Heterogeneous Phase Microwave-Assisted Reactions under CO2 or CO Pressure
Abstract
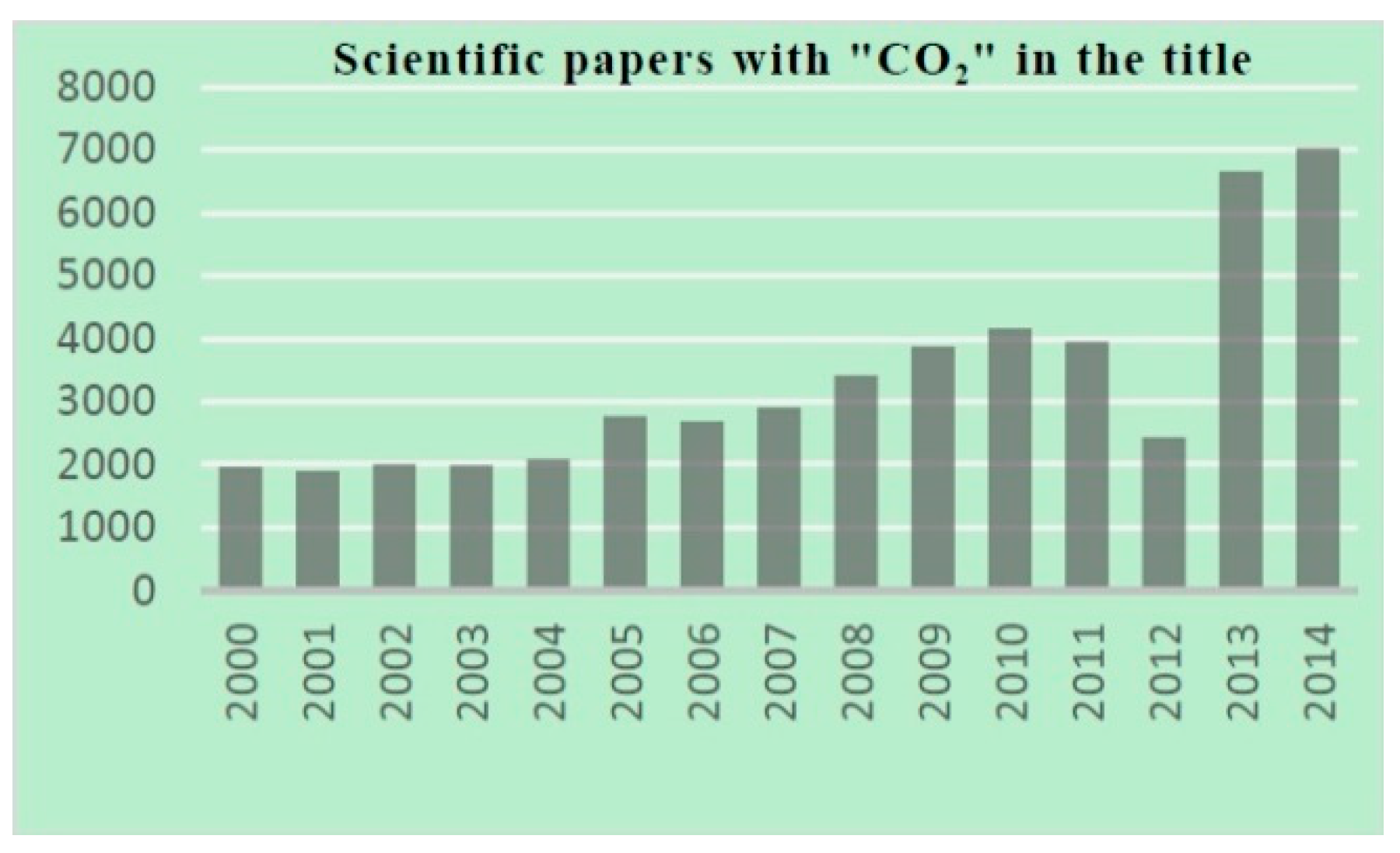
:1. Introduction
2. MW-Assisted Reactions under CO2 Pressure
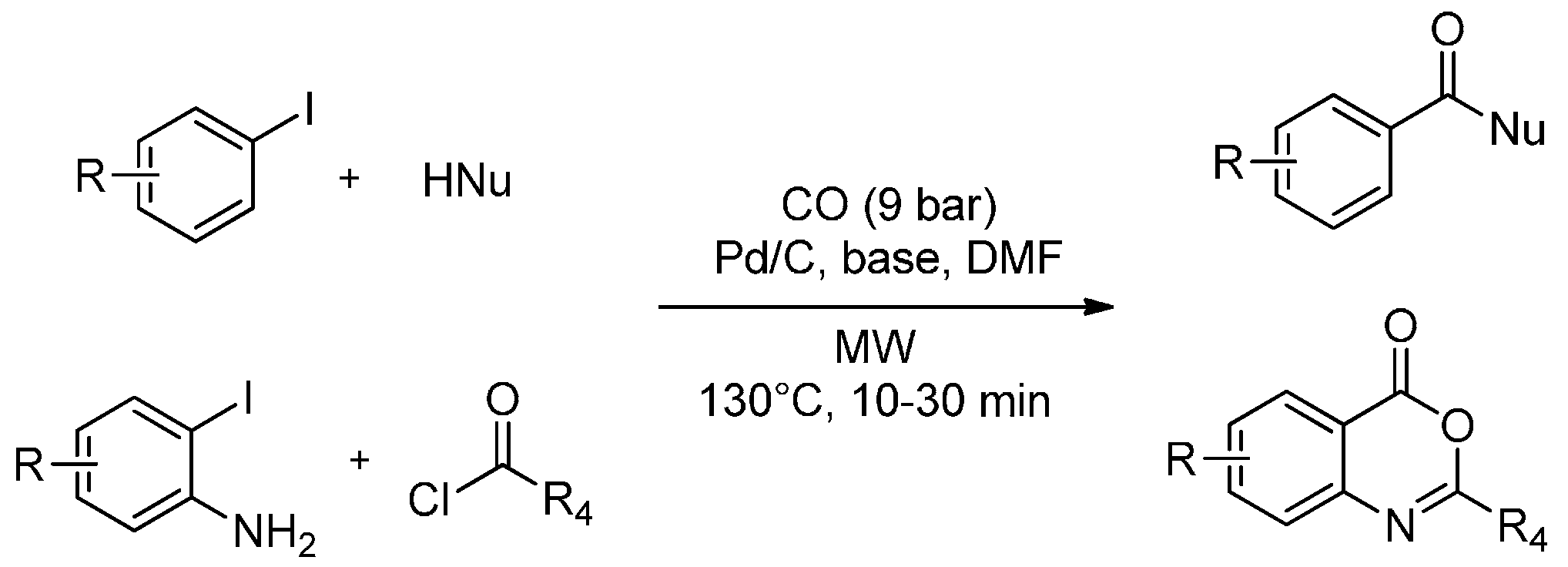
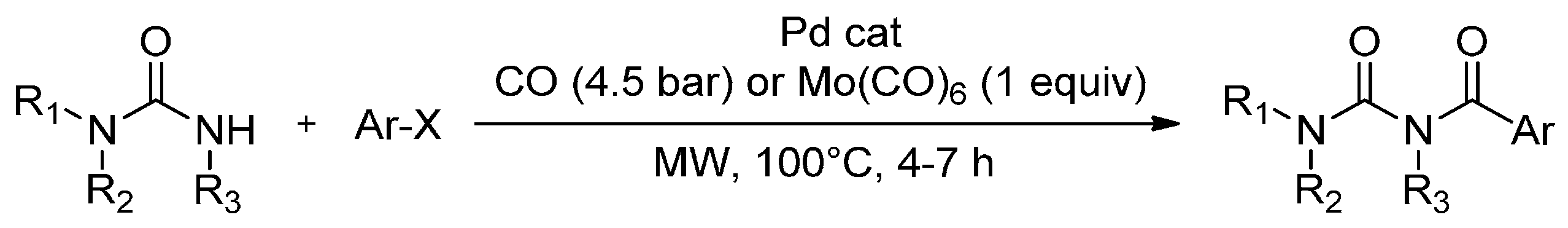
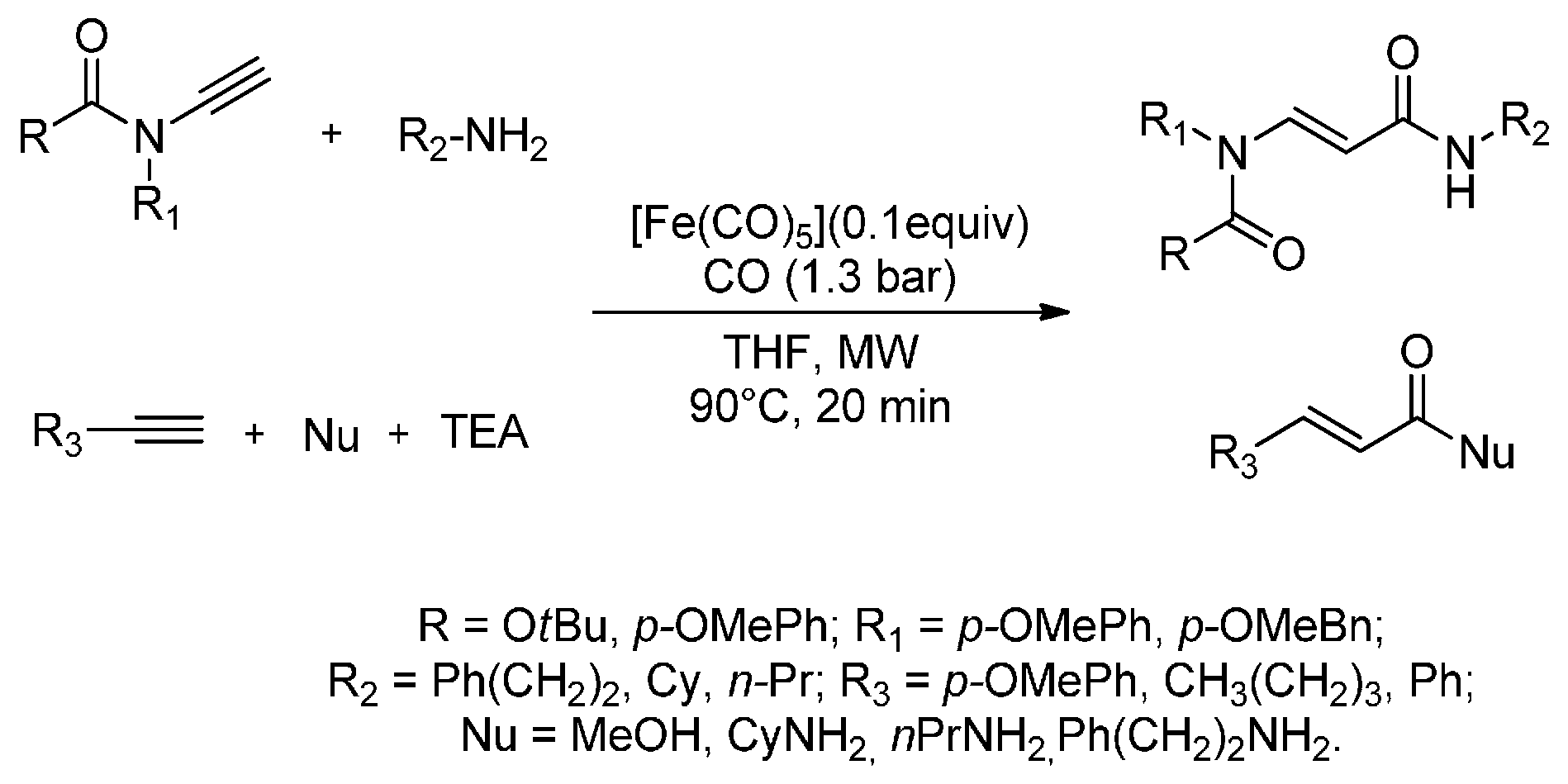
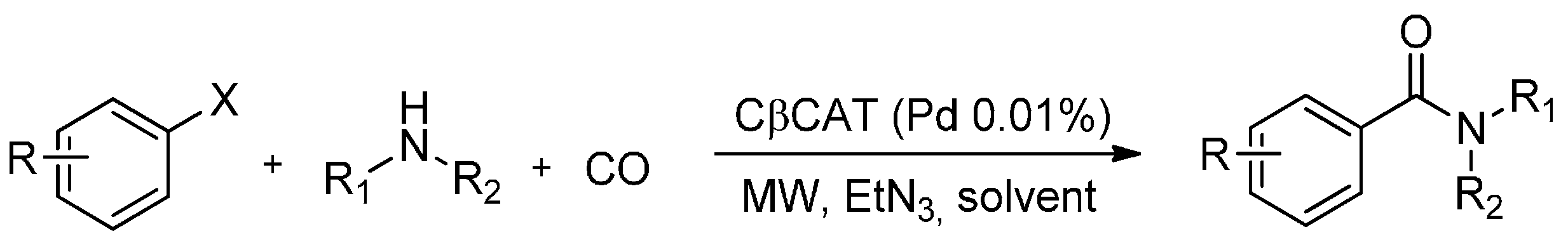
3. MW-Assisted Reactions under CO Pressure
4. A Promising Future for the Use of MW-Assisted Protocols under CO2 or CO Pressure
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Watson, W.J.W. How do the fine chemical, pharmaceutical, and related industries approach green chemistry and sustainability? Green Chem. 2012, 14, 251–259. [Google Scholar] [CrossRef]
- Beach, E.S.; Cui, Z.; Anastas, P.T. Green chemistry: A design framework for sustainability. Energy Environ. Sci. 2009, 2, 1038–1049. [Google Scholar] [CrossRef]
- Kondratenko, E.V.; Mul, G.; Baltrusaitis, J.; Larrazabal, G.O.; Pérez–Ramirez, J. Status and perspectives of CO2 conversion into fuels and chemicals by catalytic, photocatalytic and electrocatalytic processes. Energy Environ. Sci. 2013, 6, 3112–3135. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, L.; Jackstell, R.; Beller, M. Using carbon dioxide as a building block in organic synthesis. Nat. Comm. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mercadante, M.A.; Leadbeater, N.E. Development of methodologies for reactions involving gases as reagents: Microwave heating and conventionally-heated continuous-flow processing as examples. Green Process. Synth. 2012, 1, 499–507. [Google Scholar]
- Beller, M.; Wu, X.-F. Transition Metal Catalyzed Carbonylation Reactions; Springer: Berlin, Germany, 2013; pp. 13–52. [Google Scholar]
- Franke, R.; Selent, D.; Borner, A. Applied hydroformylation. Chem. Rev. 2012, 112, 5675–5732. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Jiang, H.-Z.; Shi, B.-F. Pd(II)-catalyzed oxidative alkoxycarbonylation of 2-phenoxypyridine derivatives with CO and alcohols. Org. Biomol. Chem. 2014, 12, 2538–2542. [Google Scholar] [CrossRef] [PubMed]
- Hermange, P.; Lindhardt, A.T.; Taaning, R.H.; Bjerglund, K.; Lupp, D.; Skrydstrup, T. Ex situ generation of stoichiometric and substoichiometric 12CO and 13CO and its efficient incorporation in palladium catalyzed aminocarbonylations. J. Am. Chem. Soc. 2011, 133, 6061–6071. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.F.; Neumann, H.; Spannenberg, A.; Schulz, T.; Jiao, H.; Beller, M. Development of a general palladium-catalyzed carbonylative Heck reaction of aryl halides. J. Am. Chem. Soc. 2010, 132, 14596–14602. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Zheng, G.; Zha, L.; Peng, J. Carbonylative Suzuki–Miyaura coupling of arylboronic acids with aryl iodides catalyzed by the MCM-41-supported bidentate phosphane palladium(II) complex. Eur. J. Org. Chem. 2009, 10, 1585–1591. [Google Scholar] [CrossRef]
- Fusano, A.; Fukuyama, T.; Nishitani, S.; Inouye, T.; Ryu, I. Synthesis of alkyl alkynyl ketones by Pd/light-induced three-component coupling reactions of iodoalkanes, CO, and 1-alkynes. Org. Lett. 2010, 12, 2410–2413. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Liu, Q.; Jackstell, R.; Beller, M. Carbonylations of alkenes with CO surrogates. Angew. Chem. Int. Ed. 2014, 53, 6310–6320. [Google Scholar] [CrossRef] [PubMed]
- Konishi, H.; Manabe, K. Formic acid derivatives as practical carbon monoxide surrogates for metal-catalyzed carbonylation reactions. Synlett 2014, 25, 1971–1986. [Google Scholar] [CrossRef]
- Natte, K.; Dumrath, A.; Neumann, H.; Beller, M. Palladium-catalyzed carbonylations of aryl bromides using paraformaldehyde: Synthesis of aldehydes and esters. Angew. Chem. Int. Ed. 2014, 53, 10090–10094. [Google Scholar] [CrossRef] [PubMed]
- Ueda, T.; Konishi, H.; Manabe, K. Palladium-catalyzed reductive carbonylation of aryl halides with N-formylsaccharin as a CO source. Angew. Chem. Int. Ed. 2013, 52, 8611–8615. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Cai, J.; Huang, H.; Deng, G.-J. Palladium-Catalyzed phthalazinone synthesis using paraformaldehyde as carbon source. Org. Lett. 2014, 16, 5324–5327. [Google Scholar] [CrossRef] [PubMed]
- Steffen, V.F.; Ulven, H.; Ulven, T. Oxalyl chloride as a practical carbon monoxide source for carbonylation reactions. Org. Lett. 2015, 17, 2832–2835. [Google Scholar]
- Christensen, S.H.; Olsen, E.P.K.; Rosenbaum, J.; Madsen, R. Hydroformylation of olefins and reductive carbonylation of aryl halides with syngas formed ex situ from dehydrogenative decarbonylation of hexane-1,6-diol. Org. Biomol. Chem. 2015, 13, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Schareina, T.; Zapf, A.; Gotta, C.M.; Beller, M. An improved protocol for palladium-catalyzed alkoxycarbonylations of aryl chlorides with alkyl formats. Adv. Synth. Catal. 2010, 352, 1205–1209. [Google Scholar] [CrossRef]
