tandem Mass Spectrometry. Molecules 2017, 22, 1026. https://doi.org/10.3390/molecules22061026
Li L, Luo Z, Liu Y, Wang H, Liu A, Yu G, Li M, Yang R, Chen X, Zhu J, et al. Screening and Identification of the Metabolites in Rat Plasma and Urine after Oral Administration of Areca catechu L. Nut Extract by Ultra-High-Pressure Liquid Chromatography Coupled with Linear Ion Trap–Orbitrap Tandem Mass Spectrometry. Molecules. 2017; 22(6):1026. https://doi.org/10.3390/molecules22061026
Chicago/Turabian StyleLi, Lulu, Zhiqiang Luo, Yang Liu, Hao Wang, Aoxue Liu, Guohua Yu, Mengwei Li, Ruirui Yang, Xinjing Chen, Jialian Zhu, and et al. 2017. "Screening and Identification of the Metabolites in Rat Plasma and Urine after Oral Administration of Areca catechu L. Nut Extract by Ultra-High-Pressure Liquid Chromatography Coupled with Linear Ion Trap–Orbitrap Tandem Mass Spectrometry" Molecules 22, no. 6: 1026. https://doi.org/10.3390/molecules22061026





