Diverse Phytochemicals and Bioactivities in the Ancient Fruit and Modern Functional Food Pomegranate (Punica granatum)
Abstract
:1. Introduction
2. Occurrence and Structure of Diverse Phytochemicals in Pomegranate
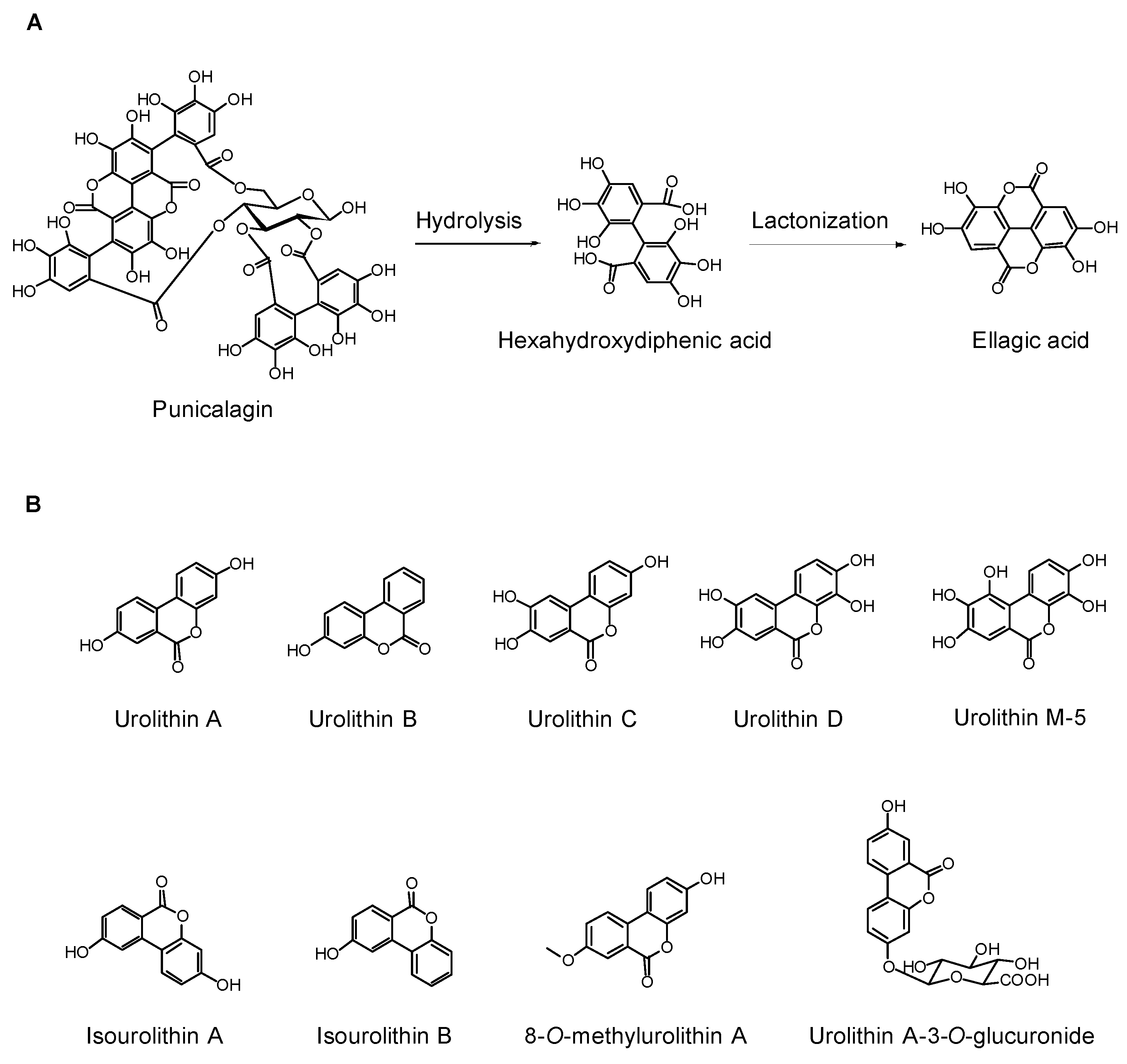
2.1. Ellagitannins, Gallotannins, and Their Derivatives
2.2. Flavonoids
2.3. Lignans
2.4. Triterpenoids and Phytosterols
2.5. Alkaloids and Indolamines
2.6. Fatty Acids and Lipids
2.7. Organic Acids and Phenolic Acids
2.8. Other Compounds
3. Interactions of Pomegranate Phytochemicals
4. Functions of Pomegranate ATs in Human Nutrition and Health
5. Roles of Urolithins, the ET-Derived Metabolites, in Human Nutrition and Health
5.1. Breast and Endometrial Cancers
5.2. Prostate Cancer
5.3. Colon and Bladder Cancers
5.4. Cardiovascular Diseases
5.5. Obesity
5.6. Aging
6. Future Perspectives
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dell’Agli, M.; Galli, G.V.; Corbett, Y.; Taramelli, D.; Lucantoni, L.; Habluetzel, A.; Maschi, O.; Caruso, D.; Giavarini, F.; Romeo, S.; et al. Antiplasmodial activity of Punica granatum L. fruit rind. J. Ethnopharmacol. 2009, 125, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Qnais, E.Y.; Elokda, A.S.; Abu Ghalyun, Y.Y.; Abdulla, F.A. Antidiarrheal activity of the aqueous extract of Punica granatum (pomegranate) peels. Pharm. Biol. 2007, 45, 715–720. [Google Scholar] [CrossRef]
- Das, A.K.; Mandal, S.C.; Banerjee, S.K.; Sinha, S.; Das, J.; Saha, B.P.; Pal, M. Studies on antidiarrhoeal activity of Punica granatum seed extract in rats. J. Ethnopharmacol. 1999, 68, 205–208. [Google Scholar] [CrossRef]
- Banihani, S.; Swedan, S.; Alguraan, Z. Pomegranate and type 2 diabetes. Nutr. Res. 2013, 33, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Bhowmik, D.; Gopinath, H.; Kumar, B.; Duraivel, S.; Aravind, G.; Kumar, K. Medicinal uses of Punica granatum and its health benefits. J. Pharmacogn. Phytochem. 2013, 1, 28–35. [Google Scholar]
- Newman, R.; Lansky, E.; Block, M. Pomegranate: The Most Medicinal Fruit; Basic Health Publications: Laguna Beach, CA, USA, 2007. [Google Scholar]
- Kaufman, M.; Wiesman, Z. Pomegranate oil analysis with emphasis on MALDI-TOF/MS triacylglycerol fingerprinting. J. Agric. Food Chem. 2007, 55, 10405–10413. [Google Scholar] [CrossRef] [PubMed]
- Melgarejo, P.; Salazar, D.M.; Artés, F. Organic acids and sugars composition of harvested pomegranate fruits. Eur. Food Res. Technol. 2000, 211, 185–190. [Google Scholar] [CrossRef]
- Pande, G.; Akoh, C.C. Antioxidant capacity and lipid characterization of six Georgia-grown pomegranate cultivars. J. Agric. Food Chem. 2009, 57, 9427–9436. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Z.; Zhou, B.; Jin, L.; Yu, H.; Liu, L.; Liu, Y.; Qin, C.; Xie, S.; Zhu, F. In vitro antioxidant and antiproliferative effects of ellagic acid and its colonic metabolite, urolithins, on human bladder cancer T24 cells. Food Chem. Toxicol. 2013, 59, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Tzulker, R.; Glazer, I.; Bar-Ilan, I.; Holland, D.; Aviram, M.; Amir, R. Antioxidant activity, polyphenol content, and related compounds in different fruit juices and homogenates prepared from 29 different pomegranate accessions. J. Agric. Food Chem. 2007, 55, 9559–9570. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.; Lee, R.; Hardy, M.; Heber, D. Rapid large scale purification of ellagitannins from pomegranate husk, a by-product of the commercial juice industry. Sep. Purif. Technol. 2005, 41, 49–55. [Google Scholar] [CrossRef]
- Ono, N.; Bandaranayake, P.C.G.; Tian, L. Establishment of pomegranate (Punica granatum) hairy root cultures for genetic interrogation of the hydrolyzable tannin biosynthetic pathway. Planta 2012, 236, 931–941. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Nonaka, G.-I.; Nishioka, I. Tannins and related compounds. XLI.: Isolation and characterization of novel ellagitannins, punicacorteins A, B, C, and D, and punigluconin from the bark of Punica granatum L. Chem. Pharm. Bull. Tokyo 1986, 34, 656–663. [Google Scholar] [CrossRef]
