Preparation, Characterization, and Biological Activities of Topical Anti-Aging Ingredients in a Citrus junos Callus Extract
Abstract
:1. Introduction
2. Results and Discussion
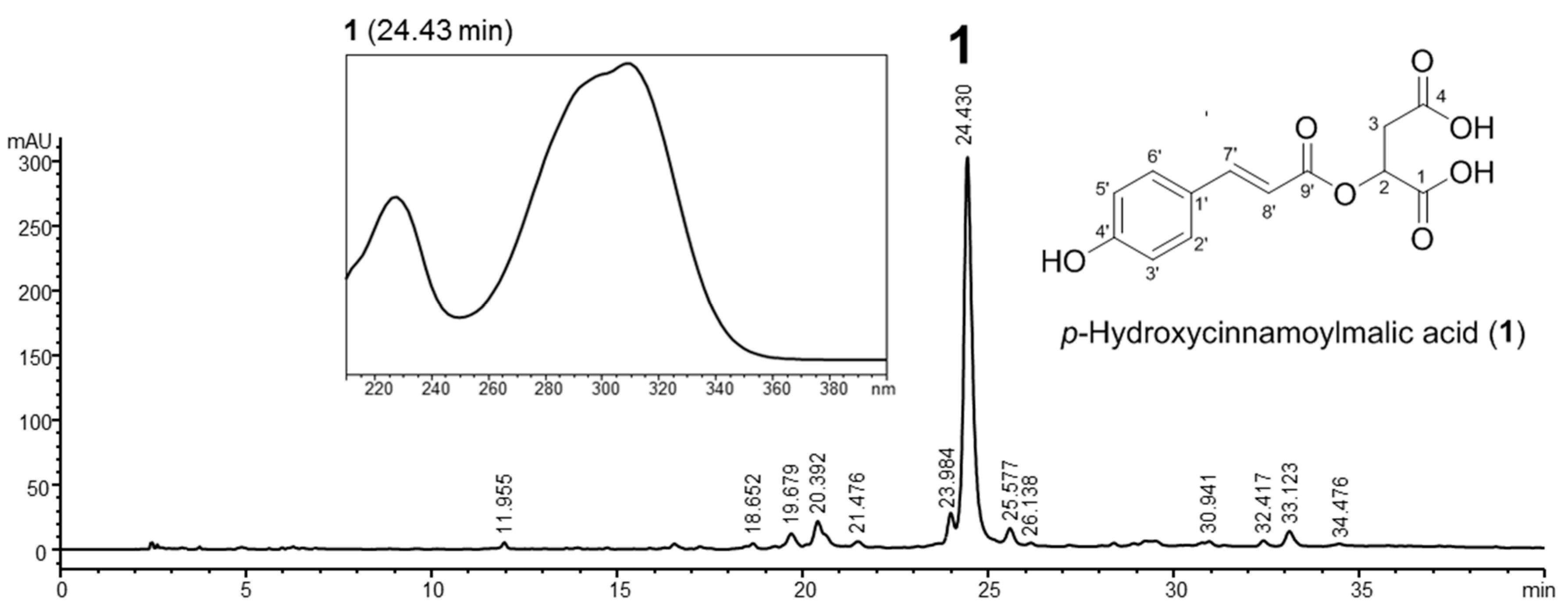
2.1. Preparation and Characterization of Callus Extract from Citrus junos
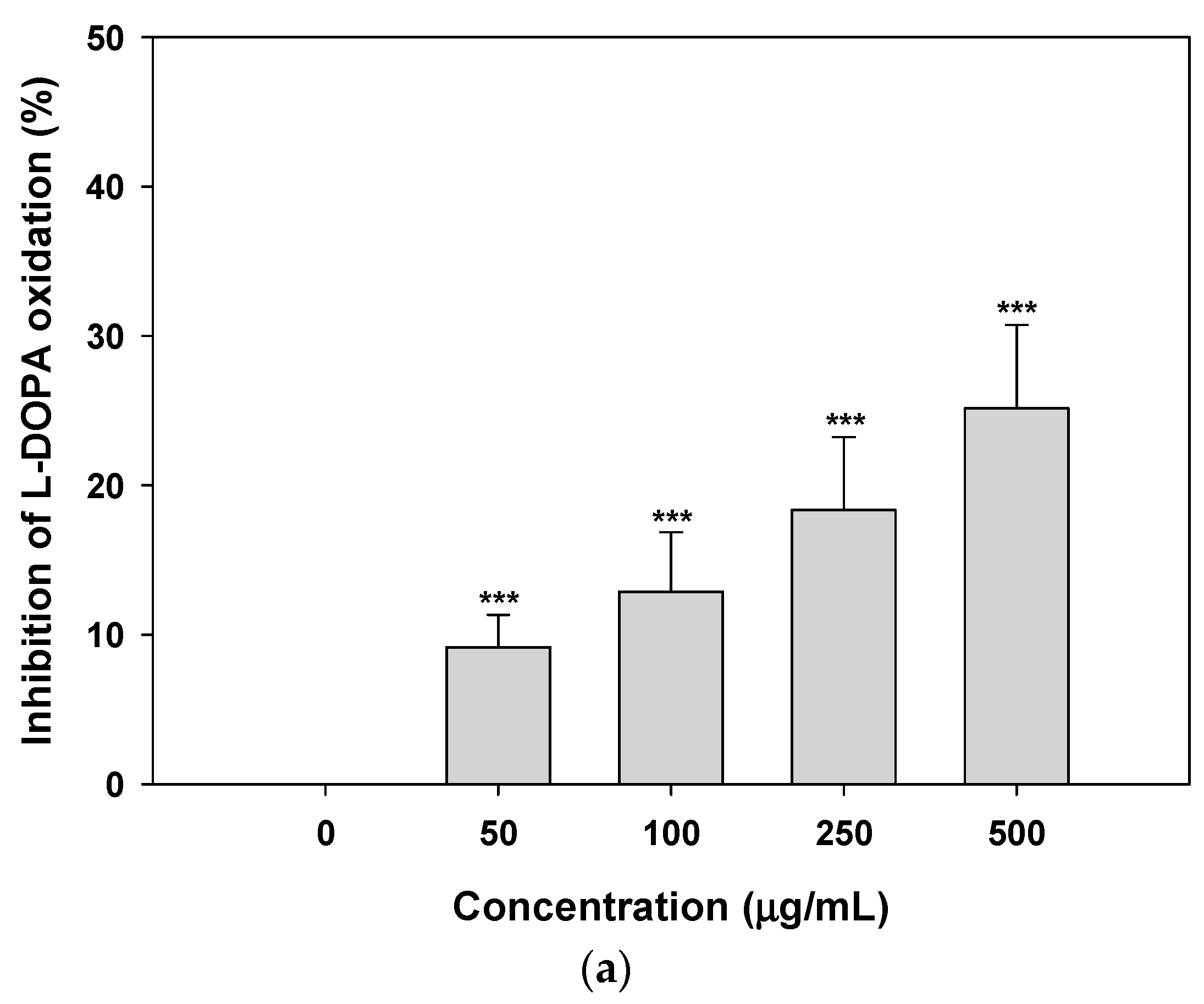
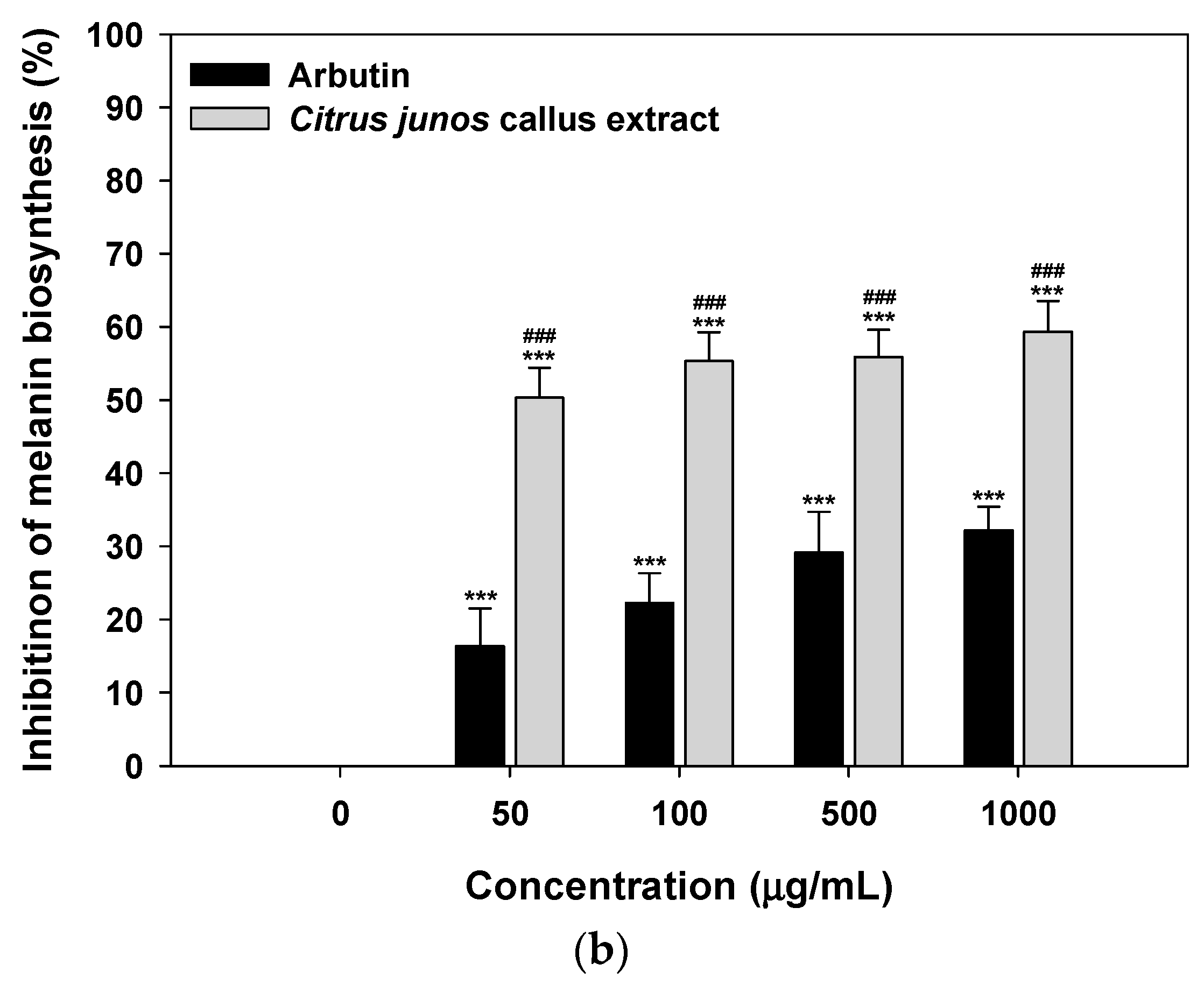
2.2. Inhibition of Tyrosinase Activity and Melanin Biosynthesis
2.3. Skin-Regeneration Activities of Citrus junos Callus Extract
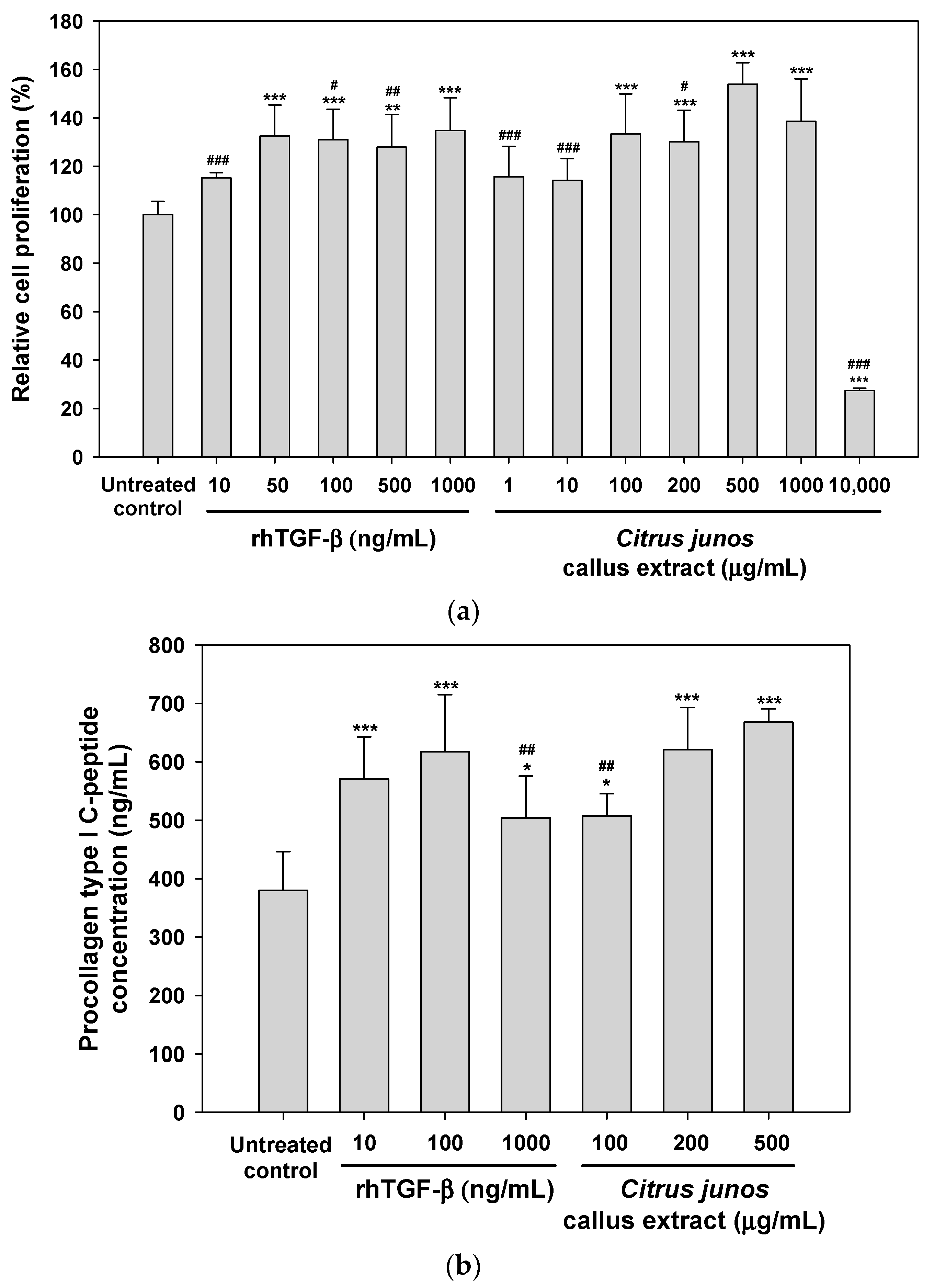
2.3.1. Effects of the Extract on Fibroblast Proliferation and Procollagen Synthesis
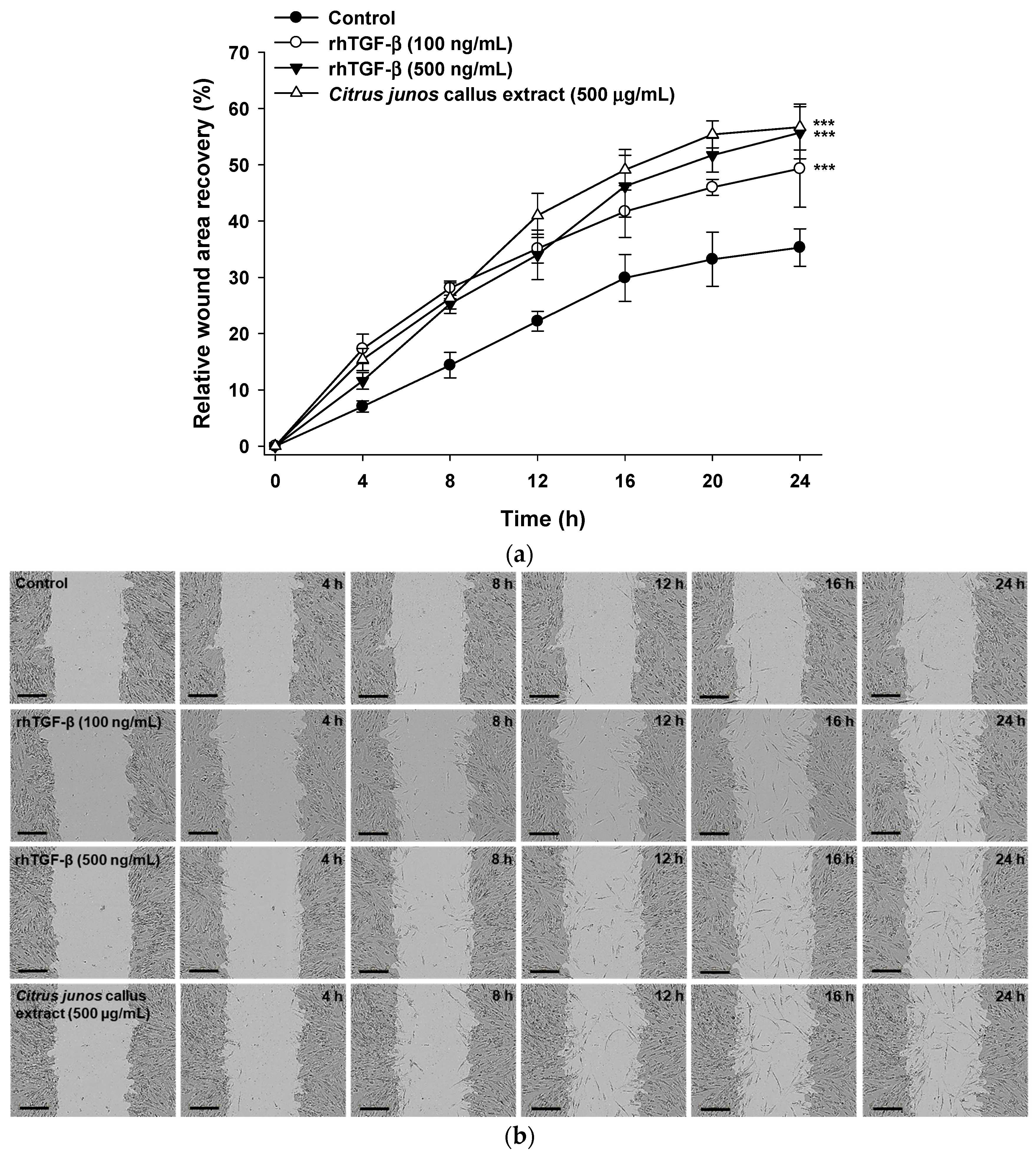
2.3.2. In Vitro Scratch Wound Recovery Effect of Citrus junos Callus Extract
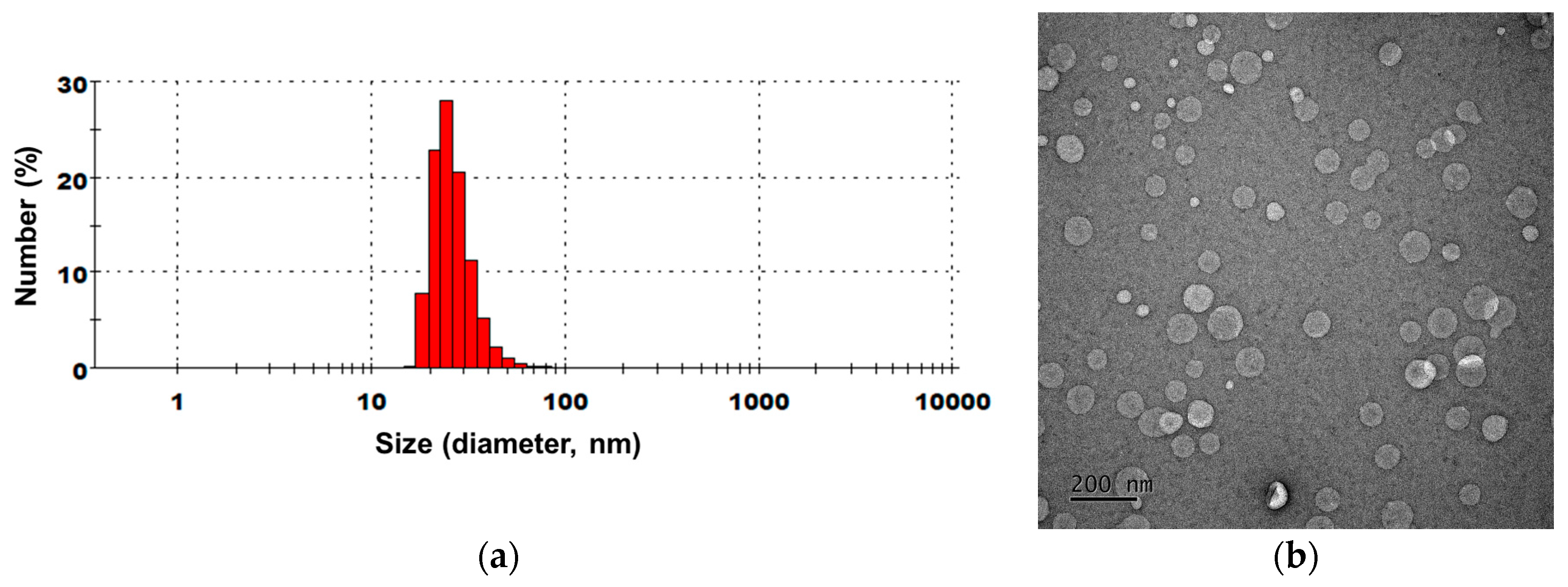
2.4. Characterization of NLs Containing Citrus junos Callus Extract
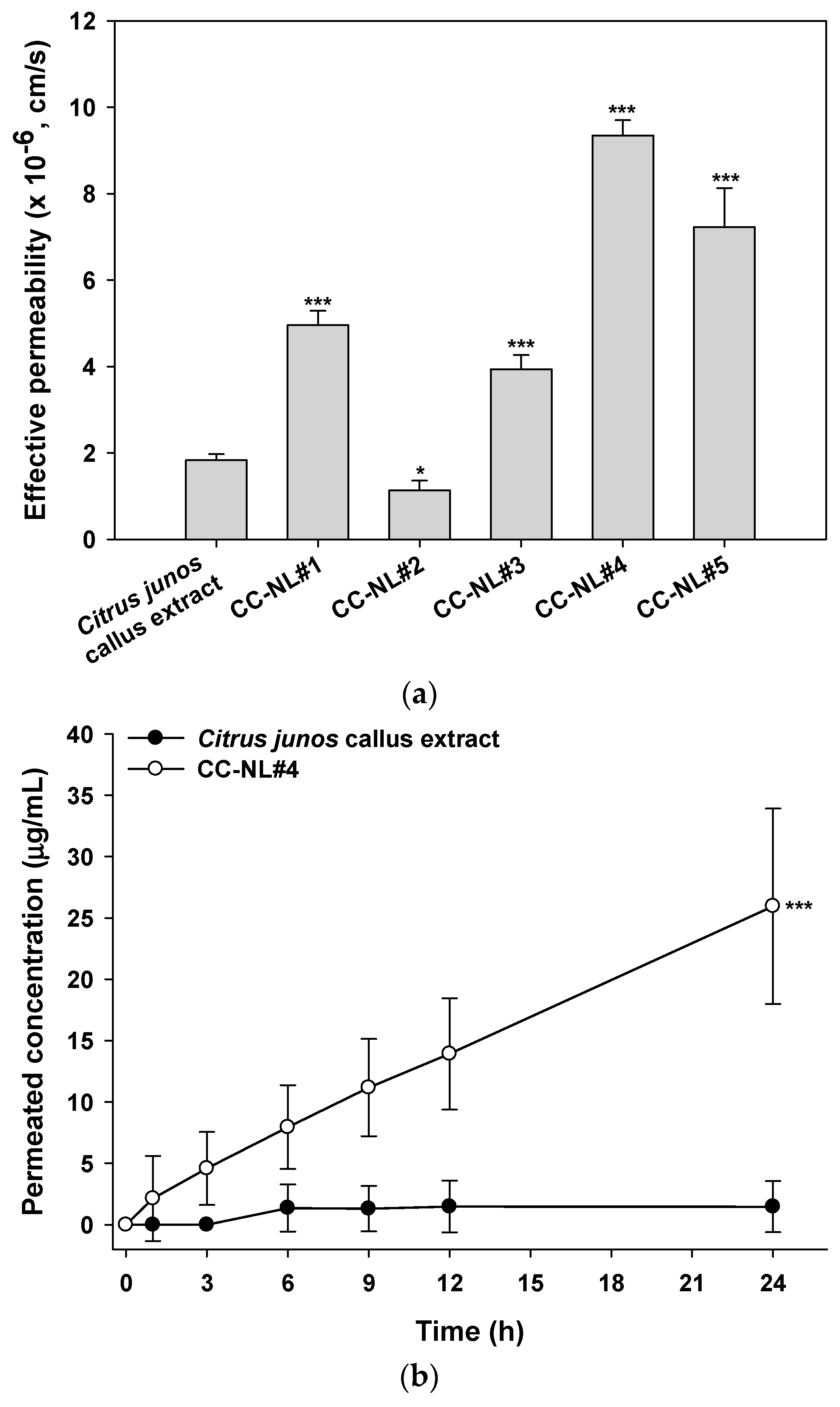
2.5. In Vitro Skin Permeability of Citrus junos Callus Extract-Loaded NLs
3. Materials and Methods
3.1. Materials
3.2. Preparation and Characterization of a Callus Extract of Citrus junos
3.2.1. Preparation of Callus Extract
3.2.2. Characterization of the Callus Extract by HPLC
3.2.3. Determination of Antioxidant Capacity for the Callus Extract
3.3. Skin-Lightening Activity
3.3.1. Inhibition of l-DOPA Oxidation
3.3.2. Inhibition of Melanin Biosynthesis in Melanocytes
