In 2001 the Kozikowski group was the first to first synthesize and evaluate urea-based PSMA inhibitors [
70]. In 2002, the Pomper group at John Hopkins School of Medicine (Baltimore, MD, USA) published the synthesis and preclinical evaluation of the first radiolabeled urea-based PSMA inhibitor, (
N-(
N-[(
S)-1,3-dicarboxypropyl]carbomoyl)-S-
11C-methyl-
l-cysteine) (
11C-DCMC), also referred to as
11C-MeCys-C(O)-Glu (
11C-MCG) [
75]. To date, a variety of urea-based PSMA inhibitors have been developed and labeled with different SPECT (
99mTc,
111In), PET (
68Ga,
64Cu), and therapeutic (
177Lu) radiometals using several chelators and showed great promise not only preclinicaly but also in the clinical assessment. Apart from the radiometals to be used as the cytotoxic units, a considerable amount of urea-based PSMA inhibitors labeled with
11C,
125I,
124I,
131I and
18F, have also been reported [
19,
76,
77,
78,
79,
80,
81,
82,
83,
84,
85,
86,
87,
88,
89,
90,
91,
92,
93,
94,
95,
96]. These radiotracers were also able to successfully image PSMA-expressing xenografted mice, but since this is beyond the focus of this review no further information will be given here with regard to this topic.
4.1. 99mTc-Labeled Urea-Based PSMA Inhibitors
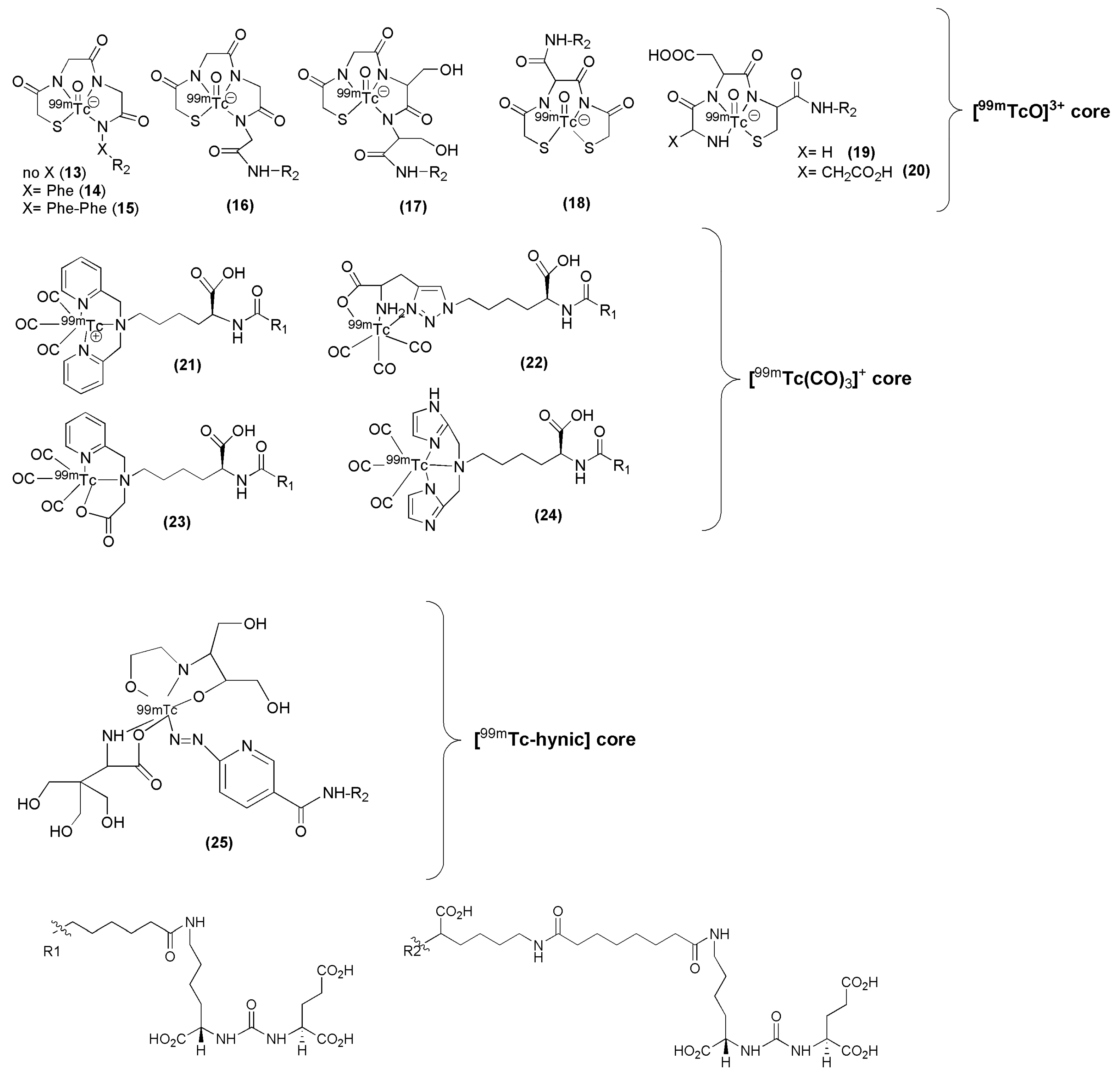
99mTc remains the radionuclide of choice for SPECT applications and a variety of PSMA inhibitors have been modified to facilitate efficient labeling with
99mTc. Banerjee et al. have published a series of PSMA inhibitors suitable for labeling with
99mTc [
98,
99]. The influence of the spacer length as well as the nature of different chelators was thoroughly investigated with respect to the overall performance of the derived
99mTc-labeled Lys-urea-Glu-based PSMA inhibitors (compounds
13–
25,
Figure 6,
Table 1). In particular, the PSMA inhibitors are attached to hydrophilic ligands for radiolabeling with the
99mTc(I)-tricarbonyl-labeled ([
99mTc(CO)
3]
+) core, traditional N
xS
y-based chelator for the
99mTc-oxo ([
99mTcO]
3+) core, and a
99mTc-organohydrazine (
99mTc-hynic) core. It was proved that parameters such the charge, polarity and hydrophilicity are crucial and contribute not only to the efficient targeting of PSMA by the
99mTc-labeled agent but also to its pharmacokinetics.
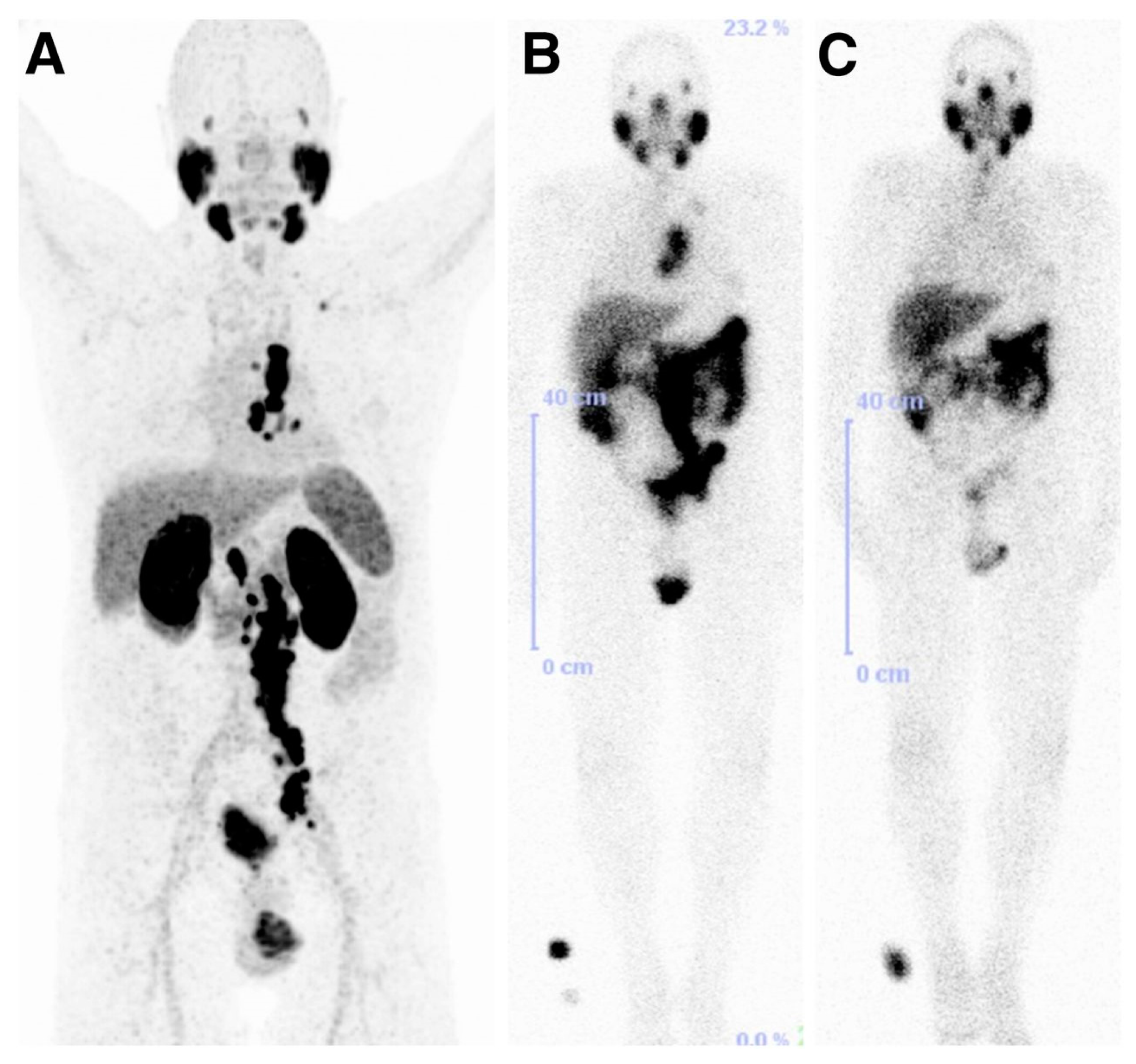
The above tested low-molecular-weight, urea-based, 99mTc-labeled, PSMA-targeted radiotracers, differ in the chelator and in the lipophilicity. Except for HYNIC-labeled 99mTc-25 irrespective of charge and lipophilicity the radioligands 99mTc-(13-14) enabled visualization of PSMA positive tumors (>20 % IA/g at 2 h p.i.) and kidneys. More specifically, the study showed that the degree of lipophilicity had a considerable effect on binding affinity as well as on in vivo performance. The ligands which showed higher lipophilic character revealed higher inhibitory activity, lower tumor and higher background uptake. Indeed, compounds 13 to 15, which