Live Fluorescent Staining Platform for Drug-Screening and Mechanism-Analysis in Zebrafish for Bone Mineralization
Abstract
:1. Introduction
2. Results
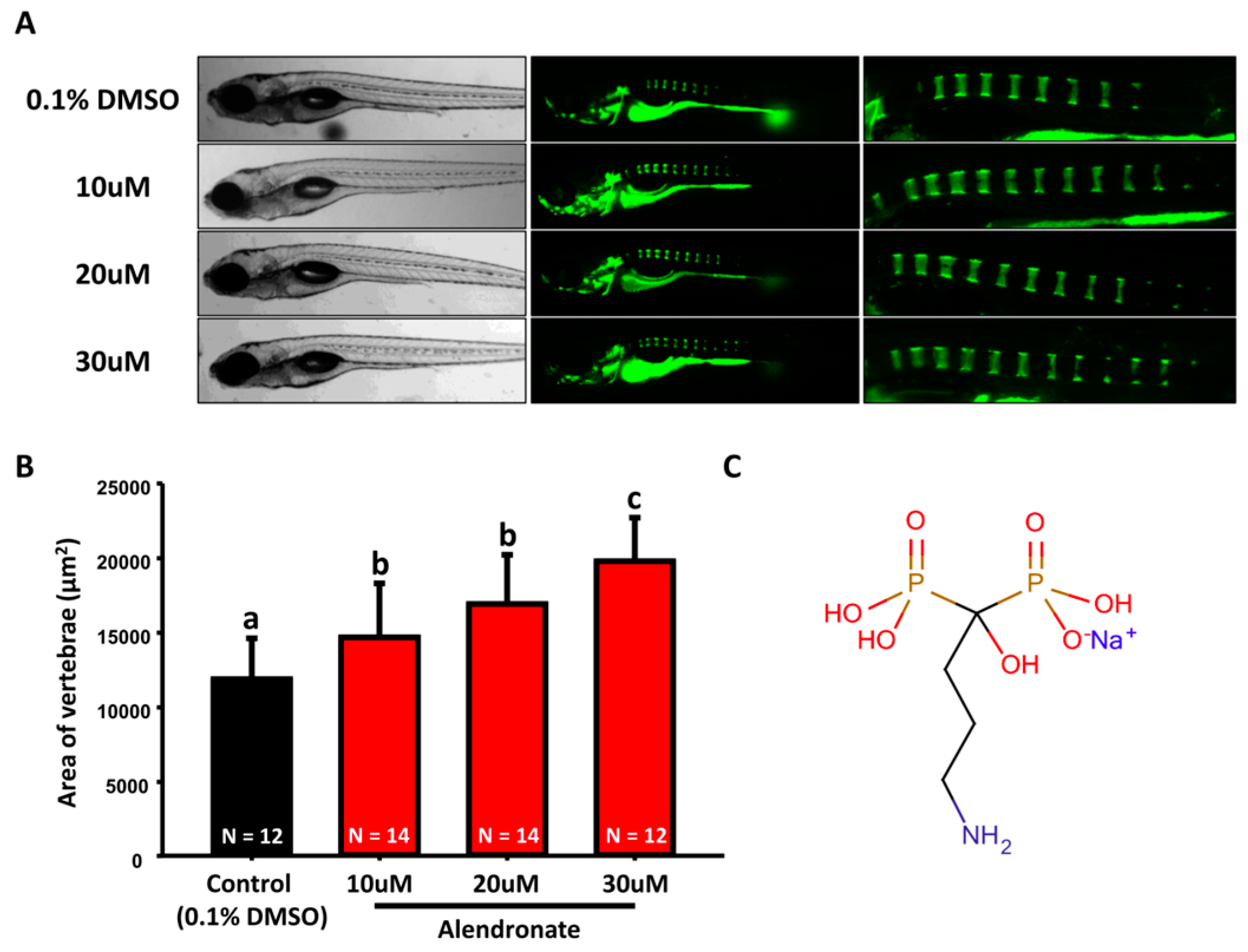
2.1. Optimization of Small Molecular Screening Platform in Zebrafish with Positive and Negative Control Compounds
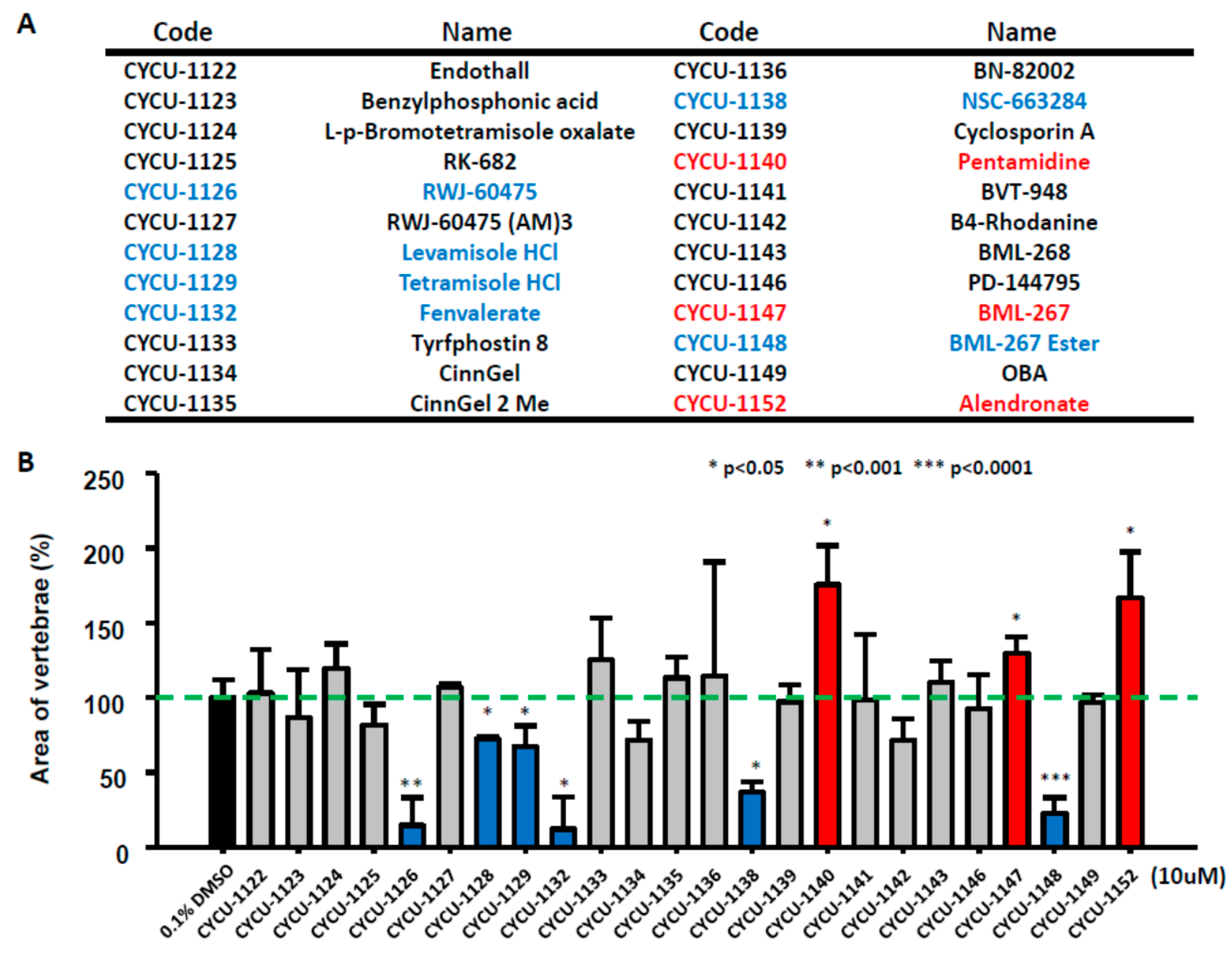
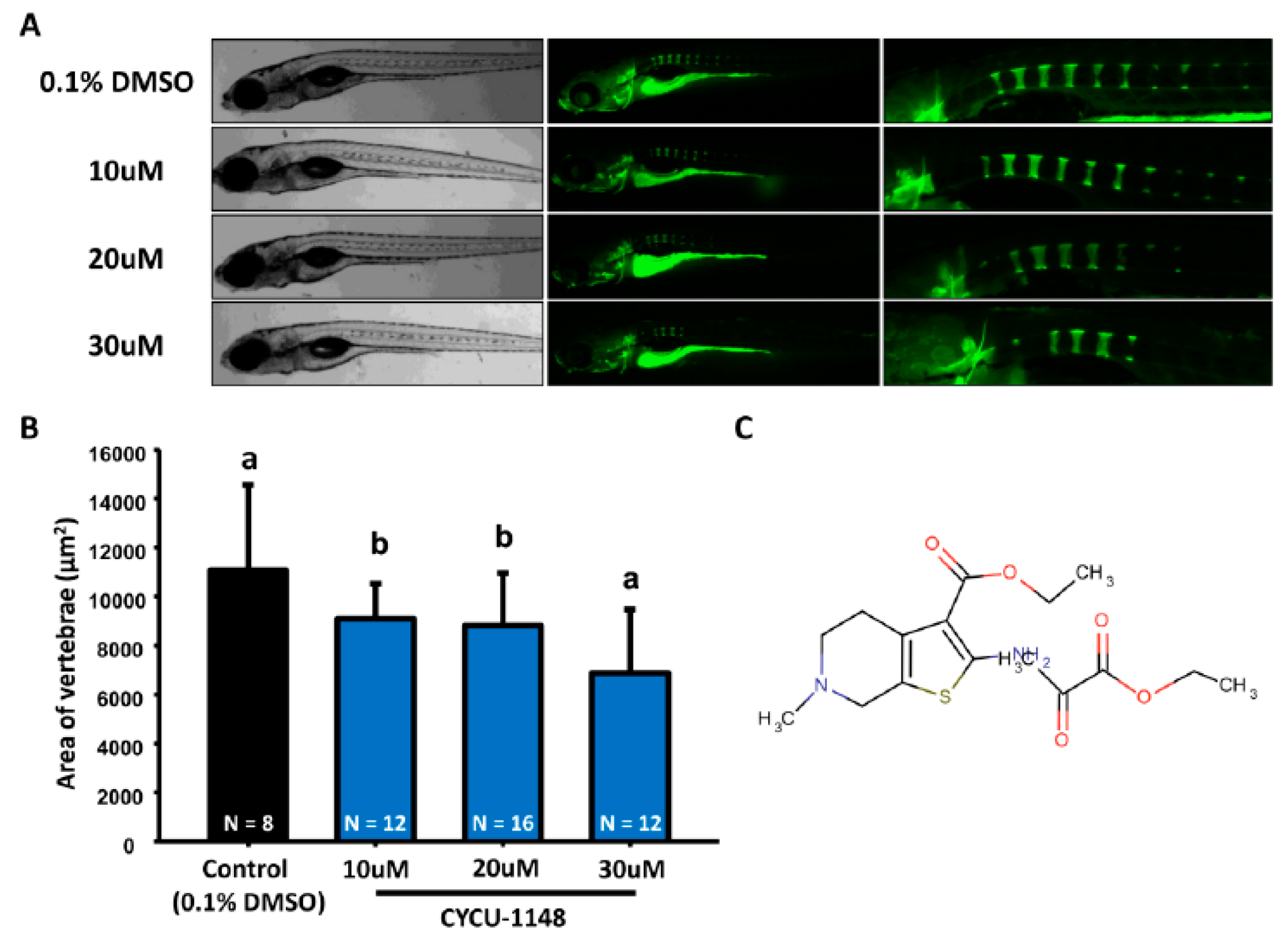
2.2. Rapid Screening on Small Molecular Library
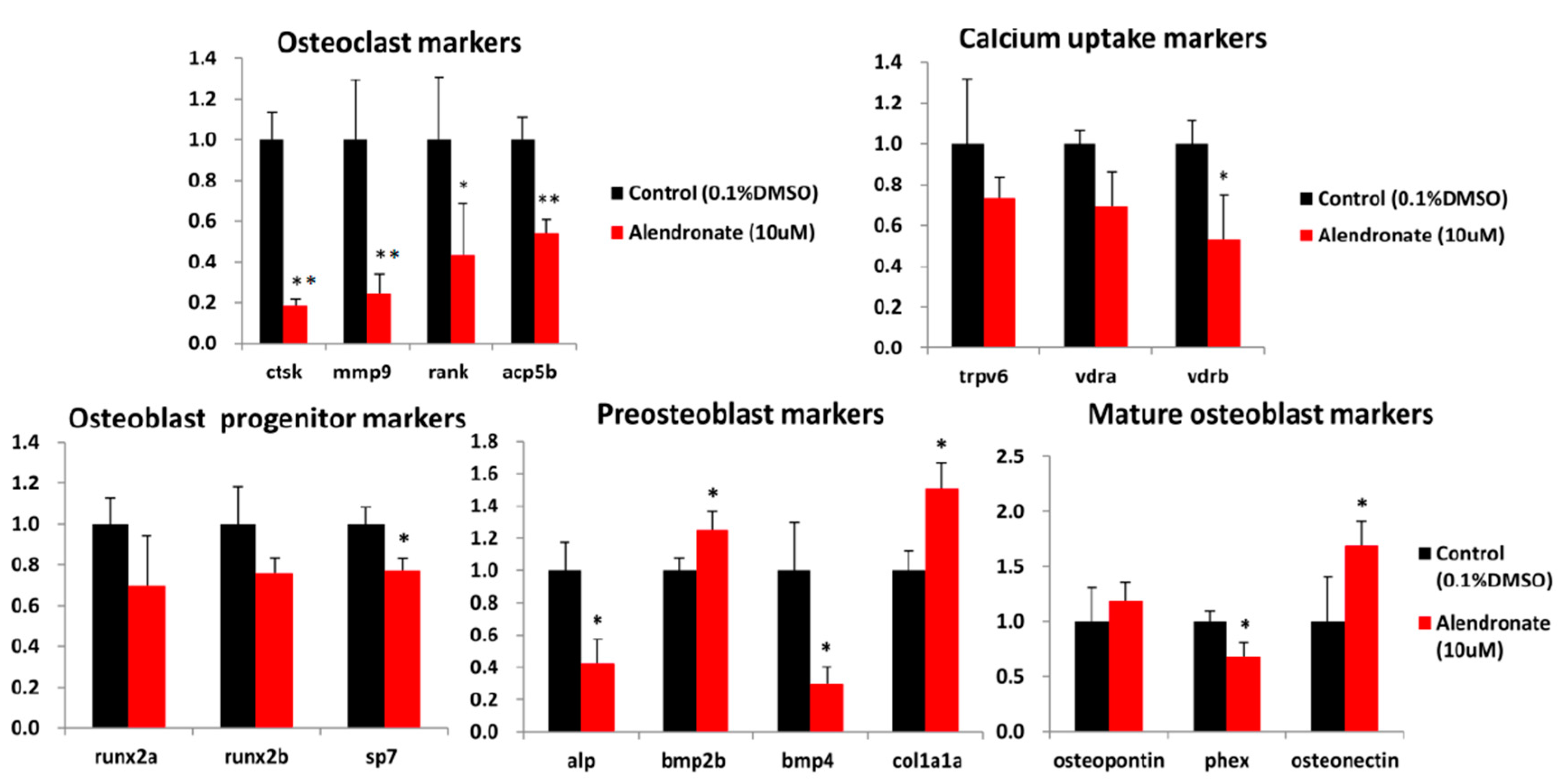
2.3. Zebrafish Embryos are A Potential Drug-Screening and Mechanism-Analysis Platform for Bone Mineralization
3. Discussion
4. Materials and Methods
4.1. Zebrafish Maintenance
4.2. Small Molecular Library
4.3. Calcein Labeling
4.4. Small Molecular Screening
4.5. Real Time Quantitative PCR (RT-qPCR)
4.6. Image Processing
4.7. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Riggs, B.L.; Khosla, S.; Melton, L.J. Sex steroids and the construction and conservation of the adult skeleton. Endocr. Rev. 2002, 23, 279–302. [Google Scholar] [CrossRef] [PubMed]
- Crockett, J.C.; Rogers, M.J.; Coxon, F.P.; Hocking, L.J.; Helfrich, M.H. Bone remodelling at a glance. J. Cell Sci. 2011, 124, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; McDonald, J.M. Disorders of bone remodeling. Annu. Rev. Pathol. 2011, 6, 121–145. [Google Scholar] [CrossRef] [PubMed]
- Lazner, F.; Gowen, M.; Pavasovic, D.; Kola, I. Osteopetrosis and osteoporosis: Two sides of the same coin. Hum. Mol. Genet. 1999, 8, 1839–1846. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, R.M. Bone health: Preventing osteoporosis. Public Health Rep. 2010, 125, 368–370. [Google Scholar] [CrossRef] [PubMed]
