Characterization of Antifungal Natural Products Isolated from Endophytic Fungi of Finger Millet (Eleusine coracana)
Abstract
:1. Introduction
2. Results
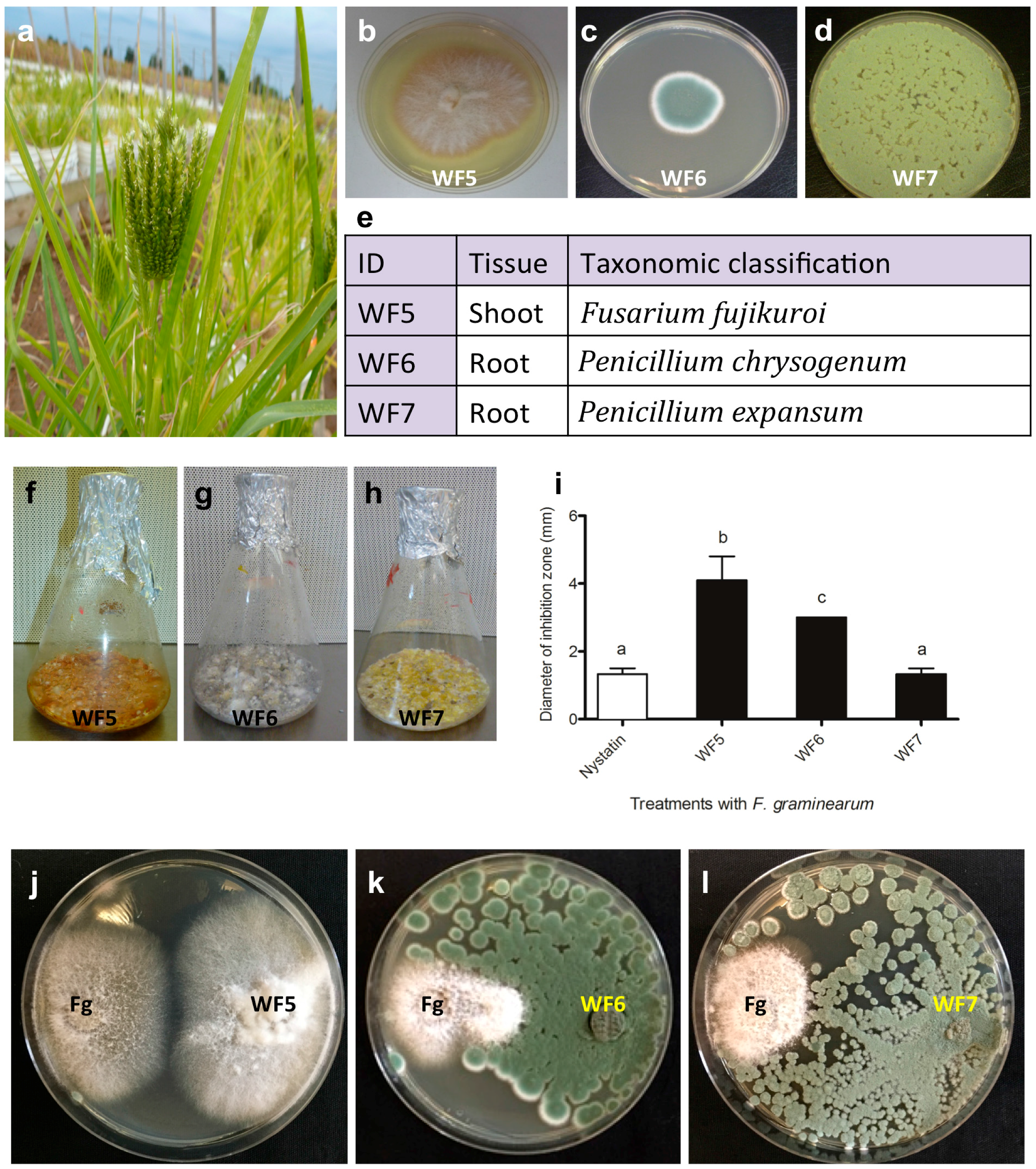
2.1. Confirming the Endophytic Behaviour of the Putative Fungal Endophytes
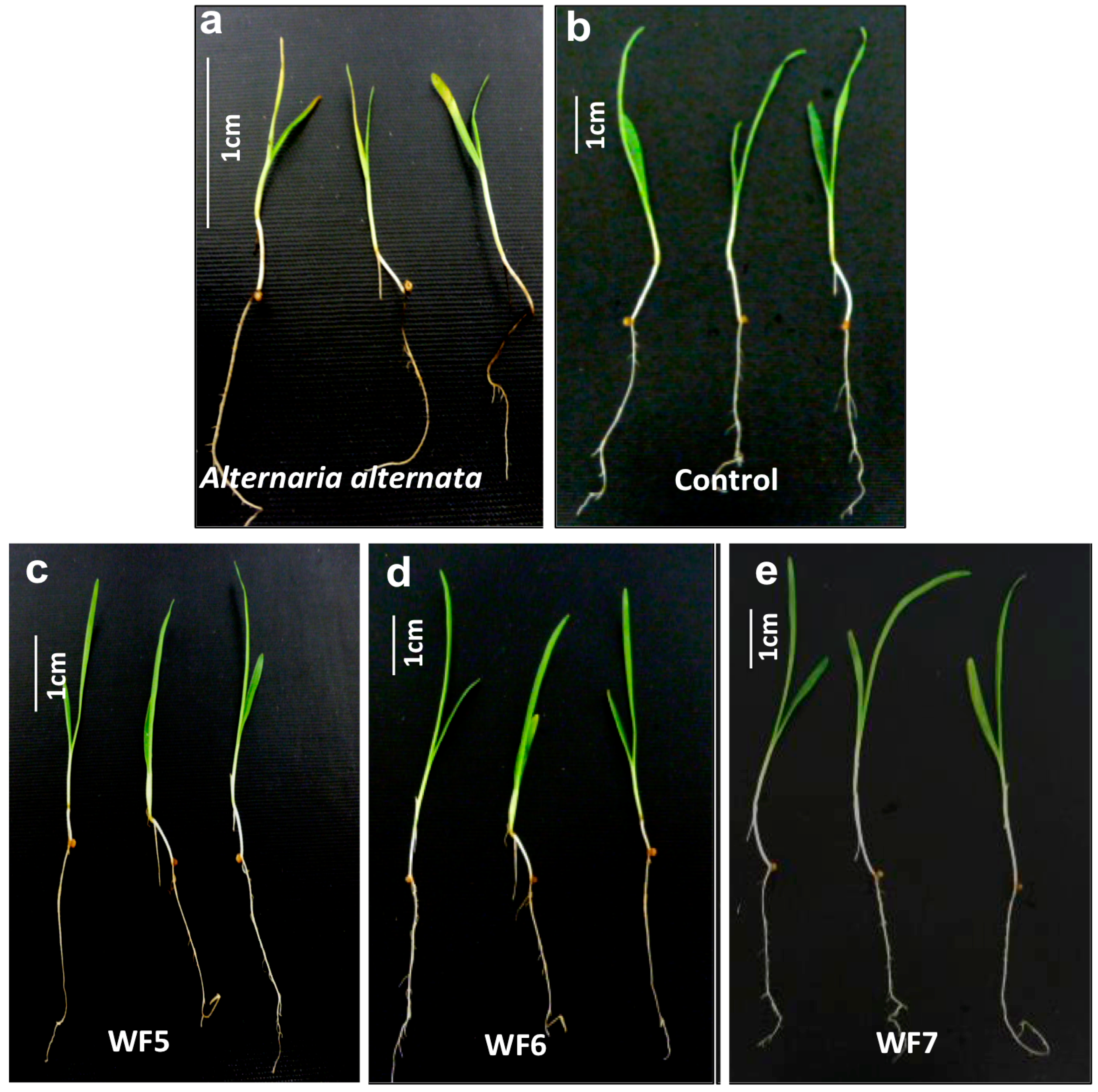
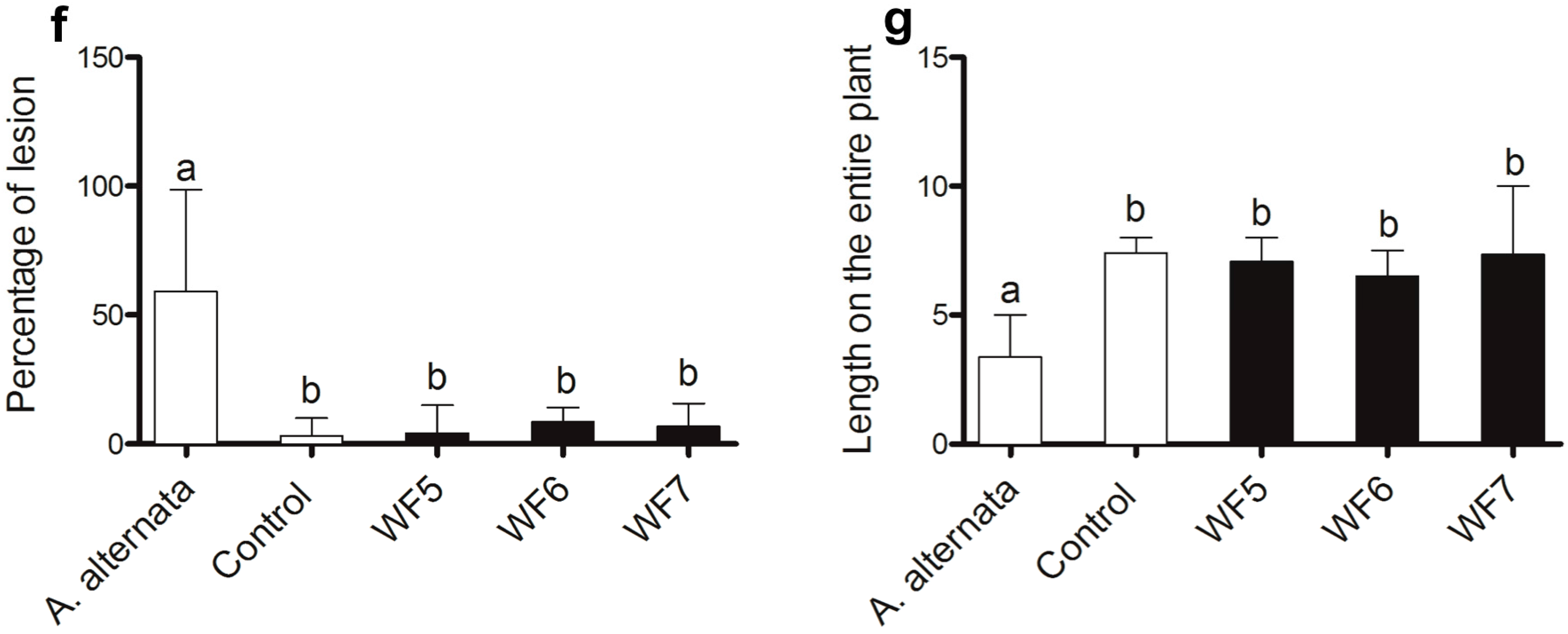
2.1.1. Plant Pathogenicity Assay
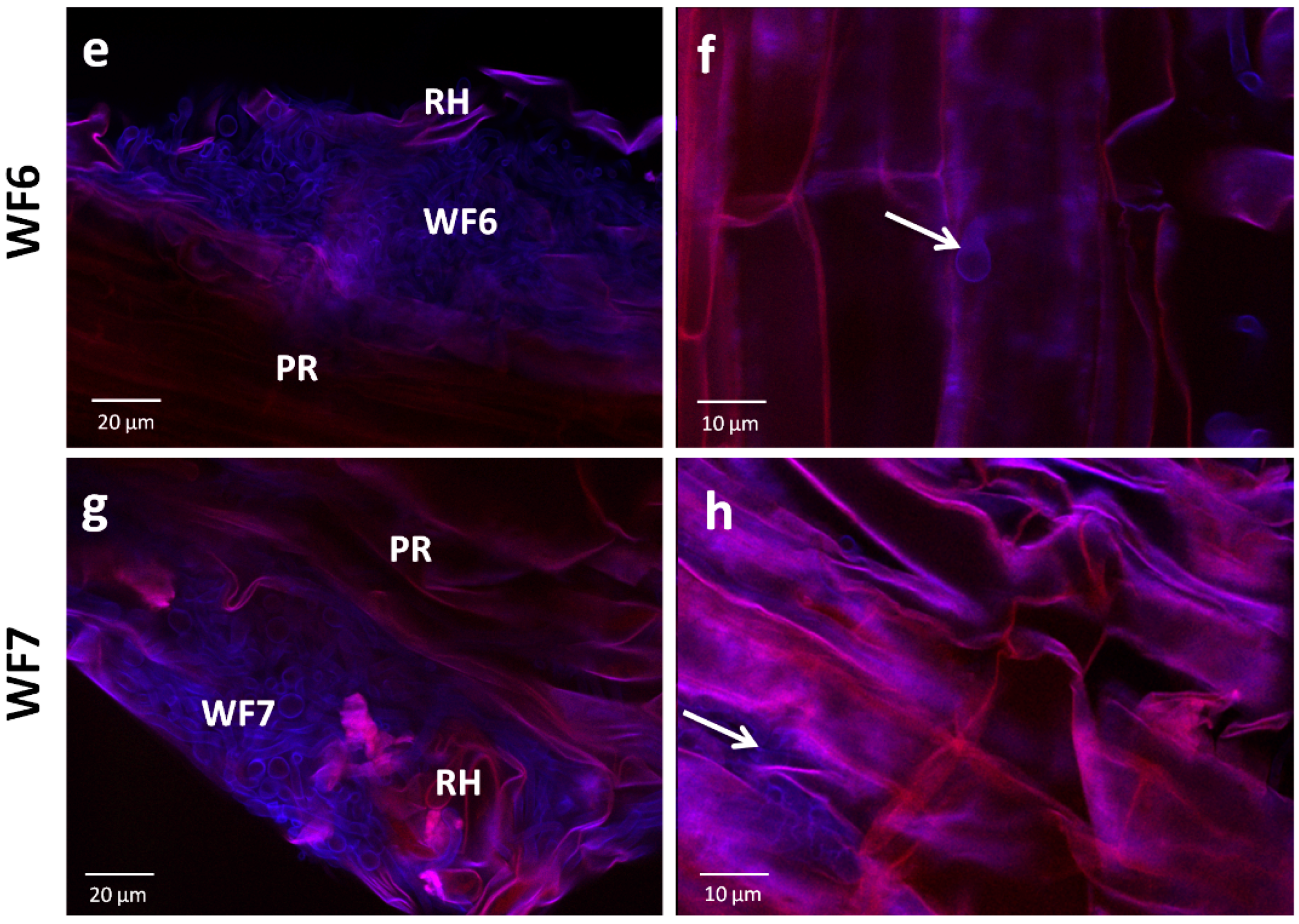
2.1.2. Root Colonization Assay
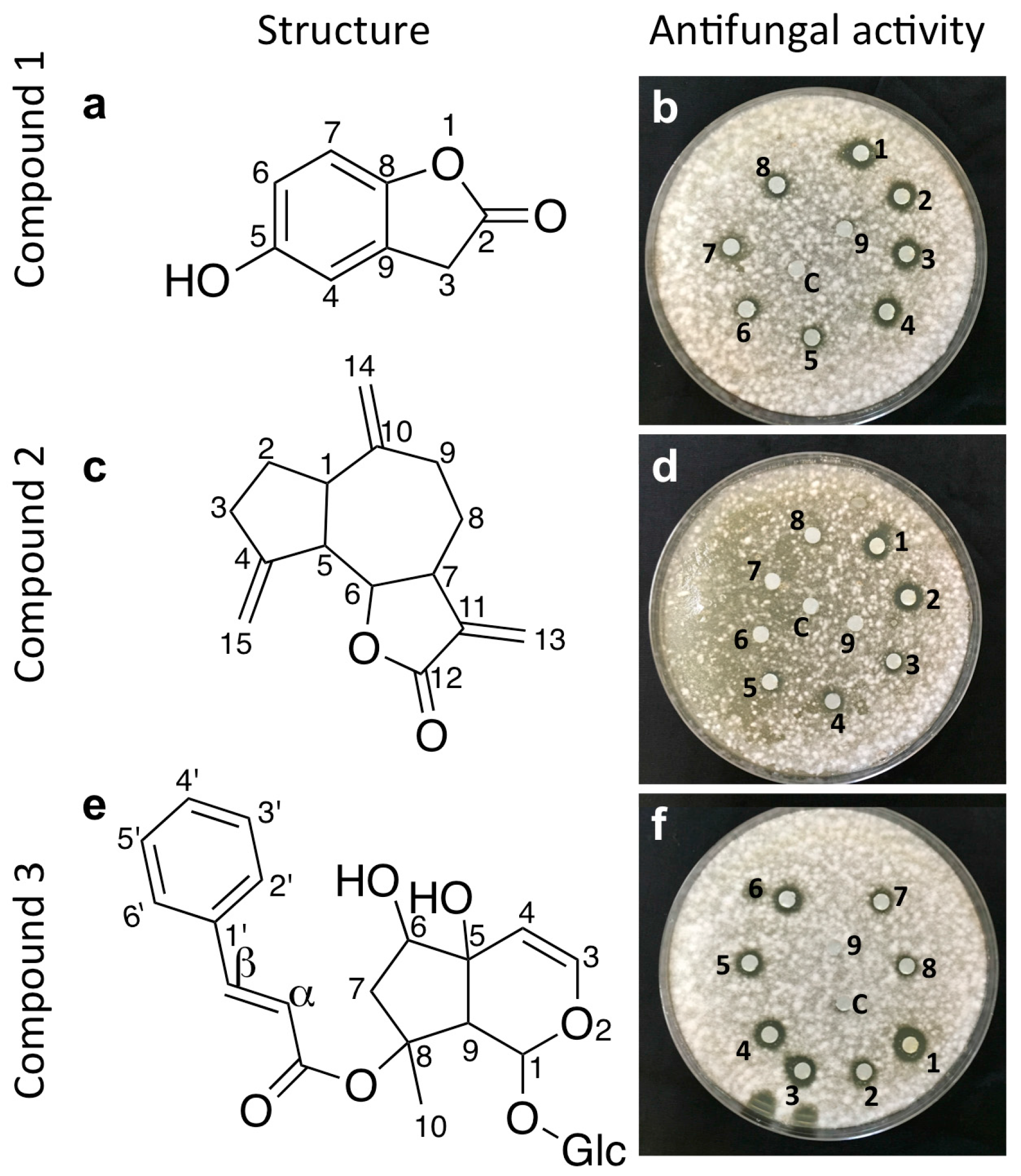
2.2. Bio-Assay Guided Purification and Structural Elucidation of Anti-Fusarium Compounds
2.3. Structural Elucidation of the Isolated Antifungal Compounds
2.3.1. Compound 1 (Isolated from Fungus WF5)
2.3.2. Compound 2 (Isolated from Fungus WF6)
2.3.3. Compound 3 (Isolated from Fungus WF7)
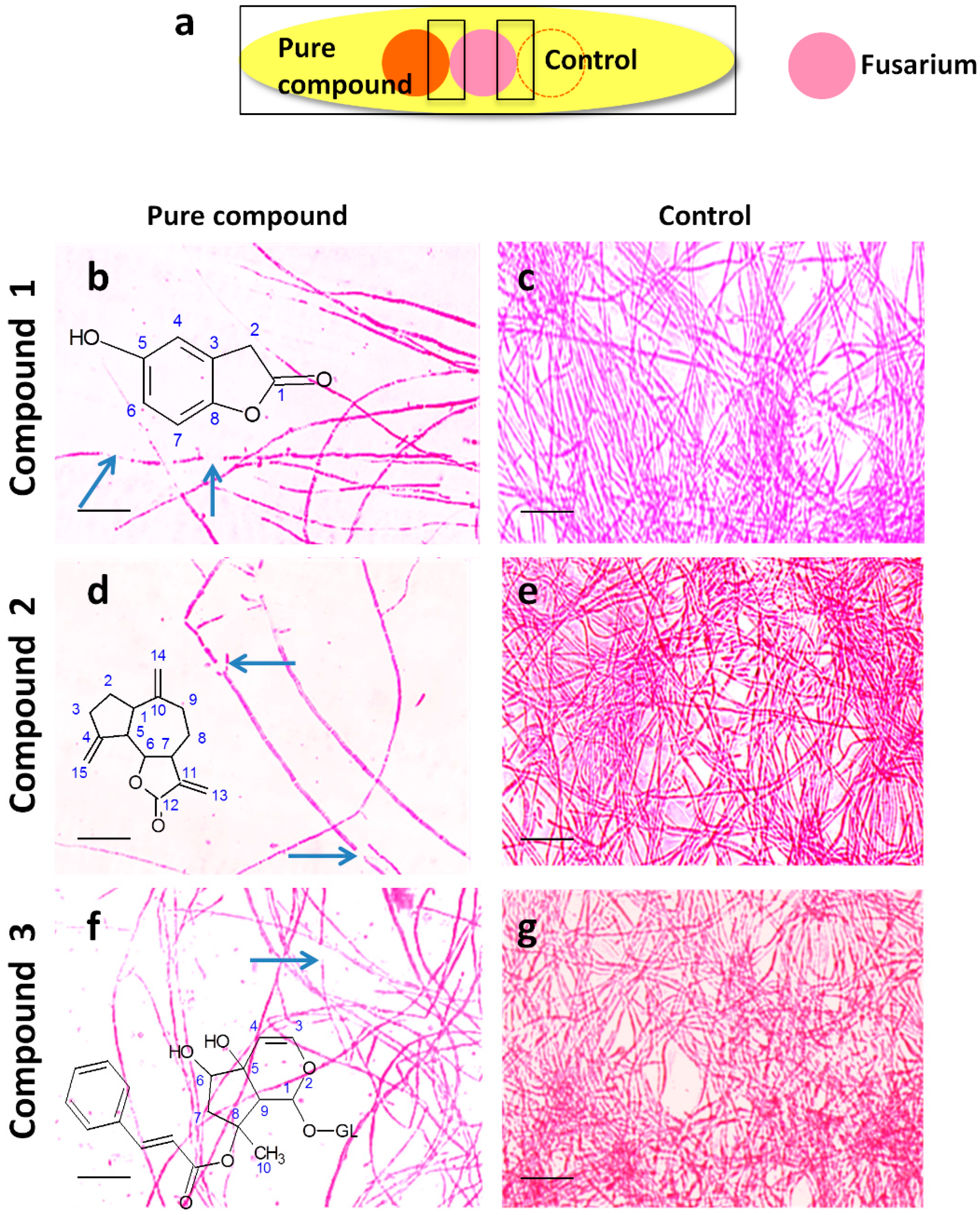
2.4. Microscopic Examination of the Interaction Between Each Pure Antifungal Compound and F. graminearum in Vitro
3. Discussion
3.1. Previous Reports of Non-Pathogenic Fusarium and Penicillium sp. as Endophytes
3.2. Anti-Fusarium Compounds Purified from Finger Millet Endophytes
3.2.1. 4-Hydroxybenzofuranone (Isolated from Fusarium sp. Strain WF5)
3.2.2. Dehydrocostus Lactone (Isolated from Penicillium sp. Strain WF6)
3.2.3. Harpagoside (Isolated from Penicillium sp. Strain WF7)
4. Experimental Section
4.1. Source of Biological Materials
4.2. Competitive Inhibition Experiment
4.3. Plant Pathogenicity Assay
4.4. Root Colonization Assay
4.5. Bio-Guided Purification of Active Anti-Fusarium Compounds
4.6. Antifungal Assay of Purified Compounds Using Agar Diffusion Assay
4.7. Structure Elucidation of Active Anti-F. graminearum Compounds
4.8. Visualization of the Interactions Between F. graminearum and the Anti-F. graminearum Compounds