possess different degree of lipophilicity due to different number of Phe moieties (from zero (13) to three (15)) on the spacer, displayed considerable differences in tumor and background uptake as measured by SPECT/CT images from male SCID mice bearing PSMA+ PC3 PIP and PSMA− PC3 flu tumors at 2 h p.i. High gallbladder, liver, and gastrointestinal uptake were observed for 99mTc-15 as compared to that of 99mTc-13. Between the radioligands bearing the tricarbonyl core, 99mTc-21 and 99mTc-22, the neutral complex 99mTc-22, exhibited favorable pharmacokinetics and promising SPECT/CT images, presumably because of the comparatively low background within the gastrointestinal tract relative to the positively charged 99mTc-21 (99mTc-21: small intestine: 1.29 ± 0.76 % IA/g, large intestine: 16.02 ± 12.39 % IA/g at 1 h p.i., 99mTc-22: small intestine: 1.35 ± 0.19 % IA/g, large intestine: 0.29 ± 0.06 % IA/g at 1 h p.i.). In addition, 99mTc-22, the most hydrophilic radioligand in the tricarbonyl series, had high, specific and retained tumor uptake in the PSMA+ PC3 PIP tumor (28.31 ± 4.38, 28.05 ± 2.04, 26.29 ± 7.45, 23.22 ± 6.02 % IA/g, at 0.5, 1, 2 and 5 h p.i. respectively). Another point which also needs to be pointed out is that the 99mTc-oxo radioliagnds showed high and prolonged spleen uptake, which was not observed with the tricarbonyl-based radioligands. 99mTc-22 proved superior with respect to tumor-to background ratios over time. Despite high tumor uptake, other compounds had either high gastrointestinal or spleen uptake, which cannot be easily attributed on their charge and lipophilicity. No clinical studies with of these 99mTc compounds have been reported.
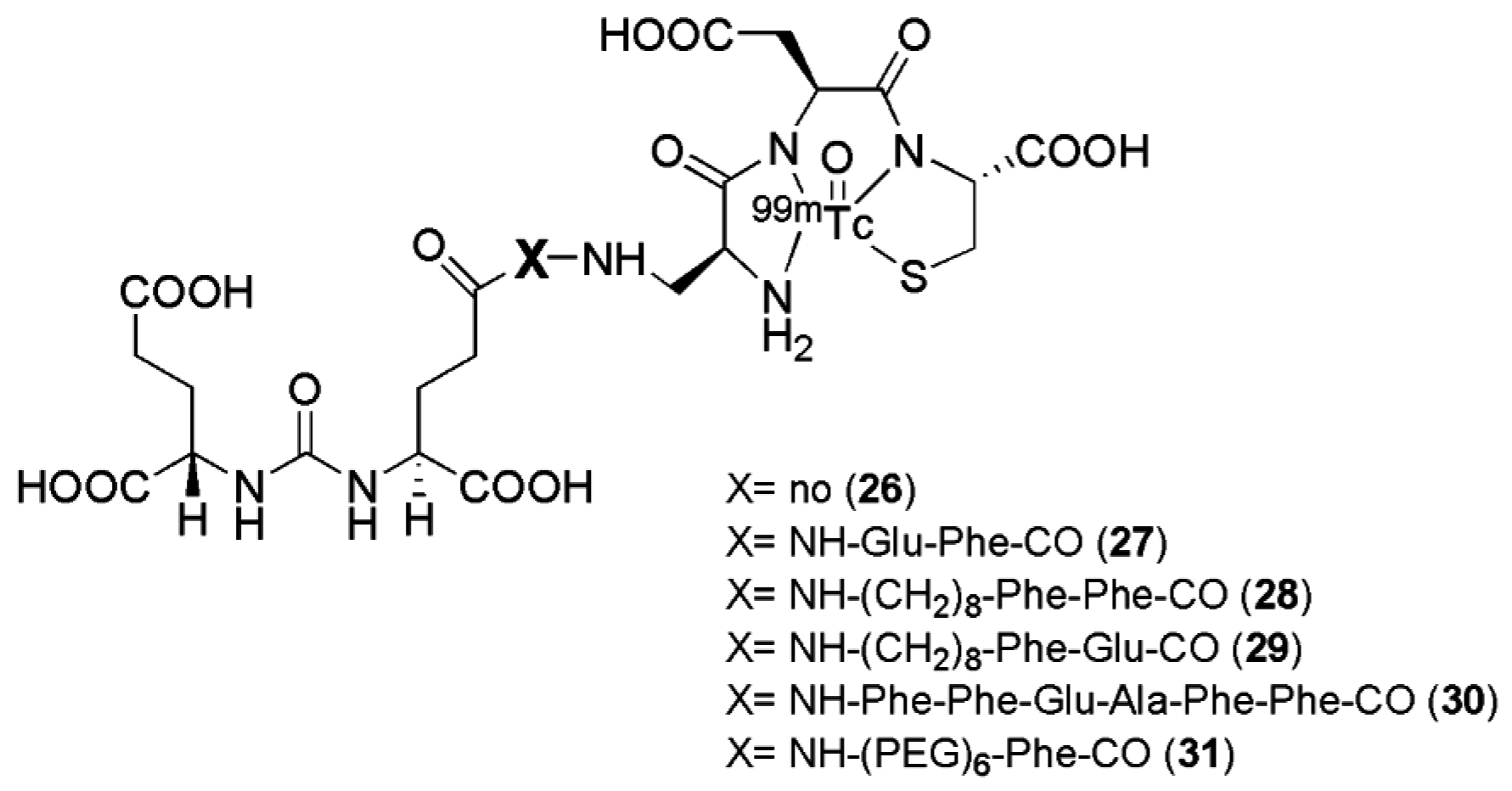
Six targeted radioimaging agents labeled with
99mTc derived from the Lys-urea-Glu peptidomimetic structure functionalized with the chelator Dap-Asp-Cys through spacers of different lengths (compounds
26–
31,
Figure 7,
Table 2) were investigated by Kularante et al. [
100].
This study demonstrates the importance of the spacer between the urea-based PSMA inhibitors and the chelator which have been used for their functionalization. Docking studies revealed that coupling the urea analogue to an 8-aminooctanoic acid moiety (to avoid steric overlap within the narrow regions at the base of the tunnel) followed by two phenylalanine residues (for maximal interaction with hydrophobic pockets near the mouth of the tunnel) generated an agent (compound 28) with high affinity and specificity. Also the most lipophilic compound was found to be the most affine towards PSMA. The in vivo data were in accordance with the findings from in vitro studies, with remarkable tumor uptake upon injection in nu/nu mice xenografted with LNCaP tumor cells (average 9.8 ± 2.4 % IA/g at 1 h p.i.) was observed for radiotracers 27, 28, and 29 with little accumulation in other tissues except the kidneys. These ligands have so far not been transferred to clinical evaluation.
