Essential Oils as an Alternative to Pyrethroids’ Resistance against Anopheles Species Complex Giles (Diptera: Culicidae)
Abstract
:1. Introduction
2. Chemical Composition
3. Toxicity to Eggs and Immature Stages in Anopheles sp.
4. Essential Oils Toxicity in Anopheles sp. Adults
5. Repellency Effects of EOs against Anopheles Mosquito Adults
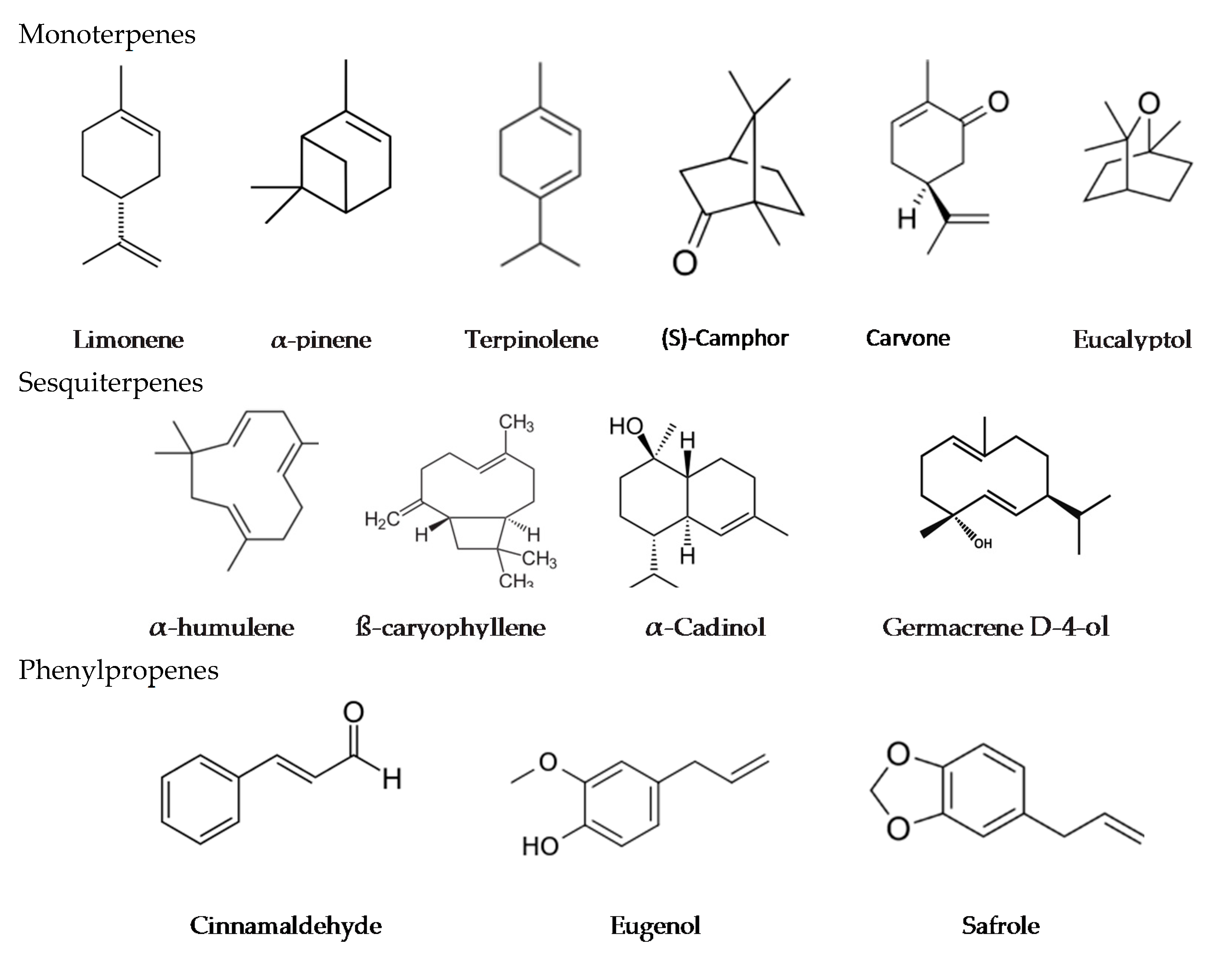
6. Biologically Active Components of Essential Oils
7. Synergistic and Antagonistic Phenomena
8. Mechanisms of Action of Essential Oil Components
9. Microencapsulation and Nanoemulsion Technologies
10. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| EOs | Essential Oils |
| IRS | Indoor residual spraying |
| LLINs | Long-lasting insecticide treated nets |
| kdr | knock down resistance |
| LD | Lethal dose |
| LC | Lethal concentration |
| US EPA | United States Environmental Protection Agency |
| WHO | World Health Organization |
| DEET | N,N-diethyl-3-methylbenzamide |
| AChE | Acetylcholinesterase |
| ACh | Acetylcholine |
| OA | Octopamine |
| GABA | Gamma aminobutyric acid |
References
- WHO. World Malaria Report 2015; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- WHO. Global Plan for Insect Management; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Kelly-Hope, L.; Ranson, H.; Hemingway, J. Lessons from the past: Managing insecticide resistance in malaria control and eradication programmes. Lancet Infect. Dis. 2008, 8, 387–389. [Google Scholar] [CrossRef]
- Namountougou, M.; Diabaté, A.; Etang, J.; Bass, C.; Sawadogo, S.P.; Gnankinié, O.; Baldet, T.; Martin, T.; Chandre, F.; Simard, F.; et al. First report of the L1014S kdr mutation in wild populations of Anopheles gambiae M and S molecular forms in Burkina Faso (West Africa). Acta Trop. 2013, 125, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Namountougou, M.; Frédéric, S.; Baldet, T.; Diabate, A.; Ouédraogo, J.-B.; Martin, T.; Dabire, R.K. Multiple Insecticide Resistance in Anopheles gambiae s.l. Populations from Burkina Faso, West Africa. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gnankiné, O.; Bassolé, I.H.N.; Chandre, F.; Glitho, I.; Akogbeto, M.; Dabiré, R.K.; Martin, T. Insecticide resistance in Bemisia tabaci Gennadius (Homoptera: Aleyrodidae) and Anopheles gambiae Giles (Diptera: Culicidae) could compromise the sustainability of malaria vector control strategies in West Africa. Acta Trop. 2013, 128, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Dabiré, R.; Namountougou, M.; Sawadogo, S.; Yaro, L.; Toé, H.; Ouari, A.; Gouagna, L.-C.; Simard, F.; Chandre, F.; Baldet, T.; et al. Population dynamics of Anopheles gambiae s.l. in Bobo-Dioulasso city: bionomics, infection rate and susceptibility to insecticides. Parasites Vectors 2012, 5, 127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, A.P.B.; Santos, J.M.M.; Martins, A.J. Mutations in the voltage-gated sodium channel gene of anophelines and their association with resistance to pyrethroids—A review. Parasites Vectors 2014, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chandre, F.; Darrier, F.; Manga, L.; Akogbeto, M.; Faye, O.; Mouchet, J.; Guillet, P. Status of pyrethroid resistance in Anopheles gambiae sensu lato. Bull. World Health Organ. 1999, 77, 230–234. [Google Scholar] [PubMed]
- Diabaté, A.; Baldet, T.; Chandre, F.; Guiguemde, R.; Brengues, C.; Guillet, P.; Hemingway, J.; Hougard, J. First report of the kdr mutation in Anopheles gambiae M form from Burkina Faso, West Africa. Parassitologia 2002, 44, 157–158. [Google Scholar] [PubMed]
- Awolola, T.S.; Oyewole, I.O.; Amajoh, C.N.; Idowu, E.T.; Ajayi, M.B.; Oduola, A.; Manafa, O.U.; Ibrahim, K.; Koekemoer, L.L.; Coetzee, M. Distribution of the molecular forms of Anopheles gambiae and pyrethroid knock down resistance gene in Nigeria. Acta Trop. 2005, 95, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Fanello, C.; Santolamazza, F.; Della-Torre, A. Simultaneous identification of species and molecular forms of the Anopheles gambiae complex by PCR-RFLP. Med. Vet. Entomol. 2002, 16, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Weill, M.; Chandre, F.; Brengues, C.; Manguin, S.; Akogbeto, M.; Pasteur, N. The kdr mutation occurs in the Mopti form of Anopheles gambiae s.s. through introgression. Insect Mol. Bol. 2000, 9, 451–455. [Google Scholar] [CrossRef]
- Della-Torre, A.; Fanello, C.; Akogbeto, M.; Favia, G.; Petrarca, V.; Coluzzi, M. Molecular evidence of incipient speciation within Anopheles gambiae s.s. in West Africa. Insect Mol. Bol. 2001, 10, 9–18. [Google Scholar] [CrossRef]
- N’Guessan, R.N.; Corbel, V.; Akogbéto, M.; Rowland, M. Reduced Efficacy of Insecticide- treated Nets and Indoor Residual Spraying for Malaria Control in Pyrethroid Resistance Area, Benin. Emerg. Infect. Dis. 2007, 13, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Diabaté, A.; Baldet, T.; Chandre, F.; Akogbeto, M.; Guiguemde, T.; Darriet, F.; Brengues, C.; Small, G.; Hougard, J. The role of agricultural use of insecticides in resistance to pyrethroids in Anopheles gambiae s.l. in Burkina Faso. Am. J. Trop. Med. Hyg. 2002, 67, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Verhaeghen, K.; Van Bortel, W.; Roelants, P.; Backeljau, T.; Coosemans, M. Detection of the East and West African kdr mutation in Anopheles gambiae and Anopheles arabiensis from Uganda using a new assay based on FRET/Melt Curve analysis. Malar. J. 2006, 9, 1–9. [Google Scholar] [CrossRef]
- Etang, J.; Fondjo, E.; Chandre, F.; Morlais, I.; Brengues, C.; Nwane, P.; Chouaibou, M.; Ndjemai, H.; Frédéric, S. Short report: First report of knockdown mutations in the malaria vector Anopheles gambiae from cameroon. Am. J. Trop. Med. Hyg. 2006, 74, 795–797. [Google Scholar] [PubMed]
- Nwane, P.; Etang, J.; Chouaibou, M.; Toto, J.; Koffi, A.; Mimpfound, R.; Simard, F. Multiple insecticide resistance mechanisms in Anopheles gambiae s.l. populations from Cameroon, Central Africa. Parasites Vectors 2013, 6, 41. [Google Scholar] [CrossRef] [PubMed]
- Djégbe, I.; Boussari, O.; Sidick, A.; Martin, T.; Ranson, H.; Chandre, F.; Akogbéto, M.; Corbel, V. Dynamics of insecticide resistance in malaria vectors in Benin: First evidence of the presence of L1014S kdr mutation in Anopheles gambiae from West Africa. Malar. J. 2011, 10, 261. [Google Scholar] [CrossRef] [PubMed]
- Padonou, G.; Sezonlin, M.; Ossè, R.; Aïzoun, N.; Agbo, F.; Oussou, O.; Gbédjissi, G.; Akogbéto, M. Impact of three years of large scale indoor residual spraying (IRS) and insecticide treated nets (ITNs) interventions on insecticide resistance in Anopheles gambiae s.l. in Benin. Parasites Vectors 2012, 5, 72. [Google Scholar] [CrossRef] [PubMed]
- Zoubiri, S.; Baaliouamer, A. Potentiality of plants as source of insecticide principles. J. Saudi Chem. Soc. 2014, 18, 925–938. [Google Scholar] [CrossRef]
- Tripathi, A.K.; Upadhyay, S.; Bhuiyan, M.; Bhattacharya, P.R. A review on prospects of essential oils as biopesticide in insect-pest management. J. Pharmacogn. Phyther. 2009, 1, 52–63. [Google Scholar]