5. Conclusions and Future Perspectives
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hilu, K.W.; Wet, J.M.J.D. Domestication of Eleusine coracana. Econ. Bot. 1976, 30, 199–208. [Google Scholar] [CrossRef]
- Mousa, W.K.; Schwan, A.; Davidson, J.; Strange, P.; Liu, H.; Zhou, T.; Auzanneau, F.-I.; Raizada, M.N. An endophytic fungus isolated from finger millet (Eleusine coracana) produces anti-fungal natural products. Front. Microbiol. 2015, 6, 1157. [Google Scholar] [CrossRef] [PubMed]
- Munimbazi, C.; Bullerman, L.B. Molds and mycotoxins in foods from burundi. J. Food Prot. 1996, 59, 869–875. [Google Scholar]
- Van der Lee, T.; Zhang, H.; van Diepeningen, A.; Waalwijk, C. Biogeography of Fusarium graminearum species complex and chemotypes. Food Add. Contam. 2015, 32, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Sundaramari, M. Rationality and adoption of indigenous cultivation practices of finger millet (Eleusine coracana (L.)gaertn.) by the tribal farmers of tamil nadu. Int. J. Mang. Soc. Sci. 2015, 2, 970–977. [Google Scholar]
- Wegulo, S.N.; Baenziger, P.S.; Nopsa, J.H.; Bockus, W.W.; Hallen-Adams, H. Management of Fusarium head blight of wheat and barley. Crop Prot. 2015, 73, 100–107. [Google Scholar] [CrossRef]
- Wong, L.; Abramson, D.; Tekauz, A.; Leisle, D.; McKenzie, R. Pathogenicity and mycotoxin production of Fusarium species causing head blight in wheat cultivars varying in resistance. Can. J. Plant Sci. 1995, 75, 261–267. [Google Scholar] [CrossRef]