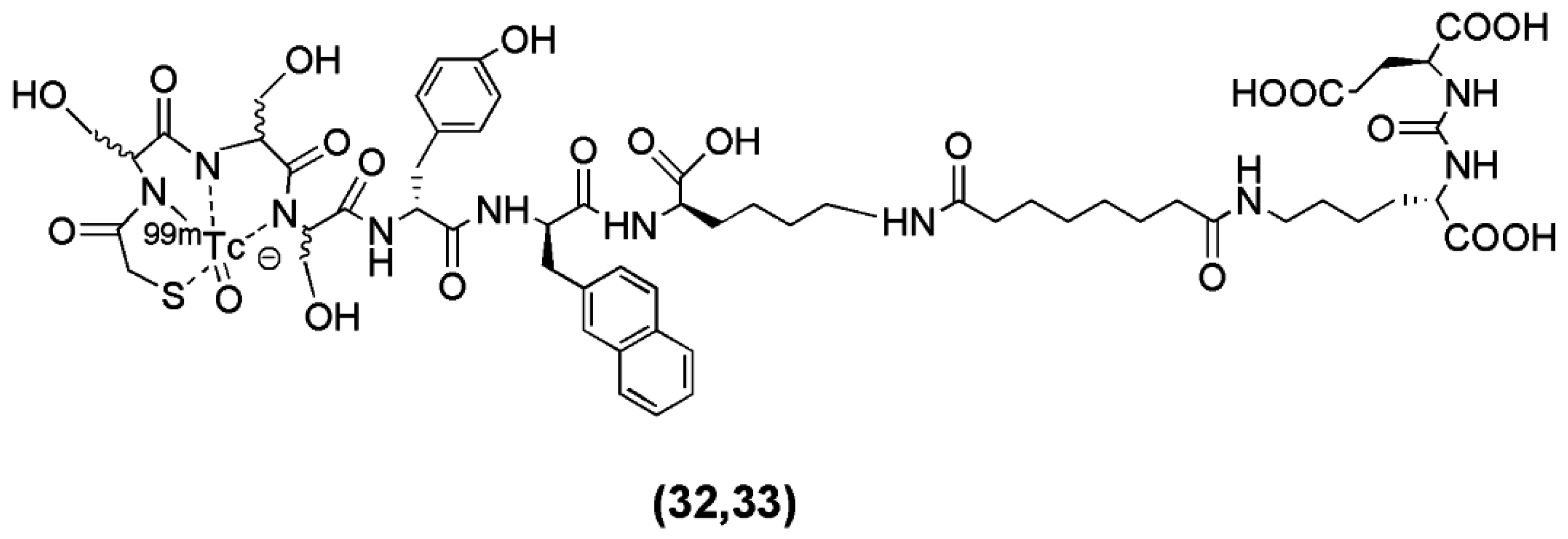
Robu et al. recently published the synthesis of two
99mTc-labeled anti-PSMA agents [
101]. In this study the Lys-urea-Glu motif was coupled to the spacer
d-Tyr-
d-2-Nal-
d-Lys-suberoyl (y-nal-k-Sub) to enhance interaction of the peptidic spacer with a remote arene binding site and the functionalization of the precursor took place through the conjugation of the chelators MAS
3 (2-mercaptoacetyl-Ser-Ser-Ser-) (compound
32,
Figure 8) and mas
3 (2-mercaptoacetyl-
d-Ser-
d-Ser-
d-Ser-) (compound
33,
Figure 8). The derived conjugates labeled with
99mTc (
Table 3).
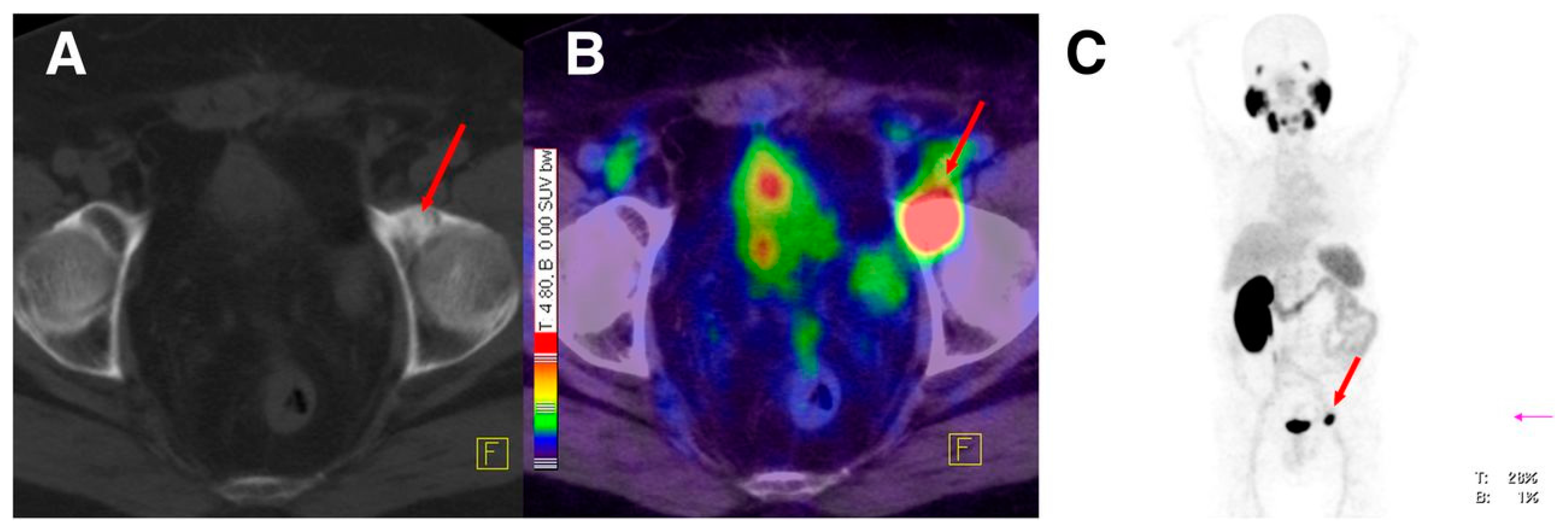
The main focus of the study was to investigate whether or not the all-L-amino acid chelator MAS3 is susceptible towards proteolytic degradation. As anticipated, no detectable influence of chelator stereochemistry on the outcome of the 99mTc-labeling reaction was observed, since the formation of the 99mTcO-MAS3/mas3-complex should be independent from the spatial orientation of the serine side chains. On the other hand, the MAS3-chelating 99mTc-labeled tracer showed substantially decreased in vivo stability compared to the mas3-derivative 99mTc-PSMA-I&S (PSMA-I&S for Imaging and Surgery), for which only intact tracer was detected in blood, urine and kidney at 1 h p.i. Furthermore, 99mTc-PSMA-I&S uptake in kidney and LNCaP-tumor was found to be high (186 ± 23, 8.28 ± 3.27 % IA/g at 1 h p.i., respectively) and specific, as shown by the blocking studies. The straightforward and reliable kit-production and the initial patient data indicate the potentiality of 99mTc-PSMA-I&S as a SPECT imaging agent.
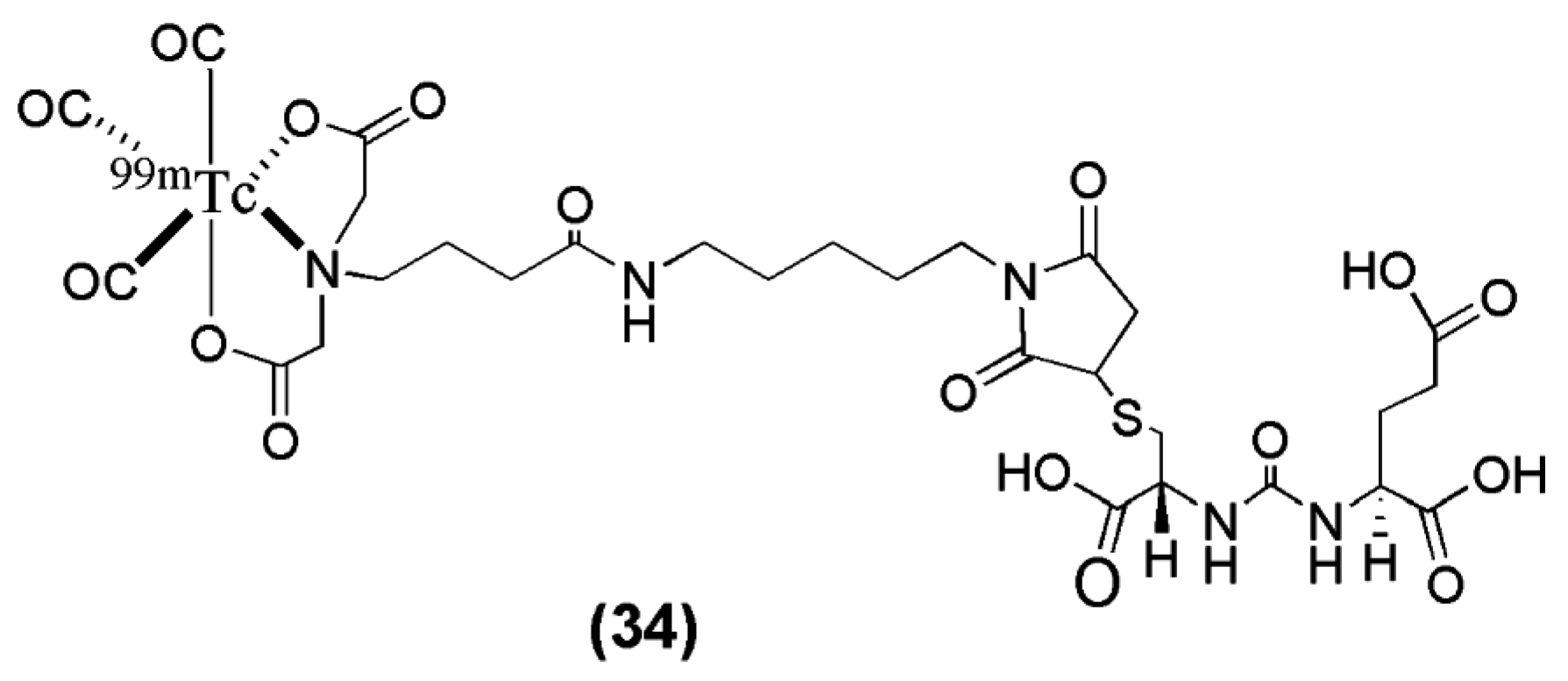
In another study, Kimura et al. [
102] synthesized and evaluated a novel anionic
99mTc-tricarbonyl complex (
99mTc-TMCE) with high hydrophilicity due to strong polarity and electric charges with the intent to further decrease non-target tissue uptake and enhance renal clearance (compound
34,
Figure 9,
Table 4).
