Evaluation of Maltose-Based Cationic Liposomes with Different Hydrophobic Tails for Plasmid DNA Delivery
Abstract
:1. Introduction
2. Results and Discussions
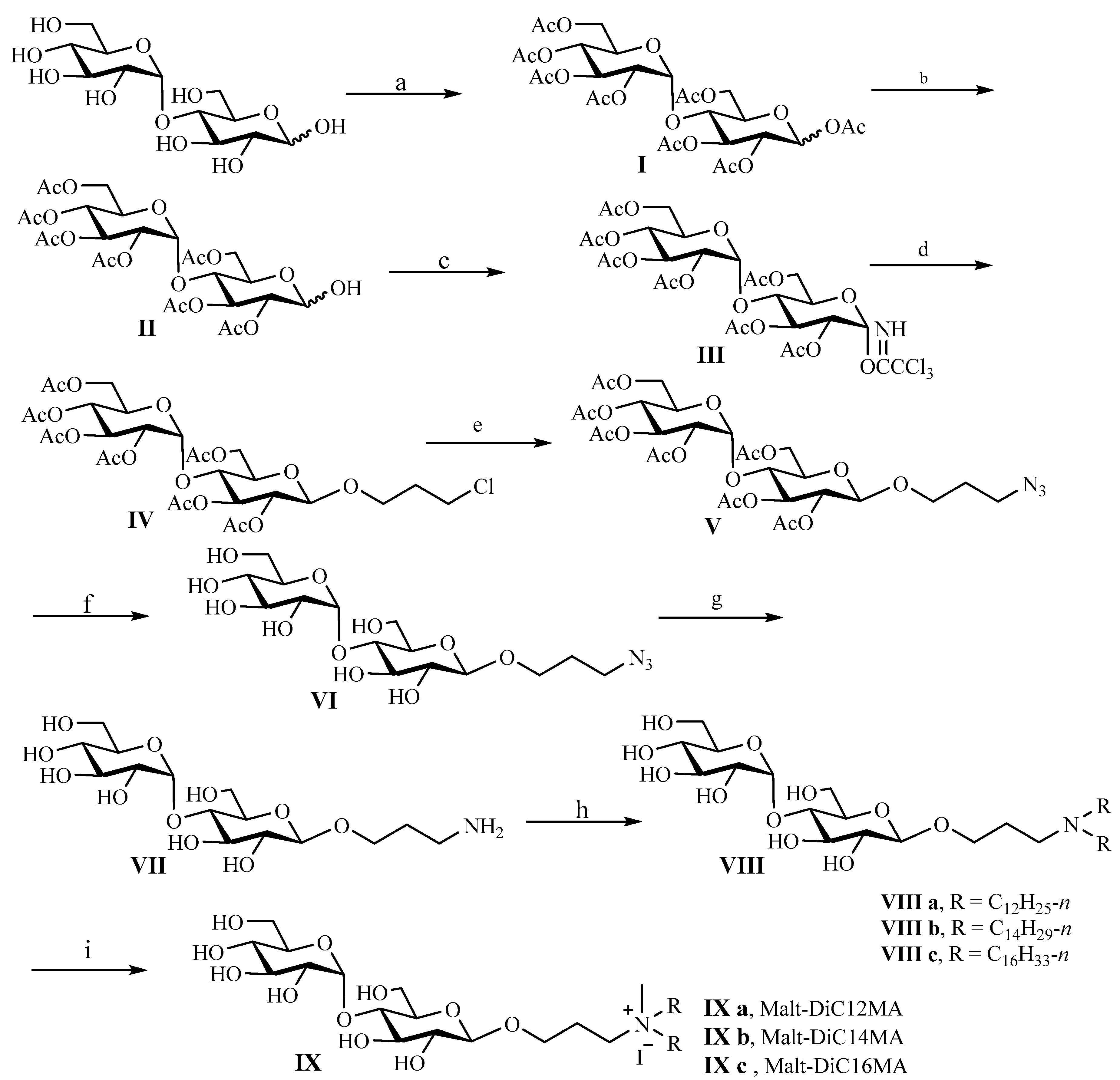
2.1. Synthesis of Cationic Glycolipids and Their Structural Elucidation
2.2. Characterization of Cationic Liposome
2.3. Gel Retardation Assay
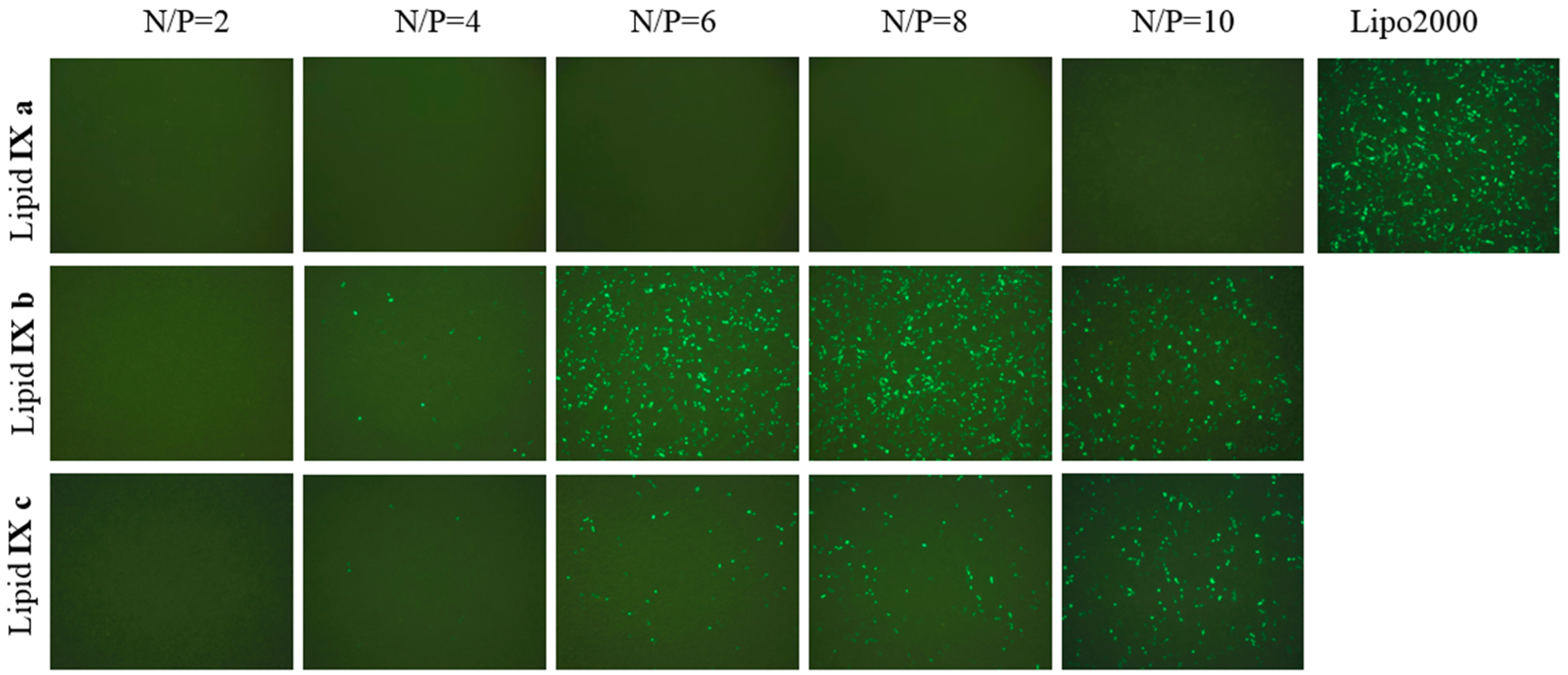
2.4. Transfection
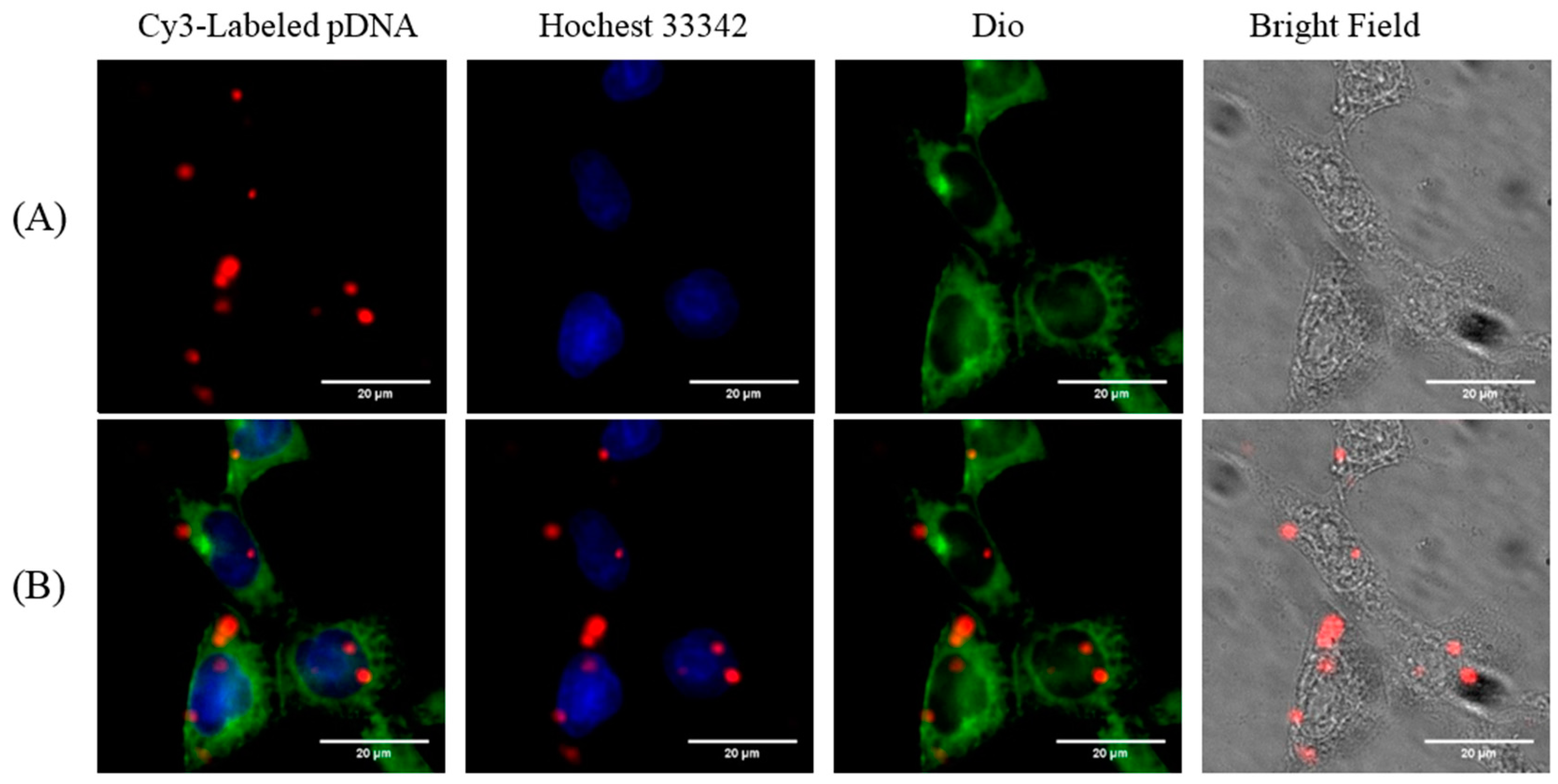
2.5. Cellular Uptake
2.6. Cytotoxicity Assay
3. Experimental Section
3.1. Reagents and General Information
3.1.1. Synthesis of Cationic Glycolipids IX a (Malt-DiC12MA)
3.1.2. Synthesis of Cationic Glycolipids IX b (Malt-DiC14MA)
3.1.3. Synthesis of Cationic Glycolipids IX c (Malt-DiC16MA)
3.2. Preparation of Liposome and Liposome/pDNA Complexes (Lipoplexes)
3.3. Cells Culture
3.4. Gel Retardation Assay
3.5. Dynamic Light Scattering
3.6. Morphology Study by Atomic Force Microscopy
3.7. Transfection Biology (GFP Expression Analysis)
3.8. Cellular Uptake
3.9. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xia, Y.; Tian, J.; Chen, X. Effect of surface properties on liposomal siRNA delivery. Biomaterials 2016, 79, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhao, B.; Jiang, H.; Wang, B.; Ma, B. Cationic lipids and polymers mediated vectors for delivery of siRNA. J. Control. Release 2007, 123, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Juliano, R.L.; Ming, X.; Nakagawa, O. Cellular uptake and intracellular trafficking of antisense and siRNA oligonucleotides. Adv. Drug Deliv. Rev. 2015, 87, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Pei, Y.T.; Wei, C.; Hedrick, J.L.; Yi, Y.Y. Co-delivery of drugs and plasmid DNA for cancer therapy. Adv. Drug Deliv. Rev. 2015, 98, 41–63. [Google Scholar]