- O’Donnell, K.; Rooney, A.P.; Proctor, R.H.; Brown, D.W.; McCormick, S.P.; Ward, T.J.; Frandsen, R.J.N.; Lysøe, E.; Rehner, S.A.; Aoki, T.; et al. Phylogenetic analyses of RPB1 and RPB2 support a middle cretaceous origin for a clade comprising all agriculturally and medically important fusaria. Fungal Genet. Biol. 2013, 52, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Mousa, W.K.; Shearer, C.R.; Limay-Rios, V.; Zhou, T.; Raizada, M.N. Bacterial endophytes from wild maize suppress Fusarium graminearum in modern maize and inhibit mycotoxin accumulation. Front. Plant Sci. 2015, 6, 805. [Google Scholar] [CrossRef] [PubMed]
- Shehata, H.R.; Lyons, E.M.; Jordan, K.S.; Raizada, M.N. Bacterial endophytes from wild and ancient maize are able to suppress the fungal pathogen Sclerotinia homoeocarpa. J. Appl. Microbiol. 2016, 120, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Venkateswarlu, S.; Panchagnula, G.K.; Guraiah, M.B.; Subbaraju, G.V. Isoaurones: Synthesis and stereochemical assignments of geometrical isomers. Tetrahedron 2006, 62, 9855–9860. [Google Scholar] [CrossRef]
- Yuuya, S.; Hagiwara, H.; Suzuki, T.; Ando, M.; Yamada, A.; Suda, K.; Kataoka, T.; Nagai, K. Guaianolides as immunomodulators. Synthesis and biological activities of dehydrocostus lactone, mokko lactone, eremanthin, and their derivatives. J. Nat. Prod. 1998, 62, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.; Yan, J.; Lou, J. Preparative isolation and purification of harpagoside from Scrophularia ningpoensis hemsley by high-speed counter-current chromatography. Phytochem. Anal. 2006, 17, 406–408. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D. Endophyte: The evolution of a term, and clarification of its use and definition. Oikos 1995, 73, 274–276. [Google Scholar]
- Saleh, A.A.; Esele, J.; Logrieco, A.; Ritieni, A.; Leslie, J.F. Fusarium verticillioides from finger millet in Uganda. Food Add. Contam. 2012, 29, 1762–1769. [Google Scholar] [CrossRef] [PubMed]