99mTc-TMCE was obtained in very low radiochemical yield (12–17%), introducing the implementation of a HPLC for purification of the radioligand. Compared to the neutral and positively charged tricarbonyl core-bearing radioligands published by Banerjee et al. the in vivo performance of the anionic
99mTc-tricarbonyl complex
99mTc-TMCE is characterized by a relatively long blood circulation and fast washout from the tumor (blood: 11.4 ± 2.3, 3.2 ± 0.5, 0.6 ± 0.6 ± 0.1 % IA/g and LNCaT-tumor: 4.0 ± 1.2, 12.8 ± 2.2 and 5.0 ± 2.7 % IA/g at 5, 30 and 120 min p.i. respectively) The outcome of the study demonstrated the effect of affinity, hydrophilicity, and electric charge in the pharmacokinetics of PSMA imaging probes. In particular, the hydrophilicity of both cationic and anionic charges led to rapid hepatobiliary clearance, although anionic charges might enhance renal clearance to a greater extent (kidney: 124.9 ± 26.2, 136 ± 6.4 and 56.8 ± 20.6 % IA/g at 5, 30 and 120 min p.i.). The implementation of this
99mTc-labeled anti-PSMA radiotracer into the clinical pathway has not taken place yet.
Four small-molecule inhibitors for PSMA labeled with
99mTc via technetium tricarbonyl chemistry were reported by Hillier et al. [
103]. The
99mTc-labeled PSMA inhibitors derived from the glutamate-urea-glutamate or glutamate-urea-lysine pharmacophores after the incorporation of the chelators 2,2′-(2,2′-(azanediylbis(methylene))bis(1
H-imidazole-2,1-diyl))diacetic acid (CIM) or 2,2′,2′′,2′′′-((2,2′-(2,2′-(azanediylbis(methylene))bis(1
H-imidazole-2,1-diyl))bis(acetyl))bis(azanetriyl))tetraacetic acid (TIM) and the labeling with
99mTc (compounds
35–
38,
Figure 10) were evaluated in vitro and in vivo (
Table 5).
The introduction of the chelators CIM and TIM to glutamate-urea-glutamate or glutamate-urea-lysine pharmacophores and the subsequent labeling with
99mTc did not influence the affinity of the derived radioligands towards PSMA (
Table 5), as their K
D values were found to be in the low nanomolar range. Furthermore, a side-by-side comparison of the affinity reveals a slightly higher affinity for PSMA of the glutamate-urea-glutamate-containing compounds,
99mTc-MIP-1404 and
99mTc-MIP-1427. The formal addition of an extra carboxylic acid group to the CIM chelator, yielding the TIM chelator, resulted in improved pharmacokinetics of the derived radioligands. Indeed,
99mTc-MIP-1404 and
99mTc-MIP-1428 displayed lower uptake in and more rapid clearance from blood, liver, kidneys (
99mTc-MIP-1404: blood: 0.13 ± 0.03 and 0.02 ± 0, liver: 0.14 ± 0.03 and 0.07 ± 0.01, kindey: 105 ± 37 and 12 ± 7 % IA/g at 1 and 4 h p.i. respectively,
99mTc-MIP-1428: blood: 0.44 ± 0.07 and 0.14 ± 0.03, liver: 0.27 ± 0.05 and 0.14 ± 0.04, kidney: 136 ± 6 and 63 ± 27 % IA/g at 1 and 4 h p.i. respectively), and most other nontarget tissues than did
99mTc-MIP-1405 and
99mTc-MIP-1427 (
99mTc-MIP-1405: blood: 0.72 ± 0.19 and 0.26 ± 0.03, liver: 0.43 ± 0.14 and 0.19 ± 0.05, kidney: 157 ± 69 and 14 ± 6 % IA/g at 1 and 4 h p.i. respectively,
99mTc-MIP-1427: blood: 0.55 ± 0.08 and 0.28 ± 0.03, liver: 0.83 ± 0.24 and 0.76 ± 0.26, kidney: 149 ± 24 and 142 ± 39 % IA/g at 1 and 4 h p.i. respectively). The chelator TIM did not diminish uptake or retention in the LNCaP xenograft tumor, as there was no statistical difference between the tumor uptake of any of the compounds at either the 1 h or the 4 h time points. More specifically, uptake in LNCaP xenografts ranged from 9.3% to 12.4% injected dose per gram at 1 h after injection and from 7.2% to 11.0% at 4 h, with tumor-to-blood ratios ranging from 29:1 to 550:1 and tumor–to–muscle ratios ranging from 31:1 to 157:1 at 4 h.
99mTc-MIP-1404 is currently under clinical investigation in a phase II trial.
4.2. 68Ga-Labeled Urea-Based PSMA Inhibitors
Because of its availability from the
68Ge/
68Ga generator systems and the relative ease of labeling chemistry, the positron emitter
68Ga (t
½ = 68 min, β
+max = 1899 keV) has gained increasing interest in the field of molecular imaging [
104] and can considered a PET-applicable counterpart of
99mTc.
Many efforts have been made with respect to the development of
68Ga-PSMA-based imaging agents in the hope of developing a cyclotron-independent nuclear probe for prostate cancer imaging. A variety of PSMA-based inhibitors have been generated where several chelators suitable for labeling with
68Ga as well as spacers have been investigated. In solution, gallium is most stable in the +3-oxidation state and forms stable complexes with several chelating agents. The HBED chelator (
N,
N′-bis[2-hydroxybenzyl] ethylenediamine-
N,
N′-diacetic acid) which contains an amine-phenol backbone which renders it suitable for complexation with +3 metal ions, and it forms a thermodynamically stable complex with
68Ga even at room temperature [
105]. A representative example of PSMA-inhibitors associated with the chelator HBED, is the peptidomimetic structure Lys-urea-NH-Glu, published by Eder et al., when coupled to the spacer 6-aminohexanoic acid (Ahx) and functionalized with the chelator
N,
N′-dis[2-hydroxy-5-(carboxyethyl)benzyl] ethylenediamine-
N,
N′-diacetic acid (HBED-CC) to obtain HBED-CC-Ahx-Lys-urea-Glu (HBED-CC-PSMA or PSMA-11) (compound
39,
Figure 11) [
106]. HBED-CC has a dual nature, it also acts as a lipophilic moiety due to the presence of the two phenolic rings and therefore an aliphatic spacer such as Aca is adequate for providing the required lipophilicity and retaining the affinity of the derived ligand towards PSMA (K
i = 12.1 ± 2.1 nM). Radiolabeling with
68Ga was performed at pH 4.2 by incubating PSMA-11 in a mixture consisting of 50–100 MBq [
68Ga]Ga
3+ and HEPES. An amount of 0.1 nmol of PSMA-11 at a final concentration of 1.7 μM led to a radiochemical yield of more than 99% in less than 1 min at room temperature, with specific activities in the range og 500–1000 GBq/μmol. These are the highest specific activities which have been reported for
68Ga-lebeled PSMA inhibitors. In order to achieve comparable high radiochemical yields with the DOTA-conjugated PSMA inhibitors, the compounds were incubated for 2 min at 80 °C using 1 nmol of precursor [
106].
68Ga-PSMA-11 exhibited fast blood clearance, relatively low liver uptake (0.87 ± 0.05 % IA/g at 1 h p.i.), and high specific uptake in PSMA-expressing tissues and tumor (tumor uptake 7.7 ± 1.5 % IA/g at 1 h p.i.). The ability to image PSMA using
68Ga-HBED-CC-PSMA showed great promise not only preclinically but also clinically [
106,
107,
108,
109,
110,
111,
112,
113,
114,
115,
116,
117,
118,
119,
120,
121,
122,
123,
124,
125,
126,
127,
128,
129]. The dimerization of the same pharmacophore, Lys-Urea-Glu, coupled to the same spacer Ahx, through the chelator HBED-CC (PSMA-10) (compound
40,
Figure 11) was also published by Schäfer et al. [
130]. The dimer was also labeled with
68Ga to obtain
68Ga-PSMA-10 and evaluated in vitro and in vivo compared to
68Ga-PSMA-11. Despite the fact that the preclinical evaluation revealed that
68Ga-PSMA-10 was superior to
68Ga-PSMA-11 in terms of binding affinity and tumor to background ratios (IC
50 = 3.9 ± 1.8 nM with a target to non-target ratio of 26.5 at 1 h p.i. as compared to a value of 9.2 for the monomer at the same time point), most of the clinical studies, so far, have been conducted using
68Ga-PSMA-11.