- Nouira, I.; Kostakis, I.K.; Dubouilh, C.; Chosson, E.; Iannelli, M.; Besson, T. Decomposition of formamide assisted by microwaves, a tool for synthesis of nitrogen-containing heterocycles. Tetrahedron Lett. 2008, 49, 7033–7036. [Google Scholar] [CrossRef]
- Loidreau, Y.; Besson, T. Microwave-assisted thermal decomposition of formamide: A tool for coupling a pyrimidine ring with an aromatic partner. Tetrahedron 2011, 67, 4852–4857. [Google Scholar] [CrossRef]
- Wan, Y.; Alterman, M.; Larhed, M.; Hallberg, A. Formamide as a combined ammonia synthon and carbon monoxide source in fast palladium-catalyzed aminocarbonylations of aryl halides. J. Comb. Chem. 2003, 5, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Sawant, D.N.; Wagh, Y.S.; Bhatte, K.D.; Bhanage, B.M. Palladium-catalyzed carbon-monoxide-free aminocarbonylation of aryl halides using N-substituted formamides as an amide source. J. Org. Chem. 2011, 76, 5489–5494. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, C.M.; Widdowson, D.A. Organic synthesis with carbon monoxide: The synthesis of carbamoylstannanes and aromatic carbamoylation. J. Chem. Soc. Perkin Trans. 1 1988, 1, 569–573. [Google Scholar] [CrossRef]
- Cunico, R.F.; Pandey, R.K. Palladium-catalyzed conversion of benzylic and allylic halides into α-aryl and β,γ-unsaturated tertiary amides by the use of a carbamoylsilane. J. Org. Chem. 2005, 70, 9048–9050. [Google Scholar] [CrossRef] [PubMed]
- Wieckowska, A.; Fransson, R.; Odell, L.R.; Larhed, M. Microwave-assisted synthesis of Weinreb and MAP aryl amides via Pd-catalyzed Heck aminocarbonylation using Mo(CO)6 or W(CO)6. J. Org. Chem. 2011, 76, 978–981. [Google Scholar] [CrossRef] [PubMed]
- Cokoja, M.; Bruckmeier, C.; Rieger, B.; Herrmann, W.A.; Kuhn, F.E. Transformation of carbon dioxide with homogeneous transition-metal catalysts: A molecular solution to a global challenge? Angew. Chem. Int. Ed. 2011, 50, 8510–8537. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.L.; Nie, Y.; Sun, J.; Cheng, W.G.; Wang, J.Q.; He, H.Y.; Zhang, S.J. Functionalized dicyandiamide-formaldehyde polymers as efficient heterogeneous catalysts for conversion of CO2 into organic carbonates. Green Chem. 2014, 16, 2771–2778. [Google Scholar] [CrossRef]
- Xu, B.H.; Wang, J.Q.; Sun, J.; Huang, Y.; Zhang, J.P.; Zhang, X.P.; Zhang, S.J. Fixation of CO2 into cyclic carbonates catalyzed by ionic liquids: A multi-scale approach. Green Chem. 2015, 17, 108–122. [Google Scholar] [CrossRef]