- Adipala, E. Seed-borne fungi of finger millet. E. Afr. Agric. Forest J. 1992, 57, 173–176. [Google Scholar]
- Penugonda, S.; Girisham, S.; Reddy, S. Elaboration of mycotoxins by seed-borne fungi of finger millet (Eleusine coracana L.). Int. J. Biotechnol. Mol. Biol. Res. 2010, 1, 62–64. [Google Scholar]
- Ramana, M.V.; Nayaka, S.C.; Balakrishna, K.; Murali, H.; Batra, H. A novel PCR-DNA probe for the detection of fumonisin-producing Fusarium species from major food crops grown in southern India. Mycology 2012, 3, 167–174. [Google Scholar]
- Cooney, J.M.; Lauren, D.R.; di Menna, M.E. Impact of competitive fungi on trichothecene production by Fusarium graminearum. J. Agric. Food Chem. 2001, 49, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Cotty, P.J. Biocompetitive exclusion of toxigenic fungi. In The Mycotoxin Factbook Food and Feed Topics; Barug, D., Bhatnagar, D., van Egmond, H.P., van der Kamp, J.W., van Osenbruggen, W.A., Visconti, A., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2006; pp. 179–197. [Google Scholar]
- Dawson, W.; Jestoi, M.; Rizzo, A.; Nicholson, P.; Bateman, G. Field evaluation of fungal competitors of Fusarium culmorum and F. graminearum, causal agents of ear blight of winter wheat, for the control of mycotoxin production in grain. Biocontrol Sci. Technol. 2004, 14, 783–799. [Google Scholar] [CrossRef]
- Fravel, D.; Olivain, C.; Alabouvette, C. Fusarium oxysporum and its biocontrol. New Phytol. 2003, 157, 493–502. [Google Scholar] [CrossRef]