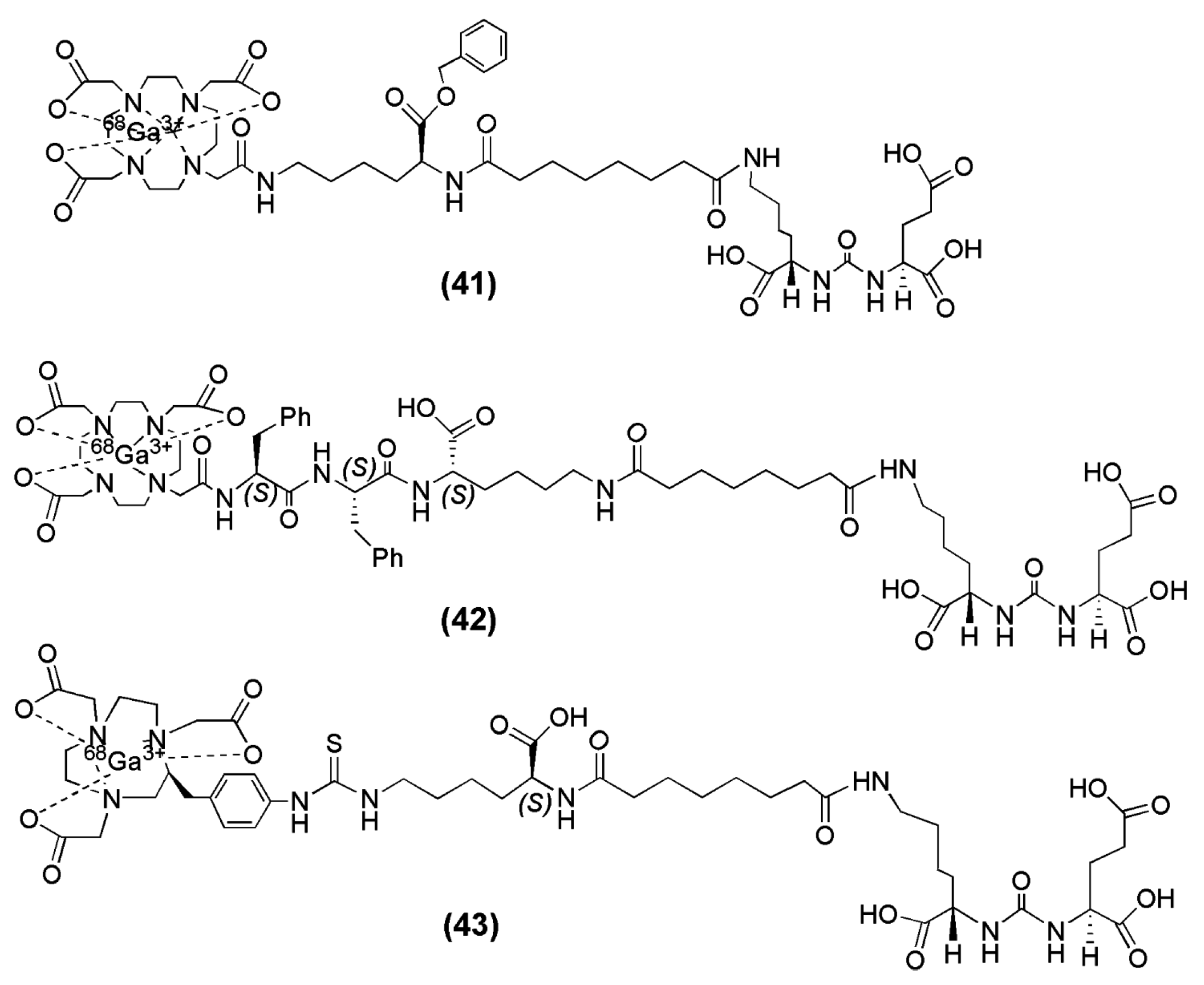
In the field of radiopharmaceutical chemistry, the cyclic chelator 1,4,7,10-tetraazacyclododecane-1,4,7,10-triacetic acid (DOTA) has been used for complexation of +3 radiometal ions such as
68Ga,
90Y and
177Lu. Banerjee et al. described two DOTA-conjugated PSMA inhibitors based on the Lys-Urea-Glu construct that confers PSMA specificity (compounds
41 and
42,
Figure 12) and are suitable for labeling with
68Ga [
131]. The difference between those two tracers is confined to the spacer unit. More specifically, a benzyl group was inserted into compound
41 (
Figure 12) in order to provide a chromophore that facilitates purification. On the other hand, two phenylalanines were added to compound
42 (
Figure 12) in order to offset its high hydrophilicity and enable sustained retention and potentially higher absolute uptake in tumor [
98]. Furthermore, in a recent study from the same group [
132], a new radiotracer containing the macrocyclic chelator NOTA was prepared, since NOTA has been shown to be an effective chelator for
68Ga (compound
43,
Figure 13,). A head-to-head preclinical comparison of
68Ga-42 with
68Ga-43 (
Figure 12) was conducted.
68Ga-PSMA-11, the radioligand which has been used throughout Europe in clinical trials, was also included in the study for comparison
68Ga-41,
68Ga-42 and
68Ga-43 were directly labeled with the eluted
68Ga from the
68Ge/
68Ga generator without the use of a buffer solution within 10 min at 90–95 °C. The yields ranged from 60% to 70% with HPLC purification and the reported radiochemical purities were >99% with specific activities >168 GBq/μmol. Both
68Ga-41 (−3.9) and
68Ga-43 (−4.01 ± 0.16) were found to be 1 order of magnitude more hydrophilic than
68Ga-42 (−3.0 ± 0.1). It is evident that
68Ga-42 is more lipophilic than
68Ga-41 and
68Ga-43, which is reasonable because of the presence of two phenylalanine residues in the spacer. The corresponding metal-labeled compounds demonstrated high binding affinity to PSMA, with Ki values ranging from 0.33 to 29 nM (compound
41 was the less affine towards PSMA).
Both compounds, 68Ga-41 and 68Ga-42, exhibited PSMA specific tumor imaging in vivo. Nevertheless, the improved tumor-to-background ratios of 68Ga-42 at later time points after injection renders it a more promising candidate for clinical translation. The PSMA+ PC3 PIP tumors as well as PSMA positive organs such as, kidney and urinary bladder were clearly delineated already at 15 min p.i. More specifically, the NOTA-conjugated radioligand 68Ga-43 exhibited the highest tumor uptake with 42.2 ± 6.7 % IA/g at 1 h p.i. and the fastest background clearance. The PSMA+ PC3 PIP-to-PSMA− PC3 flu tumor ratios were 110 ± 22 at 1 h, 232 ± 26 at 2 h, and 182 ± 15 at 3 h p.i. Renal uptake for 68Ga-43 was highest at 1 h, 106 ± 23 % IA/g, much higher than that seen for the DOTA-conjugated radioligand 68Ga-42 (26.5 ± 6.9 % IA/g), and showed faster renal clearance, which decreased to 34.7 ± 5.7 % IA/g by 2 h p.i. There was no significant difference in PSMA+ PIP tumor uptake between 68Ga-42 and 68Ga-PSMA-11 or between 68Ga-43 and 68Ga-PSMA-11 (p > 0.05) at any time-point. 68Ga-PSMA-11 demonstrated the highest and retained uptake in normal tissues, including kidney, blood, spleen, salivary glands and PSMA-negative PC3 flu tumors up to 3 h post-injection. This preclinical evaluation showed that 68Ga-43 was superior for PSMA-targeted PET imaging in clinical settings.
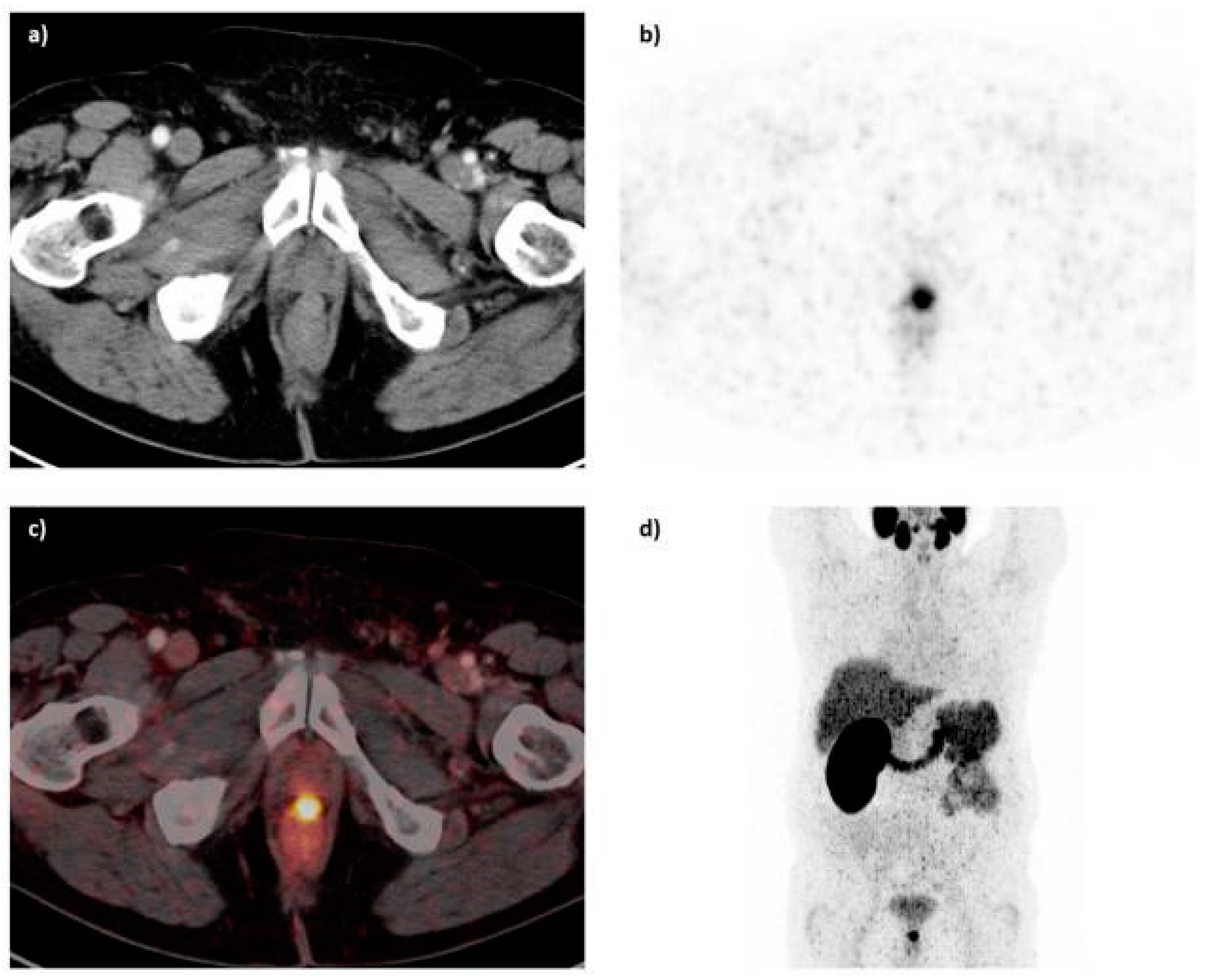
Benešová et al. [
133], published the synthesis and preclinical evaluation a novel theranostic compound termed PSMA-617 (compound
44,
Figure 13). In this case, the chelator DOTA, was conjugated to the pharmacophore Lys-urea-Glu via a naphthylic spacer. PSMA-617 was labeled with [
68Ga]Ga
3+ eluate in HEPES buffer, pH 4.0, within 15 min at 95 °C, with a radiochemical yield of more than 90% and a specific activity in the range of 14–140 GBq/μmol. The study showed that the presence of the naphthylic linker has a significant impact on the tumor-targeting as well as on the pharmacokinetics and the resulting imaging contrast. In fact,
68Ga-PSMA-617 (K
i = 6.40 ± 1.02 nM) was superior to
68Ga-PSMA-11 (K
i = 12.1 ± 2.1 nM) as far as the affinity towards PSMA and efficacy of internalization (up to 17.67 6 4.34 percentage injected activity/10
6 LNCaP cells) into the cancer cells concerns. Biodistibution studies upon injection of
68Ga-PSMA-617 on LNCaP tumor bearing mice 1 h p.i. revealed a high specific uptake in LNCaP tumors (8.47 ± 4.09 % IA/g; 0.98 ± 0.32 % IA/g by coinjection of 2-PMPA) and in the kidneys (113.3 ± 24.4 % IA/g). Other organs such as the liver (1.17 ± 0.10 % IA/g), lung (1.41 ± 0.41 % IA/g), and spleen (2.13 ± 0.16 % IA/g) showed rather low uptake. Furthermore,
68Ga-PSMA-617 dynamic PET imaging showed that the maximum kidney uptake was reached within 15 min after injection and decreased substantially as early as 20 min p.i. High and sustained tumor uptake was observed. The fast kidney clearance emboldened clinical translation of this compound.