3.4. Skin Regeneration Activity
3.4.1. Fibroblast Proliferation Assay
3.4.2. Procollagen Synthesis Assay
3.4.3. In Vitro Scratch Wound Recovery Assay
3.5. Preparation and Characterization of Citrus junos Callus Extract-Loaded NLs
3.6. In Vitro Skin Permeability of Citrus junos Callus Extract-Loaded NLs
3.6.1. Permeation across an Artificial Skin
3.6.2. In Vitro Permeability through a Human Epidermal Layer
3.7. Statistics
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rao, S.R.; Ravishankar, G.A. Plant cell cultures: Chemical factories of secondary metabolites. Biotechnol. Adv. 2002, 20, 101–153. [Google Scholar] [PubMed]
- Ochoa-Villarreal, M.; Howat, S.; Hong, S.M.; Jang, M.O.; Jin, Y.W.; Lee, E.K.; Loake, G.J. Plant cell culture strategies for the production of natural products. BMB Rep. 2016, 49, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Bourgaud, F.; Gravot, A.; Milesi, S.; Gontier, E. Production of plant secondary metabolites: A historical perspective. Plant Sci. 2001, 161, 839–851. [Google Scholar] [CrossRef]
- Barbulova, A.; Apone, F.; Colucci, G. Plant cell cultures as source of cosmetic active ingredients. Cosmetics 2014, 1, 94–104. [Google Scholar] [CrossRef]
