HPLC-DAD-ESI-MS Analysis of Flavonoids from Leaves of Different Cultivars of Sweet Osmanthus
Abstract
:1. Introduction
2. Results
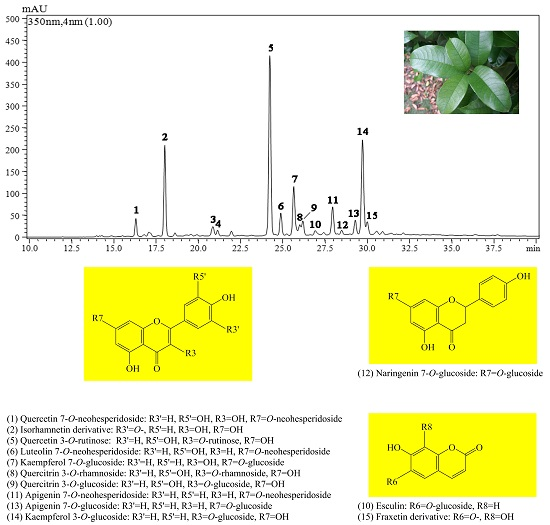
2.1. Qualitative Analysis of Leaf Flavonoids of O. fragrans
2.2. Component Analysis of Leaf Samples of O. fragrans
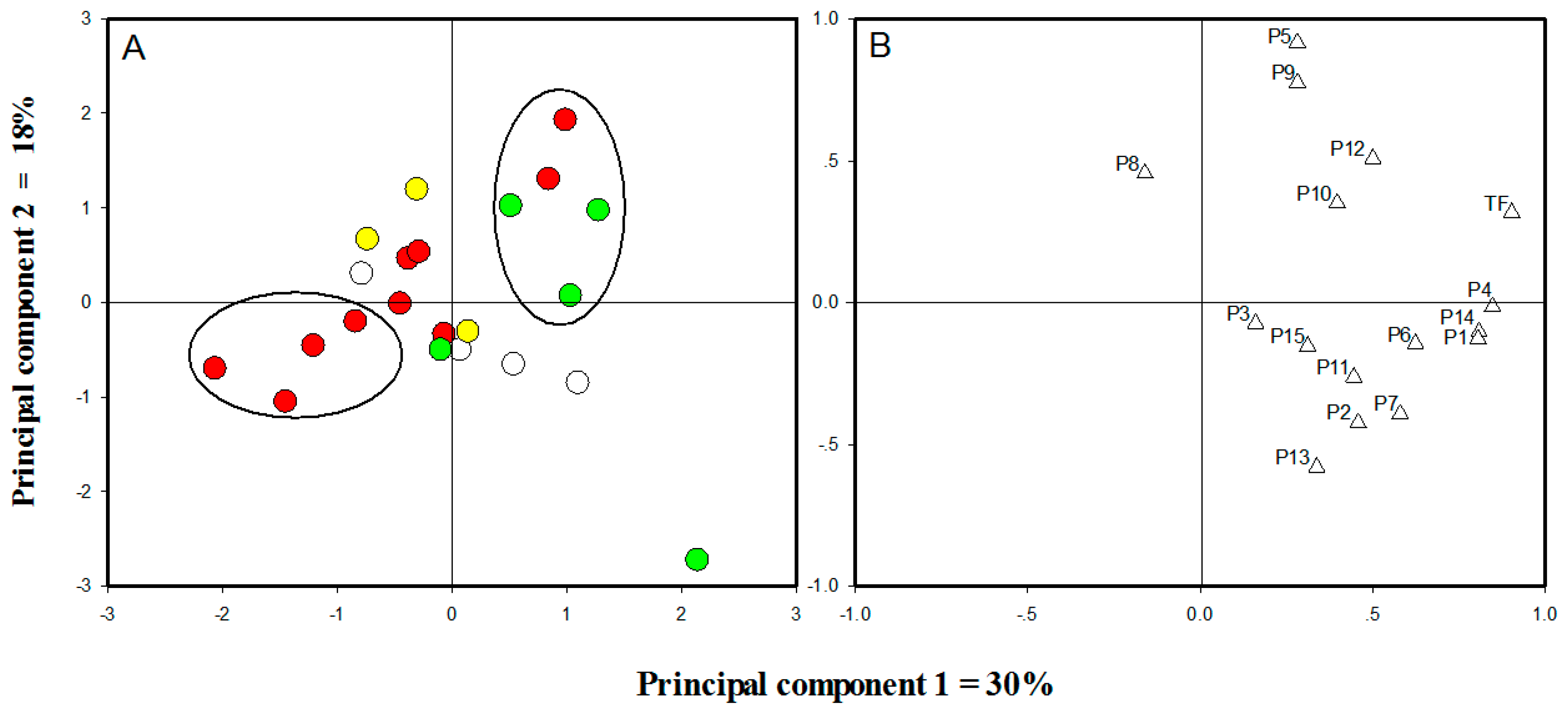
2.3. Cluster Analysis of Different Cultivars
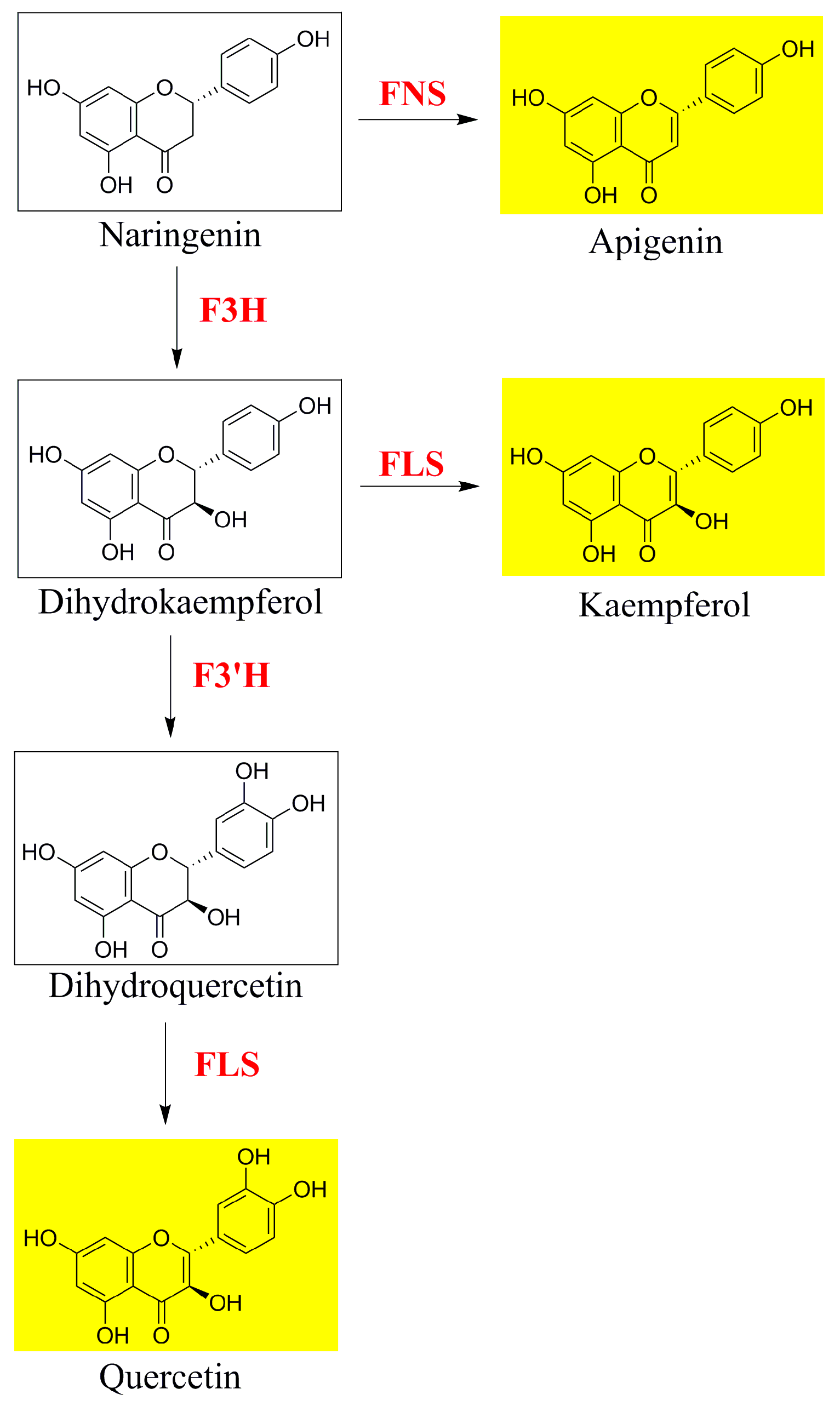
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Chemicals and Reagents
4.3. Extraction of Flavonoids
4.4. Quantitative Determination of Flavonoids
4.5. Identification of Flavonoids
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xiang, Q.; Liu, Y. An Illustrated Monograph of the Sweet Osmanthus Variety in China; Zhejiang Science & Technology Press: Hangzhou, China, 2007; pp. 86–89. [Google Scholar]
- Chen, G.L.; Chen, S.G.; Xie, Y.Q.; Chen, F.; Zhao, Y.Y.; Luo, C.X.; Gao, Y.Q. Total phenolic, flavonoid and antioxidant activity of 23 edible flowers subjected to in vitro digestion. J. Funct. Foods 2015, 17, 243–259. [Google Scholar] [CrossRef]
- Hung, C.Y.; Tsai, Y.C.; Li, K.Y. Phenolic antioxidants isolated from the flowers of Osmanthus fragrans. Molecules 2012, 17, 10724–10737. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.H.; Tsai, T.H.; Chien, Y.C.; Lee, C.W.; Tsai, P.J. In vitro antimicrobial activities against cariogenic streptococci and their antioxidant capacities: A comparative study of green tea versus different herbs. Food Chem. 2008, 110, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Lin, C.T.; Yang, L.L. Neuroprotection and free radical scavenging effects of Osmanthus fragrans. J. Biomed. Sci. 2007, 14, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.C.; Chang, L.H.; Chen, S.H.; Fan, N.C.; Ho, J.A.A. Antioxidant activity and melanogenesis inhibitory effect of the acetonic extract of Osmanthus fragrans: A potential natural and functional food flavor additive. LWT-Food Sci. Technol. 2009, 42, 1513–1519. [Google Scholar] [CrossRef]
