1. Introduction
Polyunsaturated fatty acids (PUFA), polyphenolics, including bioflavonoids, their derivatives and analogues, are important nutraceuticals and are becoming of increasing nutritional interest [
1]. The consumption of dietary PUFA and polyphenols has been shown to reduce the risk of coronary heart disease and cancer, to improve inflammatory conditions, such as arthritis, to reduce plasma triacylglycerol levels and to lower blood pressure [
2]. Although PUFA and polyphenols can be found in natural sources, these may not be enough to cover consumer´s requirements. Both food and pharmaceutical products with added PUFA and polyphenols are becoming common in the United States and European Union [
3,
4,
5]. Some previous studies in the available literature have shown that the presence of PUFA and polyphenol-enriched foods on the market, and the promotion of their consumption may significantly increase the level of their intake [
6]. These nutraceuticals have been associated with the prevention of some chronic and degenerative diseases [
7,
8,
9]. Therefore, the recovery of these compounds from natural sources for use as food additives and/or nutraceuticals and dietary supplements is an important challenge [
10].
Chaenomelessinensis (Thouin) Koehne (
C. sinensis), commonly known as “Guang Pi Mu Gua”, “Chinese-quince” and “Mingzha”, is used in the contemporary food industry for liquors and candies, but has seen thousands of years of use in Chinese medicine [
11], for the treatment of rheumatoid arthritis, hepatitis, asthma and the common cold. Most of the benefits of these plants have been attributed to their high content in PUFA, monounsaturated fatty acids (MUFA), and various polyphenolic compounds.
In recent years, the plant has been cultivated in large areas of some regions in China that have converted farmland into forestland and, as a result, yields have rapidly increased, making it an important industrial agricultural crop. Most of the studies on C. sinensis have focused on the nutritional characterization and the evaluation of the biological activity of the compounds found within the flesh. However, there is a lack of information on their seeds, which constitute an important part of these plants and have traditionally been seen as waste.
Previous studies have shown that
C. sinensis seeds are rich in unsaturated fatty acids [
12], and polyphenols. Consequently, the reutilization of seeds for functional, edible oil kills two birds with one stone and addresses both the use of waste and by-products and societal health care.
Conventional methods, such as expeller pressing and Soxhlet extraction (SE), have been used to extract plant oils in the food industry. However, these methods require long extraction times at high temperature, while the oil can often contain residual solvent and require heating, thus facilitating rancidity reactions during the following separation process. There has therefore been an increased effort to develop new methods that can improve the oil extraction process and that could potentially allow the extraction yield as well as the nutraceutical properties of food to be modulated.
The last two decades have seen cold-pressed extraction (CPE), microwave-assisted extraction (MAE), and subcritical fluid extraction (SbFE), technologies being proposed as potential tools to improve extraction processes and functional oils of high quality have thus been obtained [
2,
13,
14].
Although a number of subcritical fluids have been used in SbFE,
n-butane is the most commonly used subcritical fluid mainly because it only requires low critical pressures and temperatures. Moreover, it has an excellent capacity to extract lipophilic compounds [
15]. In addition, this solvent is inexpensive, colourless, has a low boiling point and is considered a clean solvent, as no solvent residues remain in the extracted product [
8].