- Pisani, P.; Renna, M.D.; Conversano, F.; Casciaro, E.; Di Paola, M.; Quarta, E.; Muratore, M.; Casciaro, S. Major osteoporotic fragility fractures: Risk factor updates and societal impact. World J. Orthop. 2016, 7, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.T. Osteoporosis and cancer. Curr. Osteoporos. Rep. 2013, 11, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Johnell, O.; Kanis, J.A. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 2006, 17, 1726–1733. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P. Advances in therapy for osteoporosis. Clin. Med. Res. 2003, 1, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Bodine, P.V.; Stauffer, B.; Ponce-de-Leon, H.; Bhat, R.A.; Mangine, A.; Seestaller-Wehr, L.M.; Moran, R.A.; Billiard, J.; Fukayama, S.; Komm, B.S.; et al. A small molecule inhibitor of the wnt antagonist secreted frizzled-related protein-1 stimulates bone formation. Bone 2009, 44, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.B.; Hong, C.C.; Sachidanandan, C.; Babitt, J.L.; Deng, D.Y.; Hoyng, S.A.; Lin, H.Y.; Bloch, K.D.; Peterson, R.T. Dorsomorphin inhibits bmp signals required for embryogenesis and iron metabolism. Nat. Chem. Biol. 2008, 4, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Brey, D.M.; Motlekar, N.A.; Diamond, S.L.; Mauck, R.L.; Garino, J.P.; Burdick, J.A. High-throughput screening of a small molecule library for promoters and inhibitors of mesenchymal stem cell osteogenic differentiation. Biotechnol. Bioeng. 