- Yin, L.; Song, Z.; Qu, Q.; Kim, K.H.; Zheng, N.; Yao, C.; Chaudhury, I.; Tang, H.; Gabrielson, N.P.; Uckun, F.M. Supramolecular self-assembled nanoparticles mediate oral delivery of therapeutic TNF-α siRNA against systemic inflammation. Angew. Chem. Int. Ed. Engl. 2013, 52, 7208–7225. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.R.; Wang, H.F.; Zhao, J.; Peng, Y.Y.; Wang, J.; Guinn, B.A.; Huang, L.Q. Recent developments in the use of adenoviruses and immunotoxins in cancer gene therapy. Cancer Gene Ther. 2007, 14, 599–615. [Google Scholar] [CrossRef] [PubMed]
- Elaneed, A. An overview of current delivery systems in cancer gene therapy. J. Control. Release 2004, 94, 1–14. [Google Scholar] [CrossRef]
- Aiuti, A.; Cattaneo, F.; Galimberti, S.; Benninghoff, U.; Cassani, B.; Callegaro, L.; Scaramuzza, S.; Andolfi, G.; Mirolo, M.; Brigida, I. Gene therapy for immunodeficiency due to adenosine deaminase deficiency. N. Engl. J. Med. 2009, 360, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Sobajima, S.; Kim, J.S.; Gilbertson, L.G.; Kang, J.D. Gene therapy for degenerative disc disease. Gene Ther. 2004, 11, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Hui, K.M. Synthesis of a novel series of cationic lipids that can act as efficient gene delivery vehicles through systematic heterocyclic substitution of cholesterol derivatives. Gene Ther. 2001, 8, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Kanasty, R.; Dorkin, J.R.; Vegas, A.; Anderson, D. Delivery materials for siRNA therapeutics. Nat. Mater. 2013, 12, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Verma, I.M.; Somia, N. Gene therapy—Promises, problems and prospects. Nature 1997, 389, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Mrsny, R.J. Special feature: A survey of the recent patent literature on the delivery of genes and oligonucleotides. J. Drug Target. 2000, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Knowles, M.R.; Hohneker, K.W.; Zhou, Z.; Olsen, J.C.; Noah, T.L.; Hu, P.C.; Leigh, M.W.; Engelhardt, J.F.; Edwards, L.J.; Jones, K.R. A controlled study of adenoviral-vector-mediated gene transfer in the nasal epithelium of patients with cystic fibrosis. N. Engl. J. Med. 1995, 333, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Jolly, D. Viral vector systems for gene therapy. Cancer Gene Ther. 1994, 1, 51–64. [Google Scholar] [PubMed]
