Antifungal Activity of Eucalyptus Oil against Rice Blast Fungi and the Possible Mechanism of Gene Expression Pattern
Abstract
:1. Introduction
2. Results and Discussion
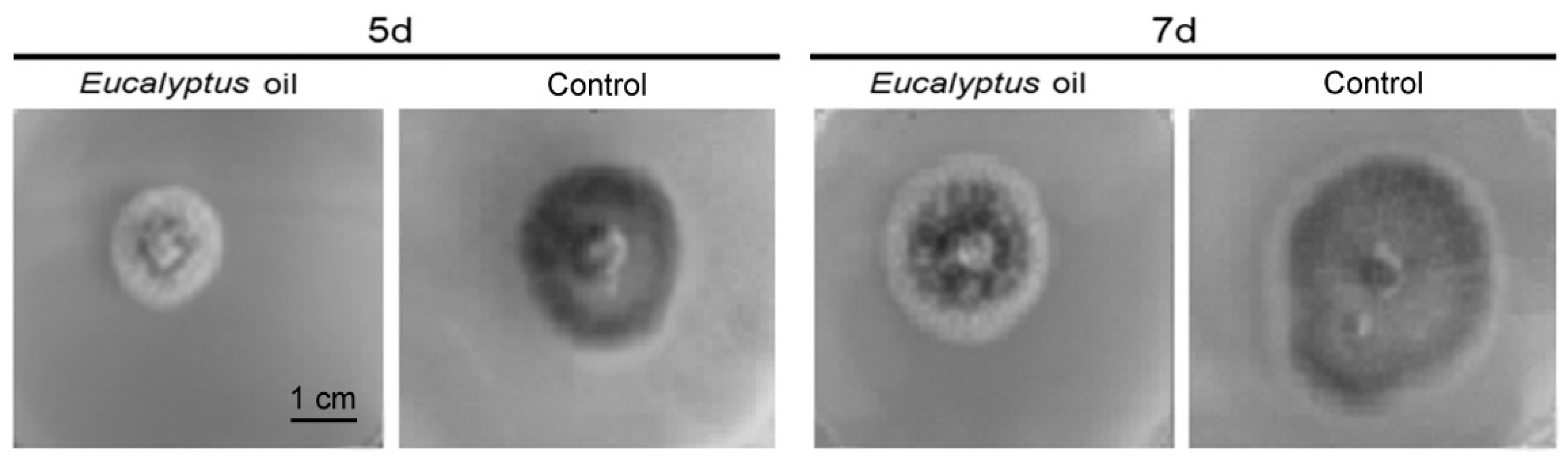
2.1. Inhibitory Activities of the Eucalyptus Oil on Plant Pathogenic Fungi
2.2. SEM Results
2.3. Hybridization Process Results
2.4. Microarray Data Analysis
2.4.1. Identification of Differentially Expressed Genes
2.4.2. GO Analysis
2.5. Analysis of Treatment Results of the Eucalyptus Oil Combined with Antifungal Drug Targets
2.5.1. Inhibition to Cell-Wall Synthesis, Chitin and Cellulose Synthesis
2.5.2. Interference with Respiration
2.5.3. Inhibition of Nucleic Acid Synthesis
2.6. Potential Application
3. Experimental Section
3.1. Extraction of Eucalyptus Oil (EO)
3.2. Inhibitory Test
3.3. Morphological Study of the Inhibitory Activities of EO on M. grisea
3.4. Total RNA Extraction
3.5. Fluorescent Dye Labelling, Chip Hybridization and Scanning
3.6. RT-PCR Analysis
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Farzaei, M.H.; Bahramsoltani, R.; Abbasabadi, Z.; Rahimi, R. A comprehensive review on phytochemical and pharmacological aspects of Elaeagnus angustifolia L. J. Pharm. Pharmacol. 2015, 67, 1467–1480. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.C.; Zhang, Z.W.; Chen, Y.E.; Yuan, M.; Yuan, S.; Bao, J.K. Antiviral and antitumor activities of the lectin extracted from Aspidistra elatior. Z. Naturforsch. 2015, 70c, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Mondal, D.; Barat, S.; Mukhopadhyay, M.K. Toxicity of neem pesticides on a fresh water loach, Lepidocephalichthys guntea (Hamilton Buchanan) of Darjeeling district in West Bengal. J. Environ. Biol. 2007, 28, 119–122. [Google Scholar] [PubMed]
- Batish, D.R.; Singh, H.P.; Kohli, R.K.; Kaur, S. Eucalyptus essential oil as a natural pesticide. For. Ecol. Manag. 2008, 256, 2166–2174. [Google Scholar] [CrossRef]
- Hajer, N.B.M.; Mehrez, R.; Ahmed, L.; Florence, M.; François, C.; Manef, A.; Mohamed, L.K.; Jalloul, B. Eucalyptus oleosa essential oils: Chemical composition and antimicrobial and antioxidant activities of the oils from different plant parts (stems, leaves, flowers and fruits). Molecules 2011, 16, 1695–1709. [Google Scholar]
- Cheng, S.S.; Huang, C.G.; Chen, Y.J.; Yu, J.J.; Chen, W.J.; Chang, S.T. Chemical compositions and larvicidal activities of leaf essential oils from two Eucalyptus species. Bioresour. Technol. 2009, 100, 452–456. [Google Scholar] [CrossRef] [PubMed]
- Gilles, M.; Zhao, J.; An, M.; Agboola, S. Chemical composition and antimicrobial properties of essential oils of three Australian Eucalyptus species. Food Chem. 2010, 119, 731–737. [Google Scholar] [CrossRef]