- El-Toumy, S.; Marzouk, M.; Rauwald, H. Ellagi- and gallotannins from Punica granatum heartwood. Pharmazie 2001, 56, 823–824. [Google Scholar] [PubMed]
- El-Toumy, S.; Rauwald, H. Two new ellagic acid rhamnosides from Punica granatum heartwood. Planta Med. 2003, 69, 682–684. [Google Scholar] [PubMed]
- El-Toumy, S.A.A.; Rauwald, H.W. Two ellagitannins from Punica granatum heartwood. Phytochemistry 2002, 61, 971–974. [Google Scholar] [CrossRef]
- Tanaka, T.; Nonaka, G.-I.; Nishioka, I. Punicafolin, an ellagitannin from the leaves of Punica granatum. Phytochemistry 1985, 24, 2075–2078. [Google Scholar] [CrossRef]
- Nawwar, M.A.M.; Hussein, S.A.M.; Merfort, I. Leaf phenolics of Punica granatum. Phytochemistry 1994, 37, 1175–1177. [Google Scholar] [CrossRef]
- Hussein, S.A.M.; Barakat, H.H.; Merfort, I.; Nawwar, M.A.M. Tannins from the leaves of Punica granatum. Phytochemistry 1997, 45, 819–823. [Google Scholar] [CrossRef]
- Wang, R.; Wei, W.; Wang, L.; Liu, R.; Yi, D.; Du, L. Constituents of the flowers of Punica granatum. Fitoterapia 2006, 77, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Ding, Y.; Wan, C.; Li, L.; Xu, J.; Liu, K.; Slitt, A.; Ferreira, D.; Khan, I.A.; Seeram, N.P. Antidiabetic ellagitannins from pomegranate flowers: Inhibition of α-glucosidase and lipogenic gene expression. Org. Lett. 2012, 14, 5358–5361. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Morikawa, T.; Ninomiya, K.; Imura, K.; Muraoka, O.; Yuan, D.; Yoshikawa, M. Medicinal flowers. XXIII. New taraxastane-type triterpene, punicanolic acid, with tumor necrosis factor-a inhibitory activity from the flowers of Punica granatum. Chem. Pharm. Bull. Tokyo 2008, 56, 1628–1631. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Wan, C.; Ma, H.; Seeram, N.P. New phenolics from the flowers of Punica granatum and their in vitro α-glucosidase inhibitory activities. Planta Med. 2013, 79, 1674–1679. [Google Scholar] [CrossRef] [PubMed]
- Bagri, P.; Ali, M.; Sultana, S.; Aeri, V. New flavonoids from Punica granatum flowers. Chem. Nat. Compd. 2010, 46, 201–204. [Google Scholar] [CrossRef]
- Chauhan, D.; Chauhan, J.S. Flavonoid diglycoside from Punica granatum. Pharm. Biol. 2001, 39, 155–157. [Google Scholar] [CrossRef]
- Srivastava, R.; Chauhan, D.; Chauhan, J. Flavonoid diglycosides from Punica granatum. Indian J. Chem. Sect. B 2001, 40B, 170–172. [Google Scholar]
- Moneam, N.M.A.; El Sharaky, A.S.; Badreldin, M.M. Oestrogen content of pomegranate seeds. J. Chromatogr. 1988, 438, 438–442. [Google Scholar] [CrossRef]
- Fischer, U.A.; Jaksch, A.V.; Carle, R.; Kammerer, D.R. Determination of lignans in edible and nonedible parts of pomegranate (Punica granatum L.) and products derived therefrom, particularly focusing on the quantitation of isolariciresinol using HPLC-DAD-ESI/MSn. J. Agric. Food Chem. 2012, 60, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Li, P.; Koreishi, M.; Nagatomo, A.; Nishida, N.; Yoshida, T. Ellagitannin oligomers and a neolignan from pomegranate arils and their inhibitory effects on the formation of advanced glycation end products. Food Chem. 2014, 152, 323–330. [Google Scholar] [CrossRef] [PubMed]
- El Wahab, S.; El Fiki, N.; Mostafa, F.; Hassan, A. Characterization of certain steroid hormones in Punica granatum L. seeds. Bull. Fac. Pharm. 1998, 36, 11–15. [Google Scholar]
- Heftmann, E.; Ko, S.-T.; Bennett, R.D. Identification of estrone in pomegranate seeds. Phytochemistry 1966, 5, 1337–1339. [Google Scholar] [CrossRef]
- Choi, D.W.; Kim, J.Y.; Choi, S.H.; Jung, H.S.; Kim, H.J.; Cho, S.Y.; Kang, C.S.; Chang, S.Y. Identification of steroid hormones in pomegranate (Punica granatum) using HPLC and GC-mass spectrometry. Food Chem. 2006, 96, 562–571. [Google Scholar] [CrossRef]
- Neuhofer, H.; Witte, L.; Gorunovic, M.; Czygan, F. Alkaloids in the bark of Punica granatum L. (pomegranate) from Yugoslavia. Pharmazie 1993, 48, 389–391. [Google Scholar]