- Bajaj, A.; Kondaiah, P.; Bhattacharya, S. Gene transfection efficacies of novel cationic gemini lipids possessing aromatic backbone and oxyethylene spacers. Biomacromolecules 2008, 9, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Nunes, F.A.; Berencsi, K.; Furth, E.E.; Gönczöl, E.; Wilson, J.M. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc. Natl. Acad. Sci. USA 1994, 91, 4407–4411. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.E.; Ehrhardt, A.; Kay, M.A. Progress and problems with the use of viral vectors for gene therapy. Nat. Rev. Genet. 2003, 4, 346–358. [Google Scholar] [CrossRef] [PubMed]
- Akinc, A.; Thomas, M.; Klibanov, A.M.; Langer, R. Exploring polyethylenimine-mediated DNA transfection and the proton sponge hypothesis. J. Gene Med. 2005, 7, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Pathak, A.; Patnaik, S.; Gupta, K.C. Recent trends in non-viral vector-mediated gene delivery. Biotechnol. J. 2009, 4, 1559–1572. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhang, S.; Zhang, Y.; Cui, S.; Chen, H.; Zhi, D.; Zhen, Y.; Zhang, S.; Huang, L. Tri-peptide cationic lipids for gene delivery. J. Mater. Chem. B 2015, 3, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Crystal, R.G. Transfer of genes to humans: Early lessons and obstacles to success. Science 1995, 270, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Huang, L. Recent advances in nonviral vectors for gene delivery. Acc. Chem. Res. 2012, 45, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Bishop, C.J.; Ketola, T.M.; Tzeng, S.Y.; Sunshine, J.C.; Urtti, A.; Lemmetyinen, H.; Vuorimaalaukkanen, E.; Yliperttula, M.; Green, J.J. The Effect and Role of Carbon Atoms in Poly(β-amino ester)s for DNA Binding and Gene Delivery. J. Am. Chem. Soc. 2013, 135, 6951–6957. [Google Scholar] [CrossRef] [PubMed]