2011, 108, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.N.; Bae, S.J.; Kwak, H.B.; Min, Y.K.; Jung, S.H.; Kim, C.H.; Kim, S.H. In vitro and in vivo osteogenic activity of licochalcone a. Amino Acids 2012, 42, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Astashkina, A.; Mann, B.; Grainger, D.W. A critical evaluation of in vitro cell culture models for high-throughput drug screening and toxicity. Pharmacol. Ther. 2012, 134, 82–106. [Google Scholar] [CrossRef] [PubMed]
- Bradaschia-Correa, V.; Barrence, F.A.; Ferreira, L.B.; Massa, L.F.; Arana-Chavez, V.E. Effect of alendronate on endochondral ossification in mandibular condyles of growing rats. Eur. J. Histochem. 2012, 56, e24. [Google Scholar] [CrossRef] [PubMed]
- Suster, M.L.; Kikuta, H.; Urasaki, A.; Asakawa, K.; Kawakami, K. Transgenesis in zebrafish with the tol2 transposon system. Methods Mol. Biol. 2009, 561, 41–63. [Google Scholar] [PubMed]
- Spaink, H.P.; Cui, C.; Wiweger, M.I.; Jansen, H.J.; Veneman, W.J.; Marin-Juez, R.; de Sonneville, J.; Ordas, A.; Torraca, V.; van der Ent, W.; et al. Robotic injection of zebrafish embryos for high-throughput screening in disease models. Methods 2013, 62, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Kashii, M.; Hashimoto, J.; Nakano, T.; Umakoshi, Y.; Yoshikawa, H. Alendronate treatment promotes bone formation with a less anisotropic microstructure during intramembranous ossification in rats. J. Bone Miner. Metab. 2008, 26, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, T.; Okimoto, N.; Okamoto, K.; Sakai, A. A comparative study of the effects of daily minodronate and weekly alendronate on upper gastrointestinal symptoms, bone resorption, and back pain in postmenopausal osteoporosis patients. J. Bone Miner. Metab. 