- Hirota, R.; Roger, N.N.; Nakamura, H.; Song, H.S.; Sawamura, M.; Suganuma, N. Anti-inflammatory effects of limonene from yuzu (Citrus junos Tanaka) essential oil on eosinophils. J. Food Sci. 2010, 75, H87–H92. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Kuraya, E.; Nakada, S.; Touyama, A.; Itoh, S. Improving the antioxidant functionality of Citrus junos Tanaka (yuzu) fruit juice by underwater shockwave pretreatment. Food Chem. 2017, 216, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Emim, J.A.; Oliveira, A.B.; Lapa, A.J. Pharmacological evaluation of the anti-inflammatory activity of a citrus bioflavonoid, hesperidin, and the isoflavonoids, duartin and claussequinone, in rats and mice. J. Pharm. Pharmacol. 1994, 46, 118–122. [Google Scholar] [CrossRef]
- Manthey, J.A.; Guthrie, N.; Grohmann, K. Biological properties of citrus flavonoids pertaining to cancer and inflammation. Curr. Med. Chem. 2001, 8, 135–153. [Google Scholar] [CrossRef] [PubMed]
- Yusof, S.; Mohd Ghazali, H.; Swee, K.G. Naringin content in local citrus fruits. Food Chem. 1990, 37, 113–121. [Google Scholar] [CrossRef]
- Lv, X.; Zhao, S.; Ning, Z.; Zeng, H.; Shu, Y.; Tao, O.; Xiao, C.; Lu, C.; Liu, Y. Citrus fruits as a treasure trove of active natural metabolites that potentially provide benefits for human health. Chem. Cent. J. 2015, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Kilci, A.; Gocmen, D. Phenolic acid composition, antioxidant activity and phenolic content of tarhana supplemented with oat flour. Food Chem. 2014, 151, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Jeon, B.K.; Mun, Y.J.; Kim, Y.M.; Lee, Y.E.; Woo, W.H. Effect of Dioscorea aimadoimo on anti-aging and skin moisture capacity. J. Orient. Physiol. Pathol. 