- Lee, D.G.; Lee, S.M.; Bang, M.H.; Park, H.J.; Lee, T.H.; Kim, Y.H.; Kim, J.Y.; Baek, N.I. Lignans from the flowers of Osmanthus fragrans var. aurantiacus and their inhibition effect on NO production. Arch. Pharm. Res. 2011, 34, 2029–2035. [Google Scholar] [PubMed]
- Yang, R.; Ouyang, X.; Gan, D.; Wang, H.; Li, Y.; Pan, Y. Isolation and determination of iridoid glycosides from the seeds of Osmanthus fragrans by HPLC. Anal. Lett. 2013, 120, 745–752. [Google Scholar] [CrossRef]
- Ouyang, X.L.; Wei, L.X.; Wang, H.S.; Pan, Y.M. Antioxidant activity and phytochemical composition of Osmanthus fragrans’ pulps. S. Afr. J. Bot. 2015, 98, 162–166. [Google Scholar] [CrossRef]
- Tang, W.; Cao, J.; Zhang, X.; Zhao, Y. Osmanthus fragrans seeds, a source of secoiridoid glucosides and its antioxidizing and novel platelet-aggregation inhibiting function. J. Funct. Foods 2015, 14, 337–344. [Google Scholar] [CrossRef]
- Asakura, K. Newly Revised Wakan-Yaku; Ishiyaku: Tokyo, Japan, 1980; p. 259. [Google Scholar]
- Sugiyama, M.; Kikuchi, M. Phenolic glycosides from Osmanthus asiaticus. Phytochemistry 1991, 30, 3147–3149. [Google Scholar] [CrossRef]
- Sugiyama, M.; Kikuchi, M. Studies on the constituents of Osmanthus Species. VII. structures of lignan glycosides from the leaves of Osmanthus asiaticus Nakai. Chem. Pharm. Bull. 1991, 39, 483–485. [Google Scholar] [CrossRef]
- Sugiyama, M.; Kikuchi, M. Studies on the consituents of Osmanthus species. VI. structures of phenylpropanoid glycosides from the leaves of Osmanthus asiaticus Nakai. Pharma. Bull. 1990, 38, 2953–2955. [Google Scholar] [CrossRef]
- Sugiyama, M.; Kikuchi, M. Phenylethanoid glycosides from Osmanthus asiaticus. Phytochemistry 1993, 32, 1553–1555. [Google Scholar] [CrossRef]
- Kikuchi, M. Studies on the constituents of Osmanthus species. I. On the components of the leaves of Osmanthus fragrans LOUR. var. aurantiacus MAKINO (1). J. Pharm. Soc. Jpn. 1984, 104, 535–539. [Google Scholar]
