Inositol Hexaphosphate Inhibits Proliferation and Induces Apoptosis of Colon Cancer Cells by Suppressing the AKT/mTOR Signaling Pathway
Abstract
:1. Introduction
2. Results
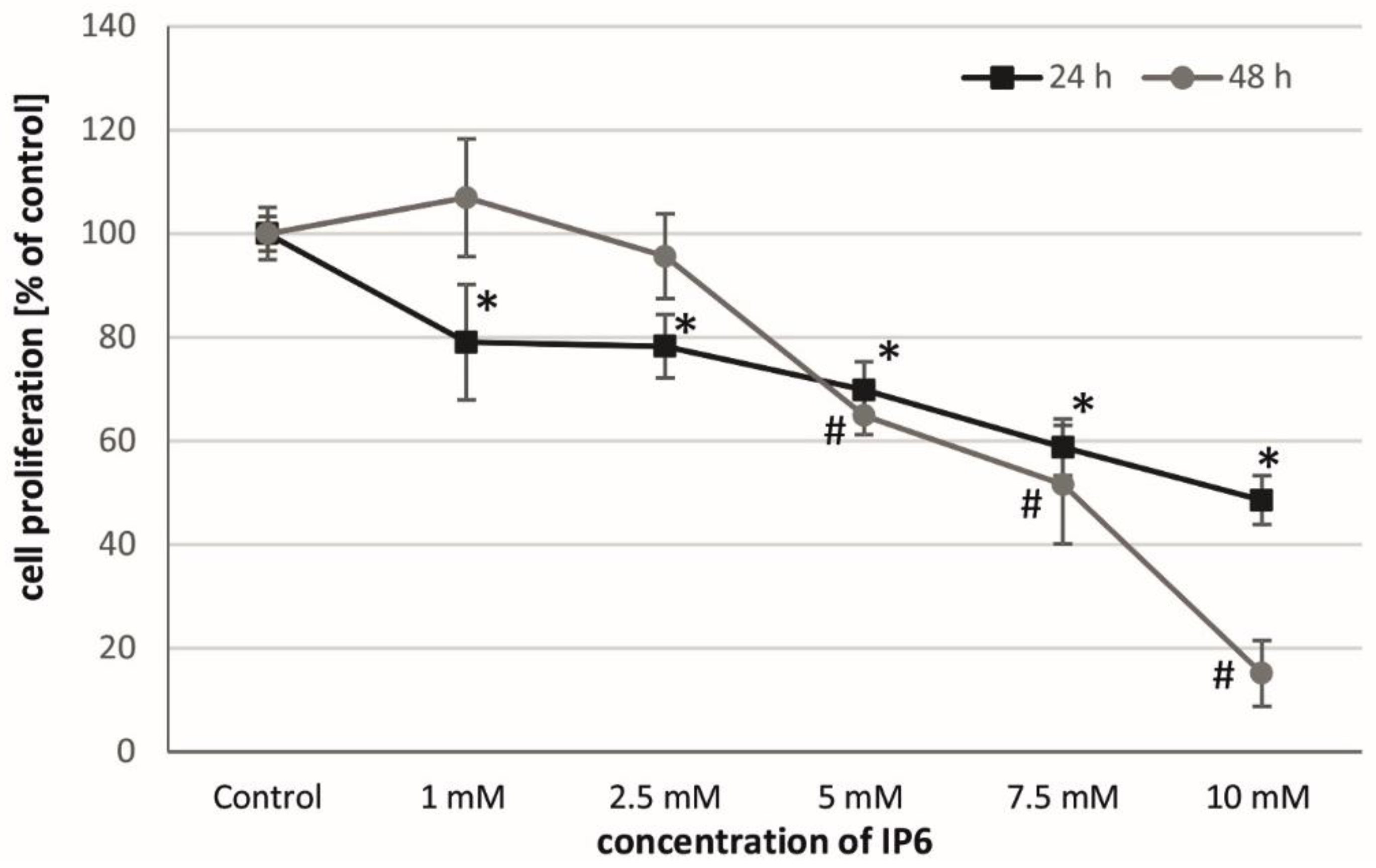
2.1. Antiproliferative Activity of InsP6 on Caco-2 Cells
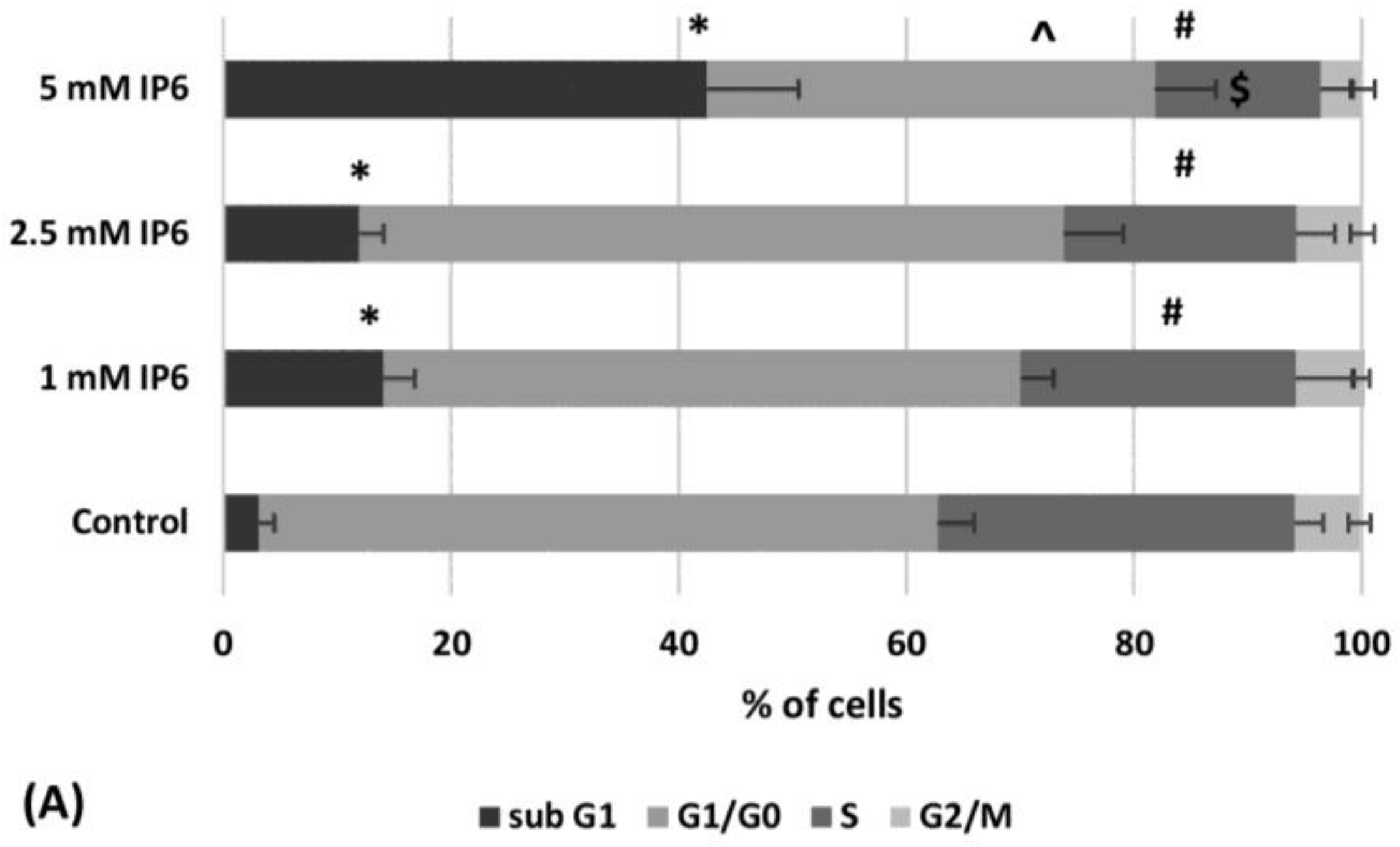
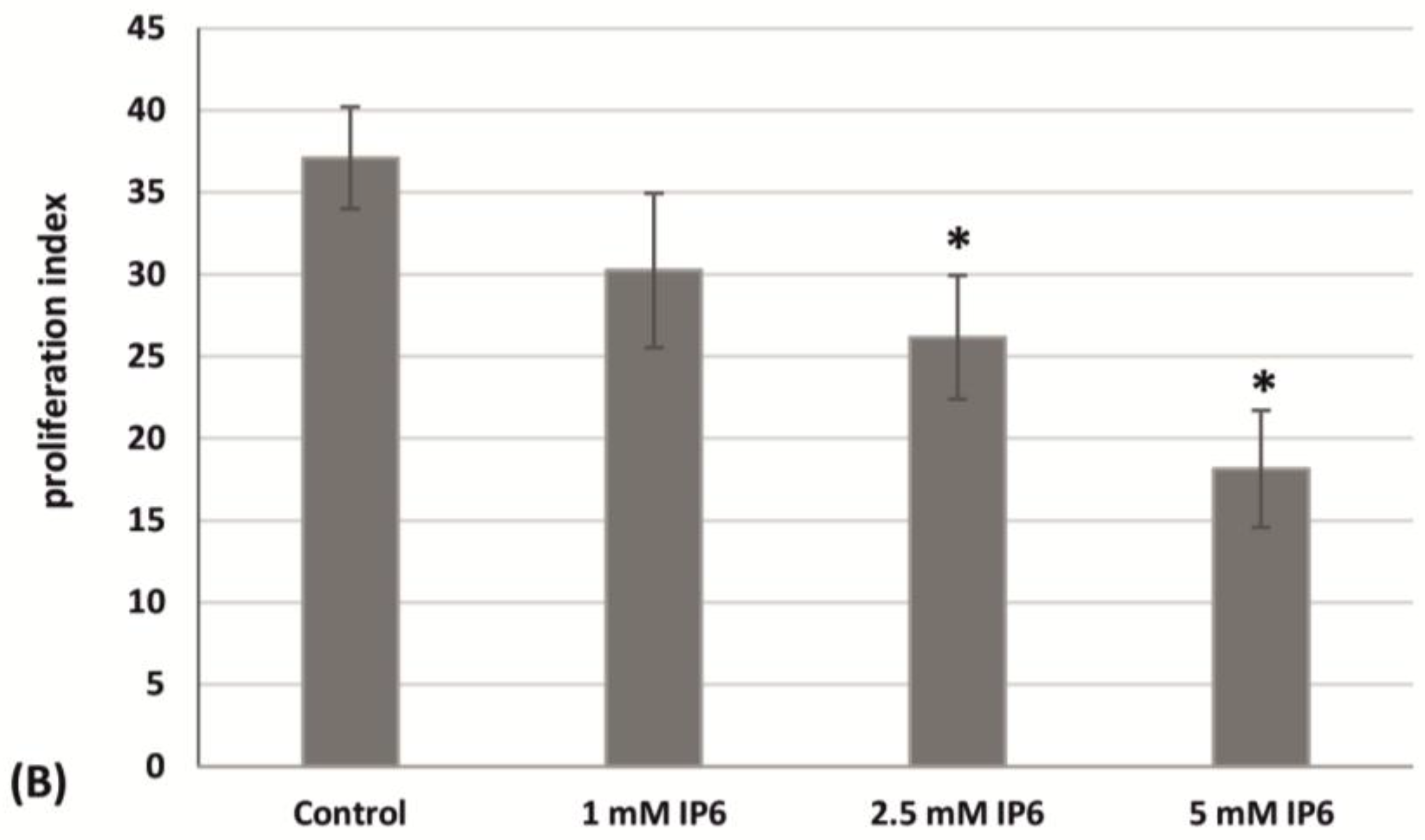
2.2. The Influence of InsP6 on Caco-2 Cell Cycle Distribution
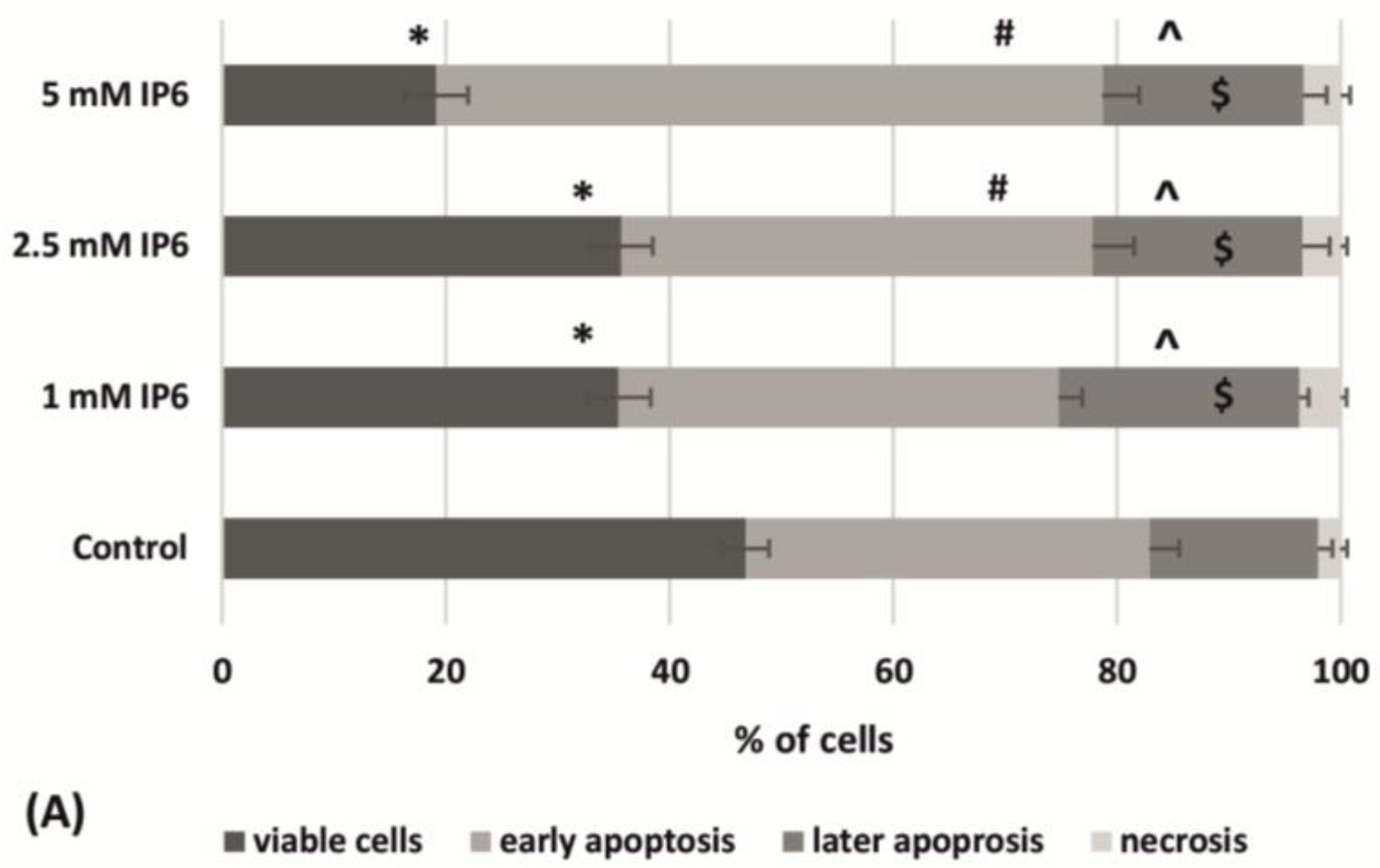
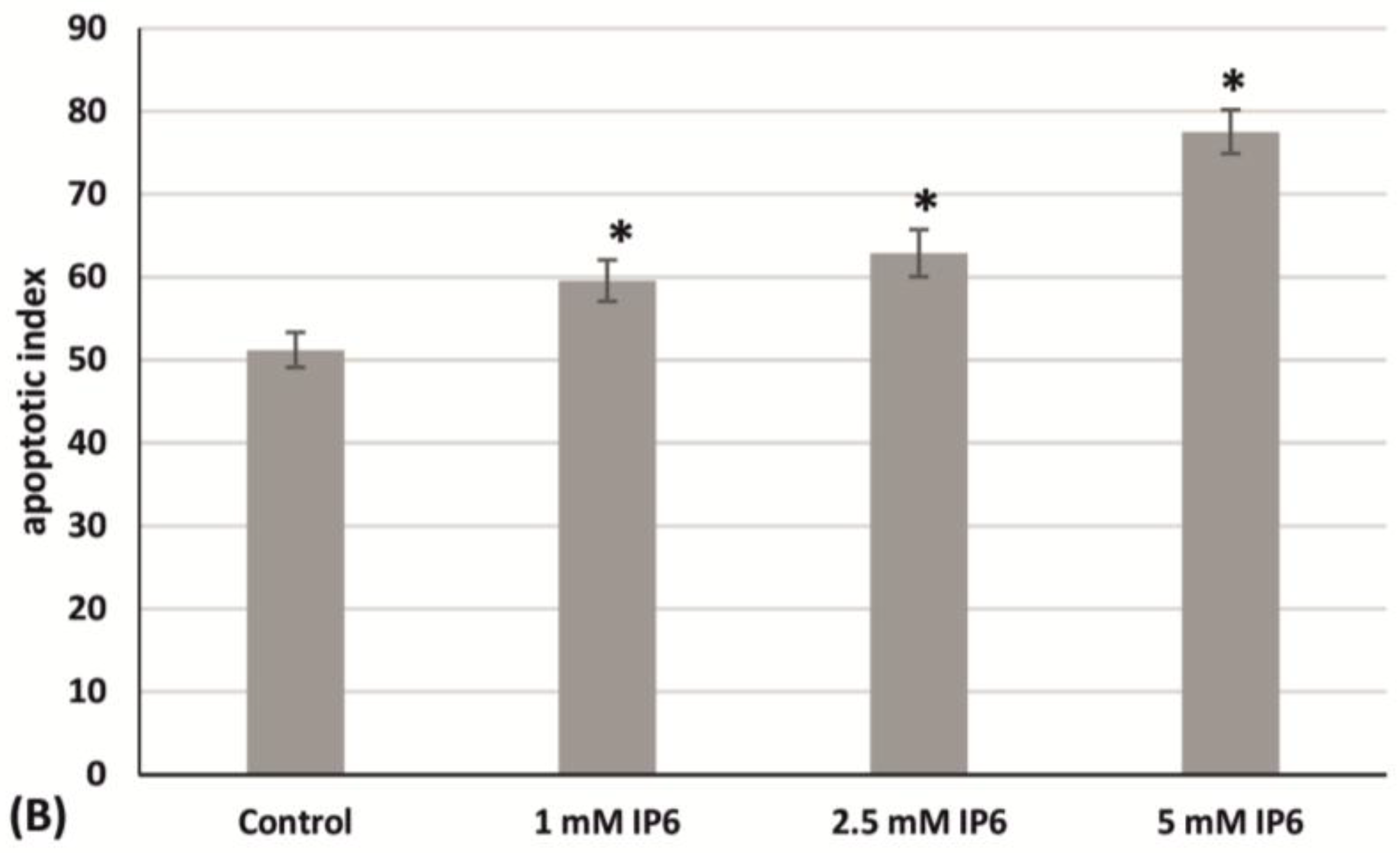
2.3. The Influence of InsP6 on Caco-2 Cells Apoptosis
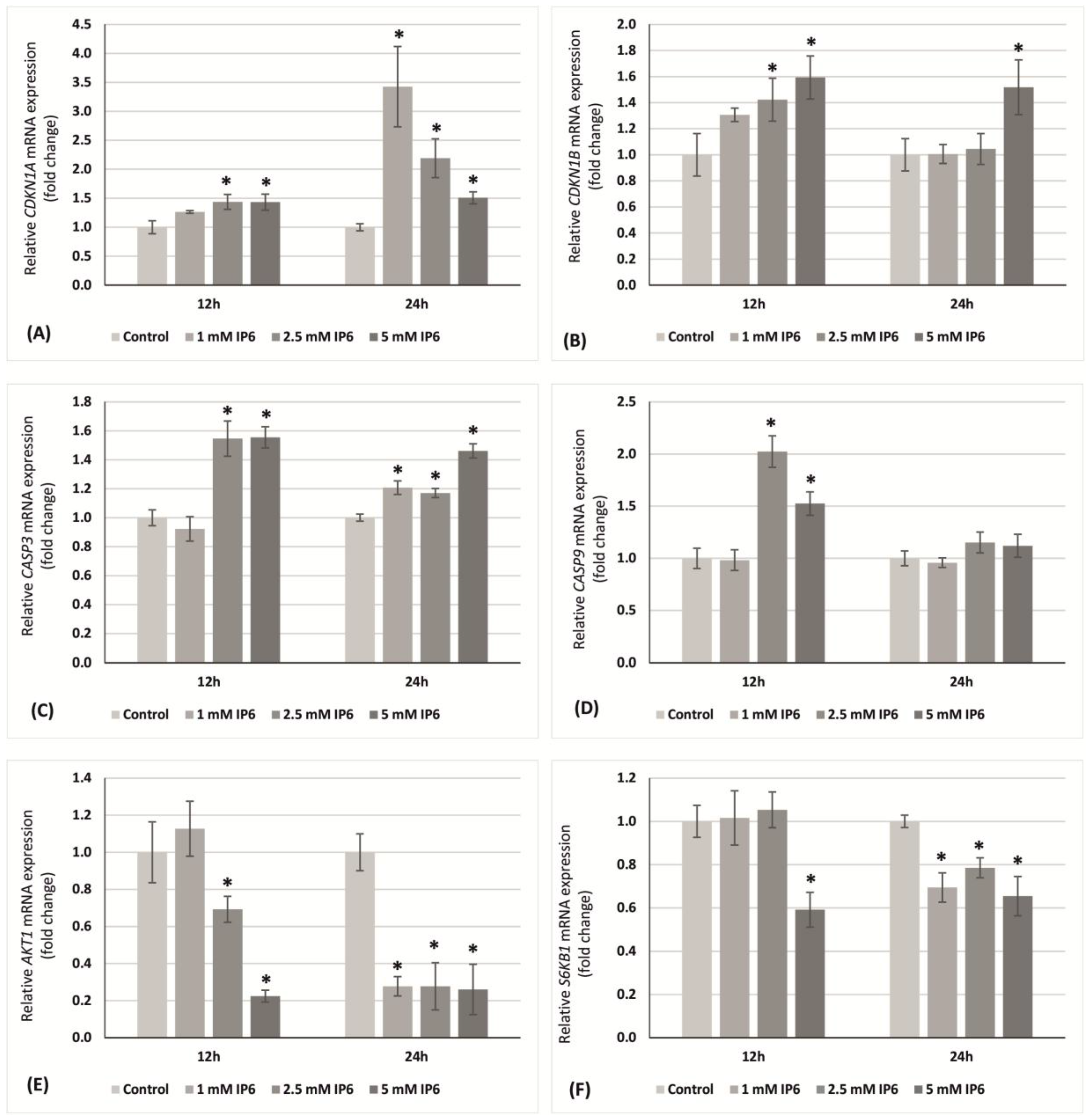
2.4. The Influence of InsP6 on Transcriptional Activity of Genes Encoding p21Waf1/Cip1, p27Kip1, Caspase 3, Caspase 9, AKT1, S6K1 in Caco-2 Cells
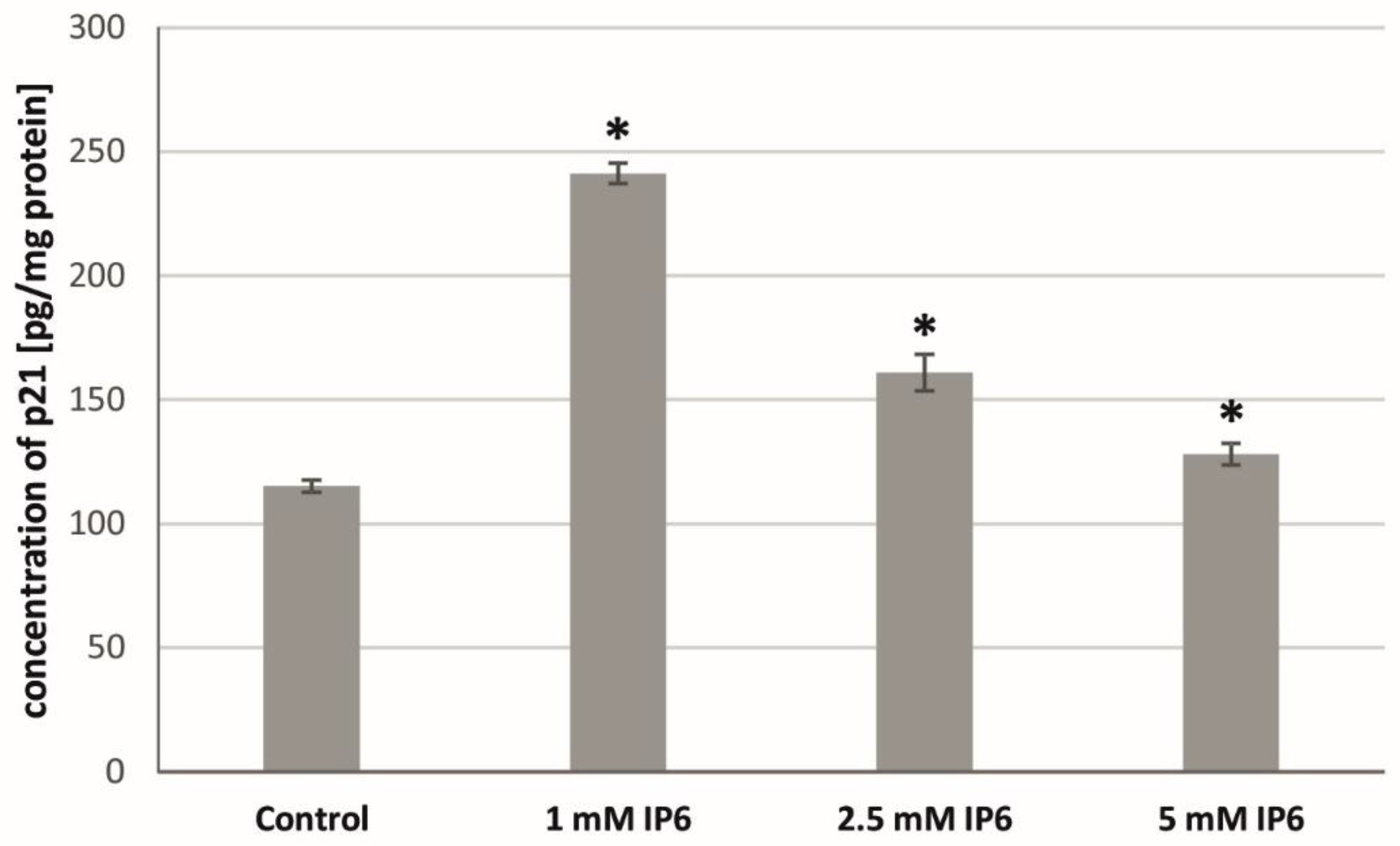
2.5. The Influence of InsP6 on the p21Waf1/Cip1 Protein Level in Caco-2 Cells
2.6. The Influence of InsP6 on Caspase-3 Activity in Caco-2 Cells
2.7. The Influence of InsP6 on AKT1 Activity in Caco-2 Cells
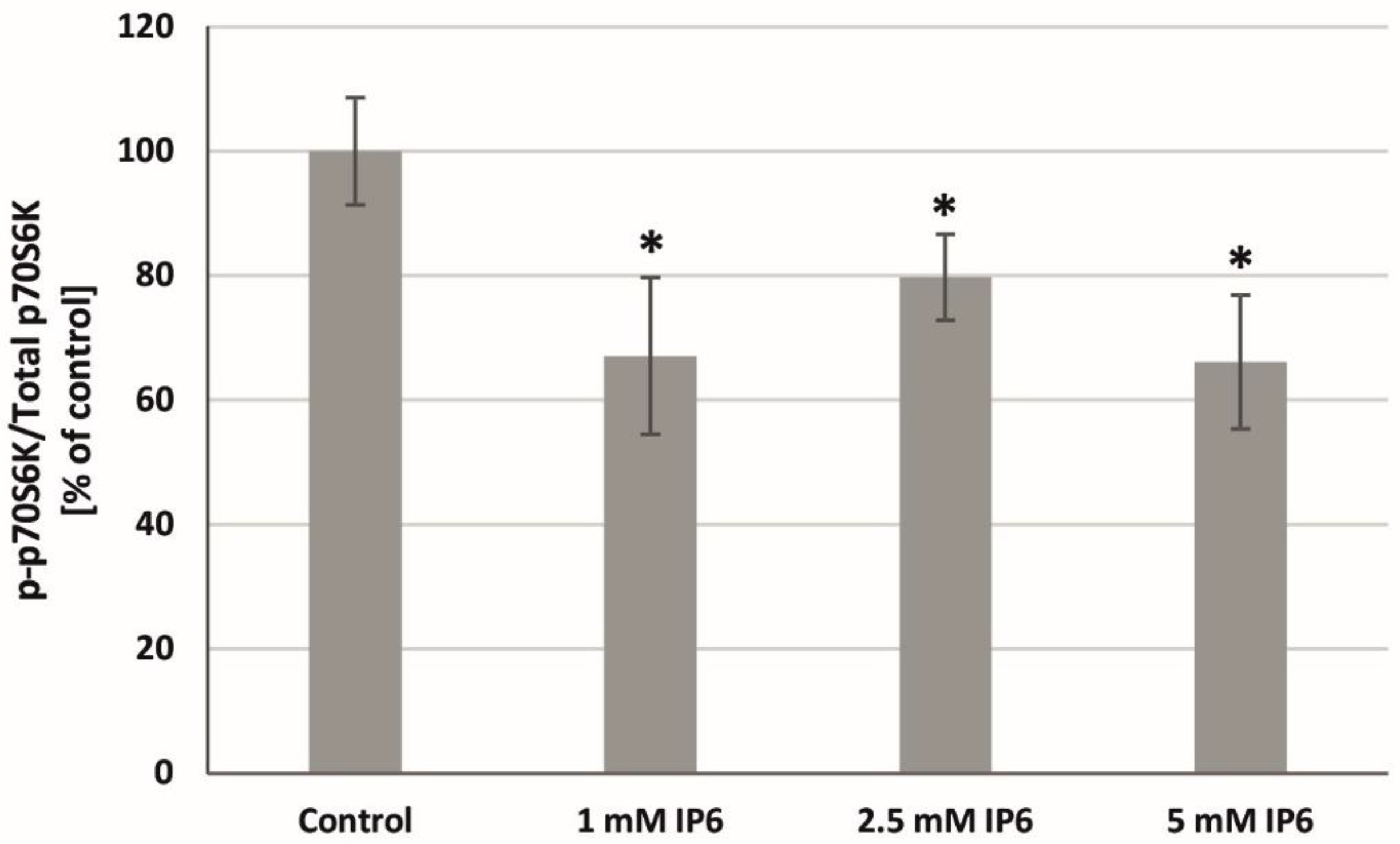
2.8. The Influence of InsP6 on p70S6K Activity in Caco-2 Cells
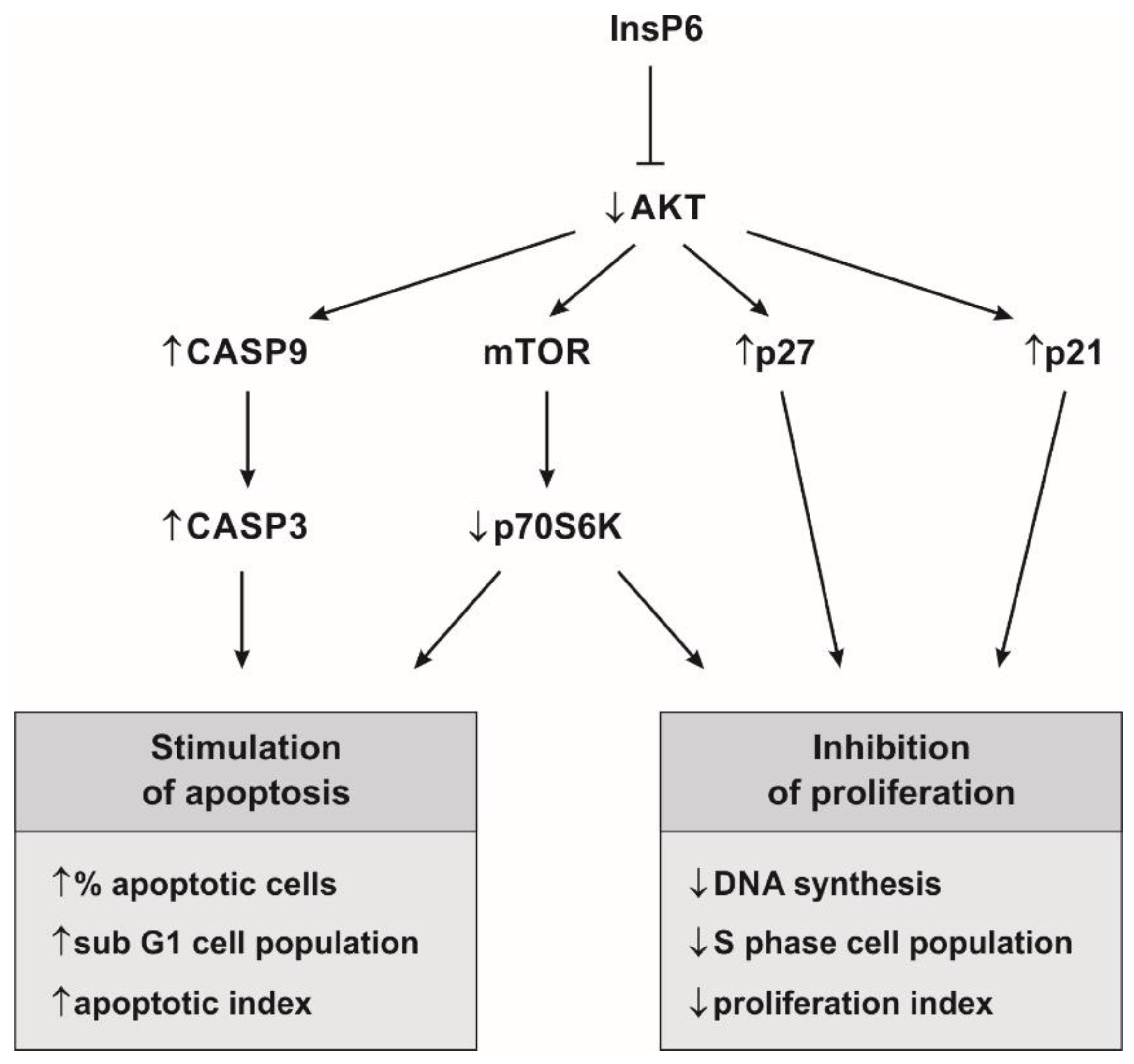
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Preparation of InsP6 Solution
4.3. BrdU Incorporation Assay
4.4. Cell Cycle Analysis
4.5. Apoptosis Assay
4.6. Total RNA Extraction and Quantitative Real-Time RT-PCR (RT-qPCR)
4.7. Measurement of the p21Waf1/Cip1 Protein Level
4.8. Measurement of Caspase-3 Activity
4.9. The p70S6K Activity Assay
4.10. The AKT1 Activity Assay
4.11. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2016. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Niu, Y.B.; Sun, Y.; Zhang, F.; Liu, C.X.; Fan, L.; Mei, Q.B. Role of phytochemicals in colorectal cancer prevention. World J. Gastroenterol. 2015, 21, 9262–9272. [Google Scholar] [CrossRef] [PubMed]