2013, 31, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Yamanaka, S.; Yoshimoto, W.; Shigematsu, T. Alendronate as an effective treatment for bone loss and vascular calcification in kidney transplant recipients. J. Transplant. 2014, 2014, 269613. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Deng, C.; Li, Y.P. Tgf-beta and bmp signaling in osteoblast differentiation and bone formation. Int. J. Biol. Sci. 2012, 8, 272–288. [Google Scholar] [CrossRef] [PubMed]
- Yavropoulou, M.P.; Yovos, J.G. The role of notch signaling in bone development and disease. Hormones (Athens) 2014, 13, 24–37. [Google Scholar] [PubMed]
- Finkelstein, J.S.; Hayes, A.; Hunzelman, J.L.; Wyland, J.J.; Lee, H.; Neer, R.M. The effects of parathyroid hormone, alendronate, or both in men with osteoporosis. N. Engl. J. Med. 2003, 349, 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Termine, J.D.; Kleinman, H.K.; Whitson, S.W.; Conn, K.M.; McGarvey, M.L.; Martin, G.R. Osteonectin, a bone-specific protein linking mineral to collagen. Cell 1981, 26, 99–105. [Google Scholar] [CrossRef]
- Hwang, P.-P.; Chou, M.-Y. Zebrafish as an animal model to study ion homeostasis. Pflüg. Arch. 2013, 465, 1233–1247. [Google Scholar] [CrossRef] [PubMed]
- Vanoevelen, J.; Janssens, A.; Huitema, L.F.; Hammond, C.L.; Metz, J.R.; Flik, G.; Voets, T.; Schulte-Merker, S. Trpv5/6 is vital for epithelial calcium uptake and bone formation. FASEB J. 2011, 25, 3197–3207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, T.-C.; Liao, B.-K.; Huang, C.-J.; Lin, L.-Y.; Hwang, P.-P. Epithelial Ca2+ channel expression and Ca2+ uptake in developing zebrafish. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2005, 289, R1202–R1211. [Google Scholar] [CrossRef] [PubMed]