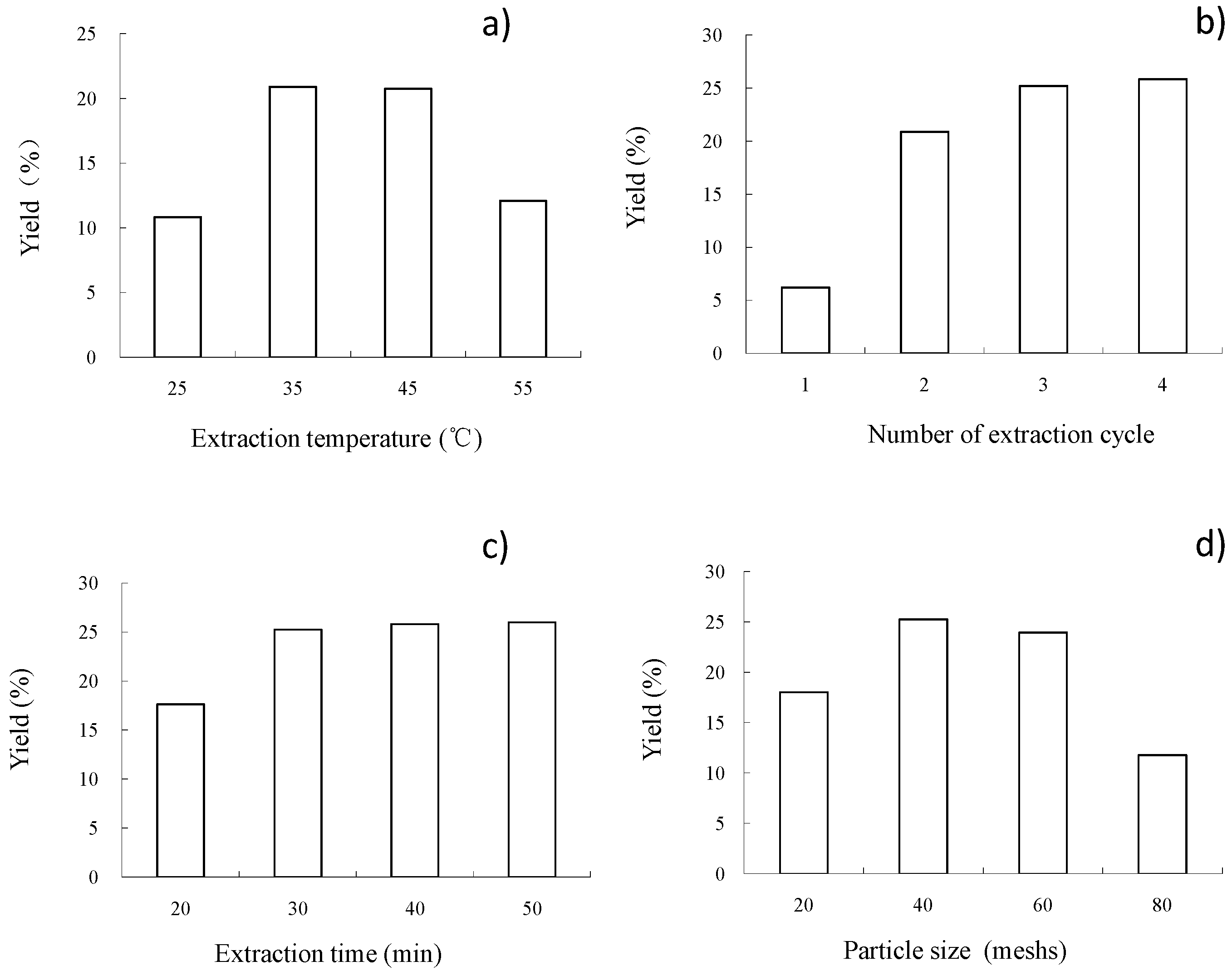
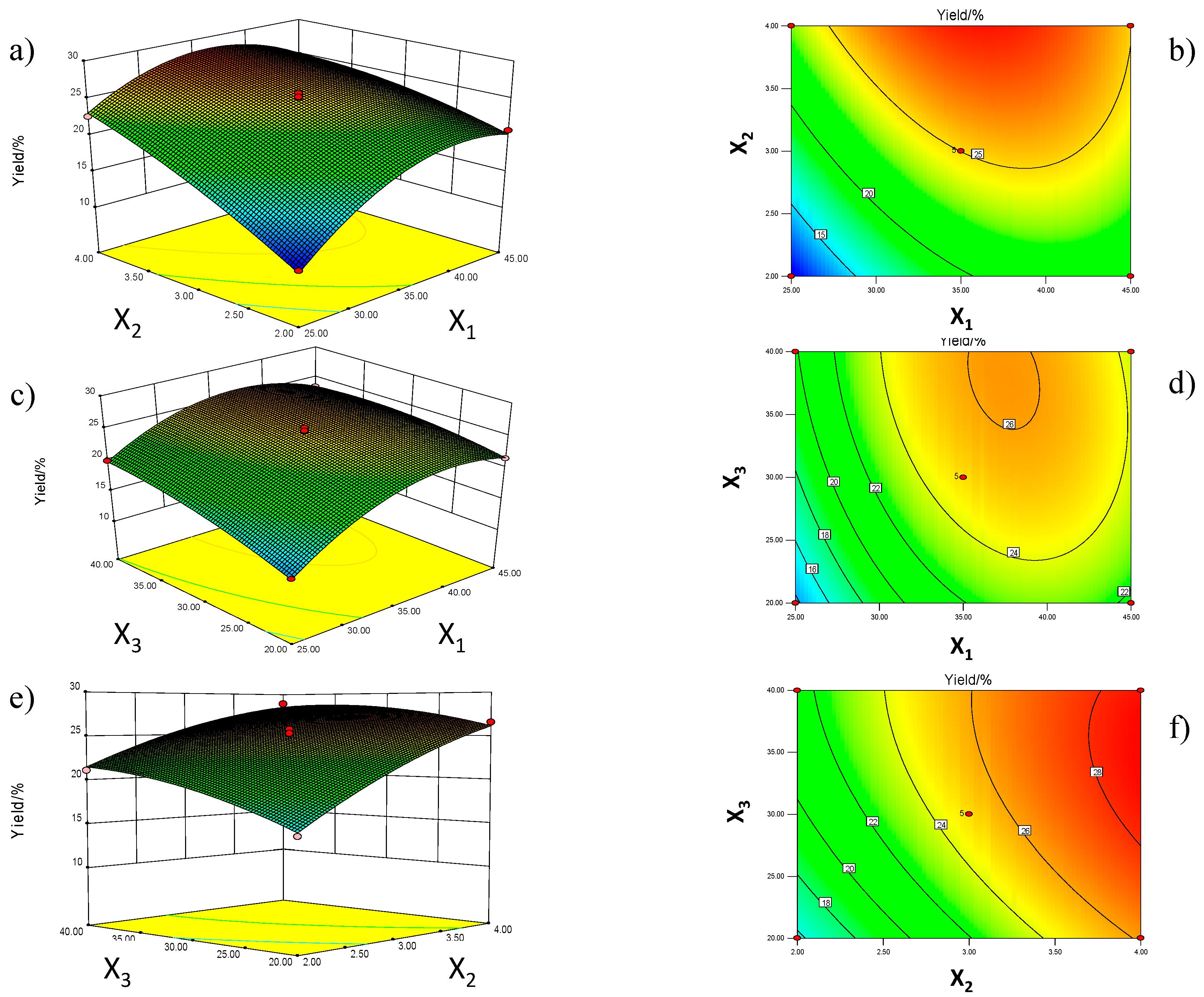
The first objective of this work is therefore to optimize SbFE processing conditions (extraction temperature, number of extraction cycles and extraction time), in order to obtain the highest oil yield, as well as the maximal content of unsaturated fatty acids and polyphenols. The response surface methodology will be used for this purpose. Secondly, the potential of SbFE to recover oil from C. sinensis seeds will be evaluated and compared with other conventional and innovative extraction methodologies.
3. Materials and Methods
3.1. Materials and Chemicals
C. sinensis seeds were provided by Yaorong papaya Bio-Tech Development Co. Ltd. (Shiyan, Hubei, China). Seed samples were dried at 40 °C for 48 h and then stored under darkness at room temperature in polyethylene bags. The dry samples were ground into a powder using a grinder (Tianjin Taisite Instruments Co., Ltd., Tianjin, China), and passed through a 40-mesh sieve just before extraction.