- Chun, K.S.; Kim, E.H.; Lee, S.; Hahm, K.B. Chemoprevention of gastrointestinal cancer: The reality and the dream. Gut Liver 2013, 7, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Terry, P.; Giovannucci, E.; Michels, K.B.; Bergkvist, L.; Hansen, H.; Holmberg, L.; Wolk, A. Fruit, vegetables, dietary fiber, and risk of colorectal cancer. J. Natl. Cancer Inst. 2001, 93, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Phytochemicals in cancer prevention and therapy: Truth or dare? Toxins 2010, 2, 517–551. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Shamsuddin, A.M. Cancer Inhibition by inositol hexaphosphate (IP6) and inositol: From laboratory to clinic. J. Nutr. 2003, 133, 3778S–3784S. [Google Scholar] [PubMed]
- Regidor, P.A.; Schindler, A.E. Myoinositol as a safe and alternative approach in the treatment of infertile PCOS women: A German observational study. Int. J. Endocrinol. 2016, 2016, 9537632. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Shamsuddin, A.M. Protection against cancer by dietary IP6 and inositol. Nutr. Cancer 2006, 55, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Shamsuddin, A.M. [3H]inositol hexaphosphate (phytic acid) is rapidly absorbed and metabolized by murine and human malignant cells in vitro. J. Nutr. 1994, 124, 861–868. [Google Scholar] [PubMed]
- Zi, X.; Singh, R.P.; Agarwal, R. Impairment of erbB1 receptor and fluid-phase endocytosis and associated mitogenic signaling by inositol hexaphosphate in human prostate carcinoma DU145 cells. Carcinogenesis 2000, 21, 2225–2235. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Roy, S.; Raina, K.; Agarwal, C.; Agarwal, R. Inositol hexaphosphate suppresses growth and induces apoptosis in prostate carcinoma cells in culture and nude mouse xenograft: PI3K-Akt pathway as potential target. Cancer Res. 2009, 69, 9465–9472. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Ramakrishna, G.; Tantivejkul, K.; Anderson, L.M.; Ramljak, D. Inositol hexaphosphate (IP6) blocks proliferation of human breast cancer cells through a PKCδ-dependent increase in p27Kip1 and decrease in retinoblastoma protein (pRb) phosphorylation. Breast Cancer Res. Treat. 2005, 91, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Tantivejkul, K.; Vucenik, I.; Shamsuddin, A.M. Inositol hexaphosphate (IP6) inhibits key events of cancer metastasis: I. In vitro studies of adhesion, migration and invasion of MDA-MB 231 human breast cancer cells. Anticancer Res. 2003, 23, 3671–3679. [Google Scholar] [PubMed]