- Cokoja, M.; Wilhelm, M.E.; Anthofer, M.H.; Herrmann, W.A.; Kuhn, F.E. Synthesis of cyclic carbonates from epoxides and carbon dioxide by using organocatalysts. ChemSusChem 2015, 10, 2436–2454. [Google Scholar] [CrossRef] [PubMed]
- North, M.; Pasquale, R.; Young, C. Synthesis of cyclic carbonates from epoxides and CO2. Green Chem. 2010, 12, 1514–1539. [Google Scholar] [CrossRef]
- Hu, J.; Ma, J.; Zhu, Q.; Zhang, Z.; Wu, C.; Han, B. Transformation of atmospheric CO2 catalyzed by protic ionic liquids: Efficient synthesis of 2-oxazolidinones. Angew. Chem. Int. Ed. 2015, 54, 5399–5403. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.H.; Himeda, Y.; Muckerman, J.T.; Manbeck, G.F.; Fujita, E. CO2 Hydrogenation to formate and methanol as an alternative to photo- and electrochemical CO2 reduction. Chem. Rev. 2015, 115, 12936–12973. [Google Scholar] [CrossRef] [PubMed]
- Kortlever, R.; Shen, J.; Schouten, K.J.P.; Calle-Vallejo, F.; Koper, M.T.M. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J. Phys. Chem. Lett. 2015, 6, 4073–4082. [Google Scholar] [CrossRef] [PubMed]
- Bai, D.; Wang, Q.; Song, Y.; Li, B.; Jing, H. Synthesis of cyclic carbonate from epoxide and CO2 catalyzed by magnetic nanoparticle-supported porphyrin. Catal. Commun. 2011, 12, 684–688. [Google Scholar] [CrossRef]
- Mikkelsen, M.; Jørgensen, M.; Krebs, F.C. The teraton challenge. A review of fixation and transformation of carbon dioxide. Energy Environ. Sci. 2010, 3, 43–81. [Google Scholar] [CrossRef]
- Kirschning, A.; Solodenko, W.; Mennecke, K. Combining enabling techniques in organic synthesis: Continuous flow processes with heterogenized catalysts. Chem. Eur. J. 2006, 12, 5972–5990. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Bonrath, W.; Tagliapietra, S.; Speranza, C.; Calcio Gaudino, E.; Barge, A. Intensification of organic reactions with hybrid flow-reactors. Chem. Eng. Process. Process Intensif. 2010, 49, 930–935. [Google Scholar] [CrossRef]
- Gedye, R.; Smith, F.; Westaway, K.; Ali, H.; Baldisera, L.; Laberge, L.; Rousell, J. The use of microwave ovens for rapid organic synthesis. Tetrahedron Lett. 1986, 27, 279–282. [Google Scholar] [CrossRef]
- Giguere, R.J.; Bray, T.L.; Duncan, S.M.; Majetich, G. Application of commercial microwave ovens to organic synthesis. Tetrahedron Lett. 1986, 27, 4945–4948. [Google Scholar] [CrossRef]
- De La Hoz, A.; Loupy, A. Microwaves in Organic Synthesis, 3rd ed.; Springer: New York, NY, USA, 2012. [Google Scholar]
- Moseley, J.D.; Kappe, C.O. A critical assessment of the greenness and energy efficiency of microwave-assisted organic synthesis. Green Chem. 2011, 13, 794–806. [Google Scholar] [CrossRef]