- Kikuchi, M.; Yamauchi, Y. Studies on the constituents of Osmanthus Species. III. On the components of the leaves of Osmanthus ilicifolius (HASSK.) Mouillefert. J. Pharm. Soc. Jpn. 1985, 105, 442–448. [Google Scholar]
- Benkrief, R.; Ranarivelo, Y.; Skaltsounis, A.L.; Tillequin, F.; Koch, M.; Pusset, J.; Sévenet, T. Monoterpene alkaloids, iridoids and phenylpropanoid glycosides from Osmanthus austrocaledonica. Phytochemistry 1998, 47, 825–832. [Google Scholar] [CrossRef]
- Machida, K.; Sakamoto, S.; Kikuchi, M. Two new neolignan glycosides from leaves of Osmanthus heterophyllus. J. Nat. Med. 2009, 63, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, S.; Machida, K.; Kikuchi, M. Studies on the constituents of Osmanthus species. XVII. On the chemical constituents from the leaves of Osmanthus ilicifolius. J. Tohoku Pharm. Univ. 2007, 54, 63–67. [Google Scholar]
- Escriche, I.; Kadar, M.; Juan-Borrás, M.; Domenech, E. Suitability of antioxidant capacity, flavonoids and phenolic acids for floral authentication of honey. Impact of industrial thermal treatment. Food Chem. 2014, 142, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Deng, Z.; Liu, R.; Loewen, S.; Tsao, R. Bioaccessibility, in vitro antioxidant activities and in vivo anti-inflammatory activities of a purple tomato (Solanum lycopersicum L.). Food Chem. 2014, 159, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Li, S.; Wang, W.; Hong, Y.; Tang, K.; Luo, Q. Screening and identification of the antibacterial bioactive compounds from Lonicera japonica Thunb. leaves. Food Chem. 2013, 138, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Xian, X.; Chen, X.; Xiao, J.; Wang, Q. Characterization of flavonoids from Dryopteris erythrosora and evaluation of their antioxidant, anticancer and acetylcholinesterase inhibition activities. Food Chem. Toxicol. 2012, 51, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Marchand, L.L. Cancer preventive effects of flavonoids—A review. Biomed. Pharmacother. 2002, 56, 296–301. [Google Scholar] [CrossRef]