- Olsen, B.R.; Reginato, A.M.; Wang, W. Bone development. Annu. Rev. Cell Dev. Biol. 2000, 16, 191–220. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Kim, J.E.; Nakashima, K.; Balmes, G.; Iwai, N.; Deng, J.M.; Zhang, Z.; Martin, J.F.; Behringer, R.R.; Nakamura, T.; et al. Osteo-chondroprogenitor cells are derived from sox9 expressing precursors. Proc. Natl. Acad. Sci. USA 2005, 102, 14665–14670. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.; Keynes, R.; Tannahill, D. A central role for the notochord in vertebral patterning. Development 2004, 131, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Mabee, P.M.; Noordsy, M. Development of the paired fins in the paddlefish, polyodon spathula. J. Morphol. 2004, 261, 334–344. [Google Scholar] [CrossRef] [PubMed]
- Flores, M.V.; Tsang, V.W.; Hu, W.; Kalev-Zylinska, M.; Postlethwait, J.; Crosier, P.; Crosier, K.; Fisher, S. Duplicate zebrafish runx2 orthologues are expressed in developing skeletal elements. Gene Expr. Patterns 2004, 4, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.; Halpern, M.E. Patterning the zebrafish axial skeleton requires early chordin function. Nat. Genet. 1999, 23, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Melton, L.J.; Johnell, O.; Lau, E.; Mautalen, C.A.; Seeman, E. Osteoporosis and the global competition for health care resources. J. Bone Miner. Res. 2004, 19, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.; Sato, M.; Goldsmith, P. High-throughput in vivo screening for bone anabolic compounds with zebrafish. J. Biomol. Screen. 