- Graf, E.; Eaton, J.W. Antioxidant functions of phytic acid. Free Radic. Biol. Med. 1990, 8, 61–69. [Google Scholar] [CrossRef]
- Zhang, Z.; Song, Y.; Wang, X.L. Inositol hexaphosphate-induced enhancement of natural killer cell activity correlates with suppression of colon carcinogenesis in rats. World J. Gastroenterol. 2005, 11, 5044–5046. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Song, Y.; Cui, L.; Wen, Z.; Lu, X. Inositol hexaphosphate suppresses growth and induces apoptosis in HT-29 colorectal cancer cells in culture: PI3K/Akt pathway as a potential target. Int. J. Clin. Exp. Pathol. 2015, 8, 1402–1410. [Google Scholar] [PubMed]
- Tian, Y.; Song, Y. Effects of inositol hexaphosphate on proliferation of HT-29 human colon carcinoma cell line. J. World J. Gastroenterol. 2006, 26, 4137–4142. [Google Scholar] [CrossRef]
- Huang, C.; Ma, W.Y.; Hecht, S.S.; Dong, Z. Inositol hexaphosphate inhibits cell transformation and activator protein 1 activation by targeting phosphatidylinositol-3′ kinase. Cancer Res. 1997, 57, 2873–2878. [Google Scholar] [PubMed]
- Ferry, S.; Matsuda, M.; Yoshida, H.; Hirata, M. Inositol hexakisphosphate blocks tumor cell growth by activating apoptotic machinery as well as by inhibiting the Akt/NFκB-mediated cell survival pathway. Carcinogenesis 2002, 23, 2031–2041. [Google Scholar] [CrossRef] [PubMed]
- Guertin, D.A.; Sabatini, D.M. Defining the role of mTOR in cancer. Cancer Cell 2007, 12, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.M.; Gulhati, P.; Rampy, B.A.; Han, Y.; Rychahou, P.G.; Doan, H.Q.; Weiss, H.L.; Evers, B.M. Novel expression patterns of PI3K/Akt/mTOR signaling pathway components in colorectal cancer. J. Am. Coll. Surg. 2010, 210, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Manning, B.D.; Cantley, L.C. AKT/PKB signaling: Navigating downstream. Cell 2007, 129, 1261–1274. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Ouyang, G.; Bao, S. The activation of Akt/PKB signaling pathway and cell survival. J. Cell Mol. Med. 2005, 9, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.M.; Evangelisti, C.; Chiarini, F.; Grimaldi, C.; McCubrey, J.A. The emerging role of the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin signaling network in cancer stem cell biology. Cancers. 2010, 2, 1576–1596. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.M.; Blenis, J. Molecular mechanisms of mTOR-mediated translational control. Nat. Rev. Mol. Cell Biol. 2009, 10, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Ekim, B.; Magnuson, B.; Acosta-Jaquez, H.A.; Keller, J.A.; Feener, E.P.; Fingar, D.C. mTOR kinase domain phosphorylation promotes mTORC1 signaling, cell growth, and cell cycle progression. Mol. Cell Biol. 2011, 31, 2787–2801. [Google Scholar] [CrossRef] [PubMed]
- Amaral, C.L.; Freitas, L.B.; Tamura, R.E.; Tavares, M.R.; Pavan, I.C.; Bajgelman, M.C.; Simabuco, F.M. S6Ks isoforms contribute to viability, migration, docetaxel resistance and tumor formation of prostate cancer cells. BMC Cancer 2016, 16, 602. [Google Scholar] [CrossRef] [PubMed]
- Melling, N.; Simon, R.; Izbicki, J.R.; Terracciano, L.M.; Bokemeyer, C.; Sauter, G.; Marx, A.H. Expression of phospho-mTOR kinase is abundant in colorectal cancer and associated with left-sided tumor localization. Int. J. Clin. Exp. Pathol. 2015, 8, 7009–7015. [Google Scholar] [PubMed]
- Lu, Q.; Wang, J.; Yu, G.; Guo, T.; Hu, C.; Ren, P. Expression and clinical significance of mammalian target of rapamycin/P70 ribosomal protein S6 kinase signaling pathway in human colorectal carcinoma tissue. Oncol. Lett. 2015, 10, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Shafie, N.H.; Esa, N.M.; Ithnin, H.; Saad, N.; Pandurangan, A.K. Pro-apoptotic effect of rice bran inositol hexaphosphate (IP6) on HT-29 colorectal cancer cells. Int. J. Mol. Sci. 2013, 14, 23545–23558. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.L.; Cantley, L.C. PI3K pathway alterations in cancer: Variations on a theme. Oncogene 2008, 27, 5497–5510. [Google Scholar] [CrossRef] [PubMed]