- Peng, Z.F.; Strack, D.; Baumert, A.; Subramaniam, R.; Goh, N.K.; Chia, T.F.; Tan, S.N.; Lian, S.C. Antioxidant flavonoids from leaves of Polygonum hydropiper L. Phytochemistry 2003, 62, 219–228. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, J.; Lu, X.; Zhang, L.; Zhang, Y. Evaluation to the antioxidant activity of total flavonoids extract from persimmon (Diospyros kaki L.) leaves. Food Chem. Toxicol. 2011, 49, 2689–2696. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.X.; Yao, Z.R.; Li, J.; Tu, P.F. Flavonoids from the leaves of Ilex cornuta. Chin. J. Nat. Med. 2012, 10, 84–87. [Google Scholar] [CrossRef]
- Liu, G.; Rajesh, N.; Wang, X.; Zhang, M.; Wu, Q.; Li, S.; Chen, B.; Yao, S. Identification of flavonoids in the stems and leaves of Scutellaria baicalensis Georgi. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B. Plant polyphenols. VL The flavonol glycosides of wild and cultivated potatoes. Biochem. J. 1962, 84, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.L.; Cuyckens, F.; Heuvel, H.V.D.; Claeys, M. Mass spectrometric methods for the characterisation and differentiation of isomeric O-diglycosyl flavonoids. Phytochem. Anal. 2001, 12, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Kashiwada, Y.; Aoshima, A.; Ikeshiro, Y.; Chen, Y.P.; Furukawa, H.; Itoigawa, M.; Fujioka, T.; Mihashi, K.; Cosentino, L.M.; Morris-Natschke, S.L.; et al. Anti-HIV benzylisoquinoline alkaloids and flavonoids from the leaves of Nelumbo nucifera, and structure-activity correlations with related alkaloids. Bioorg. Med. Chem. 2005, 36, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Tattini, M.; Ferdinando, M.D.; Brunetti, C.; Goti, A.; Pollastri, S.; Bellasio, C.; Giordano, C.; Fini, A.; Agati, G. Esculetin and esculin (esculetin 6-O-glucoside) occur as inclusions and are differentially distributed in the vacuole of palisade cells in Fraxinus ornus leaves: A fluorescence microscopy analysis. J. Photochem. Photobiol. B Biol. 2014, 140, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Stanić, G.; Juričić, B.; Brkić, D. HPLC analysis of esculin and fraxin in horse-chestnut bark (Aesculus hippocastanum L.). Croat. Chem. Acta 1999, 72, 827–834. [Google Scholar]
- Chen, S.; Wu, B.H.; Fang, J.B.; Liu, Y.L.; Zhang, H.H.; Fang, L.C.; Guan, L.; Li, S.H. Analysis of flavonoids from lotus (Nelumbo nucifera) leaves using high performance liquid chromatography/photodiode array detector tandem electrospray ionization mass spectrometry and an extraction method optimized by orthogonal design. J. Chromatogr. A 2012, 1227, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Sasaki, N.; Ohmiya, A. Biosynthesis of plant pigments: Anthocyanins, betalains and carotenoids. Plant J. 2008, 54, 733–749. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B. The Biochemical Systematics of Flavonoids; Springer: New York, NY, USA, 1975; pp. 1056–1095. [Google Scholar]
- Harborne, J.B.; Green, P.S. A chemotaxonomic survey of flavonoids in leaves of the Oleaceae. Bot. J. Linn. Soc. 1980, 81, 155–167. [Google Scholar] [CrossRef]
- Lee, H.S.; Park, S.H.; Wallander, E.; Chang, C.S. A flavonoid survey of Fraxinus (Oleaceae) in eastern Asia, and the overlooked species Fraxinus hopeiensis T. Tang in northern China. Biochem. Syst. Ecol. 2012, 41, 150–156. [Google Scholar] [CrossRef]
- Yang, K. Chinese Osmanthus; China Forestry Publishing: Beijing, China, 2013; pp. 90–108. [Google Scholar]
- Han, Y.; Wang, X.; Chen, W.; Dong, M.; Yuan, W.; Liu, X.; Shang, F. Differential expression of carotenoid-related genes determines diversified carotenoid coloration in flower petal of Osmanthus fragrans. Tree Genet. Genomes 2014, 10, 329–338. [Google Scholar] [CrossRef]
- Qiu, Y.; Hu, S.; Chen, Y.; Chen, X.; Wu, G. Studies on cultivar classification of Osmanthus fragrans by ISSR-PCR analysis. Acta Hortic. Sin. 2004, 31, 529–532. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