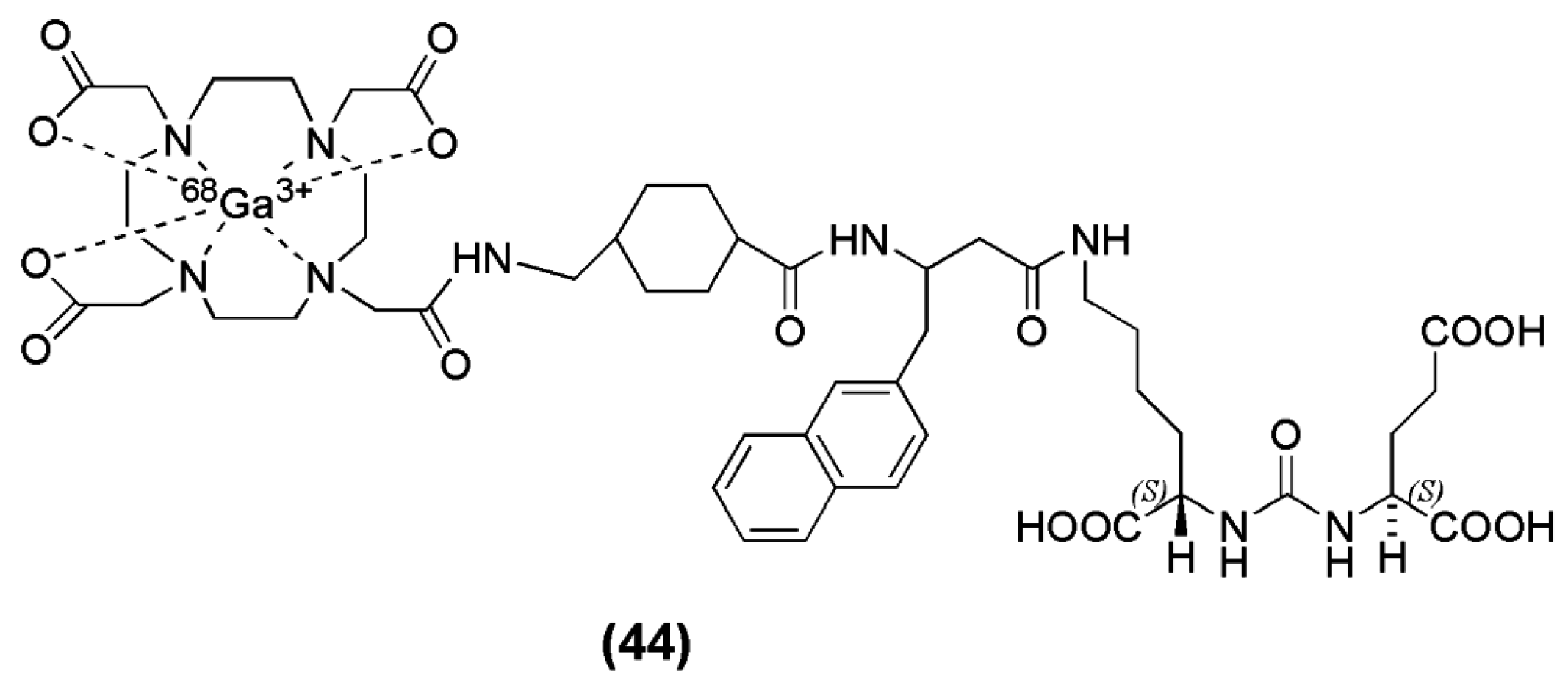
When Lys-Urea-Glu (KuE) was coupled to the spacer Phe-Phe-Lysine-suberoyl (
l-amino acid spacer, FFK-Sub) and functionalized with the chelator (1-(1,3-carboxypropyl)-4,7,10(carboxymethyl)-1,4,7,10 tetraazacyclododecane (DOTAGA) DOTAGA-FFK(Sub-KuE) was obtained [
134]. HEPES buffer was used for the labeling with
68Ga (pH 4.5). The reaction completed within 5 min at 95 °C and resulted in a radioligand with a specific activity of 250–300 GBq/μmol. However, a rapid in vivo metabolysis of the
68Ga-labeled radiovector was demonstrated. In the same report by Weineisen et al. the inclusion of a D-amino acids spacer led to an in vivo metabolic stable radiotracer (DOTAGA-ffk(Sub-KuE)) (compounds
45–
47,
Figure 14).
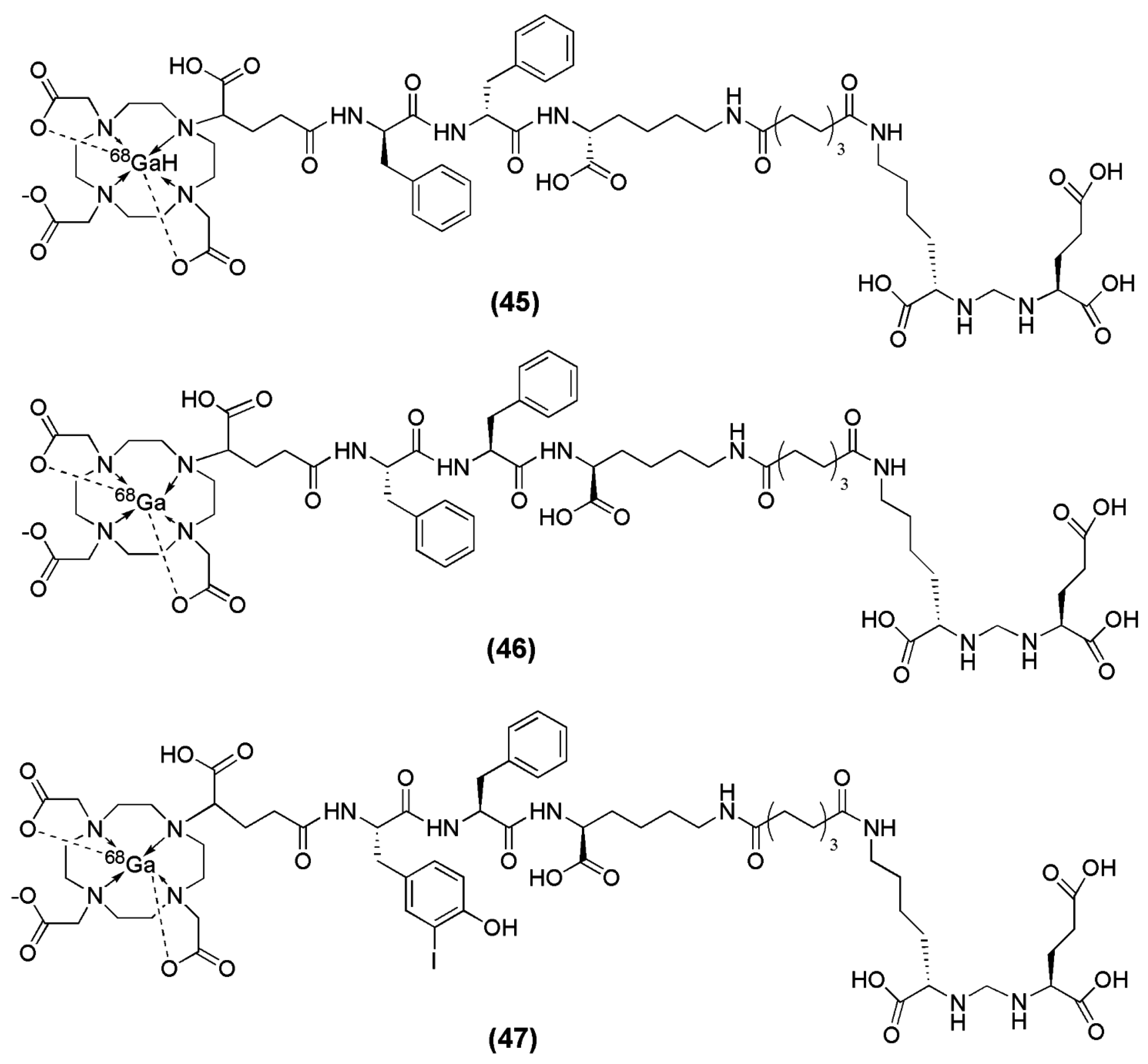
In an attempt from the same group to further optimize this second-generation of the PSMA inhibitor with respect to its affinity towards the PSMA enzyme, additional modifications on the spacer unit were undertaken. [
135]. For that purpose,
d-Phe (f) was substituted with
d-I-Tyr (I-y) and DOTAGA-(I-y)fk(Sub-KuE) was synthesized (
Figure 14). DOTAGA-(I-y)fk(Sub-KuE) is also termed PSMA I&T (PSMA I&T for Imaging &Therapy), since it can be labeled with both the diagnostic radionuclide
68Ga and the therapeutic radionuclide
177Lu. The necessary modifications for the generation of a metabolically stable radioligand did not alter the affinity of the derived ligands towards PSMA, since all the precursors as well as the corresponding metalloconjugates exhibited IC
50 values within the range of 8 to 16 nM.