2005, 10, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Tarasco, M.; Laizé, V.; Cardeira, J.; Cancela, M.L.; Gavaia, P.J. The zebrafish operculum: A powerful system to assess osteogenic bioactivities of molecules with pharmacological and toxicological relevance. Comp. Biochem. Physiol. Part C Toxicol Pharmacol. 2017, 197, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Westerfield, M. The Zebrafish Book: A Guide for the Laboratory Use of Zebrafish (Brachydanio Rerio); University of Oregon Press: Eugene, OR, USA, 1995. [Google Scholar]
- Du, S.J.; Frenkel, V.; Kindschi, G.; Zohar, Y. Visualizing normal and defective bone development in zebrafish embryos using the fluorescent chromophore calcein. Dev. Biol. 2001, 238, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time pcr data by the comparative ct method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |



| Gene Name | Symbol | Forward Primer | Reverse Primer |
|---|---|---|---|
| Osteoclast markers | |||
| cathepsin K | ctsk | GGACTCAATCACTATCACT | AGAACAAGACATCTAAGACA |
| matrix metallopeptidase 9 | mmp9 | TCGGCCTACCAAGCGACTT | TCATGTGAATCAATGGGCACTC |
| receptor activator of nuclear factor kappa-B ligand | rank | GCACGGTTATTGTTGTTA | TATTCAGAGGTGGTGTTAT |
| acid phosphatase 5b | acp5b | GCTGCTGCTAACAAACAAT | GACCAACCACGATGACAA |
| Calcium uptake markers | |||
| transient receptor potential cation channel subfamily V member 6 | trpv6 | GATCGCAATGACATAATG | CTCCATCACTCTTAGAAG |
| vitamin D receptor a | vdra | CTTCAGACTCATTCAACCAT | GATACATCATCAGCAGATTACT |
| vitamin D receptor b | vdrb | CTCATCAGACTCCTTCAG | TACATCATCAGCAGGTTAC |
| Osteoblast progenitor markers | |||
| runt-related transcription factor 2a | runx2a | GACGGTGGTGACGGTAATGG | TGCGGTGGGTTCGTGAATA |
| runt-related transcription factor 2b | runx2b | CGGCTCCTACCAGTTCTCCA | CCATCTCCCTCCACTCCTCC |