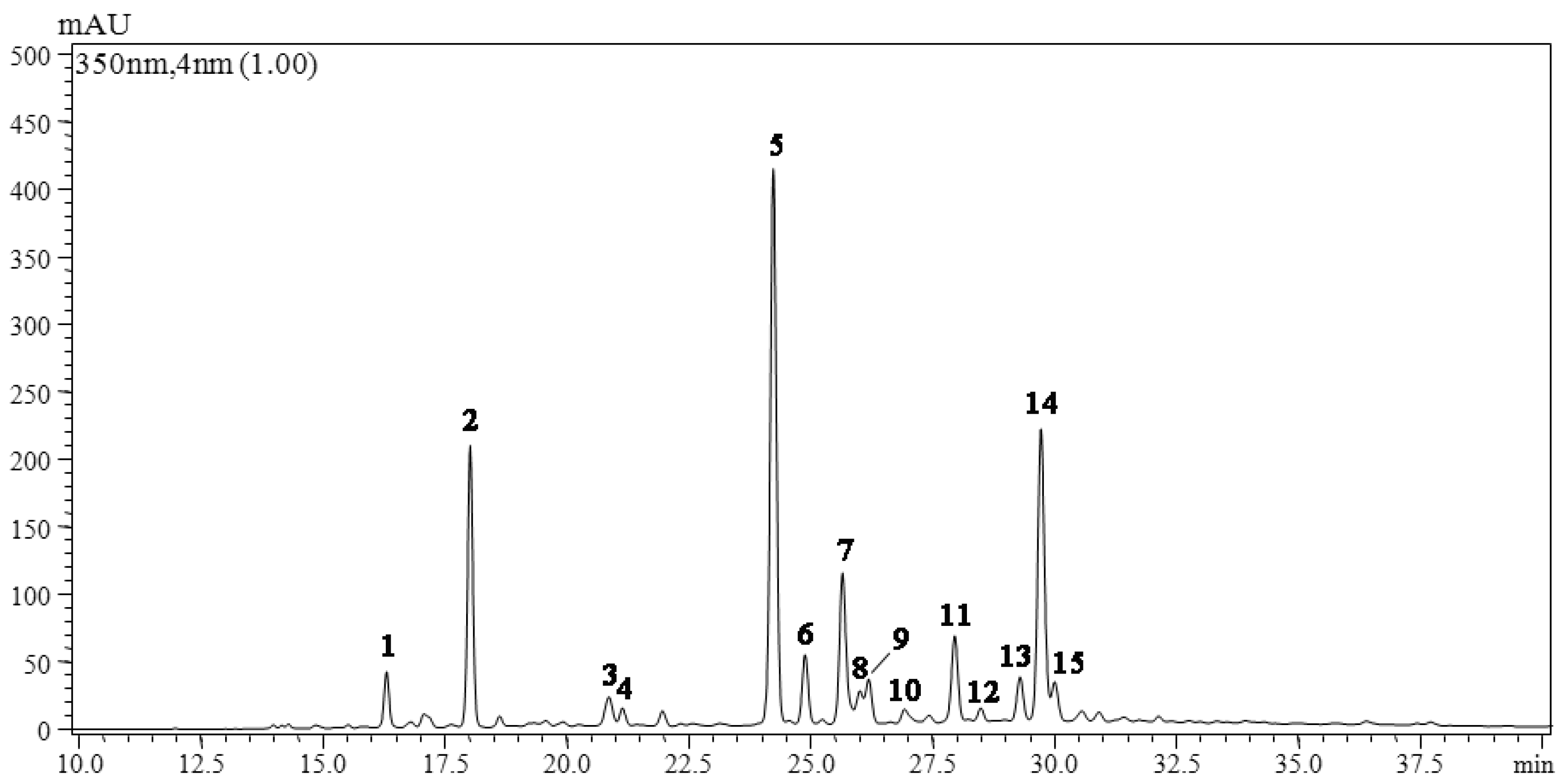




| Peaks a | tR (min) | λmax (nm) | ESI-PI MS/MS2 (m/z) | Tentative Identification |
|---|---|---|---|---|
| 1 | 16.26 | 272, 326 | 611.1[M + H]+, 427.1, 303[Y0+] | Quercetin 7-O-neohesperidoside |
| 2 | 17.93 | 270, 334 | 595.1[M + H]+, 457.1, 385.1, 317.0[Y0+] | Isorhamnetin derivative |
| 3 | 20.78 | 238, 325 | 564.3[M + H]+, 410.1 | No tentative identification |
| 4 | 20.99 | 289, 332 | 449.1[M + H]+, 265.0, 211.0 | No tentative identification |
| 5 | 24.02 | 252, 334 | 611.1[M + H]+, 465.0, 303[Y0+] | Quercetin 3-O-rutinose |
| 6 | 24.66 | 253, 345 | 595.1[M + H]+, 448.8 (82) [Y1+], 433 (4), 287.1[Y0+] (100) | Luteolin 7-O-neohesperidoside |
| 7 | 25.43 | 266, 345 | 448.8[M + H]+, 287.1[Y0+] | Kaempferol 7-O-glucoside |
| 8 | 25.80 | 284, 330 | 449.1[M + H]+, 303[Y0+] | Quercitrin 3-O-rhamnoside |
| 9 | 25.94 | 281, 329 | 465.0[M + H]+, 303.1[Y0+] | Quercitrin 3-O-glucoside |
| 10 | 26.68 | 265, 332 | 341.1[M + H]+, 209.0[Y0+] | Esculin |
| 11 | 27.69 | 266, 334 | 578.9[M + H]+, 432.8(65) [Y1+], 271.1[Y0+](100) | Apigenin 7-O-neohesperidoside |
| 12 | 28.23 | 284, 332 | 435.2[M + H]+, 273.2[Y0+] | Naringenin 7-O-glucoside |
| 13 | 29.04 | 267, 333 | 432.8[M + H]+, 271.1[Y0+] | Apigenin 7-O-glucoside |
| 14 | 29.46 | 268, 335 | 448.9[M + H]+, 287.1[Y0+] | Kaempferol 3-O-glucoside |
| 15 | 29.74 | 268, 324 | 435.1[M + H]+, 322.1, 209.0[Y0+] | Fraxetin derivative |
| Sample a No. | Contents (mg/g, DW) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 b | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | TF | |
| C1 | 0.59 | 6.11 | 1.03 | 0.33 | 9.04 | 1.96 | 3.25 | 0.84 | 0.77 | 1.03 | 4.06 | 0.67 | 2.33 | 5.73 | 2.01 | 39.75 |
| C2 | 0.71 | 4.25 | 0.63 | 0.36 | 7.94 | 2.37 | 2.86 | 0.45 | 0.76 | 0.82 | 3.25 | 0.58 | 0.91 | 6.40 | 1.24 | 33.52 |
| C3 | 0.55 | 5.02 | 0.51 | 0.26 | 8.02 | 1.43 | 3.30 | 0.94 | 0.77 | 0.81 | 2.98 | 0.70 | 1.78 | 4.53 | 1.35 | 32.93 |
