Cytotoxic and Hypoglycemic Activity of Triterpenoid Saponins from Camellia oleifera Abel. Seed Pomace
Abstract
:1. Introduction
2. Results
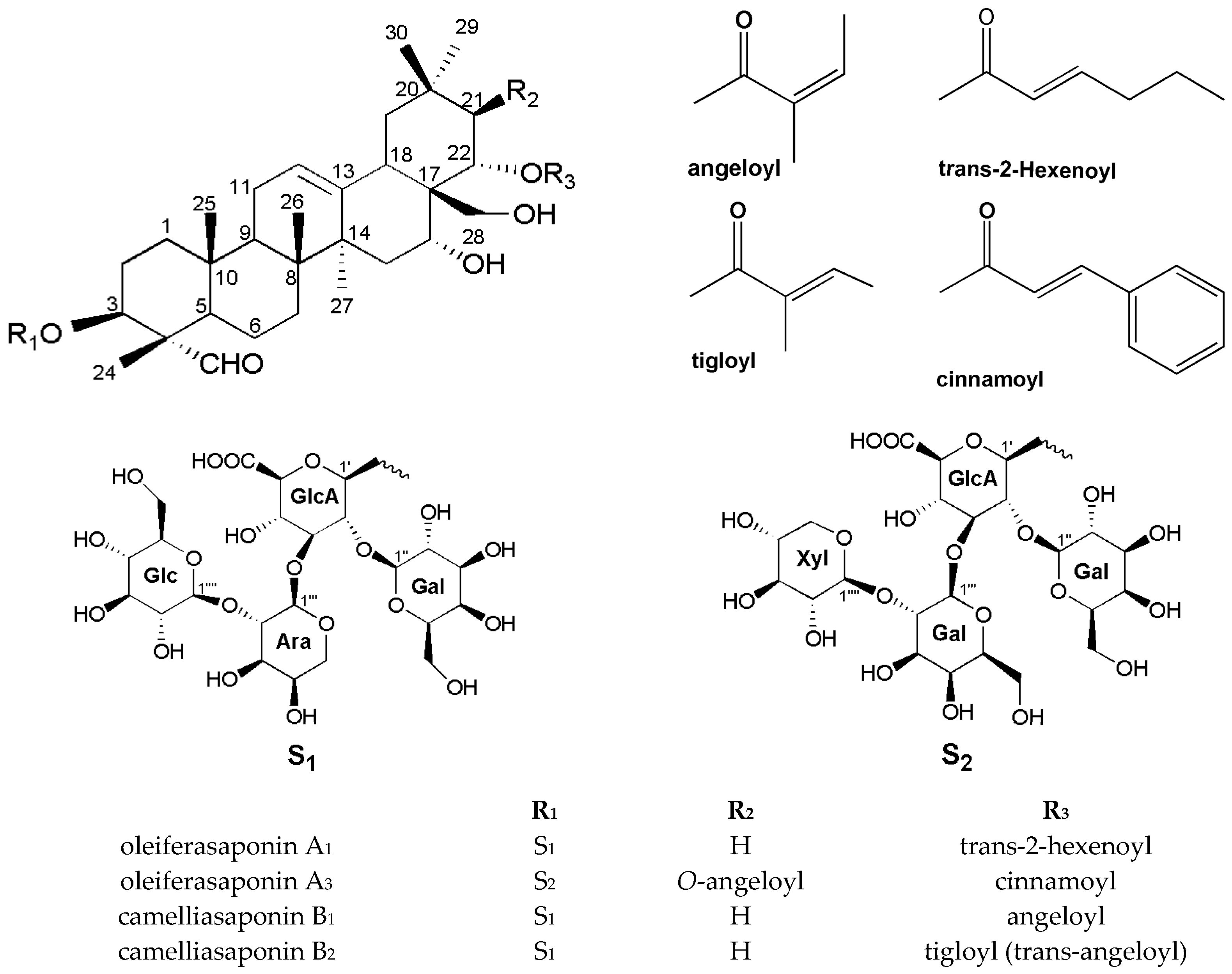
2.1. The Characterization of the Oleiferasaponin A3
2.2. Anti-Proliferative Activity
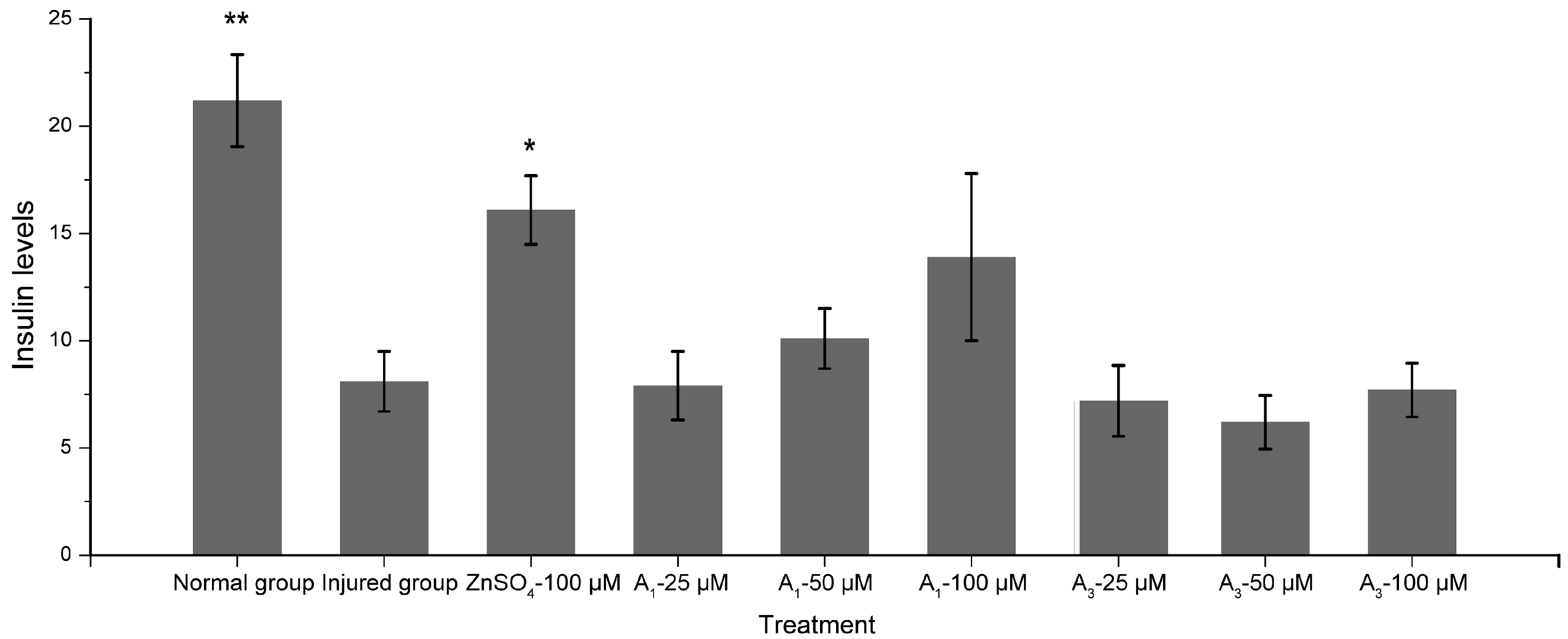
2.3. Hypoglycemic Activity
3. Materials and Methods
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Acid Hydrolysis and GC-MS Analysis
3.5. Cytotoxic Activity Assay
3.5.1. Cell Culture
3.5.2. Cell Viability Assay
3.6. Hypoglycemic Activity Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jian, H.L.; Liao, X.X.; Zhu, L.W.; Zhang, W.M.; Jiang, J.X. Synergism and foaming properties in binary mixtures of a biosurfactant derived from camellia oleifera abel and synthetic surfactants. J. Coll. Interface Sci. 2011, 359, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.F.; Yang, C.H.; Chang, M.S.; Ciou, Y.P.; Huang, Y.C. Foam properties and detergent abilities of the saponins from camellia oleifera. Int. J. Mol. Sci. 2010, 11, 4417–4425. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Z.; Li, M.A.; Liu, Z.C.; Peng, S.F.; Chen, X.L.; Chen, L.S.; Wang, X.N.; Wang, R. Molluscicidal effect of camellia oleifera saponin. J. Central South. Univ. For. Technol. 2011, 31, 147–150. [Google Scholar]
- Wang, Y.; Liu, Y.F.; Zhang, C.L.; Liang, D.; Luo, H.; Hao, Z.Y.; Chen, R.Y.; Yu, D.Q. Four new triterpenoid saponins isolated from schefflera kwangsiensis and their inhibitory activities against fbpase1. Phytochem. Lett. 2016, 15, 204–209. [Google Scholar] [CrossRef]
- Liao, Z.; Yin, D.; Wang, W.; Zeng, G.; Liu, D.; Chen, H.; Huang, Q.; He, M. Cardioprotective effect of sasanquasaponin preconditioning via bradykinin-no pathway in isolated rat heart. Phytother. Res. 2009, 23, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, C.Z.; Ye, J.Z.; Chen, H.X. New triterpene saponins from the seed cake of camellia oleifera and their cytotoxic activity. Phytochem. Lett. 2014, 8, 46–51. [Google Scholar] [CrossRef]