- Qiao, W.; Zhou, M. Hydroxyl-modified cationic lipids with a carbamate linkage as gene delivery vehicles. Eur. J. Lipid Sci. Technol. 2013, 115, 483–489. [Google Scholar] [CrossRef]
- Felgner, P.L.; Ringold, G.M. Cationic liposome-mediated transfection. Nature 1989, 337, 387–388. [Google Scholar] [CrossRef] [PubMed]
- Felgner, P.L.; Danielsen, M. Lipofection: A Highly Efficient, Lipid-Mediated DNA-Transfection Procedure. Proc. Natl. Acad. Sci. USA 1987, 84, 7413–7417. [Google Scholar] [CrossRef] [PubMed]
- Ong, Z.Y.; Yang, C.; Gao, S.J.; Ke, X.Y.; Hedrick, J.L.; Yan, Y.Y. Galactose-functionalized cationic polycarbonate diblock copolymer for targeted gene delivery to hepatocytes. Macromol. Rapid Commun. 2013, 34, 1714–1720. [Google Scholar] [CrossRef] [PubMed]
- Han, S.E.; Kang, H.; Shim, G.Y.; Sun, J.K.; Choi, H.G.; Kim, J.; Hahn, S.K.; Oh, Y.K. Cationic derivatives of biocompatible hyaluronic acids for delivery of siRNA and antisense oligonucleotides. J. Drug. Target. 2009, 17, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, S.; Sato, A.; Nishikawa, M.; Yamashita, F.; Hashida, M. Mannose receptor-mediated gene transfer into macrophages using novel mannosylated cationic liposomes. Gene Ther. 2000, 7, 292–299. [Google Scholar] [CrossRef]
- Nishikawa, M.; Kawakami, S.; Yamashita, F.; Hashida, M. Glycosylated Cationic Liposomes for Carbohydrate Receptor-Mediated Gene Transfer. Methods. Enzymol. 2003, 373, 384–399. [Google Scholar] [PubMed]
- Tauber, R.; Reher, K.; Helling, K.; Scherer, H. Complex carbohydrates—Structure and function with respect to the glycoconjugate composition of the cupula of the semicircular canals. Biol. Sci. Space 2002, 15, 363–366. [Google Scholar]
- El-Boubbou, K.; Zhu, D.C.; Vasileiou, C.; Borhan, B.; Prosperi, D.; Li, W.; Huang, X. Magnetic glyco-nanoparticles: A tool to detect, differentiate, and unlock the glyco-codes of cancer via magnetic resonance imaging. J. Am. Chem. Soc. 2010, 132, 4490–4499. [Google Scholar] [CrossRef] [PubMed]