68Ga-PSMA I&T is characterized by rapid tumor targeting (4.95 ± 1.57 % IA/g at 1 h p.i.) and pharmacokinetics with high uptake in PSMA-positive organs such as the tumor and the kidneys (53.26 ± 9.02 % IA/g at 1 h p.i.).
68Ga-PSMA I&T is currently under clinical investigation.
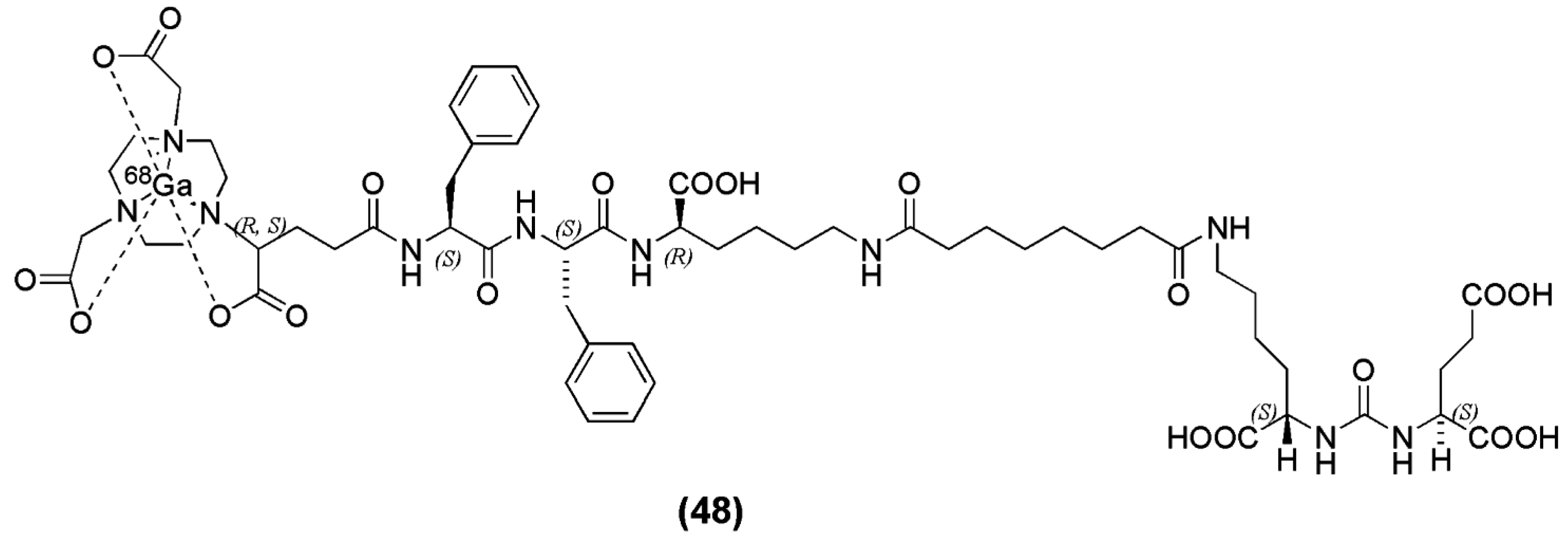
The enantiomerically pure prochelator (
R)-1-(1-carboxy-3-carbotertbutoxypropyl)-4,7-carbotartbutoxymethyl)-1,4,7-triazacyclononane ((
R)-NODAGA(tBu)3) was reported by Gourni et al. for the functionalization of urea-based PSMA inhibitor derived from the Lys-urea-Glu peptidomimetic structure. The spacer Phe-Phe-
d-Lys(suberoyl) was included to obtain (
R)-NODAGA-Phe-Phe-
d-Lys(suberoyl)-Lys-urea-Glu (CC34) (compound
48,
Figure 15) [
136].
68Ga-CC34 was obtained in high specific activity (75–80 MBq/nmol) and evaluated in vitro and in vivo in LNCaP tumor xenografts by biodistribution and PET imaging studies.
68Ga-PSMA-11 was also evaluated for comparison.
68/natGa-CC34 exhibited high affinity for the LNCaP cells, with a K
D value of 19.3 ± 2.5 nM. Tumor uptake of
68Ga-CC34 (14.5 ± 2.9 % IA/g) in LNCaP xenografts at 1 h p.i. was comparable to
68Ga-PSMA-11 (15.8 ± 1.4 % IA/g) (
p = 0.67). The tumor-to-normal tissue ratios at 1 and 2 h p.i of
68Ga-CC34 were also comparable to that of
68Ga-PSMA-11 (
p > 0.05).
In another recent preclinical study, the cyclohexyl-diethylene triamine pentaacetic acid (CHX-A″-DTPA) was used as the chelator for the Glu-urea-GLu-based peptide (Pep) bearing a 2-[3-(1,3-dicarboxypropyl)-ureido]pentanedioic acid (DUPA) moiety to obtain CHX-A″-DTPA-DUPA-Pep (compound
49,
Figure 16). The study describes the efficient labeling of CHX-A″-DTPA-DUPA-Pep with
68Ga,
90Y and
177Lu and the first in vitro characterization which shows high affinity of the tested radiotracers towards PSMA with K
D values of ≤14.67 ± 1.95 nM. Labelling with
68Ga was performed at room temperature under neutral conditions (HEPES, pH 7.4). Significant differences in radiochemical yields were observed. Radio labelling with
68Ga succeeded in 10 min with high radiochemical yields; ≥95% when 50 μg (36 nM) of the peptide was used. However, in vivo images or biodistribution data of these agents have so far not been published [
137].
4.3. 64Cu-Labeled Urea-Based PSMA Inhibitors
Amongst the PET-radioisotopes,
64Cu has gained particular attention because of its decay characteristics (t
1/2 = 12.7 h; β
+, Emax = 0.653 MeV [17.8%]; β
−, Emax = 0.579 MeV [38.4%]) and the well-established coordination chemistry with a variety of chelators. The longer half-life of
64Cu relative to
68Ga (t
1/2 = 67.8 min) and
18F (t
1/2 = 109.8 min) is particularly attractive, as it allows PET-images at later time points with improved tumor-to-background ratios [
138]. Additionally,
64Cu-labeled radiopharmaceuticals can be produced at a central facility and distributed to remote hospitals. The successful application of
64Cu as a diagnostic agent may also lead to potential targeted radionuclide therapy with
67Cu (t
1/2 = 61.9 h; β
−, E
max = 0.141 MeV [100%]) providing a promising theranostic pair.
Banerjee et al. reported the synthesis and preclinical evaluation of a series of five PSMA inhibitors labeled with
64Cu [
139]. The main focus of the study was given on the effect of various chelators on PSMA-targeted ureas with respect to their pharmacokinetics for in vivo PET imaging. The investigated chelators are 1,4,7,10-tetraazacyclodoadecane-
N,
N′,
N″,
N′′′-tetraacetic acid, (DOTA), oxa-4,7,1-tetraazacyclododecane-4,7,10-triacetic acid, (Oxo-DO3A), 1,4,8,11-tetraazabicyclo[6.6.2]-hexadecane-4,11-diacetic acid, (CB-TE2A), 3,6,9,15-tetraazabicyclo[9.3.1]-pentadeca-1(15),11,13-triene)-3,6,9-triacetic acid (PCTA) and 1,4,7-triazacyclononane-1,4,7-triacetic acid (NOTA) (compounds
50–
54,
Figure 17). The radiolabeling with
64Cu was performed in acetate buffer and the conditions differed based on the chelator (
Table 6). The metal-free CB-TE2A-conjugated ligand
53 contained two nearly inseparable isomeric peaks by HPLC. This is most probably caused by the presence of an asymmetric center at the Phe moiety of the spacer, located very close to CB-TE2A. Radiolabeling of
53 resulted in two HPLC separable products, named
64Cu-53A and
64Cu-53B. All radioligands were obtained in specific activities in the range 2.9–9.1 GBq/μmol.