- Durka, T.; Gerven, T.V.; Stankiewicz, A. Microwaves in heterogeneous gas-phase catalysis: Experimental and numerical approaches. Chem. Eng. Technol. 2009, 32, 1301–1312. [Google Scholar] [CrossRef]
- Cravotto, G.; Cintas, P. The combined use of microwaves and ultrasound: Improved tools in process chemistry and organic synthesis. Chem. Eur. J. 2007, 13, 1902–1909. [Google Scholar] [CrossRef] [PubMed]
- Taktak, F.; Bulduk, I. Microwave-assisted hydrogenation of codeine in aqueous media. ISRN Org. Chem. 2012. [Google Scholar] [CrossRef] [PubMed]
- Petricci, E.; Mann, A.; Schoenfelder, A.; Rota, A.; Taddei, M. Microwaves make hydroformylation a rapid and easy process. Org. Lett. 2006, 8, 3725–3727. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.A.; Fan, X.; Buchwald, S.L. Carbonylation of aryl chlorides with oxygen nucleophiles at atmospheric pressure. Preparation of phenyl esters as acyl transfer agents and the direct preparation of alkyl esters and carboxylic acids. J. Org. Chem. 2008, 73, 7096–7101. [Google Scholar] [CrossRef] [PubMed]
- Friis, S.D.; Skrydstrup, T.; Buchwald, S.L. Mild Pd-catalyzed aminocarbonylation of (hetero)aryl bromides with a palladacycle precatalyst. Org. Lett. 2014, 16, 4296–4299. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, M.; Cravotto, G.; Georgakopoulos, S.; Heropoulos, G.; Martina, K.; Tagliapietra, S. Pd/C-catalyzed aerobic oxidative esterification of alcohols and aldehydes: A highly efficient microwave-assisted green protocol. Beilstein J. Org. Chem. 2014, 10, 1454–1461. [Google Scholar] [CrossRef] [PubMed]
- Bergamelli, F.; Iannelli, M.; Marafie, J.A.; Moseley, D. A commercial continuous flow microwave reactor evaluated for scale-up. Org. Process Res. Dev. 2010, 14, 926–930. [Google Scholar] [CrossRef]
- Rinaldi, L.; Carnaroglio, D.; Rotolo, L.; Cravotto, G. A microwave-based chemical factory in the lab: From milligram to multigram preparations. J. Chem. 2015. [Google Scholar] [CrossRef]
- Choedkiatsakul, I.; Ngaosuwan, K.; Assabumrungrat, S.; Mantegna, S.; Cravotto, G. Biodiesel production in a novel continuous flow microwave reactor. Renew. Energy 2015, 83, 25–29. [Google Scholar] [CrossRef]
- Matsuzawa, M.; Togashi, S. Microwaves in Catalysis: Methodology and Applications; Wiley-VCH: Weinheim, Gremany, 2015; pp. 141–154. [Google Scholar]