- Isman, M.B.; Miresmailli, S.; Machial, C. Commercial opportunities for pesticides based on plant essential oils in agriculture, industry and consumer products. Phytochem. Rev. 2011, 10, 197–204. [Google Scholar] [CrossRef]
- Rehman, J.U.; Ali, A.; Khan, I.A. Fitoterapia Plant based products: Use and development as repellents against mosquitoes: A review. Fitoterapia 2014, 95, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Gnankiné, O. Use of biopesticides in the perspective of chemical resistance management in Western Africa: The cases of Bemisia tabaci (Homotera: Aleyrodidae) and Anopheles gambiae (Diptera: Culicidae). Trends Entomol. 2012, 8, 85–95. [Google Scholar]
- Nerio, S.L.; Olivero-verbel, J.; Stashenko, E. Bioresource Technology Repellent activity of essential oils: A review. Bioresour. Technol. 2010, 101, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Katz, T.; Miller, J.H.; Hebert, A. Insects repellents: Historical perspectives and new developments. J. Am. Acad. Dermatol. 2008, 58, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Guenther, E. The Essential Oils; D. Van Nostrand Co. Inc.: New York, NY, USA, 1948; p. 214. [Google Scholar]
- ASTA. Official Analytical Methods of the American Spice Trade Association; ASTA: Englewood Cliffs, NJ, USA, 1968. [Google Scholar]
- Chialva, F.; Gabri, G.; Liddle, P.A.P.; Ulian, F. Qualitative evaluation of aromatic herbs by direct headspace GC analysis. Application of the method and comparison with the traditional analysis of essential oil. J. High Resolut. Chromatogr. 1982, 5, 182–188. [Google Scholar] [CrossRef]
- Burbott, A.J.; Loomis, W.D. Effects of light and temperature on the monoterpenes of peppermint. Plant Physiol. 1967, 42, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Takeoka, G.; Ebeler, S.; Jennings, W. Capillary gas chromatographic analysis of volatile flavor compounds. In American Chemical Society Symposium; American Chemical Society: Washington, DC, USA, 1985; pp. 96–108. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Glasby, J. Encyclopédia of the Terpenoids; Wiley: New York, NY, USA, 1982; pp. 125–187. [Google Scholar]
- Prajapati, V.; Tripathi, A.K.; Aggarwal, K.K.; Khanuja, S.P.S. Insecticidal, repellent and oviposition-deterrent activity of selected essential oils against Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus. Bioresour. Technol. 2005, 96, 1749–1757. [Google Scholar] [CrossRef] [PubMed]
- Bassolé, I.; Guelbeogo, W.; Nébié, R.; Costantini, C.; Sagnon, N.; Kaboré, Z.; Traoré, S. Ovicidal and larvicidal activity against Aedes aegypti and Anopheles gambiae complex mosquitoes of essential oils extracted from three spontaneous plants of Burkina Faso. Parassitologia 2003, 45, 23–26. [Google Scholar] [PubMed]
- Tchoumbougnang, F.; Dongmo, P.; Sameza, L.; Mbanjo, N.; Fotso, G.; Zollo, P.; Menut, C. Activité larvicide sur Anopheles gambiae Giles et composition chimique des huiles essentielles extraites de quatre plantes cultivées au Cameroun. Biotechnol. Agron. Soc. Environ. 2009, 13, 77–84. [Google Scholar]
- Deletre, E.; Martin, T.; Campagne, P.; Bourguet, D.; Cadin, A.; Menut, C.; Bonafos, R.; Chandre, F. Repellent, Irritant and Toxic Effects of 20 Plant Extracts on Adults of the Malaria Vector Anopheles gambiae Mosquito. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Govindarajan, M.; Rajeswary, M.; Senthilmurugan, S.; Vijayan, P.; Alharbi, N.S.; Km, S.; Khaled, J.M.; Benelli, G. Larvicidal activity of the essential oil from Amomum subulatum Roxb.(Zingiberaceae) against Anopheles subpictus, Aedes albopictus and Culex tritaeniorhynchus (Diptera: Culicidae), and non-target impact on four mosquito natural enemies. Physiol. Mol. Plant Pathol. 2017. [Google Scholar] [CrossRef]
- Pitasawat, B.; Champakaew, D.; Choochote, W.; Jitpakdi, A.; Chaithong, U. Aromatic plant-derived essential oil: An alternative larvicide for mosquito control. Fitoterapia 2007, 78, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Tian, Y. Chemical composition and larvicidal activity of essential oil of Artemisia gilvescens against Anopheles anthropophagus. Parsitol. Res. 2013, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Sanei-dehkordi, A.; Vatandoost, H.; Abaei, M.R. Chemical Composition and Larvicidal Activity of Bunium persicum Essential Oil Against Two Important Mosquitoes Vectors. J. Essen. Oil Bear. Plants 2016, 19, 349–357. [Google Scholar] [CrossRef]
- Massebo, F.; Tadesse, M.; Bekele, T.; Balkew, M.; Gebre-michael, T. Evaluation on larvicidal effects of essential oils of some local plants against Anopheles arabiensis Patton and Aedes aegypti Linnaeus (Diptera, Culicidae) in Ethiopia. Afr. J. Biotechnol. 2009, 8, 4183–4188. [Google Scholar]
- Kiran, S.R.; Bhavani, K.; Devi, P.S.; Rao, B.R.R.; Reddy, K.J. Composition and larvicidal activity of leaves and stem essential oils of Chloroxylon swietenia DC against Aedes aegypti and Anopheles stephensi. Bioresour. Technol. 2006, 97, 2481–2484. [Google Scholar] [CrossRef] [PubMed]
- Mozaffari, E.; Abai, M.R.; Khanavi, M.; Vatandoost, H.; Sedaghat, M.M.; Moridnia, A.; Saber-Navaei, M.; Sanei-Dehkordi, A.; Rafi, F. Chemical composition, larvicidal and repellency properties of Cionura erecta (L.) Griseb. against malaria vector, Anopheles stephensi liston (Diptera: Culicidae). J. Arthropod-Borne Dis. 2014, 8, 147–155. [Google Scholar] [PubMed]
- Sanei-dehkordi, A.; Sedaghat, M.M.; Vatandoost, H. Original Article Chemical Compositions of the Peel Essential Oil of Citrus aurantium and Its Natural Larvicidal Activity against the Malaria Vector Anopheles stephensi (Diptera: Culicidae) in Comparison with Citrus paradisi. J. Arthropod-Borne Dis. 2016, 10, 577–585. [Google Scholar] [PubMed]
- Govindarajan, M.; Sivakumar, R. Mosquito larvicidal activity of thymol from essential oil of Coleus aromaticus Benth. against Culex tritaeniorhynchus, Aedes albopictus, and Anopheles subpictus (Diptera: Culicidae). Parasitol. Res. 2013, 112, 3713–3721. [Google Scholar] [CrossRef] [PubMed]
- Mdoe, F.P.; Cheng, S.; Lyaruu, L.; Nkwengulila, G.; Chang, S.; Kweka, E.J. Larvicidal efficacy of Cryptomeria japonica leaf essential oils against Anopheles gambiae. Parasites Vectors 2014, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Wang, Y.; Khan, I.A. Larvicidal and Biting Deterrent Activity of Essential Oils of Curcuma longa, Ar-turmerone, and Curcuminoids Against Aedes aegypti and Anopheles quadrimaculatus (Culicidae: Diptera). J. Med. Entomol. 2015, 1–8. [Google Scholar] [CrossRef]
- Ntonga, P.A.; Baldovini, N.; Mouray, E.; Mambu, L.; Belong, P.; Grellier, P. Activity of Ocimum basilicum, Ocimum canum, and Cymbopogon citratus essential oils against Plasmodium falciparum and mature-stage larvae of Anopheles funestus s. s. Parasite 2014, 21, 33. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, A.; Jayaraman, M.; Venkatesalu, V. Chemical constituents and larvicidal potential of Feronia limonia leaf essential oil against Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus. Parsitol. Res. 2013, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Golfakhrabadi, F.; Khanavi, M.; Ostad, S.N.; Saeidnia, S. Original Article Biological Activities and Composition of Ferulago carduchorum Essential Oil. J. Arthropod Borne Dis. 2015, 9, 104–115. [Google Scholar] [PubMed]