| sp7 transcription factor 7 | sp7 | GGCTATGCTAACTGCGACCTG | GCTTTCATTGCGTCCGTTTT |
| Preosteoblast markers | |||
| alkaline phosphatase | alp | CAAGAACTCAACAAGAAC | TGAGCATTGGTGTTATAC |
| bone morphogenetic protein 2b | bmp2b | CGGCTCCTACCAGTTCTCCA | CCATCTCCCTCCACTCCTCC |
| bone morphogenetic protein 4 | bmp4 | TTGTGCTGTGCATGTTTGAA | GGTCGCTTGGCTATGTGTTT |
| collagen type II, alpha 1 (cartilage collagen) | cola1a | CTGTGCCAATCCCATCATTTC | ATATCGCCTGGTTCTCCTTTC |
| Mature osteoblast markers | |||
| osteopontin | opn | GCCTCCATCATCATCGTA | AATCACCAAGCACCAGTA |
| phosphate regulating endopeptidase homolog, X-linked | phex | GAGAATGAATGGATGGATGA | TTGATGTCTTCGTTAATATAGGT |
| osteonectin | on | ACTAACAACAAGACCTAC | TCCGATGTAATCTATGTG |
| House keeping gene | |||
| β-actin | actb1 | CCCAAAGCCAACAGAGAGAA | ACCAGAAGCGTACAGAGAGA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.-R.; Lai, Y.-H.; Tsai, J.-J.; Hsiao, C.-D. Live Fluorescent Staining Platform for Drug-Screening and Mechanism-Analysis in Zebrafish for Bone Mineralization. Molecules 2017, 22, 2068. https://doi.org/10.3390/molecules22122068
Chen J-R, Lai Y-H, Tsai J-J, Hsiao C-D. Live Fluorescent Staining Platform for Drug-Screening and Mechanism-Analysis in Zebrafish for Bone Mineralization. Molecules. 2017; 22(12):2068. https://doi.org/10.3390/molecules22122068
Chicago/Turabian StyleChen, Jung-Ren, Yu-Heng Lai, Jhih-Jie Tsai, and Chung-Der Hsiao. 2017. "Live Fluorescent Staining Platform for Drug-Screening and Mechanism-Analysis in Zebrafish for Bone Mineralization" Molecules 22, no. 12: 2068. https://doi.org/10.3390/molecules22122068