- Dwek, R.A. Glycobiology: Toward Understanding the Function of Sugars. Chem. Rev. 1996, 96, 683–720. [Google Scholar] [CrossRef] [PubMed]
- Collins, B.E.; Paulson, J.C. Cell surface biology mediated by low affinity multivalent protein-glycan interactions. Curr. Opin. Chem. Biol. 2004, 8, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Bucior, I.; Burger, M.M. Carbohydrate-carbohydrate interactions in cell recognition. Curr. Opin. Struct. Biol. 2004, 14, 631–637. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Wang, S.; Liu, M.; Zhao, C.; Xiang, S.; Zeng, Y. Design, synthesis and in vitro evaluation of d-glucose-based cationic glycolipids for gene delivery. Org. Biomol. Chem. 2015, 14, 1611–1622. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, Z.; Blessington, D.; Li, H.; Busch, T.M.; Madrak, V.; Miles, J.; Chance, B.; Glickson, J.D.; Zheng, G. Pyropheophorbide 2-deoxyglucosamide: A new photosensitizer targeting glucose transporters. Bioconjug. Chem. 2003, 14, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Zhang, Z.; Zhang, Y.; Lv, H.; Zhou, J.; Li, C.; Hou, L.; Zhang, Q. Dual-functional liposomes based on pH-responsive cell-penetrating peptide and hyaluronic acid for tumor-targeted anticancer drug delivery. Biomaterials 2012, 33, 9246–9258. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Kuo, J.H.; Li, C.H.; Barhate, N.B.; Hon, S.W.; Li, T.W.; Chao, S.D.; Liu, C.C.; Li, Y.C.; Chang, I. Catalytic Nucleophilic Acyl Substitution of Anhydrides by Amphoteric Vanadyl Triflate. Org. Lett. 2001, 3, 3729–3732. [Google Scholar] [CrossRef] [PubMed]
- Wendeln, C.; Heile, A.; Arlinghaus, H.F.; Ravoo, B.J. Carbohydrate microarrays by microcontact printing. Langmuir 2010, 26, 4933–4940. [Google Scholar] [CrossRef] [PubMed]
- Zu, R.; Zuhorn, I.S.; Hoekstra, D. How cationic lipids transfer nucleic acids into cells and across cellular membranes: Recent advances. J. Control. Release 2012, 166, 46–56. [Google Scholar]