- Rafiq, Z.; Narasimhan, S.; Vennila, R.; Vaidyanathan, R. Punigratane, a novel pyrrolidine alkaloid from Punica granatum rind with putative efflux inhibition activity. Nat. Prod. Res. 2016, 25, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Badria, F. Melatonin, serotonin, and tryptamine in some egyptian food and medicinal plants. J. Med. Food 2004, 5, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Fatope, M.O.; Al Burtomani, S.K.S.; Takeda, Y. Monoacylglycerol from Punica granatum seed oil. J. Agric. Food Chem. 2002, 50, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Lal, C.; Sharma, M.; Shakyawar, D.; Raja, A.; Sharma, K.; Pareek, P. Natural Dye constituents from rind of Punica granatum and its application on Pashmina fabrics. Arch. Appl. Sci. Res. 2011, 3, 350–357. [Google Scholar]
- Yusuph, M.; Mann, J. A triglyceride from Punica granatum. Phytochemistry 1997, 44, 1391–1392. [Google Scholar] [CrossRef]
- Tsuyuki, H.; Ito, S.; Nakatsukasa, Y. Studies on the lipids in pomegranate seeds. Bull. Coll. Agric. Vet. Med. Nihon Univ. 1981, 38, 141–148. [Google Scholar]
- Mena, P.; Calani, L.; Dall’Asta, C.; Galaverna, G.; García-Viguera, C.; Bruni, R.; Crozier, A.; Del Rio, D. Rapid and comprehensive evaluation of (poly)phenolic compounds in pomegranate (Punica granatum L.) juice by UHPLC-MSn. Molecules 2012, 17, 14821–14840. [Google Scholar] [CrossRef] [PubMed]
- Poyrazoğlu, E.; Gökmen, V.; Artιk, N. Organic acids and phenolic compounds in pomegranates (Punica granatum L.) grown in Turkey. J. Food Compos. Anal. 2002, 15, 567–575. [Google Scholar] [CrossRef]
- Artik, N.; Murakami, H.; Mori, T. Determination of phenolic compounds in pomegranate juice by using HPLC. Fruit Process. 1998, 8, 492–499. [Google Scholar]
- Wang, R.-F.; Xie, W.-D.; Zhang, Z.; Xing, D.-M.; Ding, Y.; Wang, W.; Ma, C.; Du, L.-J. Bioactive compounds from the seeds of Punica granatum (pomegranate). J. Nat. Prod. 2004, 67, 2096–2098. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.; Jiang, W.; Mo, H.; Bravo, L.; Froom, P.; Yu, W.; Harris, N.; Neeman, I.; Campbell, M. Possible synergistic prostate cancer suppression by anatomically discrete pomegranate fractions. Investig. New Drugs 2005, 23, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.; Harrison, G.; Froom, P.; Jiang, W. Pomegranate (Punica granatum) pure chemical show possible synergistic inhibition of human PC-3 prostate cancer cell invasion across Matrigel. Investig. New Drugs 2005, 23, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Adams, L.S.; Henning, S.M.; Niu, Y.; Zhang, Y.; Nair, M.G.; Heber, D. In vitro antiproliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. J. Nutr. Biochem. 2005, 16, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.W.; Chen, Y.-Y.; Chen, C.Y.O. Contributions of phenolics and added vitamin C to the antioxidant capacity of pomegranate and grape juices: Synergism and antagonism among constituents. Int. J. Food Sci. Technol. 2013, 48, 2650–2658. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.W.; Chen, Y.-Y.; Kamil, A.G.; Chen, C.Y.O. Assay dilution factors confound measures of total antioxidant capacity in polyphenol-rich juices. J. Food Sci. 2012, 77, H69–H75. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.; Debnath, S.; Hazra, S.; Ghosh, S.; Ray, R.; Hazra, B. Pomegranate pericarp extract enhances the antibacterial activity of ciprofloxacin against extended-spectrum β-lactamase (ESBL) and metallo-β-lactamase (MBL) producing Gram-negative bacilli. Food Chem. Toxicol. 2012, 50, 4302–4309. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.F.; Monteiro, V.V.S.; de Souza Gomes, R.; do Carmo, M.M.; da Costa, G.V.; Ribera, P.C.; Monteiro, M.C. Action mechanism and cardiovascular effect of anthocyanins: A systematic review of animal and human studies. J. Transl. Med. 2016, 14, 315. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.-W.; Gong, C.-C.; Song, H.-F.; Cui, Y.-Y. Effects of anthocyanins on the prevention and treatment of cancer. Br. J. Pharmacol. 2017, 174, 1226–1243. [Google Scholar] [CrossRef] [PubMed]
- Azzini, E.; Giacometti, J.; Russo, G.L. Antiobesity effects of anthocyanins in preclinical and clinical studies. Oxid. Med. Cell. Longev. 2017, 2017, 2740364. [Google Scholar] [CrossRef] [PubMed]