- Macie, M.V.; Morais, S.M.; Bevilaqua, C.M.L.; Silva, R.A.; Barros, R.S.; Sousa, R.N.; Brito, E.S.; Souza-Neto, M.A. Chemical composition of Eucalyptus spp. essential oils and their insecticidal effects on Lutzomyia longipalpis. Vet. Parasitol. 2010, 167, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Dušan, B.; Slavenko, G.; Dejan, O.; Dragana, M.C.; Jelena, K.V.; Neda, M.D. Essential Oil of Eucalyptus Gunnii. Hook. As a Novel Source of Antioxidant, Antimutagenic and Antibacterial Agents. Molecules 2014, 19, 19007–19020. [Google Scholar]
- Ameur, E.; Zyed, R.; Samia, M.; Karima, B.H.S.; Mahjoub, A.; Mohamed, L.K.; Farhat, F.; Rachid, C.; Fethia, H.S. Correlation between chemical composition and antibacterial activity of essential oils from fifteen Eucalyptus species growing in the Korbous and Jbel Abderrahman arboreta (North East Tunisia). Molecules 2012, 17, 3044–3057. [Google Scholar]
- Kumar, P.; Mishra, S.; Malik, A.; Satya, S. Compositional analysis and insecticidal activity of Eucalyptus globulus (family: Myrtaceae) essential oil against housefly (Musca domestica). Acta Trop. 2012, 122, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Cimanga, K.; Kambu, K.; Tona, L.; Apers, S.; De Bruyne, T.; Hermans, N.; Totté, J.; Pieters, L.; Vlietinck, A.J. Correlation between chemical composition and antibacterial activity of essential oils of some aromatic medicinal plants growing in the Democratic Republic of Congo. J. Ethnopharmacol. 2002, 79, 213–220. [Google Scholar] [CrossRef]
- Ramezani, H.; Singh, H.P.; Batish, D.R.; Kohli, R.K. Antifungal activity of the volatile oil of Eucalyptus citriodora. Fitoterapia 2002, 73, 261–262. [Google Scholar] [CrossRef]
- Tyagi, A.K.; Malik, A. Antimicrobial potential and chemical composition of Eucalyptus globulus oil in liquid and vapour phase against food spoilage microorganisms. Food Chem. 2011, 126, 228–235. [Google Scholar] [CrossRef]
- Rocha Caldas, G.F.; Oliveira, A.R.; Araújo, A.V.; Lafayette, S.S.; Albuquerque, G.S.; Silva-Neto Jda, C.; Costa-Silva, J.H.; Ferreira, F.; Costa, J.G.; Wanderley, A.G. Gastroprotective mechanisms of the monoterpene 1,8-cineole (eucalyptol). PLoS ONE 2015, 10, e0134558. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.J.; Huang, L.J.; Yang, Z.R.; Bai, L.H. Optimization of supercritical CO2 extraction conditions for essential oil from Eucalyptus grandis × Eucalyptus urophylla using Box-Behnken design-response surface methodology. J. Sichuan Univ. (Nat. Sci. Ed.) 2014, 51, 1319–1324. [Google Scholar]
- Armaka, A.; Papanikolaou, E.; Sivropoulou, A.; Arsenakis, M. Antiviral properties of isoborneol, a potent inhibitor of herpes simplex virus type 1. Antivir. Res. 1999, 43, 79–92. [Google Scholar] [CrossRef]