- Szoka, F.C.; Xu, Y.; Zelphati, O. How are Nucleic Acids Released in Cells from Cationic Lipid-Nucleic Acid Complexes? J. Liposome Res. 2008, 24, 291. [Google Scholar]
- Morille, M.; Passirani, C.; Vonarbourg, A.; Clavreul, A.; Benoit, J.P. Progress in developing cationic vectors for non-viral systemic gene therapy against cancer. Biomaterials 2008, 29, 3477–3496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, E.H. The Effect of the Particle Size on Cationic Lipid-Mediated Gene Delivery; University of Wisconsin: Madison, WI, USA, 1998. [Google Scholar]
- Leclerc, L.; Rima, W.; Boudard, D.; Pourchez, J.; Forest, V.; Bin, V.; Mowat, P.; Perriat, P.; Tillement, O.; Grosseau, P. Size of submicrometric and nanometric particles affect cellular uptake and biological activity of macrophages in vitro. Inhal. Toxicol. 2012, 24, 580–588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rumschöttel, J.; Kosmella, S.; Prietzel, C.; Appelhans, D.; Koetz, J. Change in size, morphology and stability of DNA polyplexes with hyperbranched poly(ethyleneimines) containing bulky maltose units. Colloids Surf. B Biointerfaces 2016, 138, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, A.; Kondaiah, P.; Bhattacharya, S. Effect of the nature of the spacer on gene transfer efficacies of novel thiocholesterol derived gemini lipids in different cell lines: A structure-activity investigation. J. Med. Chem. 2008, 51, 2533–2540. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.Q.; Chen, J.L.; Lv, T.F.; He, C.X.; Tang, G.P.; Liang, W.Q.; Tabata, Y.; Gao, J.Q. N/P ratio significantly influences the transfection efficiency and cytotoxicity of a polyethylenimine/chitosan/DNA complex. Biol. Pharm. Bull. 2009, 32, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Hansen, M.B.; Nielsen, S.E.; Berg, K. Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J. Immunol. Methods 1989, 119, 203–210. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds IX a–IX c are available from the authors.