- Morais, C.A.; de Rosso, V.V.; Estadella, D.; Pisani, L.P. Anthocyanins as inflammatory modulators and the role of the gut microbiota. J. Nutr. Biochem. 2016, 33, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Liobikas, J.; Skemiene, K.; Trumbeckaite, S.; Borutaite, V. Anthocyanins in cardioprotection: A path through mitochondria. Phytother. Res. 2016, 113, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wang, P.; Luo, Y.; Zhao, M.; Chen, F. Health benefits of anthocyanins and molecular mechanisms: Update from recent decade. Crit. Rev. Food Sci. Nutr. 2017, 57, 1729–1741. [Google Scholar] [CrossRef] [PubMed]
- Borges, F.; Fernandes, E.; Roleira, F. Progress towards the discovery of xanthine oxidase inhibitors. Curr. Med. Chem. 2002, 9, 195–217. [Google Scholar] [CrossRef] [PubMed]
- Pergola, C.; Rossi, A.; Dugo, P.; Cuzzocrea, S.; Sautebin, L. Inhibition of nitric oxide biosynthesis by anthocyanin fraction of blackberry extract. Nitric Oxide 2006, 15, 30–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dreiseitel, A.; Korte, G.; Schreier, P.; Oehme, A.; Locher, S.; Hajak, G.; Sand, P.G. sPhospholipase A2 is inhibited by anthocyanidins. J. Neural Transm. 2009, 116, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- Speciale, A.; Canali, R.; Chirafisi, J.; Saija, A.; Virgili, F.; Cimino, F. Cyanidin-3-O-glucoside protection against TNF-α-induced endothelial dysfunction: Involvement of nuclear factor-κB signaling. J. Agric. Food Chem. 2010, 58, 12048–12054. [Google Scholar] [CrossRef] [PubMed]
- DeFuria, J.; Bennett, G.; Strissel, K.J.; Perfield, J.W.; Milbury, P.E.; Greenberg, A.S.; Obin, M.S. Dietary blueberry attenuates whole-body insulin resistance in high fat-fed mice by reducing adipocyte death and its inflammatory sequelae. J. Nutr. 2009, 139, 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Mykkänen, O.T.; Huotari, A.; Herzig, K.-H.; Dunlop, T.W.; Mykkänen, H.; Kirjavainen, P.V. Wild blueberries (Vaccinium myrtillus) alleviate inflammation and hypertension associated with developing obesity in mice fed with a high-fat diet. PLoS ONE 2014, 9, e114790. [Google Scholar] [CrossRef] [PubMed]
- Baum, J.; Howard, L.; Prior, R.; Lee, S. Effect of Aronia melanocarpa (black chokeberry) supplementation on the development of obesity in mice fed a high-fat diet. J. Berry Res. 2016, 6, 203–212. [Google Scholar] [CrossRef]
- Wright, O.R.L.; Netzel, G.A.; Sakzewski, A.R. A randomized, double-blind, placebo-controlled trial of the effect of dried purple carrot on body mass, lipids, blood pressure, body composition, and inflammatory markers in overweight and obese adults: The QUENCH trial. Can. J. Physiol. Pharmacol. 2013, 91, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Fimognari, C.; Berti, F.; Nüsse, M.; Cantelli-Forti, G.; Hrelia, P. Induction of apoptosis in two human leukemia cell lines as well as differentiation in human promyelocytic cells by cyanidin-3-O-β-glucopyranoside. Biochem. Pharmacol. 2004, 67, 2047–2056. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Zhao, C.; Schoene, N.; Guisti, M.; Moyer, M.; Magnuson, B. Anthocyanin-rich extract from Aronia meloncarpa E induces a cell cycle block in colon cancer but not normal colonic cells. Nutr. Cancer 2003, 46, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Park, S.; Park, S.; Park, J.; Shin, D.; Kim, G.; Ryu, C.; Shin, S.; Jung, J.; Kang, H.; Lee, W.; Choi, Y. Induction of apoptosis in human leukemia U937 cells by anthocyanins through down-regulation of Bcl-2 and activation of caspases. Int. J. Oncol. 2009, 34, 1077–1083. [Google Scholar] [PubMed]
- De Nigris, F.; Balestrieri, M.L.; Williams-Ignarro, S.; D’Armiento, F.P.; Fiorito, C.; Ignarro, L.J.; Napoli, C. The influence of pomegranate fruit extract in comparison to regular pomegranate juice and seed oil on nitric oxide and arterial function in obese Zucker rats. Nitric Oxide 2007, 17, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Mohan, M.; Waghulde, H.; Kasture, S. Effect of pomegranate juice on Angiotensin II-induced hypertension in diabetic wistar rats. Phytother. Res. 2010, 24, S196–S203. [Google Scholar] [CrossRef] [PubMed]