- Cardone, M.H.; Roy, N.; Stennicke, H.R.; Salvesen, G.S.; Franke, T.F.; Stanbridge, E.; Frisch, S.; Reed, J.C. Regulation of cell death protease caspase-9 by phosphorylation. Science 1998, 282, 1318–1321. [Google Scholar] [CrossRef] [PubMed]
- Slee, E.A.; Adrain, C.; Martin, S.J. Executioner caspase-3, -6, and -7 perform distinct, non-redundant roles during the demolition phase of apoptosis. J. Biol. Chem. 2001, 276, 7320–7326. [Google Scholar] [CrossRef] [PubMed]
- Abbas, T.; Dutta, A. p21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, B.; Ekim, B.; Fingar, D.C. Regulation and function of ribosomal protein S6 kinase (S6K) within mTOR signalling networks. Biochem. J. 2012, 441, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.P.; Agarwal, C.; Agarwal, R. Inositol hexaphosphate inhibits growth, and induces G1 arrest and apoptotic death of prostate carcinoma DU145 cells: Modulation of CDKI-CDK-cyclin and pRb-related protein-E2F complexes. Carcinogenesis 2003, 24, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, C.; Dhanalakshmi, S.; Singh, R.P.; Agarwal, R. Inositol hexaphosphate inhibits growth and induces G1 arrest and apoptotic death of androgen-dependent human prostate carcinoma LNCaP cells. Neoplasia 2004, 6, 646–659. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Gu, M.; Ramasamy, K.; Singh, R.P.; Agarwal, C.; Siriwardana, S.; Sclafani, R.A.; Agarwal, R. p21/Cip1 and p27/Kip1 Are essential molecular targets of inositol hexaphosphate for its antitumor efficacy against prostate cancer. Cancer Res. 2009, 69, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Somasundar, P.; Riggs, D.R.; Jackson, B.J.; Cunningham, C.; Vona-Davis, L.; McFadden, D.W. Inositol hexaphosphate (IP6): A novel treatment for pancreatic cancer. J. Surg. Res. 2005, 126, 199–203. [Google Scholar] [CrossRef] [PubMed]