- Larkin, R.P.; Fravel, D.R. Mechanisms of action and dose-response relationships governing biological control of fusarium wilt of tomato by nonpathogenic Fusarium spp. Phytopathology 1999, 89, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.-S.; Li, X.-M.; Du, F.-Y.; Li, C.-S.; Proksch, P.; Wang, B.-G. Secondary metabolites from a marine-derived endophytic fungus Penicillium chrysogenum qen-24s. Mar. Drugs 2010, 9, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Lai, D.; Brotz-Oesterhelt, H.; Muller, W.E.G.; Wray, V.; Proksch, P. Bioactive polyketides and alkaloids from Penicillium citrinum, a fungal endophyte isolated from Ocimum tenuiflorum. Fitoterapia 2013, 91, 100–106. [Google Scholar] [CrossRef] [PubMed]
- El-Neketi, M.; Ebrahim, W.; Lin, W.H.; Gedara, S.; Badria, F.; Saad, H.E.A.; Lai, D.W.; Proksch, P. Alkaloids and polyketides from penicillium citrinum, an endophyte isolated from the Moroccan plant Ceratonia siliqua. J. Nat. Prod. 2013, 76, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Kusari, P.; Kusari, S.; Spiteller, M.; Kayser, O. Endophytic fungi harbored in Cannabis sativa L.: Diversity and potential as biocontrol agents against host plant-specific phytopathogens. Fungal Divers. 2013, 60, 137–151. [Google Scholar] [CrossRef]
- Zheng, C.J.; Xu, L.L.; Li, Y.Y.; Han, T.; Zhang, Q.Y.; Ming, Q.L.; Rahman, K.; Qin, L.P. Cytotoxic metabolites from the cultures of endophytic fungi from Panax ginseng. Appl. Microbiol. Biotechnol. 2013, 97, 7617–7625. [Google Scholar] [CrossRef] [PubMed]
- Lenné, J.; Takan, J.; Mgonja, M.; Manyasa, E.; Kaloki, P.; Wanyera, N.; Okwadi, J.; Muthumeenakshi, S.; Brown, A.; Tamale, M. Finger millet blast disease management: A key entry point for fighting malnutrition and poverty in East Africa. Outlook Agric. 2007, 36, 101–108. [Google Scholar] [CrossRef]