- Schaffner, B.; Schaffner, V.; Verevkin, S.P.; Borner, A. Organic carbonates as solvents in synthesis and catalysis. Chem. Rev. 2010, 110, 4554–4581. [Google Scholar] [CrossRef] [PubMed]
- Nüchter, M.; Ondruschka, B.; Hoffmann, J.; Tied, A. Microwave-assisted reactions of oxiranes with carbon dioxide in ionic liquids. Stud. Surf. Sci. Catal. 2004, 153, 131–136. [Google Scholar]
- Leadbeater, N.E.; Torenius, H.M.; Tye, H. Microwave–promoted organic synthesis using ionic liquids: A mini review. Comb. Chem. High Throughput Screen. 2004, 7, 511–528. [Google Scholar] [CrossRef]
- Ono, F.; Oiao, K.; Tomida, D.; Yokoyama, C.J. Rapid synthesis of cyclic carbonates from CO2 and epoxides under microwave irradiation with controlled temperature and pressure. Mol. Catal. A Chem. 2007, 263, 223–226. [Google Scholar] [CrossRef]
- Dharmanm, M.M.; Hye-Ji Choi, H.J.; Park, S.W.; Park, D.W. Microwave assisted synthesis of cyclic carbonate using homogeneous and heterogeneous ionic liquid catalysts. Top. Catal. 2010, 53, 462–469. [Google Scholar]
- Dharman, M.M.; Choi, H.J.; Kim, D.W.; Park, D.W. Synthesis of cyclic carbonate through microwave irradiation using silica-supported ionic liquids: Effect of variation in the silica support. Catal. Today 2011, 164, 544–547. [Google Scholar] [CrossRef]
- Palmisano, G.; Bonrath, W.; Boffa, L.; Garella, D.; Barge, A.; Cravotto, G. Heck Reactions with Very Low Ligandless Catalyst Loads Accelerated by Microwaves or Simultaneous Microwaves/Ultrasound Irradiation. Adv. Synth. Catal. 2007, 349, 2338–2344. [Google Scholar] [CrossRef]
- Tharun, J.; Mathai, G.; Kathalikkattil, A.C.; Roshan, R.; Kwaka, J.Y.; Park, D.W. Microwave-assisted synthesis of cyclic carbonates by a formic acid/KI catalytic system. Green Chem. 2013, 15, 1673–1677. [Google Scholar] [CrossRef]
- Tharun, J.; Kathalikkattil, A.C.; Roshan, R.; Kang, D.H.; Woo, H.C.; Park, D.W. Microwave-assisted, rapid cycloaddition of allyl glycidyl ether and CO2 by employing pyridinium-based ionic liquid catalysts. Catal. Commun. 2014, 54, 31–34. [Google Scholar] [CrossRef]
- Chun, Y.; He, Y.G.; Zhu, J.H. Microwave-assisted synthesis of dimethyl carbonate. React. Kinet. Catal. Lett. 2001, 74, 23–27. [Google Scholar] [CrossRef]
- Du, J.; Shi, J.; Li, Z.; Liu, Z.; Fan, X.; Tao, C. Ionic liquid mediated CO2 activation for DMC synthesis. J. Nat. Gas Chem. 2012, 21, 476–479. [Google Scholar] [CrossRef]
- Carnaroglio, D.; Martina, K.; Palmisano, G.; Penoni, A.; Domini, C.; Cravotto, G. One-pot sequential synthesis of isocyanates and urea derivatives via a microwave-assisted Staudinger-aza-Wittig reaction. Beilstein J. Org. Chem. 2013, 9, 2378–2386. [Google Scholar] [CrossRef] [PubMed]