- El-Sherbiny, Y.M.; Cox, M.C.; Ismail, Z.A.; Shamsuddin, A.M.; Vucenik, I. G0/G1 arrest and S phase inhibition of human cancer cell lines by inositol hexaphosphate (IP6). Anticancer Res. 2001, 21, 2393–2403. [Google Scholar] [PubMed]
- Rizvi, I.; Riggs, D.R.; Jackson, B.J.; Ng, A.; Cunningham, C.; McFadden, D.W. Inositol hexaphosphate (IP6) inhibits cellular proliferation in melanoma. J. Surg. Res. 2006, 133, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Schröterová, L.; Hasková, P.; Rudolf, E.; Cervinka, M. Effect of phytic acid and inositol on the proliferation and apoptosis of cells derived from colorectal carcinoma. Oncol. Rep. 2010, 23, 787–793. [Google Scholar] [PubMed]
- Nurul-Husna, S.; Norhaizan, M.E.; Hairuszah, I.; Abdah, M.A.; Norazalina, S.; Norsharina, I. Rice bran phytic acid (IP6) induces growth inhibition, cell cycle arrest and apoptosis on human colorectal adenocarcinoma cells. J. Med. Plant. Res. 2010, 4, 2283–2289. [Google Scholar]
- Verbsky, J.; Majerus, P.W. Increased levels of inositol hexakisphosphate (InsP6) protect HEK293 cells from tumor necrosis factor α- and Fas-induced apoptosis. J. Biol. Chem. 2005, 280, 29263–29268. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Liu, C.; Li, X.; Yang, F.; Cheng, L.; Liu, C.; Song, Y. Inositol hexaphosphate suppresses colorectal cancer cell proliferation via the Akt/GSK-3β/β-catenin signaling cascade in a 1,2-dimethylhydrazine-induced rat model. Eur. J. Pharmacol. 2017, 805, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Rafiee, P.; Binion, D.G.; Wellner, M.; Behmaram, B.; Floer, M.; Mitton, E.; Nie, L.; Zhang, Z.; Otterson, M.F. Modulatory effect of curcumin on survival of irradiated human intestinal microvascular endothelial cells: Role of Akt/mTOR and NF-κB. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G865–G877. [Google Scholar] [CrossRef] [PubMed]
- Kapral, M.; Sośnicki, S.; Wawszczyk, J.; Węglarz, L. Influence of inositol hexaphosphate on the expression of selected proliferation markers in IL-1β-stimulated intestinal epithelial cells. Acta Pol. Pharm. 2014, 71, 987–993. [Google Scholar] [PubMed]