- Park, S.N.; Lim, Y.K.; Freire, M.O.; Cho, E.; Jin, D.; Kook, J.K. Antimicrobial effect of linalool and α-terpineol against periodontopathic and cariogenic bacteria. Anaerobe 2012, 18, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Gwak, K.S.; Yang, I.; Kim, K.W.; Jeung, E.B.; Chang, J.W.; Choi, I.G. Effect of citral, eugenol, nerolidol and α-terpineol on the ultrastructural changes of Trichophyton mentagrophytes. Fitoterapia 2009, 80, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Marei, G.I.Kh.; Rasoul, M.A.A.; Abdelgaleil, S.A.M. Comparative antifungal activities and biochemical effects of monoterpenes on plant pathogenic fungi. Pestic. Biochem. Phys. 2012, 103, 56–61. [Google Scholar] [CrossRef]
- Abrahim, D.; Francischini, A.C.; Pergo, E.M.; Kelmer-Bracht, A.M.; Ishii-Iwamoto, E.L. Effects of α-pinene on the mitochondrial respiration of maize seedlings. Plant. Physiol. Biochem. 2003, 41, 985–991. [Google Scholar] [CrossRef]
- Hammer, K.; Carson, C.F.; Riley, T.V. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J. Appl. Microbiol. 2003, 95, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Ojeda-Sana, A.M.; Van Baren, C.M.; Elechosa, M.A.; Juárez, M.A.; Moreno, S. New insights into antibacterial and antioxidant activities of rosemary essential oils and their main components. Food Control 2013, 31, 189–195. [Google Scholar] [CrossRef]
- Georgopapadakou, N.H. Update on antifungals targeted to the cell wall: Focus on beta-1,3-glucan synthase inhibitors. Expert Opin. Investig. Drugs 2001, 10, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Li, G.X.; Pang, Y.; Wang, P. A novel chitin-binding protein identified from the peritrophic membrane of the cabbage looper, Trichoplusia ni, Insect Biochem. Mol. Biol. 2005, 35, 1224–1234. [Google Scholar]
- Lenardon, M.D.; Munro, C.A.; Gow, N.A. Chitin synthesis and fungal pathogenesis. Curr. Opin. Microbiol. 2010, 13, 416–423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vanlerberghe, G.C.; Robson, C.A.; Yip, J.Y.H. Induction of mitochondrial alternative oxidase in response to a cell signal pathway down-regulation the cytochrome c pathway prevent programmed cell death. Plant Physiol. 2002, 129, 1829–1842. [Google Scholar] [CrossRef] [PubMed]
- Fiorani, F.; Umbach, A.L.; Siedow, J.N. The alternative oxidase of plant mitochondria is involved in the acclimation of shoot growth at low temperature. A study of Arabidopsis AOX1a transgenic plants. Plant Physiol. 2005, 139, 1795–1805. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, D.W.; Clough, J.M.; Godwin, J.R.; Hall, A.A.; Hamer, M.; Parr-Dobrzanski, B. The strobilurin fungicides. Pest. Manag. Sci. 2002, 58, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.; Williams, T.; Morgan, M.A.; Ratcliffe, R.; Sweetlove, L. Glycolytic enzymes associate dynamically with mitochondria in response to respiratory demand and support substrate channeling. Plant Cell 2007, 19, 3723–3738. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.M.; Wong, K.H.; Chen, X.D. Glucose oxidase: Natural occurrence, function, properties and industrial applications. Appl. Microbiol. Biotechnol. 2008, 78, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Scopes, R.K. 3-Phosphoglycerate kinase. Enzyme 1973, 8, 335–351. [Google Scholar]