- Karunamoorthi, K.; Girmay, A.; Hayleeyesus, S.F. Mosquito repellent activity of essential oil of Ethiopian ethnomedicinal plant against Afro-tropical malarial vector Anopheles arabiensis. J. King Saud Univ. Sci. 2014, 26, 305–310. [Google Scholar] [CrossRef]
- Kulkarni, R.R.; Pawar, P.V.; Joseph, M.P.; Akulwad, A.K.; Sen, A.; Joshi, S.P. Lavandula gibsoni and Plectranthus mollis essential oils: Chemical analysis and insect control activities against Aedes aegypti, Anopheles stephensi and Culex quinquefasciatus. J. Pest Sci. 2013, 86, 713–718. [Google Scholar] [CrossRef]
- Govindarajan, M.; Sivakumar, R.; Rajeswary, M.; Yogalakshmi, K. Experimental Parasitology Chemical composition and larvicidal activity of essential oil from Ocimum basilicum (L.) against Culex tritaeniorhynchus, Aedes albopictus and Anopheles subpictus (Diptera: Culicidae). Exp. Parasitol. 2013, 134, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.K.; Prajapati, V.; Ahmad, A.; Aggarwal, K.K.; Khanuja, S.P.S. Piperitenone Oxide as Toxic, Repellent, and Reproduction Retardant Toward Malarial Vector Anopheles stephensi (Diptera: Anophelinae) J. Med. Entomol. 2004, 41, 691–698. [Google Scholar] [CrossRef]
- Krishnamoorthy, S.; Chandrasekaran, M. Identification of chemical constituents and larvicidal activity of essential oil from Murraya exotica L. (Rutaceae) against Aedes aegypti, Anopheles stephensi and Culex quinquefasciatus (Diptera: Culicidae). Parasitol. Res. 2015, 114, 1839–1845. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Rajeswary, M.; Arivoli, S. Larvicidal and repellent potential of Zingiber nimmonii (J. Graham) Dalzell (Zingiberaceae) essential oil: An eco-friendly tool against malaria, dengue, and lymphatic filariasis mosquito vectors? Parasitol. Res. 2016, 115, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Matasyoh, J.C.; Wathuta, E.M.; Kariuki, S.T.; Chepkorir, R. Journal of Asia-Paci fi c Entomology Chemical composition and larvicidal activity of Piper capense essential oil against the malaria vector, Anopheles gambiae. J. Asia Pac. Entomol. 2011, 14, 26–28. [Google Scholar] [CrossRef]
- Kweka, J.E.; Senthilkumar, A.; Venkatesalu, V. Toxicity of essential oil from Indian borage on the larvae of the African malaria vector mosquito, Anopheles gambiae. Parasites Vectors 2012, 5, 277. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, A.; Venkatesalu, V. Chemical composition and larvicidal activity of the essential oil of Plectranthus amboinicus (Lour.) Spreng against Anopheles stephensi: a malarial vector mosquito. Parsitol. Res. 2010, 107, 1275–1278. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Rajeswary, M.; Veerakumar, K.; Muthukumaran, U.; Hoti, S.L.; Mehlhorn, H.; Barnard, D.R. Novel synthesis of silver nanoparticles using Bauhinia variegata: A recent eco-friendly approach for mosquito control. Parasitol. Res. 2015, 115, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Demirci, B.; Kiyan, H.T.; Bernier, U.R.; Tsikolia, M.; Wedge, D.E.; Khan, I.A.; Husnu, K.; Bas, C.A.N. Biting Deterrence, Repellency, and Larvicidal Activity of Ruta chalepensis (Sapindales: Rutaceae) Essential Oil and Its Major Individual Constituents Against Mosquitoes. J. Med. Entomol. 2013, 50, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Benelli, G. α-Humulene and β-elemene from Syzygium zeylanicum (Myrtaceae) essential oil: Highly effective and eco-friendly larvicides against Anopheles subpictus, Aedes albopictus, and Culex tritaeniorhynchus (Diptera: Culicidae). Parasitol. Res. 2016, 115, 2771–2778. [Google Scholar] [CrossRef] [PubMed]
- Dharmagadda, V.S.S.; Naik, S.N.; Mittal, P.K.; Vasudevan, P. Larvicidal activity of Tagetes patula essential oil against three mosquito species. Bioresour. Technol. 2005, 96, 1235–1240. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.C.; Dong, H.W.; Zhou, L.; Du, S.S.; Liu, Z.L. Essential oil composition and larvicidal activity of Toddalia asiatica roots against the mosquito Aedes albopictus (Diptera: Culicidae). Parasitol. Res. 2013, 112, 1197–1203. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.K.; Upadhyay, S.; Tripathi, A.K. Insecticidal and repellent activities of thymol from the essential oil of Trachyspermum ammi (Linn) Sprague seeds against Anopheles stephensi. Parasitol. Res. 2009, 105, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Tiwary, M.; Naik, S.N.; Tewary, D.K.; Mittal, P.K.; Yadav, S. Chemical composition and larvicidal activities of the essential oil of Zanthoxylum armatum DC (Rutaceae) against three mosquito vectors. J. Vector Borne Dis. 2007, 44, 198–204. [Google Scholar] [PubMed]
- Sanei-dehkordi, A.; Soleimani-ahmadi, M.; Akbarzadeh, K.; Abadi, Y.S.; Paksa, A.; Gorouhi, M.A. Chemical Composition and Mosquito Larvicidal Properties of Essential Oil from Leaves of an Iranian Indigenous Plant Zhumeria majdae. J. Essen. Oil Bear. Plant. 2016, 19, 1454–1461. [Google Scholar] [CrossRef]
- Kweka, E.J.; Lima, T.C.; Marciale, C.M.; Sousa, D.P. De Asian Paci fi c Journal of Tropical Biomedicine. Asian Pac. J. Trop. Biomed. 2016, 6, 290–294. [Google Scholar] [CrossRef]
- Govindarajan, M.; Mathivanan, T.; Elumalai, K. Mosquito larvicidal, ovicidal, and repellent properties of botanical extracts against Anopheles stephensi, Aedes aegypti, and Culex quinquefasciatus (Diptera: Culicidae). Parsitol. Res. 2011, 109, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Benelli, G. Facile biosynthesis of silver nanoparticles using Barleria cristata: Mosquitocidal potential and biotoxicity on three non-target aquatic organisms. Parsitol. Res. 2015, 115, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Pavela, R.; Pavela, R. Essential oils for the development of eco-friendly mosquito larvicides: A review. Ind. Crop. Prod. 2016, 76, 174–187. [Google Scholar] [CrossRef]
- Dias, C.N.; Moraes, F.D.C. Essential oils and their compounds as Aedes aegypti L. (Diptera: Culicidae) larvicides: review. Parsitol. Res. 2014, 113, 565–592. [Google Scholar] [CrossRef] [PubMed]
- Norris, E.J.; Gross, A.D.; Dunphy, B.M.; Bessette, S. Comparison of the Insecticidal Characteristics of Commercially Available Plant Essential Oils Against Aedes aegypti and Anopheles gambiae (Diptera: Culicidae). J. Med. Entomol. 2015, 52, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Bossou, A.D.; Mangelinckx, S.; Yedomonhan, H.; Boko, P.M.; Akogbeto, M.C.; De Kimpe, N.; Avlessi, F.; Sohounhloue, D.C.K. Chemical composition and insecticidal activity of plant essential oils from Benin against Anopheles gambiae (Giles). Parasites Vectors 2013, 6, 337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- WHO. Test Procedures; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Choochote, W.; Chaithong, U.; Kamsuk, K.; Jitpakdi, A.; Tippawangkosol, P.; Tuetun, B.; Champakaew, D.; Pitasawat, B. Repellent activity of selected essential oils against Aedes aegypti. Fitoterapia 2007, 78, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Deletre, E.; Schatz, B.; Chandre, F.; Ratnadass, A. Prospects for repellent in pest control: Current developments and future challenges. Chemoecology 2016, 26, 127–142. [Google Scholar] [CrossRef]