Rutin (>98%), and gallic acid (>98%), were obtained from Chengdu preferred Biological Technology Co., Ltd. (Chengdu, China). Folin–Ciocalteu reagents, purchased from Sigma, were of analytical grade (Sigma-Aldrich Co., Saint Louis, MO, USA). Methanol of chromatographic grade was purchased from Thermo Scientific Co., Ltd. (Waltham, MA, USA). The other chemicals used in the present study were of analytical grade and were purchased from Sinopharm Chemical Reagent Co., Ltd., (Shanghai, China).
3.2. Oil extraction Methodologies
3.2.1. Soxhlet Extraction (SE)
Soxhlet extraction (SE) is a traditional method, adopted by the Association of Official Analytical Chemists (AOAC), for extracting fats and oils and thus for lipid determination [
8]. The SE of oil from
C. sinensis seed was performed as described previously with some modifications [
13]. Briefly, approximately 4 g of grounded
C. sinensis seed powder was packed in a Soxhlet apparatus and the oil was extracted using petroleum ether (boiling point: 30–60 °C) at 70 °C for 8 h. When the extraction process was completed, the mixture was evaporated using a BUCHI R-3 rotavapor (BUCHI Labortechnik AG, Flawil, Switzerland) at 38 °C. The oil sample was kept at 4 °C until needed for analysis.
3.2.2. Cold-Pressed Extraction (CPE)
The cold-pressed extraction (CPE) of oil from
C. sinensis seeds was carried out as previously described by Yang et al. [
25]. In brief, 500 g of ground
C. sinensis seed powder was squeezed three times at room temperature in a CA59G screw type expeller (German Monforts Group, Moenchengladbach, Germany). Subsequently, the pressed oil was centrifuged at 5000 rpm for 10 min in a TGL 205 centrifuge (Changsha Pingfan Instrument Co., Ltd., Changsha, Hunan, China). The extracted oil was kept at 4 °C until needed for analysis.
3.2.3. Microwave-Assisted Extraction (MAE)
The microwave-assisted extraction (MAE) of oil from
C. sinensis seeds was performed according to the method described by Li et al. [
26], with some modifications. Ethyl acetate was selected as the extraction solvent. Generally, ground
C. sinensis seed powder (4 g) was mixed with 20 mL of extraction solvent in a 100 mL three neck flask. A 500 W microwave apparatus (Uwave-1000, Shanghai SINEO Microwave Chemistry Technology Co., Ltd., Shanghai, China) was used at 30 °C for 30 min at 1200 rpm. After microwave treatment, the mixture was centrifuged at 5000 rpm for 10 min. Subsequently, the supernatant was evaporated using a rotavapor at 38 °C. The oil sample was kept at 4 °C until needed for analysis.