- Hove-Jensen, B. Mutation in the phosphoribosylpyrophosphate synthetase gene (prs) that results in simultaneous requirements for purine and pyrimidine nucleosides, nicotinamide nucleotide, histidine, and tryptophan in Escherichia coli. J. Bacteriol. 1988, 170, 1148–1152. [Google Scholar] [PubMed]
- Nenoff, P.; Haustein, U.F.; Brandt, W. Antifungal activity of the essential oil of Melaleuca alternifolia (tea tree oil) against pathogenic fungi in vitro. Skin Pharmacol. 1996, 9, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, D. Supercritical CO2 extraction of Eucalyptus leaves oil and comparison with Soxhlet extraction and hydro-distillation methods. Sep. Purif. Technol. 2014, 133, 443–451. [Google Scholar] [CrossRef]
- Scheckhuber, C.Q. Penicillium chrysogenum as a model system for studying cellular effects of methylglyoxal. BMC Microbiol. 2015, 15, 138. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, D.; Liu, S.; Li, S.; Xu, G.; Zheng, G.; Xie, L.; Zhang, R. Microarray: A global analysis of biomineralization-related gene expression profiles during larval development in the pearl oyster, Pinctada fucata. BMC Genom. 2015, 16, 325. [Google Scholar] [CrossRef] [PubMed]
- Eisen, M.B.; Spellman, P.T.; Brown, P.O.; Botstein, D. Cluster analysis and display of genome-wide expression patterns. Proc. Natl. Acad. Sci. USA 1998, 95, 14863–14868. [Google Scholar] [CrossRef] [PubMed]
- Conesa, A.; Gotz, S.; Garcia-Gomez, J.M.; Terol, J.; Talon, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds from Eucalyptus leaves oil are available from the authors.


| Strain | Control Group (cm) | EO-Treated Group (cm) | Inhibition Rate (%) |
|---|---|---|---|
| Setosphaeria turcica | 1.7 ± 0.3 | 0.6 ± 0.2 | 91 ± 9 |
| Magnaporthe grisea | 2.4 ± 0.4 | 1.5 ± 0.3 | 81 ± 7 |
| Botrytis cinerea | 3.0 ± 0.8 | 1.1 ± 0.3 | 75 ± 7 |
| Fusarium graminearum | 5.2 ± 1.7 | 1.7 ± 0.4 | 74 ± 6 |
| Bipolaris maydis | 5.9 ± 1.6 | 2.6 ± 0.6 | 62 ± 6 |
| Rhizoctonia solani | 5.4 ± 1.5 | 2.6 ± 0.8 | 56 ± 5 |
| Rhizoctonia solani | 3.5 ± 1.0 | 2.1 ± 0.5 | 46 ± 5 |
| Colletotrichum gloeosporioides | 2.9 ± 0.6 | 2.0 ± 0.4 | 45 ± 5 |
| Alternuria longipes | 4.0 ± 1.1 | 2.9 ± 0.7 | 31 ± 5 |
| Alternaria solani | 3.6 ± 1.0 | 2.8 ± 0.7 | 26 ± 4 |
| Fusarium moniliforme | 4.3 ± 1.1 | 3.3 ± 0.7 | 25 ± 4 |
| GO Terms | Terms Type | Genes_In_Term | DEG | Up | Down | p-Value |
|---|---|---|---|---|---|---|
| GO:0055114 (oxidation-reduction process) | P | 777 | 109 | 51 | 58 | 1.53 × 10−10 |