- Kuo, P.C.; Lin, T.C.; Yang, C.W.; Lin, C.L.; Chen, G.F.; Huang, J.W. Bioactive saponin from tea seed pomace with inhibitory effects against rhizoctonia solani. J. Agric. Food Chem. 2010, 58, 8618–8622. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Chen, J.; Xu, H. Sasanquasaponin from camellia oleifera abel. Induces cell cycle arrest and apoptosis in human breast cancer mcf-7 cells. Fitoterapia 2013, 84, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Wu, H.Y.; Bingchung, L.; Chang, C.M.J.; Tingting, J.; Wu, L.C. Identification and evaluation of antioxidants defatted camellia oleifera seeds by isopropanol salting-out pretreatment. Food Chem. 2010, 121, 1246–1254. [Google Scholar] [CrossRef]
- Zhang, X.F.; Han, Y.Y.; Bao, G.H.; Ling, T.J.; Zhang, L.; Gao, L.P.; Xia, T. A new saponin from tea seed pomace (camellia oleifera abel) and its protective effect on pc12 cells. Molecules 2012, 17, 11721–11728. [Google Scholar] [CrossRef] [PubMed]
- Zong, J.; Wang, R.; Bao, G.; Ling, T.; Zhang, L.; Zhang, X.; Hou, R. Novel triterpenoid saponins from residual seed cake of camellia oleifera abel. Show anti-proliferative activity against tumor cells. Fitoterapia 2015, 104, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Zong, J.F.; Peng, Y.R.; Bao, G.H.; Hou, R.Y.; Wan, X.C. Two new oleanane-type saponins with anti-proliferative activity from camellia oleifera abel. Seed cake. Molecules 2015, 21, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Zong, J.; Wang, D.; Jiao, W.; Zhang, L.; Bao, G.; Ho, C.T.; Hou, R.; Wan, X. Oleiferasaponin C6 from the seeds of camellia oleifera abel.: A novel compound inhibits proliferation through inducing cell-cycle arrest and apoptosis on human cancer cell lines in vitro. RSC Adv. 2016, 6, 91386–91393. [Google Scholar] [CrossRef]