- Mägerlein, W.; Indolese, A.F.; Beller, M. A More Efficient Catalyst for the Carbonylation of Chloroarenes. Angew. Chem. Int. Ed. 2001, 40, 2856–2859. [Google Scholar] [CrossRef]
- Brennführer, A.; Neumann, H.; Beller, M. Palladium-Catalyzed Carbonylation Reactions of Aryl Halides and Related Compounds. Angew. Chem. Int. Ed. 2009, 48, 4114–4133. [Google Scholar] [CrossRef] [PubMed]
- Kormos, C.M.; Leadbeater, N.E. Microwave-promoted hydroxycarbonylation in water using gaseous carbon monoxide and pre-pressurized reaction vessels. Synlett 2006, 11, 1663–1666. [Google Scholar] [CrossRef]
- Iannelli, M.; Bergamelli, F.; Kormos, C.M.; Paravisi, S.; Leadbeater, N.E. Application of a batch microwave unit for scale-up of alkoxycarbonylation reactions using a near-stoichiometric loading of carbon monoxide. Org. Process Res. Dev. 2009, 13, 634–637. [Google Scholar] [CrossRef]
- Kormos, C.M.; Leadbeater, N.E. Alkoxycarbonylation reaction performed using near stoichiometric quantities of CO. Synlett 2007, 13, 2006–2010. [Google Scholar] [CrossRef]
- Kormos, C.M.; Leadbeater, N.E. Alkoxycarbonylation of aryl iodides using gaseous carbon monoxide and pre-pressurized reaction vessels in conjunction with microwave heating. Org. Biomol. Chem. 2007, 5, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Kappe, C.O.; Stadler, A.; Dallinger, D. Microwaves in Organic and Medicinal Chemistry, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2013; pp. 41–81. ISBN 3527647848. [Google Scholar]
- Salvadori, J.; Balducci, E.; Zaza, S.; Petricci, E.; Taddei, M. Microwave-assisted carbonylation and cyclocarbonylation of aryl iodides under ligand free heterogeneous catalysis. J. Org. Chem. 2010, 75, 1841–1847. [Google Scholar] [CrossRef] [PubMed]
- Liptrot, D.; Alcaraz, L.; Roberts, B. New synthesis of aryl and heteroaryl N-acylureas via microwave-assisted palladium-catalysed carbonylation. Adv. Synth. Catal. 2010, 352, 2183–2188. [Google Scholar] [CrossRef]
- Pizzetti, M.; Russo, A.; Petricci, E. Microwave-assisted aminocarbonylation of ynamides by using catalytic [Fe3(CO)12] at low pressures of carbon monoxide. Chem. Eur. J. 2011, 17, 4523–4528. [Google Scholar] [CrossRef] [PubMed]
- Calcio Gaudino, E.; Carnaroglio, D.; Martina, K.; Palmisano, G.; Penoni, A.; Cravotto, G. Highly efficient microwave-assisted CO aminocarbonylation with a recyclable Pd(II)/TPP-β-cyclodextrin cross-linked catalyst. Org. Process Res. Dev. 2015, 19, 499–505. [Google Scholar] [CrossRef]