| C4 | 0.49 | 4.20 | 0.98 | 0.23 | 9.54 | 0.40 | 1.42 | 0.46 | 1.85 | 0.84 | 2.87 | 0.52 | 1.17 | 1.79 | 1.16 | 27.91 |
| C5 | 0.35 | 2.29 | 0.46 | 0.19 | 14.86 | 1.06 | 2.75 | 1.10 | 1.96 | 0.55 | 1.93 | 0.81 | 2.42 | 4.18 | 1.45 | 36.01 |
| C6 | 0.85 | 4.11 | 0.76 | 0.39 | 9.70 | 1.32 | 2.99 | 0.75 | 0.92 | 0.63 | 1.89 | 0.46 | 1.09 | 5.70 | 0.99 | 32.54 |
| C7 | 0.39 | 2.04 | 0.32 | 0.22 | 12.27 | 0.97 | 2.35 | 1.30 | 0.82 | 0.68 | 2.23 | 0.72 | 1.34 | 4.38 | 1.17 | 31.19 |
| C8 | 0.38 | 2.25 | 0.73 | 0.17 | 9.67 | 1.38 | 3.96 | 1.07 | 0.74 | 1.12 | 1.77 | 0.52 | 2.14 | 5.34 | 0.95 | 32.18 |
| C9 | 0.36 | 2.49 | 0.35 | 0.19 | 9.53 | 1.14 | 2.98 | 0.75 | 1.51 | 0.85 | 2.13 | 0.81 | 1.81 | 5.05 | 1.44 | 30.89 |
| C10 | 0.33 | 2.44 | 0.59 | 0.16 | 4.41 | 1.18 | 3.88 | n.d. | 0.42 | 0.77 | 1.50 | 0.53 | 1.77 | 2.15 | 0.48 | 20.60 |
| C11 | 0.77 | 2.67 | 0.53 | 0.33 | 14.74 | 1.48 | 4.56 | 0.88 | 1.54 | 1.63 | 2.24 | 1.00 | 2.00 | 3.85 | 0.94 | 39.15 |
| C12 | 0.58 | 3.26 | 0.67 | 0.23 | 8.09 | 3.16 | 3.55 | 1.01 | 0.77 | 0.73 | 3.37 | 0.73 | 1.70 | 2.72 | 0.56 | 31.12 |
| C13 | 0.20 | 1.97 | 0.72 | 0.18 | 5.85 | 1.54 | 2.23 | 0.88 | 0.58 | 0.51 | 3.25 | 0.67 | 1.53 | 3.18 | 0.88 | 24.18 |
| C14 | 0.10 | 1.79 | 0.65 | 0.09 | 5.25 | 1.02 | 1.77 | 0.79 | 0.64 | 0.27 | 3.25 | 0.41 | 1.35 | 1.56 | 0.87 | 19.80 |
| C15 | 0.54 | 2.32 | 0.66 | 0.36 | 11.22 | 1.25 | 2.74 | 0.72 | 1.13 | 1.19 | 1.73 | 0.53 | 0.99 | 4.52 | 0.97 | 30.85 |
| C16 | 0.68 | 2.74 | 0.29 | 0.31 | 7.16 | 2.09 | 2.48 | 0.53 | 0.52 | 0.56 | 2.35 | 0.82 | 0.63 | 1.52 | 0.39 | 23.05 |
| C17 | 0.59 | 2.09 | 0.43 | 0.35 | 16.33 | 2.20 | 3.87 | 1.10 | 1.97 | 0.91 | 2.80 | 1.22 | 0.82 | 6.44 | 0.82 | 41.57 |
| C18 | 0.60 | 2.17 | 1.33 | 0.44 | 16.06 | 1.48 | 3.28 | n.d. | 2.09 | 0.53 | 2.32 | 0.75 | 0.97 | 5.33 | 0.67 | 38.01 |
| C19 | 0.93 | 4.12 | 0.68 | 0.53 | 2.08 | 2.53 | 7.34 | n.d. | 0.62 | 0.63 | 3.49 | 0.71 | 5.67 | 9.00 | 1.35 | 39.72 |