3.2.4. Subcritical Fluid Extraction (SbFE)
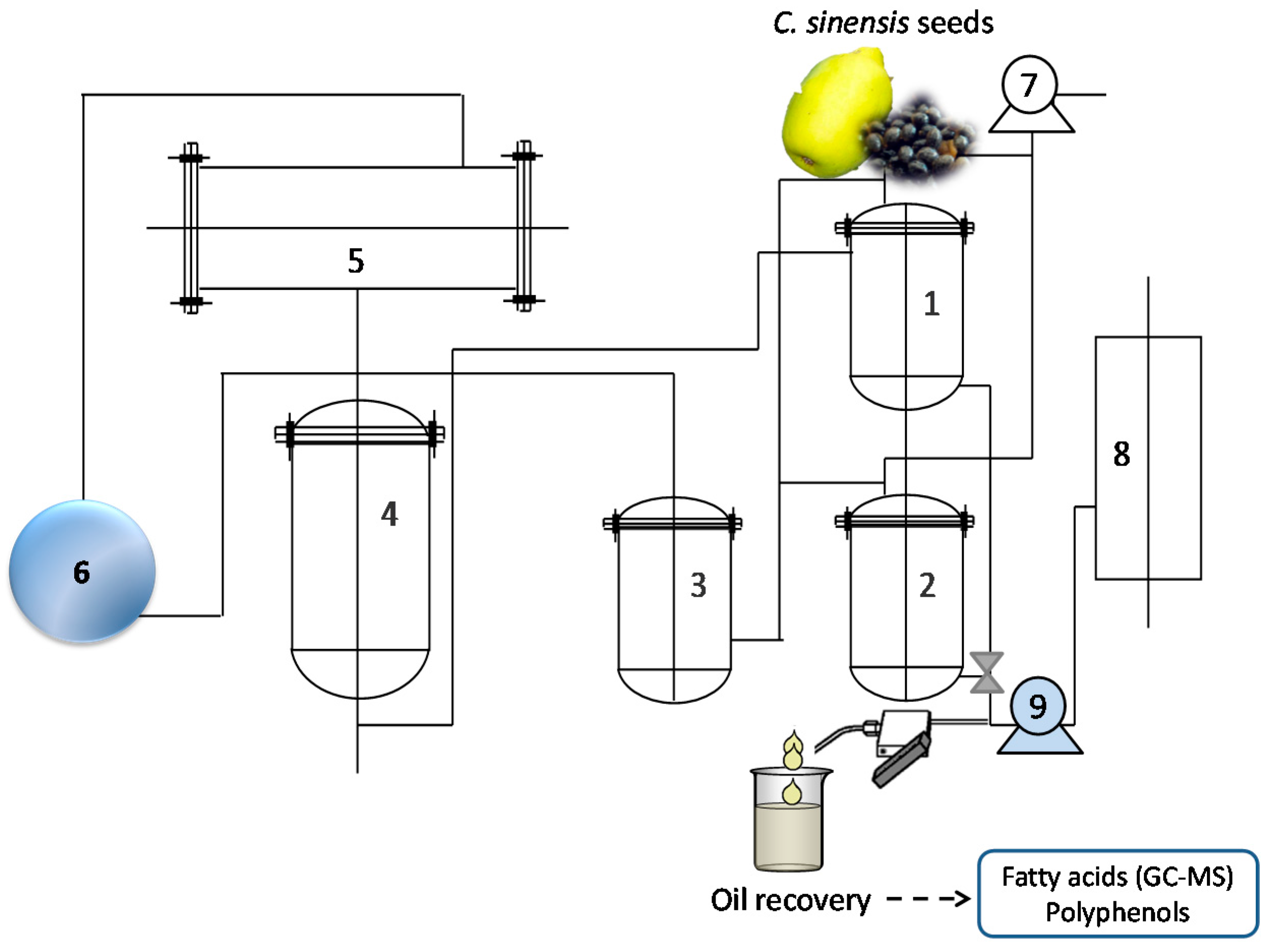
Subcritical fluid extraction (SbFE), was performed onpilot-scale CBE-10L apparatus (Subcritical Biological Technology Co., Ltd., Shanxi, Henan Province, China). A schematic diagram of the SbFE apparatus is shown in
Figure 4. A G445-5/6-13 vacuum pump (Beijing Huizi Mechanical and Electrical Equipment Co., Ltd., Beijing, China) (7) was used to drive the
n-butane extractant through the system. The maximum extraction capacity was 5 L. Extractor temperature was regulated using a hot water cylinder (8), and hot water pump (9). After extraction, the extractant fluid became gaseous and reached the
n-butane storage pot (4), via the compressor (6), and condenser (5).
For the SbFE procedure, a 300 g quantity of
C. sinensis seed powder was mixed in an extraction pot with about 1.5 L of pure
n-butane (1). In the verification experiment, 1000 grams of
C. sinensis seed powder were used for each extraction. SbFE was carried out at 35 ± 5 °C, −0.1 MPa for 30 min and then repeated twice. The mixture was immediately pumped into the separation pot (2), after extraction [
27]. Oil and solvent were then separated in the separation pot by depressurization from the compressor. The oil was collected and centrifuged at 5000 rpm for 10 min and then stored at 4 °C until needed for analysis.