- Huang, Q.; He, M.; Chen, H.; Shao, L.; Liu, D.; Luo, Y.; Dai, Y. Protective effects of sasanquasaponin on injury of endothelial cells induced by anoxia and reoxygenation in vitro. Basic Clin. Pharmacolo. 2007, 101, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.L.; Nie, S.P.; Huang, D.F.; Li, C.; Xie, M.Y. Extraction of saponin from camellia oleifera cake and evaluation of its antioxidant activity. Int. J. Food Sci. Technol. 2012, 47, 1676–1687. [Google Scholar] [CrossRef]
- Hu, J.L.; Nie, S.P.; Huang, D.F.; Chang, L.; Xie, M.Y.; Yin, W. Antimicrobial activity of saponin-rich fraction from camellia oleifera cake and its effect on cell viability of mouse macrophage raw 264.7. J. Sci. Food Agric. 2012, 92, 2443–2449. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.F.; Yang, S.L.; Han, Y.Y.; Zhao, L.; Lu, G.L.; Xia, T.; Gao, L.P. Qualitative and quantitative analysis of triterpene saponins from tea seed pomace (camellia oleifera abel) and their activities against bacteria and fungi. Molecules 2014, 19, 7568–7580. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Xia, T.S.; Wang, Y.F.; Zhou, W.; Liang, X.Q.; Xue, J.Q.; Shi, L.; Wang, Y.; Ding, Q. The apoptotic effect of d rhamnose β-hederin, a novel oleanane-type triterpenoid saponin on breast cancer cells. PLoS ONE 2014, 9, e90848. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Teruya, T.; Suenaga, K.; Matsui, Y.; Masuda, H.; Kigoshi, H. Isotheasaponins B1–B3 from camellia sinensis var. Sinensis tea leaves. Phytochemistry 2006, 67, 1385–1389. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Harada, E.; Murakami, T.; Matsuda, H.; Yamahara, J.; Murakami, N. Camelliasaponins B1, B2, C1 and C2, new type inhibitors of ethanol absorption in rats from the seeds of camellia Japonica L. Chem. Pharm. Bull. 1994, 42, 742–744. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.K.; Choi, S.U.; Lee, K.R. Triterpene saponins from pleurospermum kamtschaticum and their biological activity. Chem. Pharm. Bull. 2012, 60, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Ownby, S.; Zhang, Z.; Yuan, W.; Li, S. Cytotoxicity and inhibition of DNA topoisomerase i of polyhydroxylated triterpenoids and triterpenoid glycosides. Bioorg. Med. Chem. Lett. 2010, 20, 2790–2796. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Wang, T.; Sugimoto, S.; Nakamura, S.; Nagatomo, A.; Matsuda, H.; Harima, S. Functional saponins in tea flower (flower buds of camellia sinensis): Gastroprotective and hypoglycemic effects of floratheasaponins and qualitative and quantitative analysis using HPLC. Yakugaku Zasshi-J. Pharm. Soc. Japan 2008, 128, 141–151. [Google Scholar] [CrossRef]
Sample Availability: Not available. |


| Position | δC | δH | Position | δC | δH |
|---|---|---|---|---|---|
| 1 | 38 | 1.15 m, 1.73 m | 21-O-Ang | ||
| 2 | 24.3 | 1.83 m, 2.09 m | Ang-1 | 168.1 | |
| 3 | 84.7 | 3.90 m | Ang-2 | 128 | |
| 4 | 55 | Ang-3 | 137.2 | 6.02, q (7.2) | |
| 5 | 47.4 | 1.38 m | Ang-4 | 14.6 | 1.85, d (7.2) |
| 6 | 19.8 | 0.95 m, 1.55 m | Ang-5 | 19.5 | 1.82 |
| 7 | 31.8 | 1.29 m, 1.68 m | 22-O-Cin | ||
| 8 | 39.9 | Cin-1 | 167.6 | ||
| 9 | 46.6 | 1.82 m | Cin-2 | 117.4 | 6.49 d (16.2) |
| 10 | 35.6 | Cin-3 | 145.3 | 7.72 d (16.2) | |
| 11 | 23.2 | 1.95 m, 2.00m | Cin-4 | 134.4 | |
| 12 | 123.6 | 5.44 m | Cin-5, 9 | 127.8 | 7.60 m |
| 13 | 141.6 | Cin-6, 8 | 128.6 | 7.42 overlap | |
| 14 | 41.1 | Cin-7 | 130.1 | 7.42 overlap | |
| 15 | 33.4 | 1.39 m, 1.72 m | GlcA-1′ | 103.4 | 4.38 (7.8) |
| 16 | 68.1 | 4.06 brs | GlcA-2’ | 77 | 3.79 overlap |