| C20 | 0.45 | 4.80 | 0.42 | 0.30 | 5.80 | 1.63 | 2.33 | n.d. | 0.91 | 0.88 | 3.46 | 0.96 | 1.04 | 3.94 | 0.98 | 27.88 |
| C21 | 0.66 | 4.03 | 0.80 | 0.50 | 12.66 | 3.27 | 1.69 | 0.97 | 1.48 | 1.31 | 4.43 | 0.93 | 0.56 | 5.24 | 0.87 | 39.38 |
| C22 | 0.91 | 2.74 | 0.36 | 0.56 | 12.19 | 2.48 | 3.71 | n.d. | 1.17 | 0.61 | 3.69 | 0.86 | 0.80 | 4.76 | 0.78 | 35.62 |
| Cultivar Groups | Sample No. | Cultivars |
|---|---|---|
| Albus Group | C1 | O. fragrans ‘Xiaoye Sugui’ |
| C2 | O. fragrans ‘Zaoyin Gui’ | |
| C3 | O. fragrans ‘Yu Linglong’ | |
| C4 | O. fragrans ‘Ziyin Gui’ | |
| Luteus Group | C5 | O. fragrans ‘Boye Jingui’ |
| C6 | O. fragrans ‘Hangzhou Huang’ | |
| C7 | O. fragrans ‘Jin Qiu’ | |
| Aurantiacus Group | C8 | O. fragrans ‘Ruanye Dangui’ |
| C9 | O. fragrans ‘Chilian Jindan’ | |
| C10 | O. fragrans ‘Mantiao Hong’ | |
| C11 | O. fragrans ‘Wuyi Dangui’ | |
| C12 | O. fragrans ‘Xionghuang’ | |
| C13 | O. fragrans ‘Zhuangyuan Hong’ | |
| C14 | O. fragrans ‘Chenghong Dangui’ | |
| C15 | O. fragrans ‘Zhusha Dangui’ | |
| C16 | O. fragrans ‘Yan Hong’ | |
| C17 | O. fragrans ‘Yingye Dangui’ | |
| Asiaticus Group | C18 | O. fragrans ‘Danzhuang’ |
| C19 | O. fragrans ‘Sijigui’ | |
| C20 | O. fragrans ‘Tiannu Sanhua’ | |
| C21 | O. fragrans ‘Foding Zhu’ | |
| C22 | O. fragrans ‘Tianxiang Taige’ |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Fu, J.; Zhang, C.; Zhao, H. HPLC-DAD-ESI-MS Analysis of Flavonoids from Leaves of Different Cultivars of Sweet Osmanthus. Molecules 2016, 21, 1224. https://doi.org/10.3390/molecules21091224
Wang Y, Fu J, Zhang C, Zhao H. HPLC-DAD-ESI-MS Analysis of Flavonoids from Leaves of Different Cultivars of Sweet Osmanthus. Molecules. 2016; 21(9):1224. https://doi.org/10.3390/molecules21091224
Chicago/Turabian StyleWang, Yiguang, Jianxin Fu, Chao Zhang, and Hongbo Zhao. 2016. "HPLC-DAD-ESI-MS Analysis of Flavonoids from Leaves of Different Cultivars of Sweet Osmanthus" Molecules 21, no. 9: 1224. https://doi.org/10.3390/molecules21091224