- Amer, A.; Mehlhorn, H. Repellency effect of forty-one essential oils against Aedes, Anopheles, and Culex mosquitoes. Parasitol. Res. 2006, 99, 478–490. [Google Scholar] [CrossRef] [PubMed]
- WHO. Report of the Who Informal Consultation on the Evaluation and Testing of Insecticides CTD/WHOPES/IC/96.1 Geneva: Control of Tropical Diseases; World Health Organization: Geneva, Switzerland, 1996. [Google Scholar]
- Rajikumar, S.; Jebanessan, A. Repellent activity of selected plant essential oils against the malarial fever mosquito Anopheles stephensi. Trop. Biomed. 2007, 24, 71–75. [Google Scholar]
- Phasomkusolsil, S.; Soonwera, M. Comparative mosquito repellency of essential oils against Aedes aegypti (Linn.), Anopheles dirus (Peyton and Harrison) and Culex quinquefasciatus (Say). Asian Pac. J. Trop. Biomed. 2011, 1, S113–S118. [Google Scholar] [CrossRef]
- Abagli, A.Z.; Alavo, T.B.C. Essential Oil from Bush Mint, Hyptis suaveolens, is as Effective as DEET for Personal Protection against Mosquito Bites. Open Entomol. 2011, 5, 45–48. [Google Scholar] [CrossRef]
- Ipek, E.; Zeytinoglu, H.; Okay, S.; Tuylu, B.A.; Kurkcuoglu, M.; Baser, K.H.C. Food Chemistry Genotoxicity and antigenotoxicity of Origanum oil and carvacrol evaluated by Ames Salmonella/microsomal test. Food Chem. 2005, 93, 551–556. [Google Scholar] [CrossRef]
- Govindarajan, M.; Sivakumar, R.; Rajeswari, M. Chemical composition and larvicidal activity of essential oil from Mentha spicata (Linn.) against three mosquito species. Parasitol. Res. 2012, 110, 2023–2032. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Rajeswary, M.; Hoti, S.L.; Benelli, G. Research in Veterinary Science Larvicidal potential of carvacrol and terpinen-4-ol from the essential oil of Origanum vulgare (Lamiaceae) against Anopheles stephensi, Anopheles subpictus, Culex quinquefasciatus and Culex tritaeniorhynchus (Diptera: Culicidae). Res. Vet. Sci. 2016, 104, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Rajeswary, M.; Hoti, S.L.; Bhattacharyya, A.; Benelli, G. Eugenol, α-pinene and β-caryophyllene from Plectranthus barbatus essential oil as eco-friendly larvicides against malaria, dengue and Japanese encephalitis mosquito vectors. Parasitol. Res. 2016, 115, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Deletre, E.; Chandre, F.; Williams, L.; Duménil, C.; Menut, C.; Martin, T. Electrophysiological and behavioral characterization of bioactive compounds of the Thymus vulgaris, Cymbopogon winterianus, Cuminum cyminum and Cinnamomum zeylanicum essential oils against Anopheles gambiae and prospects for their use as bednet treatmen. Parasites Vectors 2015, 8, 316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaenson, T.G.T.; Pålsson, K. Borg-karlson Anna-Karin Evaluation of Extracts and Oils of Mosquito (Diptera: Culicidae) Repellent Plants from Sweden and Guinea-Bissau. J. Med. Entomol. 2006, 43, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Sukumar, K.; Perich, M.; Boobar, L. Botanical derivatives in mosquito control: A review. J. Am. Mosq. Control Assoc. 1991, 7, 210–237. [Google Scholar] [PubMed]
- Jantan, I.; Zaki, Z. Development of environment-friendly insect repellents from the leaf oils of selected malaysian plants. Rev. Biodivers. Environ. Conserv. 1999, 6, 1–7. [Google Scholar]
- Park, B.-S.P.; Choi, W.-S.; Kim, J.-H.; Kim, K.-H.; Lee, S.-E. Monoterpenes from thyme (Thymus vulgaris) as potential mosquito repellents potential mosquito repellents. Source J. Am. Mosq. Control Assoc. 2005, 21, 80–83. [Google Scholar] [CrossRef]
- Omolo, M.O.; Okinyo, D.; Ndiege, I.O.; Hassanali, A. Repellency of essential oils of some Kenyan plants against Anopheles gambiae. Phytochemistry 2004, 65, 2797–2802. [Google Scholar] [CrossRef] [PubMed]
- Trongtokit, Y.; Rongsriyam, Y.; Komalamisra, N. Comparative Repellency of 38 Essential Oils against Mosquito Bites. Phyther. Res. 2005, 309, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Gillij, Y.G.; Gleiser, R.M.; Zygadlo, J.A. Mosquito repellent activity of essential oils of aromatic plants growing in Argentina. Bioresour. Technol. 2008, 99, 2507–2515. [Google Scholar] [CrossRef] [PubMed]
- Odalo, J.O.; Omolo, M.O.; Malebo, H.; Angira, J.; Njeru, P.M.; Ndiege, I.O.; Hassanali, A. Repellency of essential oils of some plants from the Kenyan coast against Anopheles gambiae. Acta Trop. 2005, 95, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Pridham, J. Terpenoids in Plants; Academic Press: New York, NY, USA, 1967; pp. 59–82. [Google Scholar]
- Davidson, P.; Parish, M. Methods for testing the efficacy of food antimicrobials. Food Technol. 1989, 43, 148–155. [Google Scholar]
- Abbassy, M.; Abdelgaleil, S.; Rabie, R. Insecticidal and synergistic effects of Majorana hortensis essential oil and some of its major constituents. Entomol. Exp. Appl. 2009, 131, 225–232. [Google Scholar] [CrossRef]
- Berenbaum, M.; Neal, J. Synergism between myristicin and xanthotoxin, a naturally cooccurring plant toxicant. J. Chem. Ecol. 1985, 11, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential Oils in Insect Control: Low-Risk Products in a High-Stakes World. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Prates, H.T.; Santos, J.P.; Waquil, J.M.; Fabris, J.D.; Oliveira, A.B.; Foster, J.E.; Lagoas, S.; Horizonte, B. Insecticidal Activity of Monoterpenes Against Rhyzopertha dominica (F.) and Tribolium castaneum (Herbst). J. Stored Prod. Res. 1998, 34, 5–8. [Google Scholar] [CrossRef]
- Regnault-Roger, C. The potential of botanical essential oils for insect pest control. Integr. Pest Manag. Rev. 1997, 34, 25–34. [Google Scholar] [CrossRef]
- Rattan, R. Mechanism of action of insecticidal secondary metabolites of plant origin. Crop Prot. 2010, 29, 913–920. [Google Scholar] [CrossRef]
- Fournier, D.; Mutero, A. Modification of acetylcholinesterase as a mechanism of resistance to insecticide. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1994, 108, 19–31. [Google Scholar] [CrossRef]
- Aygun, D.; Doganay, Z.; Altintop, L.; Guven, H.; Onar, M.; Deniz, T.; Sunter, T. Serum acetylcholinesterase and prognosis of acute organophosphate poisoning. J. Toxicol. Clin. Toxicol. 2002, 40, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Houghton, P.; Ren, Y.; Howes, M. Acetylcholinesterase inhibitors from plants and fungi. Nat. Prod. Rep. 2006, 23, 181–199. [Google Scholar] [CrossRef] [PubMed]
- Abdelgaleil, S.A.; Mohamed, M.I.; Badawy, M.E.; El-arami, S.A. Fumigant and contact toxicities of monoterpenes to Sitophilus oryzae (L.) and Tribolium castaneum (Herbst) and their inhibitory effects on acetylcholinesterase activity. J. Chem. Ecol. 2009, 35, 518–535. [Google Scholar] [CrossRef] [PubMed]