- Owen, R.W.; Weisgerber, U.M.; Spiegelhalder, B.; Bartsch, H. Faecal phytic acid and its relation to other putative markers of risk for colorectal cancer. Gut 1996, 38, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Passaniti, A.; Vitolo, M.I.; Tantivejkul, K.; Eggleton, P.; Shamsuddin, A.M. Anti-angiogenic activity of inositol hexaphosphate (IP6). Carcinogenesis 2004, 25, 2115–2123. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, E.; Vignati, S.; Maffucci, T.; Innominato, P.F.; Riley, A.M.; Potter, B.V.; Pandolfi, P.P.; Broggini, M.; Iacobelli, S.; Innocenti, P.; et al. Inositol pentakisphosphate promotes apoptosis through the PI 3-K/Akt pathway. Oncogene 2004, 23, 1754–1765. [Google Scholar] [CrossRef] [PubMed]
- Maffucci, T.; Piccolo, E.; Cumashi, A.; Iezzi, M.; Riley, A.M.; Saiardi, A.; Godage, H.Y.; Rossi, C.; Broggini, M.; Iacobelli, S.; et al. Inhibition of the phosphatidylinositol 3-kinase/Akt pathway by inositol pentakisphosphate results in antiangiogenic and antitumor effects. Cancer Res. 2005, 65, 8339–8349. [Google Scholar] [CrossRef] [PubMed]
- Dinicola, S.; Fabrizi, G.; Masiello, M.G.; Proietti, S.; Palombo, A.; Minini, M.; Harrath, A.H.; Alwasel, S.H.; Ricci, G.; Catizone, A.; et al. Inositol induces mesenchymal-epithelial reversion in breast cancer cells through cytoskeleton rearrangement. Exp. Cell Res. 2016, 345, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Robey, R.B.; Hay, N. Is Akt the “Warburg kinase”?-Akt-energy metabolism interactions and oncogenesis. Semin. Cancer Biol. 2009, 19, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Krześlak, A. Akt kinase: A key regulator of metabolism and progression of tumors. Postepy Hig. Med. Dosw. 2010, 64, 490–503. [Google Scholar]