3.2.5. Calculation of the Oil Extraction Yield and Recovery Rate
The extraction yield (%), was determined gravimetrically as follows [
31]:
Since the SE method was observed to have the highest extraction yield of the four methods, the recovery rate (%) could be calculated as in Equation (4).
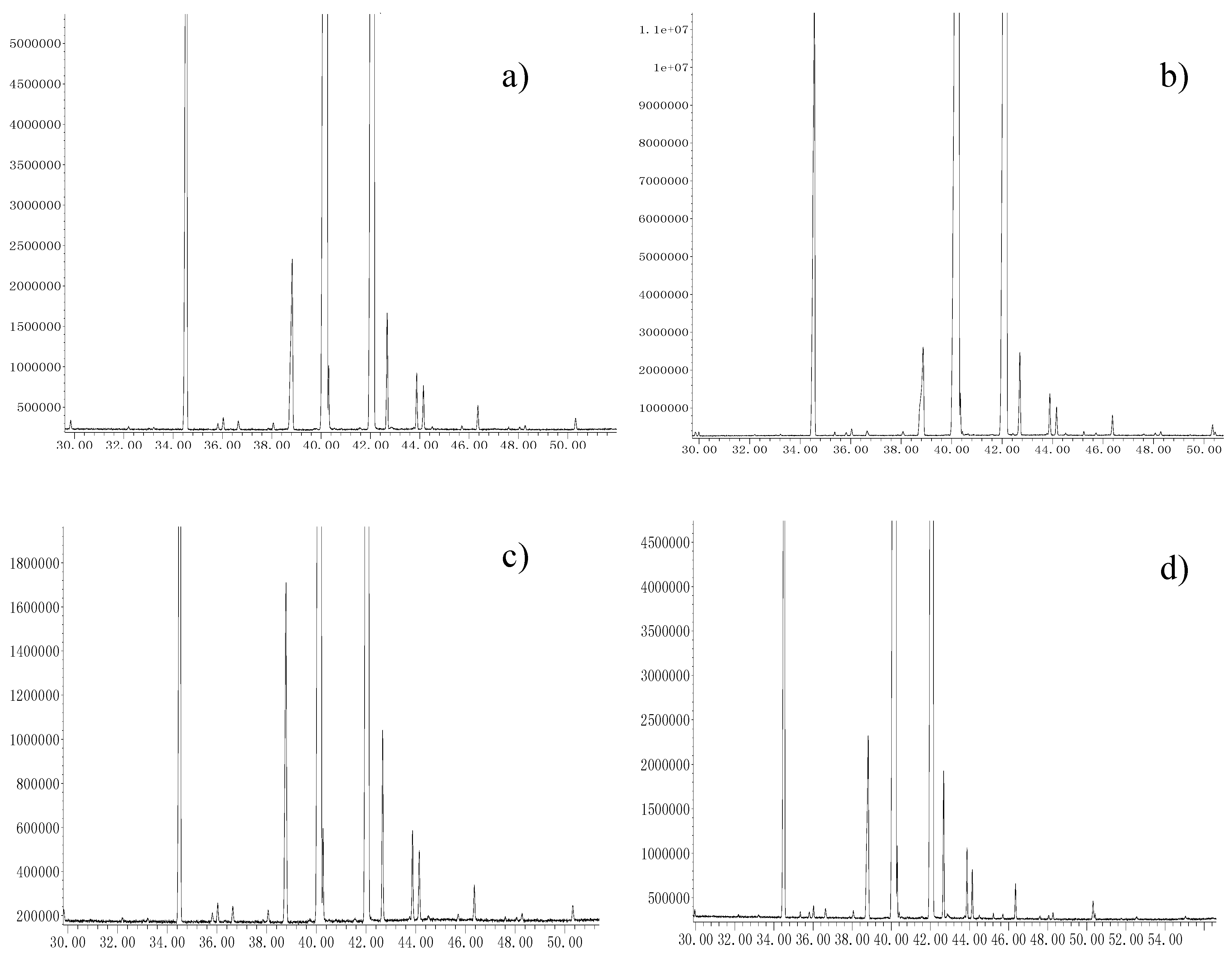
3.3. Fatty Acid Composition Analysis by GC–MS
Gas chromatography–mass spectrometry (GC–MS) was used to analyse the fatty acid composition of
C. sinensis seed oil on an Agilent 7890A/5975C GC–MSappliance (Agilent Technologies, Richardson, TX, USA), according to the procedure by Liu et al. [
4], with some modifications. Generally, gas chromatographic separation was carried out using an elastic quartz column (SP-2560, 100 m × 250 μm i.d., 0.25 μm film thickness). The GC oven was held at its initial temperature of 100 °C for 1 min, then increased to 230 °C at a rate of 3 °C /min and maintained for 20 min. Helium (purity of 99.99%), was used as the carrier gas at a flow rate of 1.0 mL/min. The injection volume was 1.0 μL at a 10:1 split. The mass spectrometer was operated at 70 eV ionization energy using electron impact ionization (EI), and a scan range from 50 to 550 amu at 2.84 scans/s.
Before GC–MS analysis, the various fatty acids of
C. sinensis seed oil extracts were derivatized into methyl esters according to Liu et al. [
6]. Briefly, 0.03 grams of
C. sinensis seed oil were added into a 10 mL centrifuge tube and dissolved in 2.5 mL of
n-hexane and 100 μL of 0.5 mol/L sodium methoxide. The mixture was then blended using an eddy instrument (model IKA-Lab Dancer S25), for 5 min and centrifuged at 4 °C at 5000 rpm for 10 min. Finally, 1.0 μL of the supernatant was injected into the GC–MS system for analysis. The identification of fatty acids was based on matching their recorded retention times and standard mass spectral library (NIST11.L), as provided by the GC–MS software. Compounds with matching rates of over 90% were taken as the target compounds. The normalization method was applied to analyse the fatty acid composition of the oils from the GC peak areas.