- Hideyuki, T.; Mitsuo, M. Inhibition of acetylcholinesterase activity by essential oil from Bergamot. Koryo Terupen oyobi Seiyu Kagaku ni Kansuru Toronkai Koen Yoshishu 2001, 45, 435–436. [Google Scholar]
- Seo, S.M.; Jung, C.S.; Kang, J.; Lee, H.R.; Kim, S.W.; Hyun, J.; Park, I.K. Larvicidal and Acetylcholinesterase Inhibitory Activities of Apiaceae Plant Essential Oils and Their Constituents against Aedes albopictus and Formulation Development. J. Agric. Food Chem. 2015, 63, 9977–9986. [Google Scholar] [CrossRef] [PubMed]
- Savelev, S.; Okello, E.; Perry, N.; Wilkins, R.; Perry, E. Synergistic and antagonistic interactions of anticholinesterase terpenoids in Salvia lavandulae folia essential oil. Pharmacol. Biochem. Behav. 2003, 75, 661–668. [Google Scholar] [CrossRef]
- Keane, S.; Ryan, M. Purification, characterisation, and inhibition bymonoterpenes of acetylcholinesterase from the waxmoth, Galleria mellonella (L.). Insect Biochem. Mol. Biol. 1999, 29, 1097–1104. [Google Scholar] [CrossRef]
- Evans, P. Multiple receptor types for octopamine in the locust. J. Physiol. 1981, 318, 99–122. [Google Scholar] [CrossRef] [PubMed]
- Evans, P. Studies on the mode of action of octopamine, 5-hydroxytryptamine and proctolin on a myogenic rhythm in the locust. J. Exp. Biol. 1984, 110, 231–251. [Google Scholar] [PubMed]
- Enan, E. Insecticidal activity of essential oils: Octopaminergic sites of action. Comp. Biochem. Physiol. 2001, 130, 325–337. [Google Scholar] [CrossRef]
- Kostyukovsky, M.; Rafaeli, A.; Gileadi, C.; Demchenko, N.; Shaaya, E. Activation of octopaminergic receptors by essential oil constituents isolated from aromatic plants: Possible mode of action against insect pests. Pest Manag. Sci. 2002, 1106, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.; Robb, S. Octopamine receptor subtypes and their modes of action. Neurochem. Res. 1993, 18, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Howell, K.; Evans, P. The characterization of presynaptic octopamine receptors modulating octopamine release from an identified neurone in the locust. J. Exp. Biol. 1998, 201, 2053–2060. [Google Scholar] [PubMed]
- Bloomquist, J.R. Chloride channels as tools for developing selective insecticides. Arch. Insect Biochem. Physiol. 2003, 54, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Tong, F.; Coats, J.R. Effects of monoterpenoid insecticides on [3H]-TBOB binding in house fly GABA receptor and 36Cl− À uptake in American cockroach ventral nerve cord. Pestic. Biochem. Physiol. 2010, 98, 317–324. [Google Scholar] [CrossRef]
- Hold, M.; Sirisoma, S.; Ikeda, T.; Narahashi, T.; Casida, E. Thujone (the active component of absinthe): aminobutyric acid type. A receptor modulation and metabolic detoxification. Proc. Natl. Acad. Sci. USA 2000, 97, 3826–3831. [Google Scholar] [CrossRef] [PubMed]
- Ratra, G.; Casida, J. GABA receptor subunit composition relative to insecticide potency and selectivity. Toxicol. Lett. 2001, 122, 215–222. [Google Scholar] [CrossRef]
- Priestley, C.; Williamson, E.; Wafford, K.; Satelle, D.B. Thymol, a constituent of thyme essential oil, is a positive allosteric modulator of human GABA receptors and a homo-oligomeric GABA receptor from Drosophila melanogaster. Br. J. Pharmacol. 2003, 140, 1363. [Google Scholar] [CrossRef] [PubMed]
- Krimer, V.; Vaštag, Ž.; Radulovi, L.; Peri, I. Microencapsulation Technology and Essential Oil Pesticides for Food Plant Production. In Essential Oils in Food Preservation, Flavor and Safety; Preedy, V., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 123–129. [Google Scholar]
- Devi, N.; Maji, T.K. Neem seed oil: Encapsulation and controlled release—Search for a greener alternative for pest control. In Pesticides in the Modern World—Pesticides Use and Management; Stoytcheva, M., Ed.; InTech: Rijeka, Croatia, 2011; pp. 191–232. [Google Scholar]
- Maji, T.; Baruah, I.; Dube, S.; Hussain, M. Microencapsulation of Zanthoxylum limonella oil (ZLO) in glutaraldehyde crosslinked gelatin for mosquito repellent application. Bioresour. Technol. 2007, 98, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Tawatsin, A.; Wratten, S.D.; Scott, R.R.; Thavara, U.; Techadamrongsin, Y. Repellency of volatile oils from plants against three mosquito vectors. J. Vector Ecol. 2001, 26, 76–82. [Google Scholar]
- Specos, M.M.; Garcia, J.J.; Tornesello, J.; Marino, P.; Vecchia, M.D.; Tesoriero, M.D.; Hermida, L.G. Transactions of the Royal Society of Tropical Medicine and Hygiene Microencapsulated citronella oil for mosquito repellent finishing of cotton textiles. Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Solomon, B.; Sahle, F.; Gebre-Mariam, T.; Asres, K.; Neubert, R. Microencapsulation of citronella oil for mosquito-repellent application: Formulation and in vitro permeation studies. Eur. J. Pharm. Biopharm. 2012, 80, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Sakulkua, U.; Nuchuchuaa, O.; Uawongyartb, N.; Puttipipatkhachornc, S.; Soottitantawatd, A.; Ruktanonchaia, U. Characterization and mosquito repellent activity of citronella oil nanoemulsion. Int. J. Pharm. Nanotechnol. 2009, 372, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Duvallet, G.; De Gentile, L. Protection Personnelle Antivectorielle; IRD: Marseille, France, 2012; p. 352. [Google Scholar]
- Zaim, M.; Aitio, A.; Nakashima, N. Safety of pyrethroid-treated mosquito nets. Med. Vet. Entomol. 2000, 14, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Palchick, S. The Biology of Disease of Disease Vectors; University Press of Colarado: Boulder, CO, USA, 1996. [Google Scholar]
- McAllister, C.; Adams, M.F. Mode of Action for Natural Products Isolated From Essential Oils of Two Trees Is Different From Available Mosquito Adulticides. J. Med. Entomol. 2010, 47, 1123–1126. [Google Scholar] [CrossRef] [PubMed]
- Chitwood, D. Phytochemical based strategies for nematode control. Annu. Rev. Phytopathol. 2002, 40, 221–249. [Google Scholar] [CrossRef] [PubMed]
- Yadouleton, A.; Martin, T.; Padonou, G.; Chandre, F.; Asidi, A.; Djogbenou, L.; Dabiré, R.; Aïkpon, R.; Boko, M.; Glitho, I.; et al. Cotton pest management practices and the selection of pyrethroid resistance in Anopheles gambiae population in Northern Benin. Parasites Vectors 2011, 4, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Plant Species | Plant Organ | Extraction Method | Species | LC50 (mg/L) | LC90 (mg/L) | LC95 (mg/L) | Reference |
|---|---|---|---|---|---|---|---|
| Cinnamomum zeylanicum | Bark | Hydrod. | An. stephensi | NI | NI | 130.0 | [36] |
| Cuminum cyminum | Seed | Hydrod. | An. stephensi | NI | NI | 150.7 | [36] |
| Curcuma longa | Rhizome | Hydrod. | An. stephensi | NI | NI | 89.9 | [36] |
| Juniperus macropoda | Fruit | Hydrod. | An. stephensi | NI | NI | 59.3 | [36] |
| Ocimum basilicum | Leaf | Hydrod. | An. stephensi | NI | NI | 172.8 | [36] |
| Pimpinella anisum | Seed | Hydrod. | An. stephensi | NI | NI | 33.3 | [36] |
| Zingiber officinalis | Rhizome | Hydrod. | An. stephensi | NI | NI | 32.2 | [36] |