- Howell, J.J.; Ricoult, S.J.H.; Ben-Sahra, I.; Manning, B.D. A growing role for mTOR in promoting anabolic metabolism. Biochem. Soc. Trans. 2013, 41, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Xu, Z.; Zhang, Z.; Li, L.; Pan, Q.; Zheng, F.; Li, H. Association of PI3K/AKT/mTOR pathway genetic variants with type 2 diabetes mellitus in Chinese. Diabetes Res. Clin. Pract. 2017, 128, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960–976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, H.; Zhu, J.; Xu, J.; Ding, K. Mollugin induces tumor cell apoptosis and autophagy via the PI3K/AKT/mTOR/p70S6K and ERK signaling pathways. Biochem. Biophys. Res. Commun. 2014, 450, 247–254. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |


| Gene | Primer Sequence | Product Size (Base Pair) |
|---|---|---|
| CDNK1A | F: 5′ AGGGATTTCTTCTGTTCAGG 3′ | 185 |
| R: 5′ GACAAAGTCGAAGTTCCATC 3′ | ||
| CDNK1B | F: 5′ AAAATGTTTCAGACGGTTCC 3′ | 93 |
| R: 5′ ATTCGAGCTGTTTACGTTTG 3′ | ||
| CASP3 | F: 5′GGCCTGCCGTGGTACAGAACTGG 3′ | 174 |
| R: 5′ AGCGACTGGATGAACCAGGAGCCA3′ | ||
| CASP9 | F: 5′GACCGGAAACACCCAGACCAGTGGA 3′ | 125 |
| R: 5′GCAGTGGCCACAGGGCTCCAT 3′ | ||
| AKT1 | F: 5′AAGTACTCTTTCCAGACCC 3′ | 197 |
| R: 5′ TTCTCCAGCTTGAGGTC 3′ | ||
| S6KB1 | F: 5′ATTCATGATGGAACAGTCAC 3′ | 121 |
| R: 5′ACATTAATGCTCCCAAACTC 3′ |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kapral, M.; Wawszczyk, J.; Jesse, K.; Paul-Samojedny, M.; Kuśmierz, D.; Węglarz, L. Inositol Hexaphosphate Inhibits Proliferation and Induces Apoptosis of Colon Cancer Cells by Suppressing the AKT/mTOR Signaling Pathway. Molecules 2017, 22, 1657. https://doi.org/10.3390/molecules22101657
Kapral M, Wawszczyk J, Jesse K, Paul-Samojedny M, Kuśmierz D, Węglarz L. Inositol Hexaphosphate Inhibits Proliferation and Induces Apoptosis of Colon Cancer Cells by Suppressing the AKT/mTOR Signaling Pathway. Molecules. 2017; 22(10):1657. https://doi.org/10.3390/molecules22101657
Chicago/Turabian StyleKapral, Małgorzata, Joanna Wawszczyk, Katarzyna Jesse, Monika Paul-Samojedny, Dariusz Kuśmierz, and Ludmiła Węglarz. 2017. "Inositol Hexaphosphate Inhibits Proliferation and Induces Apoptosis of Colon Cancer Cells by Suppressing the AKT/mTOR Signaling Pathway" Molecules 22, no. 10: 1657. https://doi.org/10.3390/molecules22101657