- Asgary, S.; Sahebkar, A.; Afshani, M.R.; Keshvari, M.; Haghjooyjavanmard, S.; Rafieian-Kopaei, M. Clinical evaluation of blood pressure lowering, endothelial function improving, hypolipidemic and anti-inflammatory effects of pomegranate juice in hypertensive subjects. Phytother. Res. 2014, 28, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Ben-Simhon, Z.; Judeinstein, S.; Trainin, T.; Harel-Beja, R.; Bar-Ya’akov, I.; Borochov-Neori, H.; Holland, D. A “white” anthocyanin-less pomegranate (Punica granatum L.) caused by an insertion in the coding region of the leucoanthocyanidin dioxygenase (LDOX; ANS) gene. PLoS ONE 2015, 10, e0142777. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yuan, Z.; Feng, L.; Fang, Y. Cloning and expression of anthocyanin biosynthetic genes in red and white pomegranate. J. Plant Res. 2015, 128, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Okuda, T.; Yoshida, T.; Hatano, T. Ellagitannins as active constituents of medicinal plants. Planta Med. 1989, 55, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.E.; Matel, H.D.; Tian, L. Glucose ester enabled acylation in plant specialized metabolism. Phytochem. Rev. 2016, 15, 1057–1074. [Google Scholar] [CrossRef]
- Seeram, N.P.; Henning, S.M.; Zhang, Y.; Suchard, M.; Li, Z.; Heber, D. Pomegranate juice ellagitannin metabolites are present in human plasma and some persist in urine for up to 48 hours. J. Nutr. 2006, 136, 2481–2485. [Google Scholar] [PubMed]
- Cerdá, B.; Espín, J.C.; Parra, S.; Martínez, P.; Tomás-Barberán, F.A. The potent in vitro antioxidant ellagitannins from pomegranate juice are metabolised into bioavailable but poor antioxidant hydroxy-6H-dibenzopyran-6-one derivatives by the colonic microflora of healthy humans. Eur. J. Nutr. 2004, 43, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Mertens-Talcott, S.U.; Jilma-Stohlawetz, P.; Rios, J.; Hingorani, L.; Derendorf, H. Absorption, metabolism, and antioxidant effects of pomegranate (Punica granatum L.) polyphenols after ingestion of a standardized extract in healthy human volunteers. J. Agric. Food Chem. 2006, 54, 8956–8961. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; García-Villalba, R.; González-Sarrías, A.; Selma, M.V.; Espín, J.C. Ellagic acid metabolism by human gut microbiota: Consistent observation of three urolithin phenotypes in intervention trials, independent of food source, age, and health status. J. Agric. Food Chem. 2014, 62, 6535–6538. [Google Scholar] [CrossRef] [PubMed]
- Lephart, E.D. Modulation of aromatase by phytoestrogens. Enzyme Res. 2015, 2015, 594656. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.S.; Zhang, Y.; Seeram, N.P.; Heber, D.; Chen, S. Pomegranate ellagitannin-derived compounds exhibit antiproliferative and antiaromatase activity in breast cancer cells. Cancer Prev. Res. 2010, 3, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Chen, J.-H.; Aguilera-Barrantes, I.; Shiau, C.-W.; Sheng, X.; Wang, L.-S.; Stoner, G.D.; Huang, Y.-W. Urolithin A suppresses the proliferation of endometrial cancer cells by mediating estrogen receptor-α-dependent gene expression. Mol. Nutr. Food Res. 2016, 60, 2387–2395. [Google Scholar] [CrossRef] [PubMed]
- Stolarczyk, M.; Piwowarski, J.P.; Granica, S.; Stefańska, J.; Naruszewicz, M.; Kiss, A.K. Extracts from Epilobium sp. herbs, their components and gut microbiota metabolites of epilobium ellagitannins, urolithins, inhibit hormone-dependent prostate cancer cells-(LNCaP) proliferation and PSA secretion. Phytother. Res. 2013, 27, 1842–1848. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Gonzalez, C.; Ciudad, C.J.; Noe, V.; Izquierdo-Pulido, M. Walnut polyphenol metabolites, urolithins A and B, inhibit the expression of the prostate-specific antigen and the androgen receptor in prostate cancer cells. Food Funct. 2014, 5, 2922–2930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-González, C.; Ciudad, C.J.; Izquierdo-Pulido, M.; Noé, V. Urolithin A causes p21 up-regulation in prostate cancer cells. Eur. J. Nutr. 2016, 55, 1099–1112. [Google Scholar] [CrossRef] [PubMed]