| Lippia multiflora | Leaf | Hydrod. | An. gambiae s.s | 17.1 | 33.5 | NI | [37] |
| Ocimum canum | Leaf | Hydrod. | An. gambiae s.s | 188.7 | 488.0 | NI | [37] |
| Cymbopogon proximus | Leaf | Hydrod. | An. gambiae s.s | 52.8 | 91.1 | NI | [37] |
| Plant Species | Plant Organ | Extraction Method | Mosquito | LC50 (mg/L) | LC90 (mg/L) | LC95 (mg/L) | LC99 (mg/L) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Amomum subulatum | Leaf | Hydrod. | An. subpictus | 41.2 | 80.3 | NI | NI | [40] |
| Apium graveolens | Whole plant | Steam-d. | An. dirus | 59.4 | 111.7 | 159.1 | [41] | |
| Artemisia gilvescens | Plant | Hydrod. | An. anthropophagus | 49.9 | 97.4 | NI | NI | [42] |
| Artemisia gilvescens | Whole | Hydrod. | An. anthropophagus | 49.0 | 97.0 | NI | NI | [42] |
| Blumea densiflora | Aerial parts | Hydrod. | An. anthropophagus | 10.0 | 33.0 | NI | NI | [42] |
| Bunium persicum | Seed | Hydrod. | An. stephensi | 27.7 | 91.3 | NI | NI | [43] |
| Carum carvi | Whole plant | Steam-d. | An. dirus | 72.2 | NI | 104.7 | 128.7 | [41] |
| Chenopodium ambrosioides | Aerial parts | Hydrod. | An. arabiensis | 17.5 | NI | NI | NI | [44] |
| Chloroxylon swietenia | Leaf and stem | Hydrod. | An. stephensi | 14.0 | 22.0 | NI | NI | [45] |
| Cionura erecta | Root | Hydrod. | An. stephensi | 77.0 | 199.0 | NI | NI | [46] |
| Citrus aurantium | Fruit | Hydrod. | A. stephensi | 31.2 | 73.8 | NI | NI | [47] |
| Citrus paradisi | Fruit | Hydrod. | An. stephensi | 35.7 | 70.2 | NI | NI | [47] |
| Coleus aromaticus | Leaf | Hydrod. | An. subpictus | 60.3 | 118.7 | NI | NI | [48] |
| Cryptomeria japonica | Leaf | Hydrod. | An. gambiae | 67.1 | 193.7 | NI | NI | [49] |
| Curcuma longa | Leaf | Hydrod. | An. quadrimaculatus | 1.8 | 4.1 | NI | NI | [50] |
| Curcuma longa | Rhizome | Hydrod. | An. quadrimaculatus | 3.7 | 9.4 | NI | NI | [50] |
| Curcuma zedoaria | Whole plant | Steam-d. | An. dirus | 29.7 | NI | 40.2 | 47.7 | [41] |
| Cymbopogon citratus | Leaf | Hydrod. | An. funestus | 34.6 | NI | NI | NI | [51] |
| Cymbopogon citratus | Leaf | Hydrod. | An. gambiae | 18.0 | NI | NI | NI | [38] |
| Cymbopogon proximus | Leaf | Hydrod. | An. gambiae | 69.7 | NI | NI | NI | [37] |
| Feronia limonia | Leaf | Hydrod. | An. stephensi | 15.0 | 36.7 | NI | NI | [52] |
| Ferulago carduchorum | Aerial parts | Hydrod. | An. stephensi | 12.0 | 47.0 | NI | NI | [53] |
| Ferulago carduchorum | Aerial part | Hydrod. | An. stephensi | 12.0 | 47.0 | NI | NI | [53] |
| Foeniculum vulgare | Whole plant | Steam-d. | An. dirus | 35.3 | NI | 38.8 | 40.9 | [41] |
| Juniperus procera | Leaf | Hydrod. | An. arabiensis | 24.5 | 34.2 | NI | NI | [54] |
| Lavandula gibsoni | Plant | Hydrod. | An. stephensi | 62.8 | 129.0 | NI | NI | [55] |
| Lippia multiflora | Leaf | Hydrod. | An. gambiae | 61.9 | NI | NI | NI | [37] |
| Mentha spicata | Aerial part | Hydrod. | An. stephensi | 49.7 | 101.0 | NI | NI | [56] |
| Mentha spicata | Leaf | Hydrod. | An. stephensi | 82.9 | NI | NI | NI | [57] |
| Murraya exotica | Leaf | Hydrod. | An. stephensi | 56.3 | 107.8 | NI | NI | [58] |
| Nigella sativa | Leaf | Hydrod. | An. arabiensis | 23.4 | NI | NI | NI | [44] |
| Ocimum basilicum | Leaf | Hydrod. | An. subpictus | 9.75 | 18.6 | NI | NI | [56] |
| Ocimum canum | Leaf | Hydrod. | An. funestus | 91.2 | NI | NI | NI | [51] |
| Ocimum lamiifolium | Leaf | Hydrod. | An. arabiensis | 20.9 | NI | NI | NI | [44] |
| Origanum vulgare | Leaf | Hydrod. | An. subpictus | 67.0 | 128.6 | NI | NI | [59] |
| Piper capense | Plant | Hydrod. | An. gambiae | 34.9 | 85.0 | NI | NI | [60] |
| Plectranthus amboinicus | Leaf | Hydrod. | An. gambiae | 55.2 | 99.1 | NI | NI | [61] |
| Plectranthus amboinicus | Leaf | Hydrod. | An. stephensi | 28.37 | 59.4 | NI | NI | [62] |
| Plectranthus barbatus | Leaf | Hydrod. | An. subpictus | 84.2 | 165.2 | NI | NI | [63] |
| Plectranthus mollis | Whole plant | Hydrod. | An. stephensi | 33.5 | NI | NI | NI | [55] |
| Ruta chalepensis | Aerial parts | An. quadrimaculatus | 15.0 | 42.0 | NI | NI | [64] | |
| Salvia elegan | Aerial parts | Hydrod. | An. quadrimaculatus | 6.2 | 15.8 | NI | NI | [50] |
| Salvia leucantha | Aerial parts | Hydrod. | An. quadrimaculatus | 10.9 | 29.1 | NI | NI | [50] |
| Salvia officinalis | Aerial parts | Hydrod. | An. quadrimaculatus | 14.1 | 35.8 | NI | NI | [50] |
| Schinus molle | Leaf and seed | Hydrod. | An. arabiensis | 21 | NI | NI | NI | [44] |
| Syzygium zeylanicum | Leaf | Hydrod. | An. subpictus | 83.1 | 164.2 | NI | NI | [65] |
| Tagetes patula | Leaf | Steam-d. | An. stephensi | 12.0 | 57.0 | NI | NI | [66] |
| Toddalia asiatica | Root | Hydrod. | An. stephensi | 69.0 | 110.0 | NI | NI | [67] |
| Trachyspermum ammi | Seed | Steam-d. | An. stephensi | 80.8 | NI | NI | 172.1 | [68] |
| Zanthoxylum armatum | Seed | Hydrod. | An. stephensi | 58.0 | 183.0 | NI | NI | [69] |
| Zanthoxylum limonella | Whole plant | Steam-d. | Anopheles dirus | 57.2 | NI | 76.2 | 89.5 | [41] |
| Zhumeria majdae | Leaf | Hydrod. | An. stephensi | 61.3 | 135.8 | NI | NI | [70] |
| Zingiber nimmoni | Rhizome | Hydrod. | An. stephensi | 41.2 | 80.3 | NI | NI | [59] |
| Components of EO | Species | LC50 (mg/L) | LC90 (mg/L) | Reference |
|---|---|---|---|---|
| Monoterpene hydrocarbons | ||||
| (+)-Limonene | An. gambiae s.s. | 270.3 | NI | [71] |
| Limonene | An. stephensi | 8.8 | 17.6 | [72] |
| α-Pinene | An. subpictus | 32.1 | 62.8 | [73] |
| Terpinolene | An. gambiae s.s. | 404.71 | NI | [71] |
| Camphor | An. anthropophagus | 129.7 | 192.4 | [42] |
| Carvacrol | An. subpictus | 21.1 | 41.9 | [65] |
| Carvone | An. stephensi | 19.3 | 37.1 | [72] |
| (−)-Carvone epoxide | Anopheles gambiae s.s. | 124.7 | NI | [71] |
| cis-Carveol | An. stephensi | 28.5 | 59.2 | [72] |
| Eucalyptol | An. anthropophagus | >200 | NI | [42] |
| (−)-Hydroxycarvone | An. gambiae s.s. | 1172.2 | NI | [71] |
| (−)-Isopulegol (2) | An. gambiae s.s. | 49.4 | NI | [71] |
| (+)-Limonene epoxide | An. gambiae s.s. | 200.8 | NI | [71] |
| (−)-Perillyl alcohol | An. gambiae s.s. | 18.4 | NI | [71] |
| Piperitenone oxide | An. stephensi | 25.8 | NI | [57] |
| Terpine-4-ol | An. anthropophagus | 76.7 | 139.4 | [42] |
| Terpinen-4-ol | An. gambiae s.s. | 337.7 | NI | [71] |
| Terpinen-4-ol | An. subpictus | 43.3 | 84.1 | [65] |
| Thymol | An. stephensi | 48.88 | NI | [68] |
| Thymol | An. subpictus | 22.06 | 40.0 | [48] |
| Sesquiterpenes | ||||
| α-Humulene | An. subpictus | 6.19 | 12.0 | [65] |
| β-Caryophyllene | An. subpictus | 41.66 | 84.9 | [65] |
| β-Elemene | An. subpictus | 10.26 | 20.0 | [65] |
| Caryophyllene | An. anthropophagus | >200 | [42] | |
| Germacrene D | An. anthropophagus | 49.81 | 106.2 | [42] |
| α-Cadinol | An. subpictus | 10.27 | 20.4 | [74] |
| Ar-turmerone | An. quadrimaculatus | 2.8 | 7.0 | [50] |
| Caryophyllene oxide | An. anthropophagus | 49.46 | 115.4 | [42] |
| Germacrene D-4-ol | An. subpictus | 6.12 | 12.1 | [74] |
| Phenylpropenes | ||||
| Eugenol | An. subpictus | 25.45 | 50.6 | [65] |
| Others | ||||
| Curcumin | An. quadrimaculatus | 32.5 | 74.6 | [50] |