3.4. Physicochemical Properties and Determination of Polyphenols
Acid value, iodine value, saponification value and peroxide value were determined according to official AOAC methods. Total phenolic content (TPC), was determined according to the Folin–Ciocalteu method [
32], with some modifications. Briefly, the reaction mixture, which was composed of 1 mL extract, 0.5 mL of Folin–Ciocalteu reagent, 1.5 mL 20% sodium carbonate and 7 mL distilled water, was stirred and placed in a volumetric flask under darkness for 2 h. The absorbance was then measured at 760 nm. The TPC was calculated according to a standard curve previously prepared with gallic acid as standard. The values were expressed as mg of gallic acid equivalents (GAE), per kg of oil (mg GAE/kg oil). The total flavonoid content (TFC), was determined according to the Al(NO
3)
3-NaNO
2-NaOH colorimetric method [
33]. The TFC content was calculated according to a standard curve previously prepared with rutin as the standard. The values were expressed as mg of rutin equivalents (RE), per kg of oil (mg RE/kg oil). The extraction of phenolics and flavonoids from oil for TPC and TFC determination was carried out as follows; one gram of
C. sinensis seed oil was mixed with 20 mL of
n-hexane in a 60 mL separating funnel and 45 mL 60% methyl alcohol was then added in three additions with vigorous mixing. The aqueous layer was collected, evaporated and justified to 5 mL with distilled water.
3.5. Statistical Analysis
An ANOVA analysis was performed to evaluate the influence of conventional Soxhlet extraction, cold-pressing, microwave-assisted extraction and subcritical fluid extraction on oil recovery and fatty acid profile. Differences at p < 0.01 were considered to be significant.