- Machida, K.; Trifonov, L.S.; Ayer, W.A.; Lu, Z.-X.; Laroche, A.; Huang, H.C.; Cheng, K.J.; Zantige, J.L. 3(2h)-benzofuranones and chromanes from liquid cultures of the mycoparasitic fungus Coniothyrium minitans. Phytochemistry 2001, 58, 173–177. [Google Scholar] [CrossRef]
- Ding, G.; Zheng, Z.; Liu, S.; Zhang, H.; Guo, L.; Che, Y. Photinides a–f, cytotoxic benzofuranone-derived γ-lactones from the plant endophytic fungus pestalotiopsis photiniae. J. Nat. Prod. 2009, 72, 942–945. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Liu, Y.; Liu, J.; Liu, Z. Inhibitory effects and chemical basis of eucalyptus orelliana wood meals on the growth of Alexandrium tamarense. Huan Jing Ke Xue 2008, 29, 2296–2301. [Google Scholar] [PubMed]
- De Souza Nunes, J.P.; da Silva, K.A.B.; da Silva, G.F.; Quintão, N.L.M.; Corrêa, R.; Cechinel-Filho, V.; de Campos-Buzzi, F.; Niero, R. The antihypersensitive and antiinflammatory activities of a benzofuranone derivative in different experimental models in mice: The importance of the protein kinase c pathway. Anesth. Analg. 2014, 119, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Milošević Ifantis, T.; Solujić, S.; Pavlović-Muratspahić, D.; Skaltsa, H. Secondary metabolites from the aerial parts of Centaurea pannonica (Heuff.) simonk. From Serbia and their chemotaxonomic importance. Phytochemistry 2013, 94, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Julianti, T.; Hata, Y.; Zimmermann, S.; Kaiser, M.; Hamburger, M.; Adams, M. Antitrypanosomal sesquiterpene lactones from Saussurea costus. Fitoterapia 2011, 82, 955–959. [Google Scholar] [CrossRef] [PubMed]
- Rao, G.V.; Reddy, M.V.B.; Pirabakaran, R.; Madhavi, M.S.L.; Kavitha, K.; Mukhopadhyay, T. Antimicrobial and melanin synthesis inhibitory activities of the roots of Inula racemosa hook f. Arch. Appl. Sci. Res. 2013, 5, 104–108. [Google Scholar]