2011, 25, 425–430. [Google Scholar]
- Damle, M.; Mallya, R. Development and evaluation of a novel delivery system containing phytophospholipid complex for skin aging. AAPS PharmSciTech 2016, 17, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Agarwal, R. Cosmeceuticals and silibinin. Clin. Dermatol. 2009, 27, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Vinardell, M.P.; Mitjans, M. Nanocarriers for delivery of antioxidants on the skin. Cosmetics 2015, 2, 342–354. [Google Scholar] [CrossRef]
- Goyal, R.; Macri, L.K.; Kaplan, H.M.; Kohn, J. Nanoparticles and nanofibers for topical drug delivery. J. Control. Release 2016, 240, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.P.; Caon, T.; Battisti, M.A.; Silva, H.B.S.; Simões, C.M.O.; Reginatto, F.H.; Campos, A.M. Antioxidant polymeric nanoparticles containing standardized extract of Ilex paraguariensis A. St.-Hil. for topical use. Ind. Crops Prod. 2015, 77, 402–408. [Google Scholar] [CrossRef]
- Ahmad, N.; Faisal, M.; Anis, M.; Aref, I.M. In vitro callus induction and plant regeneration from leaf explants of Ruta graveolens L. S. Afr. J. Bot. 2010, 76, 597–600. [Google Scholar] [CrossRef]