| GO:0044271 (cellular nitrogen compound biosynthetic process) | P | 38 | 14 | 0 | 14 | 1.01 × 10−5 |
| GO:0043581 (mycelium development) | P | 468 | 59 | 22 | 37 | 5.60 × 10−4 |
| GO:0009103 (lipopolysaccharide biosynthetic process) | P | 10 | 6 | 4 | 2 | 6.24 × 10−4 |
| GO:0006066 (alcohol metabolic process) | P | 13 | 6 | 4 | 2 | 3.37 × 10−3 |
| GO:0055085 (transmembrane transport) | P | 292 | 37 | 10 | 27 | 1.06 × 10−2 |
| GO:0052051 (interaction with host via protein secreted by type II secretion system) | P | 50 | 11 | 9 | 2 | 1.15 × 10−2 |
| GO:0008152 (metabolic process) | P | 328 | 39 | 27 | 12 | 1.69 × 10−2 |
| GO:0005375 (copper ion transmembrane transporter activity) | P | 8 | 4 | 1 | 3 | 1.69 × 10−2 |
| GO:0035434 (copper ion transmembrane transport) | P | 8 | 4 | 1 | 3 | 1.69 × 10−2 |
| Total P | 1992 | 289 | 129 | 160 | ||
| GO: 0016491 (oxidoreductase activity) | F | 268 | 50 | 23 | 27 | 6.79 × 10−9 |
| GO:0050660 (flavin adenine dinucleotide binding) | F | 90 | 18 | 11 | 7 | 2.27 × 10−3 |
| GO:0008812 (choline dehydrogenase activity) | F | 9 | 5 | 3 | 2 | 6.47 × 10−3 |
| GO:0004181 (metallocarboxypeptidase activity) | F | 6 | 4 | 1 | 3 | 9.44 × 10−3 |
| GO:0003824 (catalytic activity) | F | 175 | 24 | 11 | 13 | 2.44 × 10−2 |
| GO:0000981 (sequence-specific DNA binding RNA polymerase II transcription) factor activity) | F | 113 | 17 | 11 | 6 | 3.99 × 10−2 |
| Total F | 661 | 118 | 60 | 58 | ||
| GO:0016021 (integral to membrane) | C | 742 | 52 | 21 | 31 | 3.37 × 10−6 |
| GO:0005737 (cytoplasm) | C | 259 | 20 | 1 | 19 | 8.13 × 10−3 |
| GO:0005634 (nucleus) | C | 610 | 35 | 18 | 17 | 1.41 × 10−2 |
| Total C | 1611 | 107 | 40 | 67 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.-J.; Li, F.-R.; Huang, L.-J.; Yang, Z.-R.; Yuan, S.; Bai, L.-H. Antifungal Activity of Eucalyptus Oil against Rice Blast Fungi and the Possible Mechanism of Gene Expression Pattern. Molecules 2016, 21, 621. https://doi.org/10.3390/molecules21050621
Zhou L-J, Li F-R, Huang L-J, Yang Z-R, Yuan S, Bai L-H. Antifungal Activity of Eucalyptus Oil against Rice Blast Fungi and the Possible Mechanism of Gene Expression Pattern. Molecules. 2016; 21(5):621. https://doi.org/10.3390/molecules21050621
Chicago/Turabian StyleZhou, Li-Jun, Fu-Rong Li, Li-Jie Huang, Zhi-Rong Yang, Shu Yuan, and Lin-Han Bai. 2016. "Antifungal Activity of Eucalyptus Oil against Rice Blast Fungi and the Possible Mechanism of Gene Expression Pattern" Molecules 21, no. 5: 621. https://doi.org/10.3390/molecules21050621
APA StyleZhou, L.-J., Li, F.-R., Huang, L.-J., Yang, Z.-R., Yuan, S., & Bai, L.-H. (2016). Antifungal Activity of Eucalyptus Oil against Rice Blast Fungi and the Possible Mechanism of Gene Expression Pattern. Molecules, 21(5), 621. https://doi.org/10.3390/molecules21050621