- Go, R.-E.; Hwang, K.-A.; Choi, K.-C. Cytochrome P450 1 family and cancers. J. Steroid Biochem. Mol. Biol. 2015, 147, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Kasimsetty, S.G.; Bialonska, D.; Reddy, M.K.; Thornton, C.; Willett, K.L.; Ferreira, D. Effects of pomegranate chemical constituents/intestinal microbial metabolites on CYP1B1 in 22Rv1 prostate cancer cells. J. Agric. Food Chem. 2009, 57, 10636–10644. [Google Scholar] [CrossRef] [PubMed]
- Vicinanza, R.; Zhang, Y.; Henning, S.M.; Heber, D. Pomegranate juice metabolites, ellagic acid and urolithin A, synergistically inhibit androgen-independent prostate cancer cell growth via distinct effects on cell cycle control and apoptosis. Evid. Based Complement. Altern. Med. 2013, 2013, 247504. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Wang, J.; Zheng, G.; Qiu, Z. Methylated urolithin A, the modified ellagitannin-derived metabolite, suppresses cell viability of DU145 human prostate cancer cells via targeting miR-21. Food Chem. Toxicol. 2016, 97, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Giorgio, C.; Mena, P.; DelRio, D.; Brighenti, F.; Barocelli, E.; Hassan-Mohamed, I.; Callegari, D.; Lodola, A.; Tognolini, M. The ellagitannin colonic metabolite urolithin D selectively inhibits EphA2 phosphorylation in prostate cancer cells. Mol. Nutr. Food Res. 2015, 59, 2155–2167. [Google Scholar] [CrossRef] [PubMed]
- Krausova, M.; Korinek, V. Wnt signaling in adult intestinal stem cells and cancer. Cell. Signal 2014, 26, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Li, L.; Celver, J.; Killian, C.; Kovoor, A.; Seeram, N.P. Effects of fruit ellagitannin extracts, ellagic acid, and their colonic metabolite, urolithin A, on Wnt signaling. J. Agric. Food Chem. 2010, 58, 3965–3969. [Google Scholar] [CrossRef] [PubMed]
- Kasimsetty, S.G.; Bialonska, D.; Reddy, M.K.; Ma, G.; Khan, S.I.; Ferreira, D. Colon cancer chemopreventive activities of pomegranate ellagitannins and urolithins. J. Agric. Food Chem. 2010, 58, 2180–2187. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Jung, H.; Lee, H.; Yi, H.C.; Kwak, H.-K.; Hwang, K.T. Chemopreventive activity of ellagitannins and their derivatives from black raspberry seeds on HT-29 colon cancer cells. Food Funct. 2015, 6, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Nasimudeen, R.J.; Shams, T. Cardiovascular disease management through restrained inflammatory responses. Curr. Pharm. Des. 2016, 22, 940–946. [Google Scholar]
- Piwowarski, J.P.; Granica, S.; Kiss, A.K. Influence of gut microbiota-derived ellagitannins’ metabolites urolithins on pro-inflammatory activities of human neutrophils. Planta Med. 2014, 80, 887–895. [Google Scholar] [PubMed]
- Mele, L.; Mena, P.; Piemontese, A.; Marino, V.; López-Gutiérrez, N.; Bernini, F.; Brighenti, F.; Zanotti, I.; Del Rio, D. Antiatherogenic effects of ellagic acid and urolithins in vitro. Arch. Biochem. Biophys. 2016, 599, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Bastida, J.A.; González-Sarrías, A.; Larrosa, M.; Tomás-Barberán, F.; Espín, J.C.; García-Conesa, M.-T. Ellagitannin metabolites, urolithin A glucuronide and its aglycone urolithin A, ameliorate TNF-α-induced inflammation and associated molecular markers in human aortic endothelial cells. Mol. Nutr. Food Res. 2012, 56, 784–796. [Google Scholar] [CrossRef] [PubMed]
- Naseem, K.M. The role of nitric oxide in cardiovascular diseases. Mol. Asp. Med. 2005, 26, 33–65. [Google Scholar] [CrossRef] [PubMed]
- Spigoni, V.; Mena, P.; Cito, M.; Fantuzzi, F.; Bonadonna, R.; Brighenti, F.; Dei Cas, A.; Del Rio, D. Effects on nitric oxide production of urolithins, gut-derived ellagitannin metabolites, in human aortic endothelial cells. Molecules 2016, 21, 1009. [Google Scholar] [CrossRef] [PubMed]
- Romo-Vaquero, M.; García-Villalba, R.; González-Sarrías, A.; Beltrán, D.; Tomás-Barberán, F.A.; Espín, J.C.; Selma, M.V. Interindividual variability in the human metabolism of ellagic acid: Contribution of Gordonibacter to urolithin production. J. Funct. Food 2015, 17, 785–791. [Google Scholar] [CrossRef]