- Liang, Y.S.; Kim, H.K.; Lefeber, A.W.M.; Erkelens, C.; Choi, Y.H.; Verpoorte, R. Identification of phenylpropanoids in methyl jasmonate-treated Brassica rapa leaves using two-dimensional nuclear magnetic resonance spectroscopy. J. Chromatogr. A 2006, 1112, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Guzman, J.D. Natural cinnamic acids, synthetic derivatives and hybrids with antimicrobial activity. Molecules 2014, 19, 19292–19349. [Google Scholar] [CrossRef] [PubMed]
- Vogt, T. Phenylpropanoid biosynthesis. Mol. Plant 2010, 3, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Ververidis, F.; Trantas, E.; Douglas, C.; Vollmer, G.; Kretzschmar, G.; Panopoulos, N. Biotechnology of flavonoids and other phenylpropanoid-derived natural products. Part I: Chemical diversity, impacts on plant biology and human health. Biotechnol. J. 2007, 2, 1214–1234. [Google Scholar] [CrossRef] [PubMed]
- Solecka, D. Role of phenylpropanoid compounds in plant responses to different stress factors. Acta Physiol. Plant 1997, 19, 257–268. [Google Scholar] [CrossRef]
- De, P.; Baltas, M.; Bedos-Belval, F. Cinnamic acid derivatives as anticancer agents—A review. Curr. Med. Chem. 2011, 18, 1672–1703. [Google Scholar] [CrossRef] [PubMed]
- Tawata, S.; Taira, S.; Kobamoto, N.; Zhu, J.; Ishihara, M.; Toyama, S. Synthesis and antifungal activity of cinnamic acid esters. Biosci. Biotechnol. Biochem. 1996, 60, 909–910. [Google Scholar] [CrossRef] [PubMed]