- Cantrell, C.L.; Nuñez, I.S.; Castañeda-Acosta, J.; Foroozesh, M.; Fronczek, F.R.; Fischer, N.H.; Franzblau, S.G. Antimycobacterial activities of dehydrocostus lactone and its oxidation products. J. Nat. Prod. 1998, 61, 1181–1186. [Google Scholar] [CrossRef] [PubMed]
- Kretschmer, N.; Rinner, B.; Stuendl, N.; Kaltenegger, H.; Wolf, E.; Kunert, O.; Boechzelt, H.; Leithner, A.; Bauer, R.; Lohberger, B. Effect of costunolide and dehydrocostus lactone on cell cycle, apoptosis, and abc transporter expression in human soft tissue sarcoma cells. Planta Med. 2012, 78, 1749–1756. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Hong, J.E.; Lim, S.S.; Kwon, G.T.; Kim, J.; Kim, J.-S.; Lee, K.W.; Park, J.H.Y. The hexane extract of Saussurea lappa and its active principle, dehydrocostus lactone, inhibit prostate cancer cell migration. J. Med. Food 2012, 15, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Mncwangi, N.; Chen, W.; Vermaak, I.; Viljoen, A.M.; Gericke, N. Devil’s claw—A review of the ethnobotany, phytochemistry and biological activity of Harpagophytum procumbens. J. Ethnopharmacol. 2012, 143, 755–771. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, Y.; Xiao, L.; Jin, X.; Yang, K. Simultaneous determination of harpagoside and cinnamic acid in rat plasma by high-performance liquid chromatography: Application to a pharmacokinetic study. Anal. Bioanal. Chem. 2007, 389, 2259–2264. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, M.I.; Ivanovska, N.; Alipieva, K.; Dimitrova, P.; Verpoorte, R. Harpagoside: From Kalahari Desert to pharmacy shelf. Phytochemistry 2013, 92, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B. Phytotoxic effect of seed mycoflora associated with the genotypes of finger millet (Eleusine coracana). Prog. Agric. 2010, 10, 112–115. [Google Scholar]