| Desmethoxycurcumin | An. quadrimaculatus | 29.7 | 66.7 | [50] |
| Plant Species | Plant Organ | Extraction Method | Mosquito | Methods | LC50 (mg/L) | LD50 (µg/g Mosquito) | LC95 (mg/mat) | LC99 (mg/L) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| * Deltamethrin | An. gambiae | TA | 0.003 | [76] | |||||
| * Permethrin | An. gambiae | TA | 0.6 | [76] | |||||
| Apium graveolens L. | NI | NI | An. gambiae | TA | 600.0 | [76] | |||
| Apium graveolens L. | NI | NI | An. gambiae | TA | 4.5 | [76] | |||
| Apium graveolens L. | NI | NI | An. gambiae | TA | 18.0 | [76] | |||
| Apium graveolens L. | seed | NI | An. gambiae | TA | 6.6 | [76] | |||
| Artemisia absinthium | NI | NI | An. gambia | TA | 12.0 | [76] | |||
| Cedrus sp. | NI | NI | An. gambiae | TA | 3.8 | [76] | |||
| Chenopodium ambrosioides | Leafy stems | Hydrod. | An. gambiae | WHOTK | 0.9 | 2.1 | [77] | ||
| Cinnamomum zeylanicum | Bark | Hydrod. | An. stephensi | MA | 286.2 | [36] | |||
| Cinnamomun zeylanicum | bark | NS | An. gambiae | TA | 2.1 | [76] | |||
| Cinnamomun zeylanicum | NI | NI | An. gambiae | TA | 2.9 | [76] | |||
| Citrus sinensis | NI | NI | An. gambiae | TA | 11.1 | [76] | |||
| Cochlospermum planchonii | Root | Hydrod. | An. gambiae | WHOTK | 2.3 | 7.6 | [77] | ||
| Cuminum cyminum | Seed | Hydrod. | An. stephensi | MA | 305.2 | [36] | |||
| Curcuma longa | Rhizome | Hydrod. | An. stephensi | MA | 302.6 | [36] | |||
| Cymbopogon citratus | Leaf | Hydrod. | An. gambiae | WHO test kits | 0.2 | 0.4 | [77] | ||
| Cymbopogon citratus | NI | NI | An. gambiae | TA | 3 | [76] | |||
| Cymbopogon schoenanthus | Leafy stems | Hydrod. | An. gambiae | WHOTK | 1.57 | 2.7 | [77] | ||
| Cymbopogon winterianus | NI | NI | An. gambiae | TA | 3.9 | [76] | |||
| Eucalyptus tereticornis | Leaf | Hydrod. | An. gambiae | WHOTK | 0.148 | 1.4 | [77] | ||
| Gaultheria procumbens | NI | NI | An. gambiae | TA | 11.1 | [76] | |||
| Juniperus virginiana | NI | NI | An. gambiae | TA | 7.7 | [76] | |||
| Litsea cubeba | NI | NI | An. gambiae | TA | 4.0 | [76] | |||
| Mentha piperita | NI | NI | An. gambiae | TA | 6.8 | [76] | |||
| Myristica fragrans | NI | NI | An. gambiae | TA | 10.5 | [76] | |||
| Ocimum basilicum | Leaf | Hydrod. | An. stephensi | MA | 316.4 | [36] | |||
| Origanum vulgare | NI | NI | An. gambiae | TA | 1.6 | [76] | |||
| Pelargonium graveolens | NI | NI | An. gambiae | TA | 2.6 | [76] | |||
| Petroselinum crispum | NI | NI | An. gambiae | TA | 5.0 | [76] | |||
| Pimpinella anisum | Seed | Hydrod. | An. stephensi | MA | 378.5 | [36] | |||
| Piper nigrum | NI | NI | An. gambiae | TA | 8 | [76] | |||
| Rosmarinus officinalis | NI | NI | An. gambiae | TA | 31 | [76] | |||
| Rosmarinus officinalis | Shoot | Hydrod. | An. stephensi | MA | 282.6 | [36] | |||
| Sassafras sp. | NI | NI | An. gambiae | TA | 10 | [76] | |||
| Sesamum indicum | NI | NI | An. gambiae | TA | 5.9 | [76] | |||
| Syzygium aromaticum | leaf | NI | An. gambiae | TA | 1.5 | [76] |
| Plant Species | Plant Organ | Extraction Method | Moquito | Test Method | Concentration (mg/cm2) | Protection Time (mn) | % Repellency | RD95 (mg/mat) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Amyris balsamifera | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Anethum graveolens | NI | NI | An. stephensi | Human bait | 0.5 | 210.0 | 71.4 | [81] | |
| Aniba rosaeodora | NI | NI | An. stephensi | Human bait | 0.5 | 390.0 | 4.8 | [81] | |
| Anthemis nobilis | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 76.2 | [81] | |
| Boswellia carteri | An. stephensi | Human bait | 0.5 | 300.0 | 19.0 | [81] | |||
| Cananga odorata | Flower | Steamd. | An. dirus | Human bait | 0.2 | 24.0 | 92.0 | [84] | |
| Chamaemelum nobile | NI | NI | An. stephensi | Human bait | 0.5 | 330.0 | 47.6 | [81] | |
| Cinnamomum camphora | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 42.8 | [81] | |
| Cinnamomum zeylanicum | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Cinnamomum zeylanicum | Bark | Hydrod. | An. stephensi | Cage test | 49.6 | [36] | |||
| Citrus limon | NI | NI | An. stephensi | Human bait | 0.5 | 420.0 | 9.5 | [81] | |
| Citrus sinensis | Fruit | Steamd. | An. dirus | Human bait | 0.21 | 24 | 84 | [84] | |
| Curcuma longa | Rhizome | Hydrod. | An. stephensi | Cage test | 93.7 | [36] | |||
| Cymbopogon citratus | Leaf and stem | Steamd. | An. dirus | Human bait | 0.2 | 132.0 | 98.0 | [84] | |
| Cymbopogon citratus | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Cymbopogon nardus | Leaf | Steamd. | An. dirus | Human bait | 0.2 | 90.0 | 98.0 | [84] | |
| Cymbopogon winterianus | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 52.4 | [81] | |
| DEET | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Eucalyptus citriodora | Leaf | Steamd. | An. dirus | Human bait | 0.2 | 30.0 | 86.0 | [84] | |
| Eucalyptus citriodora | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 52.4 | [81] | |
| Eucalyptus dives | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 38.1 | [81] | |
| Eucalyptus globulus | NI | NI | An. stephensi | Human bait | 0.5 | 330.0 | 28.6 | [81] | |
| Eucalyptus radiata | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 42.8 | [81] | |
| Ferula galbaniflua | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Glycina max | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 76.2 | [81] | |
| Glycina soja | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 9.5 | [81] | |
| Helichrysum italicum | NI | NI | An. stephensi | Human bait | 0.5 | 360.0 | 47.6 | [81] | |
| Jasminum grandiflorum | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Juniperus communis | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 76.2 | [81] | |
| Juniperus virginiana | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 38.1 | [81] | |
| Lavandula angustifolia | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 80.9 | [81] | |
| Lippia citriodora | NI | NI | An. stephensi | Human bait | 0.5 | 330.0 | 38.1 | [81] | |
| Litsea cubeba | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Melaleuca leucadendron | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Melaleuca quinquenervia | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Mentha piperita | NI | NI | An. stephensi | Human bait | 0.5 | 390.0 | 57.1 | [81] | |
| Myrtus communis | NI | NI | An. stephensi | Human bait | 0.5 | 390.0 | 42.8 | [81] | |
| Nepeta cataria | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Ocimum basilicum | Leaf | Hydrod. | An. stephensi | Cage test | 75.0 | [36] | |||
| Ocimum basilicum | Leaf | Steamd. | An. dirus | Human bait | 0.2 | 96.0 | 96.0 | [84] | |
| Ocimum basilicum | NI | NI | An. stephensi | Human bait | 0.5 | 210.0 | 66.7 | [81] | |
| Olea europaea | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 71.4 | [81] | |
| Pelargonium graveolens | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 61.9 | [81] | |
| Picea excelsa | NI | NI | An. stephensi | Human bait | 0.5 | 180.0 | 19.0 | [81] | |
| Pimpinella anisum | Seed | Hydrod. | An. stephensi | Cage test | 154.1 | [36] | |||