- Sova, M. Antioxidant and antimicrobial activities of cinnamic acid derivatives. Mini Rev. Med. Chem. 2012, 12, 749–767. [Google Scholar] [CrossRef] [PubMed]
- Peperidou, A.; Pontiki, E.; Hadjipavlou-Litina, D.; Voulgari, E.; Avgoustakis, K. Multifunctional cinnamic acid derivatives. Molecules 2017, 22, 1247. [Google Scholar] [CrossRef] [PubMed]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; Oomath, B.D. Antioxidant activity and total phenolics in selected fruits, vegetables and grain products. J. Agric. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Kumar, V.; Moyo, M.; Gruz, J.; Šubrtová, M.; Staden, J.V. Phenolic acid profiles and antioxidant potential of Pelargonium sidoides callus cultures. Ind. Crops Prod. 2015, 77, 402–408. [Google Scholar] [CrossRef]
- Lee, H.S. Tyrosinase inhibitors of Pulsatilla cernua root-derived materials. J. Agric. Food Chem. 2002, 50, 1400–1403. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Are polyphenols antioxidants or pro-oxidants? What do we learn from cell culture and in vivo studies? Arch. Biochem. Biophys. 2008, 476, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.H.; Jo, Y.O.; Cho, C.W.; Son, D.; Park, S.; Rho, J.; Choi, S.Y. Inhibitory effects of cinnamic acid on melanin biosynthesis in skin. Biol. Pharm. Bull. 2008, 31, 946–948. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.I.; Kubo, I. Kinetics of mushroom tyrosinase inhibition by quercetin. J. Agric. Food Chem. 2002, 50, 4108–4112. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Ahn, S.; Chang, H.; Cho, N.S.; Joo, K.; Lee, B.G.; Chang, I.; Hwang, J.S. Influence of N-glycan processing disruption on tyrosinase and melanin synthesis in HM3KO melanoma cells. Exp. Dermatol. 2007, 16, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Mao, G.; Ng, K.Y. Mechanical properties and stability measurement of cholesterol-containing liposome on mica by atomic force microscopy. J. Colloid Interface Sci. 2004, 278, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Coderch, L.; Fonollosa, J.; De Pera, M.; Estelrich, J.; De La Maza, A.; Parra, J.L. Influence of cholesterol on liposome fluidity by EPR. Relationship with percutaneous absorption. J. Control. Release 2000, 68, 85–95. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
Sample Availability: A sample of compound 1 is available from the authors. |

| Component (%) | CC-NL#1 | CC-NL#2 | CC-NL#3 | CC-NL#4 | CC-NL#5 |
|---|---|---|---|---|---|
| Lipoid P75-3 | 5 | 0 | 2.5 | 5 | 2.5 |
| Lipoid P100-3 | 0 | 5 | 2.5 | 0 | 2.5 |
| Cholesterol | 0 | 0 | 0 | 2 | 1 |
| Squalene | 1 | 1 | 1 | 0 | 1 |
| Caprylic capryl triglyceride | 0 | 0 | 0 | 0 | 5 |
| Ethanol | 1 | 1 | 1 | 1 | 1 |