| Cationic Liposomes | Average Size (nm) | PDI a | Zeta Potential (mV) |
|---|---|---|---|
| Malt-DiC12MA IX a | 90.3 ± 3.5 | 0.221 ± 0.031 | +46.0 ± 5.6 |
| Malt-DiC14MA IX b | 63.4 ± 0.7 | 0.167 ± 0.025 | +36.2 ± 6.8 |
| Malt-DiC16MA IX c | 109.7 ± 2.6 | 0.198 ± 0.089 | +37.4 ± 4.3 |
| Lipid/DNA Complex | N/P Ratio | Average Size (nm) | PDI | Zeta Potential (mV) |
|---|---|---|---|---|
| Malt-DiC14MA/DNA | 4:1 | 88.6 ± 6.8 | 0.125 ± 0.056 | +15.1 ± 1.6 |
| Malt-DiC14MA/DNA | 6:1 | 98.6 ± 7.5 | 0.365 ± 0.036 | +14.6 ± 3.5 |
| Malt-DiC14MA/DNA | 8:1 | 78.6 ± 11.5 | 0.154 ± 0.012 | +16.0 ± 7.8 |
| Malt-DiC14MA/DNA | 10:1 | 90.3 ± 4.5 | 0.365 ± 0.046 | +20.4 ± 3.7 |
| Malt-DiC16MA/DNA | 4:1 | 120.6 ± 9.9 | 0.157 ± 0.036 | +19.3 ± 8.6 |
| Malt-DiC16MA/DNA | 6:1 | 127.6 ± 11.2 | 0.258 ± 0.014 | +21.8 ± 6.5 |
| Malt-DiC16MA/DNA | 8:1 | 146.8 ± 5.6 | 0.147 ± 0.045 | +22.6 ± 1.2 |
| Malt-DiC16MA/DNA | 10:1 | 110.6 ± 11.5 | 0.375 ± 0.046 | +26.8 ± 4.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, B.; Deng, L.; Liu, M.; Zeng, Y. Evaluation of Maltose-Based Cationic Liposomes with Different Hydrophobic Tails for Plasmid DNA Delivery. Molecules 2017, 22, 406. https://doi.org/10.3390/molecules22030406
Li B, Deng L, Liu M, Zeng Y. Evaluation of Maltose-Based Cationic Liposomes with Different Hydrophobic Tails for Plasmid DNA Delivery. Molecules. 2017; 22(3):406. https://doi.org/10.3390/molecules22030406
Chicago/Turabian StyleLi, Bo, Liangliang Deng, Meiyan Liu, and Youlin Zeng. 2017. "Evaluation of Maltose-Based Cationic Liposomes with Different Hydrophobic Tails for Plasmid DNA Delivery" Molecules 22, no. 3: 406. https://doi.org/10.3390/molecules22030406