64Cu-53A,
64Cu-53B and
64Cu-54 were found to be more hydrophilic despite the presence of two Phe moieties at the spacer. The diastereomeric metalloconjuagtes 53A and 53B were the ones with highest affinity for PSMA. PET imaging as well as ex vivo biodistribution studies showed that
64Cu-50 and
64Cu-53A/B (NOTA and CB-TE2A chelators, respectively) exhibited high in vivo stability as evidenced by their lower liver uptake than that of the other three conjugates (blood: 1.06 ± 0.29, 1.87 ± 0.6, 0.20 ± 0.03 and 0.20 ± 0.07 % IA/g; liver: 8.63 ± 0.92, 17.04 ± 1.44, 1.68 ± 0.38 and 1.63 ± 0.72 % IA/g for
64Cu-50,
64Cu-51,
64Cu-53A and
64Cu-53B at 2 h p.i.). An additional finding of this study was that liver uptake and blood concentration were much lower at all time points for
64Cu-53A/B compared to that of
64Cu-50, suggesting a higher in vivo stability of CB-TE2A-conjugated
64Cu-53A/B than of NOTA-conjugated
64Cu-50. That might also be related to the higher hydrophilicity of
64Cu-53A/B compared to
64Cu-50. On the other hand, the high liver uptake and the slow blood clearance for the
64Cu-DOTA-conjugated radiotracer were indicative of free Cu(II), which is accumulated in liver [
140,
141]. Noteworthy is also the fact that negatively charged
64Cu-labeled NOTA-conjugated PSMA inhibitor,
64Cu-50, similarly to
99mTc-oxo compounds [
99], exhibited higher kidney and spleen uptake compared to
64Cu-53A/B (kidney: 125 ± 42, 26 ± 9 and 25 ± 11 % IA/g; spleen: 13.42 ± 1.18, 0.39 ± 0.30 and 0.90 ± 0.23 % IA/g for
64Cu-50,
64Cu-53A and
64Cu-53B at 2 h p.i.) Furthermore, although the PSMA-mediated renal uptake has been shown to be specific by several groups, it is interesting that variations are observed not only in regard to the absolute kidney uptake, but also with regard to the renal elimination.
The NODAGA-conjugated PSMA inhibitor CC34 (
Figure 15), was also radiolabeled with
64Cu in ammonium acetate buffer, pH 5.4, to completion within 30 min at 95 °C, and was used without further purification. The specific activity of
64Cu-CC34 was 40 MBq/nmol and a log
P of −3.01 ± 0.06. When CC34 was labeled with
64Cu, the resulting tracer exhibited a slightly higher lipophilicity than did
68Ga-CC34 (Log
P = −3.54 ± 0.06) [
136].
64Cu-CC34 was preclinically evaluated in LNCaP xenografted mice. A high affinity towards PSMA on LNCaP cells was shown (K
D = 27.5 ± 2.7 nM). Furthermore, the versatility of NODAGA was exploited for efficient labeling also with
64Cu, which allows for the conduction of biodistribution/imaging studies at later time points compared to
68Ga-CC34, and thus a complete assessment of the pharmacokinetics of the radiotracer.
64Cu-CC34 is specifically taken up by the PSMA positive organs at early time points and washed out faster from the PSMA positive organs compared to uptake in tumor, leading to improved tumor to background ratios (tumor/blood: 41 ± 10 and 114 ± 17, tumor/muscles: 61 ± 18 and 103 ± 21 at 1 and 4 h p.i., respectively). The very low liver uptake of
64Cu-CC34 at all time points in combination with the short blood circulation, comparable to what was also reported for the CB-2A-conjugated PSMA inhibitor from Banerjee et al. [
139] is a strong indication of the excellent in vivo stability of the
64Cu-NODAGA complex.
4.4. 111In-Labeled Urea-Based PSMA Inhibitors
111In has suitable nuclear characteristics (t
½ = 2.8 d, E(γ) = 173, 245 keV) for use as an important SPECT label. Additionally, intraoperative gamma probes are now an important, well-established technology in the management of cancer, particularly in the detection of sentinel lymph nodes [
140]. So far two already established PSMA inhibitors, DOTAGA-(I-y)fk(Sub-KuE) (also named PSMA-I&T) and PSMA-617, have been labeled with
111In, as companion nuclear probes for radioguided surgery and SPECT imaging [
135,
136,
142].
111In-PSMA-I&T exhibited high PSMA-affinity (
natIn-PSMA-I&T: IC
50: 7.5 ± 1.5 nM) and enhanced internalization (104 ± 7%) compared to
177Lu-PSMA-I&T (76 ± 2%) into LNCaP cells. Biodistribution studies in LNCaP tumor-bearing mice (1 h p.i.) revealed slightly reduced background accumulation of
111In-PSMA-I&T compared to
177Lu-PSMA-I&T and comparable tumor uptake of both compounds. These findings led to a somehow improved tumor/background ratios for
111In-PSMAI&T compared to
177Lu-PSMA-I&T at 1 h p.i. (tumor/blood-, tumor/liver-, tumor/intestines-, and tumor/muscle-ratios of 34 ± 8, 32 ± 6, 53 ± 8, and 43 ± 6, respectively, versus 18 ± 9, 7 ± 3, 12 ± 3, and 14 ± 9 for
177Lu-PSMA-I&T) [
135,
142].
111In-PSMA-617 was prepared within 30 min at 95 °C in ammonium acetate buffer pH 5.4 and were used without any further purification with a specific activity of 10 MBq/nmol and a K
D value of approximately 5 nM The pharmacokinetics of
111In labeled naphthyl-containing DOTA-conjugated PSMA inhibitor, PSMA-617were excellent, with impressive tumor to background ratios over time (i.e., tumor/blood-, tumor/kidney-, and tumor-muscle ratios of 72 ± 19, 0.2 ± 0.01, 148 ± 61; 772 ± 153, 0.4 ± 0.04, 582 ± 24; 4768 ± 110, 9 ± 2, 2819 ± 864; 5343 ± 1033, 16 ± 2, 1987 ± 177 at 1-, 4-, 24-, and 48-h p.i. respectively) [
136].
4.5. 177Lu-Labeled Urea-Based PSMA Inhibitors
The development of new radiolabeled specific biomarkers is mainly focused on the early diagnosis and prognostic prediction continuing with the adaptation of their therapeutic interventions that are individually optimized. Taking into account the theranostic approach, an ideal radiopharmaceutical should be able to combine the ability to be used for both diagnosis and therapy when labeled with a diagnostic or a suitable therapeutic radionuclide respectively. The beta emitter 177Lu can be considered as an appropriate therapeutic unit during the construction of a therapeutic nuclear probe due to its favorable characteristics (t1/2 = 6.73 days, Eβmax = 497 keV, Eγ = 113 keV (6.4%) and 208 keV (11%)).
The tailor-made DOTA-conjugated PSMA inhibitor, PSMA-617, has also been reported to be efficiently labeled with
177Lu [
133] in addition to
68Ga. Radiolabeling with
177Lu reached a radiochemical yield of greater than 99% at low amount of precursor (0.5 mg, 0.5 nmol) in sodium acetate buffer, pH 5. The specific activity was in the range of 4–40 GBq/mmol.
177Lu-PSMA-617 exhibited high stability for at least 72 h, high binding affinity towards PSMA (K
i = 6.91 ± 1.32 nM for
natLu-PSMA-617) along with enhanced internalization rate into the LNCaP prostate cancer cells.
68Ga-labeled PSMA-617 was specifically internalized up to 17.67 ± 4.34% injected activity/10
6 LNCaP cells and
177Lu-labeled PSMA-617 up to 17.51 ± 3.99% injected activity/10
6 LNCaP cells, both at 37 °C. The in vivo performance of the
177Lu-PSMA-617 was characterized by high and retained tumor uptake combined with a rapid clearance from the kidneys within 24 h p.i., which led to improved tumor to background ratios over time (1058 (tumor to blood) and 529 (tumor to muscle), 24 h p.i.). It is worth noting that
177Lu-PSMA-617 exhibited similar pharmacokinetics as
111In-PSMA-617 [
131].
177Lu-PSMA-617 seems attractive for endoradiotherapy due to its higher tumor uptake at later time points, lower spleen accumulation, and the highly efficient clearance from the kidneys. The clinical evaluation of
177Lu-PSMA-617 is under way.
Under the same concept of the theranostic approach, the PSMA inhibitors DOTAGA-ffk(Sub-KuE) and DOTAGA-(I-y)fk(Sub-KuE) (also named PSMA I&T) were also labeled with
177Lu and evaluated preclinically [
135]. Radiolabeling with
177Lu performed in ammonium acetate buffer, pH 5, and the radiolabeled products were achieved in a specific activity of more than 38 GBq/μmol. Both cold lutetium analogues exhibited high affinity towards PSMA (IC
50:
natLu-DOTAGA-ffk(Sub-KuE): 13.1 ± 2.2 nM,
natLu-PSMA I&T: 7.9 ± 2.4 nM). Compared with
177Lu-DOTAGA-ffk(Sub-KuE), tumor targeting of
177Lu-PSMA I&T was fast, with the highest uptake in tumor (7.96 ± 1.76 % IA/g at 1 h p.i.) xenografts and kidneys (107 ± 16 % IA/g at 1 h p.i.) (both PSMA-specific).
177Lu-PSMA I&T exhibits suitable targeting and retention characteristics and an assessment of this compound for application in endoradiotherapy is currently ongoing.