- Selma, M.V.; Romo-Vaquero, M.; Garcia-Villalba, R.; Gonzalez-Sarrias, A.; Tomas-Barberan, F.A.; Espin, J.C. The human gut microbial ecology associated with overweight and obesity determines ellagic acid metabolism. Food Funct. 2016, 7, 1769–1774. [Google Scholar] [CrossRef] [PubMed]
- González-Sarrías, A.; García-Villalba, R.; Romo-Vaquero, M.; Alasalvar, C.; Örem, A.; Zafrilla, P.; Tomás-Barberán, F.A.; Selma, M.V.; Espín, J.C. Clustering according to urolithin metabotype explains the interindividual variability in the improvement of cardiovascular risk biomarkers in overweight-obese individuals consuming pomegranate: A randomised clinical trial. Mol. Nutr. Food Res. 2017, 61, 1600830. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Ma, H.; Liu, W.; Niesen, D.B.; Shah, N.; Crews, R.; Rose, K.N.; Vattem, D.A.; Seeram, N.P. Pomegranate’s neuroprotective effects against Alzheimer’s disease are mediated by urolithins, its ellagitannin-gut microbial derived metabolites. ACS Chem. Neurosci. 2016, 7, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ma, H.; Frost, L.; Yuan, T.; Dain, J.A.; Seeram, N.P. Pomegranate phenolics inhibit formation of advanced glycation endproducts by scavenging reactive carbonyl species. Food Funct. 2014, 5, 2996–3004. [Google Scholar] [CrossRef] [PubMed]
- Ryu, D.; Mouchiroud, L.; Andreux, P.A.; Katsyuba, E.; Moullan, N.; Nicolet-dit-Felix, A.A.; Williams, E.G.; Jha, P.; Lo Sasso, G.; Huzard, D.; et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat. Med. 2016, 22, 879–888. [Google Scholar] [CrossRef] [PubMed]
| Classes | Phytochemicals |
|---|---|
| Ellagitannins, gallotannins and derivatives | Brevifolin, Brevifolin carboxylic acid, Brevifolin carboxylic acid 10-monopotassium sulphate, Castalagin, Casuariin, Casuarinin, Corilagin, Isocorilagin, Hippomanin A, Gemin D, Diellagic acid rhamnosyl(1→4) glucopyranoside, 1,2-Di-O-galloyl-4,6-O-(S)-hexahydroxydiphenoyl β-d-glucopyranoside, Ellagic acid, 3,3′-Di-O-methylellagic acid, 3,3′,4′-Tri-O-methylellagic acid, 3-O-Methylellagic acid, 4,4′-Di-O-methylellagic acid, 3′-O-Methyl-3,4-methylenedioxy-ellagic acid, Eschweilenol C (Ellagic acid 4-O-α-l-rhamnopyranoside), Ethyl brevifolincarboxylate, Eucalbanin B, Eucarpanin T1, Pomegraniin A, Pomegraniin B, Gallagic acid, Gallic acid 3-O-β-d-(6′-O-galloyl)-glucopyranoside, 6-O-Galloyl-2,3-(S)-hexahydroxydiphenoyl-d-glucose, 5-Galloylpunicacortein D, 2-O-Galloylpunicalin (2-O-Galloyl-4,6-(S,S)-gallagyl-d-glucose), Granatin A, Granatin B, 2,3-(S)-Hexahydroxydiphenoyl-d-glucose, Lagerstannin B, Lagerstannin C, 3-O-Methylellagic acid 4-O-α-l-rhamnopyranoside, 3,4′-O-Dimethylellagic acid 4-O-α-l-rhamnopyranoside, Oenothein B, Pedunculagin I, Pedunculagin II, 1,2,3,4,6-Penta-O-galloyl-β-d-glucose, 3,4,8,9,10-Pentahydroxydibenzo [b,d] pyran-6-one (Urolithin M-5), Phyllanthusiin E, Pomegranatate, Punicacortein A, Punicacortein B, Punicacortein C, Punicacortein D, Punicafolin, Punicalagin A, Punicalagin B, Punicalin, Punicatannin A, Punicatannin B, Punigluconin, Strictinin [1-O-Galloyl-4,6-(S)-hexahydroxydiphenoyl-d-glucose], Tellimagrandin I, Tercatain [1,4-Di-O-galloyl-3,6-(R)-hexahydroxydiphenoyl-β-glucopyranose], Terminalin (Gallagyl dilactone), 1,2,4,6-Tetra-O-galloyl-β-d-glucose, 1,2,3-Tri-O-galloyl-β-glucopyranose, 1,2,4-Tri-O-galloyl-β-glucopyranose, 1,2,6-Tri-O-galloyl-β-glucopyranose, 1,3,4-Tri-O-galloyl-β-glucopyranose, 1,4,6-Tri-O-galloyl-β-glucopyranose, 3,4,6-Tri-O-galloyl-β-glucopyranose, Valoneic acid dilactone |
| Flavonoids | Hovetrichoside C, Phloretin, Phlorizin, Eriodictyol-7-O-α-l-arabinofuranosyl (1-6)-β-d-glucoside, Granatumflavanyl xyloside, Naringin (Naringenin-7-O-rhamnoglucoside), Naringenin-4′methyl ether 7-O-α-l-arabinofuranosyl(1-6)-β-d-glucoside, Pinocembrin, Punicaflavanol, Apigenin, Apigenin 4′-O-β-glucopyranoside, Luteolin, Luteolin 3′-O-β-glucopyranoside, Luteolin 4′-O-β-glucopyranoside, Cynaroside (Luteolin 7-O-glycoside), Luteolin 3′-O-β-xylopyranoside, Tricetin, Daidzein, Genistein, Amurensin (Noricaritin 7-β-d-glucopyranoside), Kaempferol, Astragalin (Kaempferol 3-O-glucoside), Kaempferol-3-O-rhamnoglucoside, Myricetin, Phellatin, Quercetin, Hirsutrin (Quercetin-3-O-glucoside), Quercimeritrin (Quercetin-7-O-glucoside), Quercetin 3-O-rhamnoside, Rutin (Quercetin-3-O-rutinoside), Quercetin-3,4′-dimethyl ether 7-O-α-l-arabinofuranosyl(1-6)-β-d-glucoside, Cyanidin, Chrysanthemin (Cyanidin-3-O-glucoside), Cyanin (Cyanidin-3,5-di-O-glucoside), Antirrhinin (Cyanidin-3-O-rutinoside), Catechin-cyanidin-3-hexoside, Delphinidin, Myrtillin (Delphinidin-3-O-glucoside), Delphinidin-3,5-di-O-glucoside, Pelargonidin, Callistephin (Pelargonidin-3-O-glucoside), Pelargonin (Pelargonidin-3,5-di-O-glucoside), Catechin, Epicatechin, Epicatechin gallate, Epigallocatechin-3-O-gallate, Gallocatechin-(4→8)-catechin, Gallocatechin-(4→8)-gallocatechin, Catechin-(4→8)-gallocatechin, Procyanidin A2, Procyanidin B1, Procyanidin B2, Procyanidin B3 |
| Lignans | Conidendrin, Isohydroxymatairesinol, Isolariciresinol, Matairesinol, Medioresinol, Phylligenin, Pinoresinol, Secoisolariciresinol, Syringaresinol, Pomegralignan, Punicatannin C |
| Triterpenoids and phytosterols | Asiatic acid, Betulinic acid (Betulic acid), Friedooleanan-3-one (Friedelin), Maslinic acid, Oleanolic acid, Punicanolic acid, Ursolic acid, Campesterol, Cholesterol, Daucosterol, β-Sitosterol, β-Sitosterol laurate, β-Sitosterol myristate, Stigmasterol |
| Alkaloids and indolamines | N-(2′,5′-Dihydroxyphenyl)pyridinium chloride, Hygrine, Norhygrine, Pelletierine, N-Methylpelletierine, Norpseudopelletierine, Pseudopelletierine, 2-(2′-Hydroxypropyl)-∆1piperideine, 2-(2′-Propenyl)-∆1piperideine, Punigratane (2,5-Diheptyl-N-methylpyrrolidine), Sedridine, Melatonin, Serotonin, Tryptamine |
| Fatty acids and lipids | Caproic acid (Hexanoic acid), Caprylic acid (Octanoic acid), Capric acid (Decanoic acid), Lauric acid (Dodecanoic acid), Myristic acid (Tetradecanoic acid), Myristoleic acid (9-cis-Tetradecanoic acid), Palmitic acid (Hexadecanoic acid), Palmitoleic acid (Hexadec-9-enoic acid), Punicic acid (9Z, 11E, 13Z-octadecatrienoic acid), Linoleic acid (cis, cis-9,12-Octadecadienoic acid), α-Linolenic acid (All-cis-9,12,15-octadecatrienoic acid), γ-Linolenic acid (All-cis-6,9,12-octadecatrienoic acid), Oleic acid (9Z-octadecenoic acid), Stearic acid (Octadecanoic acid), α-Eleostearic acid (9Z, 11E, 13E-octadecatrienoic acid), β-Eleostearic acid (9E, 11E, 13E-octadecatrienoic acid), Catalpic acid (9E, 11E, 13Z-octadecatrienoic acid), Arachidic acid (Eicosanoic acid), Gadoleic acid (9Z-icosenoic acid), Behenic acid (Docosanoic acid), Nervonic acid (cis-15-Tetracosenoic acid), 1-O-9E,11Z,13E-Octadecatrienoyl glycerol, 1-O-Isopentyl-3-O-octadec-2-enoyl glycerol, Tri-O-punicylglycerol, Di-O-punicyl-O-octadeca-8Z, 11Z, 13E-trienylglycerol, N-palmitoyl cerebroside |
| Organic acids and phenolic acids | Ascorbic acid, Citric acid, Fumaric acid, l-Malic acid, Oxalic acid, Quinic acid, Succinic acid, Tartaric acid, Caffeic acid, Chlorogenic acid, Cinnamic acid, o-Coumaric acid, p-Coumaric acid, cis-p-Coumaric acid, Coutaric acid, 7,8-Dihydroxy-3-carboxymethylcoumarin-5-carboxylic acid, Ferulic acid, Gallic acid, Methyl gallate, Neochlorogenic acid (5-O-Caffeoylquinic acid), Protocatechuic acid, Vanillic acid, Coniferyl 9-O-[β-d-apiofuranosyl(1→6)]-O-β-d-glucopyranoside, Sinapyl 9-O-[β-d-apiofuranosyl(1→6)]-O-β-d-glucopyranoside |
| Other compounds | Catechol, Coumestrol, Icariside D1, Phenylethylrutinoside, Syringaldehyde |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.; Tian, L. Diverse Phytochemicals and Bioactivities in the Ancient Fruit and Modern Functional Food Pomegranate (Punica granatum). Molecules 2017, 22, 1606. https://doi.org/10.3390/molecules22101606
Wu S, Tian L. Diverse Phytochemicals and Bioactivities in the Ancient Fruit and Modern Functional Food Pomegranate (Punica granatum). Molecules. 2017; 22(10):1606. https://doi.org/10.3390/molecules22101606
Chicago/Turabian StyleWu, Sheng, and Li Tian. 2017. "Diverse Phytochemicals and Bioactivities in the Ancient Fruit and Modern Functional Food Pomegranate (Punica granatum)" Molecules 22, no. 10: 1606. https://doi.org/10.3390/molecules22101606