| 17 | 47.2 | GlcA-3’ | 81.6 | 3.90 overlap | |
| 18 | 39.4 | 2.71 m | GlcA-4’ | 69.6 | 3.56 overlap |
| 19 | 46.4 | 1.25 m, 2.68 m | GlcA-5’ | 75.6 | 3.64 overlap |
| 20 | 35.4 | GlcA-6’ | 174.9 | ||
| 21 | 78.4 | 6.04 d (10.2) | Gal-1′′ | 101.2 | 5.05 d (7.8) |
| 22 | 73.8 | 5.62 d (10.2) | Gal-2′′ | 73.6 | 3.51 m |
| 23 | 209.3 | 9.50 s | Gal-3′′ | 75.1 | 3.82overlap |
| 24 | 9.4 | 1.19 s | Gal-4′′ | 69.6 | 3.84 overlap |
| 25 | 15 | 1.05 s | Gal-5′′ | 76.4 | 3.33 m |
| 26 | 15.9 | 0.97 s | Gal-6′′ | 63 | 3.02 d (10.8), 3.30 m |
| 27 | 26.3 | 1.53 s | Gal-1′′′ | 100.2 | 5.07 d (7.8) |
| 28 | 63.1 | 3.02 d (10.8), 3.33 d (10.8) | Gal-2′′′ | 82.4 | 3.68 overlap |
| 29 | 28.2 | 0.93 s | Gal-3′′′ | 75.5 | 3.57 overlap |
| 30 | 18.8 | 1.14 s | Gal-4′′′ | 69.1 | 3.83 m |
| Gal-5′′′ | 76.4 | 3.65 overlap | |||
| Gal-6′′′ | 61.2 | 3.71 overlap, 3.81overlap | |||
| Xyl-1′′′′ | 106.2 | 4.53 d (7.8) | |||
| Xyl-2′′′′ | 74.9 | 3.31 m | |||
| Xyl-3′′′′ | 76.9 | 3.90 m | |||
| Xyl-4′′′′ | 70.3 | 3.55 m | |||
| Xyl-5′′′′ | 65.9 | 3.99 m, 3.21 m |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di, T.-M.; Yang, S.-L.; Du, F.-Y.; Zhao, L.; Xia, T.; Zhang, X.-F. Cytotoxic and Hypoglycemic Activity of Triterpenoid Saponins from Camellia oleifera Abel. Seed Pomace. Molecules 2017, 22, 1562. https://doi.org/10.3390/molecules22101562
Di T-M, Yang S-L, Du F-Y, Zhao L, Xia T, Zhang X-F. Cytotoxic and Hypoglycemic Activity of Triterpenoid Saponins from Camellia oleifera Abel. Seed Pomace. Molecules. 2017; 22(10):1562. https://doi.org/10.3390/molecules22101562
Chicago/Turabian StyleDi, Tai-Mei, Shao-Lan Yang, Feng-Yu Du, Lei Zhao, Tao Xia, and Xin-Fu Zhang. 2017. "Cytotoxic and Hypoglycemic Activity of Triterpenoid Saponins from Camellia oleifera Abel. Seed Pomace" Molecules 22, no. 10: 1562. https://doi.org/10.3390/molecules22101562