| Piper nigrum | NI | NI | An. stephensi | Human bait | 0.5 | 180.0 | 61.9 | [81] | |
| Rosmarinus officinalis | Shoot | Hydrod. | An. stephensi | Cage test | 38.9 | [36] | |||
| Rosmarinus officinalis | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Salvia sclarea | NI | NI | An. stephensi | Human bait | 0.5 | 300.0 | 19.0 | [81] | |
| Santalum album | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Syzygium aromaticum | Flower | Steamd. | An. dirus | Human bait | 0.2 | 96.0 | 98.0 | [84] | |
| Tagetes minuta | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] | |
| Thymus serpyllum | NI | NI | An. stephensi | Human bait | 0.5 | 450.0 | 33.3 | [81] | |
| Viola odorata | NI | NI | An. stephensi | Human bait | 0.5 | 480.0 | 100.0 | [81] |
| Plant Species | Essential Oil Major Components (%) | Mono Hydro | Mono Oxy | Sesqui Hydro | Sesqui Oxy | Mosquito Species | Larvicidal LC50 (mg/L) | Adulcidal LC50 (mg/L) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Ocimum basilicum | Linalool (52.4), Methyl eugenol (18.7) | 9.9 | 79.8 | 13.4 | An. subpictus | 9.7 | [56] | ||
| Blumea densiflora | Borneol (11.4), germacrene D. (8.66), β-caryophyllene (6.6), γ-terpinene (4.3), sabinene (4.3), and β-bisabolene (4.2) | 25.9 | 25.2 | 29.3 | 10.7 | An. anthropophagus | 10.0 | [42] | |
| Tagetes patula | Limonene (13.60), terpinolene (11.2), Z-β-ocimene (8.3), E-caryophyllene (8.0), piperitone (6.1), p-cymen-8-ol (5.4), piperitenone (4.9) | 40.6 | 21.2 | 9.3 | 3.1 | An. stephensi | 12.1 | [66] | |
| Ferulago carduchorum | (Z)-β-ocimene (43.3), α-pinene (18.2), bornyl acetate (3.9) | 78.6 | 5.9 | 5.9 | 0.7 | An. stephensi | 12.8 | [53] | |
| Chloroxylon swietenia | Geijerene (26.9), limonene (15.2), germacrene D (10.6), pregeijerene (7.8) | 22.9 | 2.7 | 53.9 | 3.0 | An. stephensi | 14.9 | [77] | |
| Feronia limonia | Estragole (34.7), β-pinene (23.6), (Z)-caryophyllene (11.0), methyl (Z)-caryophyllene (11.0). | 32.5 | 46.9 | 13.0 | 7.2 | An. Stephensi | 15.0 | [52] | |
| Cymbopogon citratus | Geranial (39.3), neral (21.9), geraniol (15.6), myrcene (14.00) | 15.8 | 81.6 | 0.5 | An. gambiae | 18.0 | [38] | ||
| Chloroxylon swietenia | Limonene (12.9), geijerene (17.7), pregeijerene (9.92), germacrene D (8.84) | 21.1 | 5.47 | 41.5 | 8.6 | An. stephensi | 19.0 | [45] | |
| Bunium persicum | p-Cuminaldehyde (23.5), α-methylbenzenemethanol (14.6), camphor (13.5), γ-terpinene (13.1), β-cymene (8.5) | 33.3 | 53.7 | 5.8 | 1.5 | An. stephensi | 27.7 | [43] | |
| Plectranthus amboinicus | Carvacrol (28.6), thymol (21.7), α-humulene (9.7), undecanal (8.3) | 15.1 | 64.2 | 11.7 | 8.6 | An. stephensi | 28.4 | [62] | |
| Citrus aurantium | d,l-limonene(94.8) | 97.4 | 0.7 | 0.1 | 0.06 | A. stephensi | 31.2 | [47] | |
| Plectranthus mollis | Piperitone oxide (23.7), fenchone (19.2), piperitenone oxide (13.0), β-caryophyllene (10.3) | 5.6 | 60.2 | 18.3 | 0.9 | An. stephensi | 33.5 | [55] | |
| Cymbopogon citratus | Myrcene (11.4), neral (30.2), geranial (32.8) | 14.1 | 74.5 | 1.3 | 0.1 | An. funestus | 34.6 | [51] | |
| Piper capense | δ-Cadinene (16.8), β-pinene (7.2), β-bisabolene (5.6), α-phellandrene (4.7), myristicin (4.), α-pinene (3.9), sabinene (3.8), β-cubebene (3.3), bicyclogermacrene (3.3), limonene (3.1), β-phellandrene (2.5), linalool (2.4), spathulenol (2.4) | 30.6 | 4.3 | 43.9 | 6.1 | An. gambiae | 34.9 | [60] | |
| Zingiber nimmoni | β-Caryophyllene (26.9), α-humulene (19.6), α-cadinol (5.20), myrcene (5.10) | 19.7 | 9.5 | 51.9 | 16.2 | An. stephensi | 41.2 | [59] | |
| Amomum subulatum | 1.8-cineole (39.8), α-terpineol (11.5), β-pinene (4.2), terpinen-4-ol (3.9) | 50.7 | 23.7 | 9.0 | 11.0 | An. subpictus | 41.2 | [40] | |
| Mentha spicata | carvone (48.6), cis-carveol (21.3), limonene (11.3) | 11.3 | 83.2 | 3.2 | 0.7 | An. stephensi | 49.7 | [87] | |
| Artemisia gilvescens | Camphor (13.5), eucalyptol (12.1), terpine-4-ol (9.6), germacrene D (8.6) | 10.1 | 53.9 | 18.9 | 9.0 | An. anthropophagus | 49.9 | [42] | |
| Plectranthus amboinicus | Carvacrol (29.2), thymol (21.7), α-humulene (9.7), undecanal (8.3) | 14.9 | 64.2 | 9.7 | 7.9 | An. gambiae | 55.2 | [61] | |
| Murraya exotica | β-Humulene (40.6), benzyl benzoate (23.9) | 5.66 | 1.0 | 61.5 | 6.01 | An. stephensi | 56.3 | [58] | |
| Zanthoxylum armatum | Linalool (57.0), limonene (19.8), E-methyl cinnamate (5.7) | 21.7 | 75.0 | An. stephensi | 58.0 | [69] | |||
| Coleus aromaticus | thymol (82.6) | 2.8 | 88.7 | 3.2 | 1.3 | An. subpictus | 60.3 | [48] | |
| Zhumeria majdae | Linalool (31.2), camphor (38.5) | 18.9 | 77.4 | 0.6 | 0.7 | An. stephensi | 61.3 | [70] | |
| Lavandula gibsoni | Thymol (10.4), α-terpinolene (22.2) | 5.6 | 60.2 | 18.3 | 0.9 | An. stephensi | 62.8 | [55] | |
| Origanum vulgare | Carvacrol (38.3), terpiene-4-ol (28.7) | 7.7 | 80.6 | 5.2 | 3.7 | An. stephensi | 67.0 | [88] | |
| Cryptomeria japonica | Kau-16-rene (23.3), β-elemol (18.3) | 28.9 | 6.5 | 0.4 | 39.0 | An. gambiae | 67.1 | [49] | |
| Origanum vulgare | Carvacrol (38.30), terpiene-4-ol (28.7) | 7.7 | 80.6 | 5.2 | 3.7 | An. subpictus | 74.1 | [88] | |
| Trachyspermum ammi | Thymol (66.7), p-cymene (17.4), γ-terpenene (10.1) | 29.8 | 67.9 | An. stephensi | 80.8 | [68] | |||
| Mentha spicata | Piperitenone oxide (71.1), carvone (5.8), β-caryophyllene (2.3), limonene (1.3) | 1.4 | 76.9 | 2.3 | An. stephensi | 82.9 | [57] | ||
| Syzygium zeylanicum | α-Humulene (37.8), β-elemene (10.7) | 1.2 | 0.9 | 64.6 | 24.9 | An. subpictus | 83.1 | [65] | |
| Plectranthus barbatus | Eugenol (31.1), α-pinene (19.4), β-caryophyllene (18.4). | 25.2 | 31.1 | 38.9 | 2.6 | An. subpictus | 84.2 | [89] | |
| Ocimum canum | 1.8-Cineole (29.4), linalool (19.1), Perpinen-4-ol (7.5) | 10.4 | 35.2 | 8.4 | An. funestus | 91.2 | [51] | ||
| Eucalyptus tereticornis | p-Cymene (16.7), caryophyllene oxide (14.2), spathulenol (13.5). cryptone (11.4) | 23.9 | 20.0 | 2.0 | 34.7 | An. gambiae | 0.1 | [77] | |
| Cymbopogon citratus | Neral (33.1), geranial (44.3) | 12.9 | 81.1 | 0.2 | 0.5 | An. gambiae | 0.2 | [77] | |
| Chenopodium ambrosioides | Ascaridole (41.9), α-terpinene (16.5), p-cymene (14.4), isoascridole (7.5) | 31.6 | 51.5 | An.gambiae | 1.0 | [44] | |||
| Cymbopogon schoenanthus | piperitone (58.9), δ-2-carene (15.5) | 19.8 | 63.5 | 2.3 | 12.8 | An. gambiae | 1.6 | [77] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gnankiné, O.; Bassolé, I.H.N. Essential Oils as an Alternative to Pyrethroids’ Resistance against Anopheles Species Complex Giles (Diptera: Culicidae). Molecules 2017, 22, 1321. https://doi.org/10.3390/molecules22101321
Gnankiné O, Bassolé IHN. Essential Oils as an Alternative to Pyrethroids’ Resistance against Anopheles Species Complex Giles (Diptera: Culicidae). Molecules. 2017; 22(10):1321. https://doi.org/10.3390/molecules22101321
Chicago/Turabian StyleGnankiné, Olivier, and Imaël Henri Nestor Bassolé. 2017. "Essential Oils as an Alternative to Pyrethroids’ Resistance against Anopheles Species Complex Giles (Diptera: Culicidae)" Molecules 22, no. 10: 1321. https://doi.org/10.3390/molecules22101321