| Glycerol | 50 | 50 | 50 | 50 | 50 |
| Citrus callus extract | 10 | 10 | 10 | 10 | 10 |
| Water | 33 | 33 | 33 | 32 | 27 |
| Formulation Code | Particle Size (nm) | Polydispersity Index | Zeta Potential (mV) |
|---|---|---|---|
| CC-NL#1 | 44.12 ± 0.889 | 0.482 ± 0.026 | −63.4 ± 0.55 |
| CC-NL#2 | 51.04 ± 0.404 | 0.083 ± 0.001 | 0.59 ± 0.08 |
| CC-NL#3 | 122.3 ± 1.818 | 0.285 ± 0.015 | −48.1 ± 0.85 |
| CC-NL#4 | 146.8 ± 12.66 | 0.419 ± 0.062 | −65.8 ± 2.29 |
| CC-NL#5 | 73.15 ± 1.226 | 0.280 ± 0.006 | −54.5 ± 3.10 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adhikari, D.; Panthi, V.K.; Pangeni, R.; Kim, H.J.; Park, J.W. Preparation, Characterization, and Biological Activities of Topical Anti-Aging Ingredients in a Citrus junos Callus Extract. Molecules 2017, 22, 2198. https://doi.org/10.3390/molecules22122198
Adhikari D, Panthi VK, Pangeni R, Kim HJ, Park JW. Preparation, Characterization, and Biological Activities of Topical Anti-Aging Ingredients in a Citrus junos Callus Extract. Molecules. 2017; 22(12):2198. https://doi.org/10.3390/molecules22122198
Chicago/Turabian StyleAdhikari, Deepak, Vijay Kumar Panthi, Rudra Pangeni, Hyun Jung Kim, and Jin Woo Park. 2017. "Preparation, Characterization, and Biological Activities of Topical Anti-Aging Ingredients in a Citrus junos Callus Extract" Molecules 22, no. 12: 2198. https://doi.org/10.3390/molecules22122198