- Hussain, H.; Krohn, K.; Ullah, Z.; Draeger, S.; Schulz, B. Bioactive chemical constituents of two endophytic fungi. Biochem. Syst. Ecol. 2007, 35, 898–900. [Google Scholar] [CrossRef]
- Soliman, S.S.; Greenwood, J.S.; Bombarely, A.; Mueller, L.A.; Tsao, R.; Mosser, D.D.; Raizada, M.N. An endophyte constructs fungicide-containing extracellular barriers for its host plant. Curr. Biol. 2015, 25, 2570–2576. [Google Scholar] [CrossRef] [PubMed]
- Waqas, M.; Khan, A.L.; Kamran, M.; Hamayun, M.; Kang, S.-M.; Kim, Y.-H.; Lee, I.-J. Endophytic fungi produce gibberellins and indoleacetic acid and promotes host-plant growth during stress. Molecules 2012, 17, 10754–10773. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Trace samples of the compounds 1 and 2 are available from the authors.

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mousa, W.K.; Schwan, A.L.; Raizada, M.N. Characterization of Antifungal Natural Products Isolated from Endophytic Fungi of Finger Millet (Eleusine coracana). Molecules 2016, 21, 1171. https://doi.org/10.3390/molecules21091171
Mousa WK, Schwan AL, Raizada MN. Characterization of Antifungal Natural Products Isolated from Endophytic Fungi of Finger Millet (Eleusine coracana). Molecules. 2016; 21(9):1171. https://doi.org/10.3390/molecules21091171
Chicago/Turabian StyleMousa, Walaa Kamel, Adrian L. Schwan, and Manish N. Raizada. 2016. "Characterization of Antifungal Natural Products Isolated from Endophytic Fungi of Finger Millet (Eleusine coracana)" Molecules 21, no. 9: 1171. https://doi.org/10.3390/molecules21091171