- Tlili, A.; Blondiaux, E.; Frogneux, X.; Cantat, T. Reductive functionalization of CO2 with amines: An entry to formamide, formamidine and methylamine derivatives. Green Chem. 2015, 17, 157–168. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Han, Z.; Zhao, X.; Wang, Z.; Ding, K. Highly Efficient Ruthenium-Catalyzed N-Formylation of Amines with H2 and CO2. Angew. Chem. Int. Ed. 2015, 54, 6186–6189. [Google Scholar] [CrossRef] [PubMed]
- Takada, Y.; Foo, S.W.; Yamazakia, Y.; Saito, S. Catalytic fluoride triggers dehydrative oxazolidinone synthesis from CO2. RSC Adv. 2014, 4, 50851–50857. [Google Scholar] [CrossRef]
- Higashimae, S.; Tamai, T.; Nomoto, A.; Ogawa, A. Selective Thiolative Lactonization of Internal Alkynes Bearing a Hydroxyl Group with Carbon Monoxide and Organic Disulfides Catalyzed by Transition-Metal Complexes. J. Org. Chem. 2015, 80, 7126–7133. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Wu, L.; Zhang, X.; Mei, H.; Gu, Y.; Li, G. Palladium supported on triphenylphosphine functionalized porous organic polymer: A highly active and recyclable catalyst for alkoxycarbonylation of aryl iodides. J. Mol. Catal. A Chem. 2015, 398, 164–169. [Google Scholar] [CrossRef]
- Chen, J.; Neumann, H.; Beller, M.; Wu, X.-F. Palladium-catalyzed synthesis of isoindoloquinazolinones via dicarbonylation of 1,2-dibromoarenes. Org. Biomol. Chem. 2014, 12, 5835–5838. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Xia, C.; Huang, H. Palladium-catalyzed oxidative aminocarbonylation: A new entry to amides via C-H activation. Org. Lett. 2013, 15, 3370–3373. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, Y.; Higashimae, S.; Tamai, T.; Ogawa, A. A highly selective cobalt-catalyzed carbonylative cyclization of internal alkynes with carbon monoxide and organic thiols. Tetrahedron 2013, 69, 11197–11202. [Google Scholar] [CrossRef]
- Natte, K.; Neumann, H.; Wu, X.-F. Pd/C as an efficient heterogeneous catalyst for carbonylative four-component synthesis of 4(3H)-quinazolinones. Catal. Sci. Technol. 2015, 5, 4474–4480. [Google Scholar] [CrossRef]
- Mallia, C.J.; Baxendale, I.R. The use of gases in flow synthesis. Org. Process Res. Dev. 2015. [Google Scholar] [CrossRef] [Green Version]
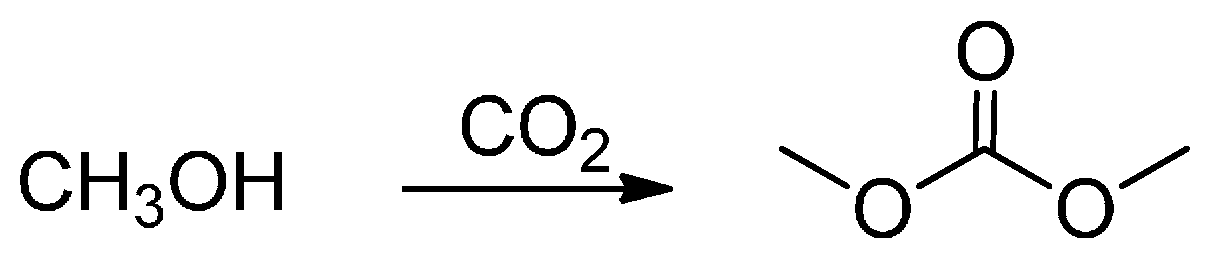
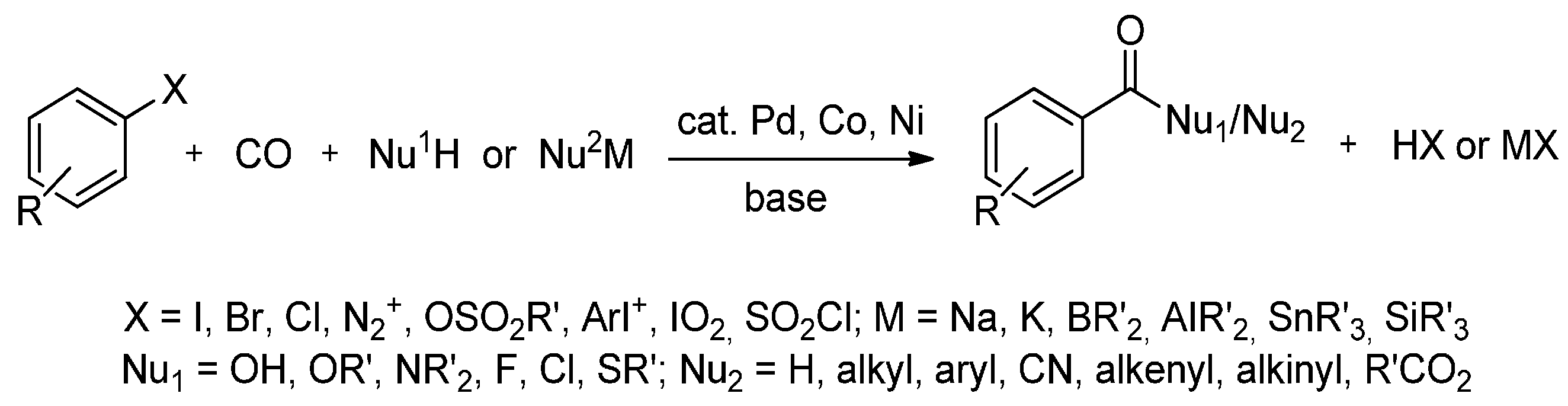
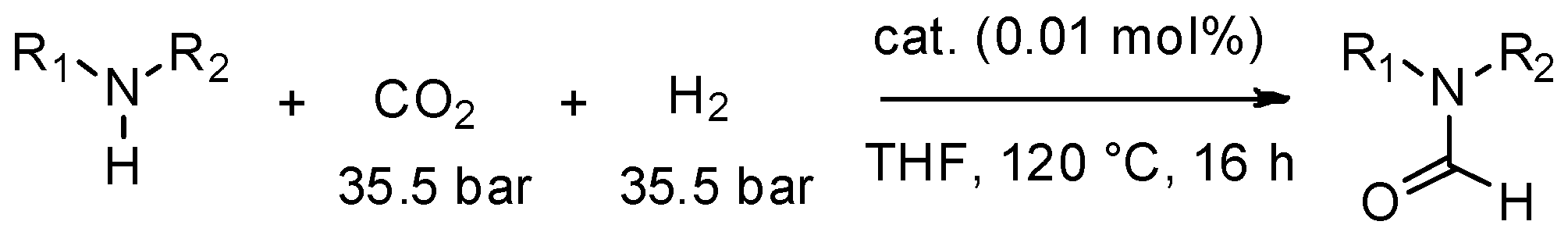

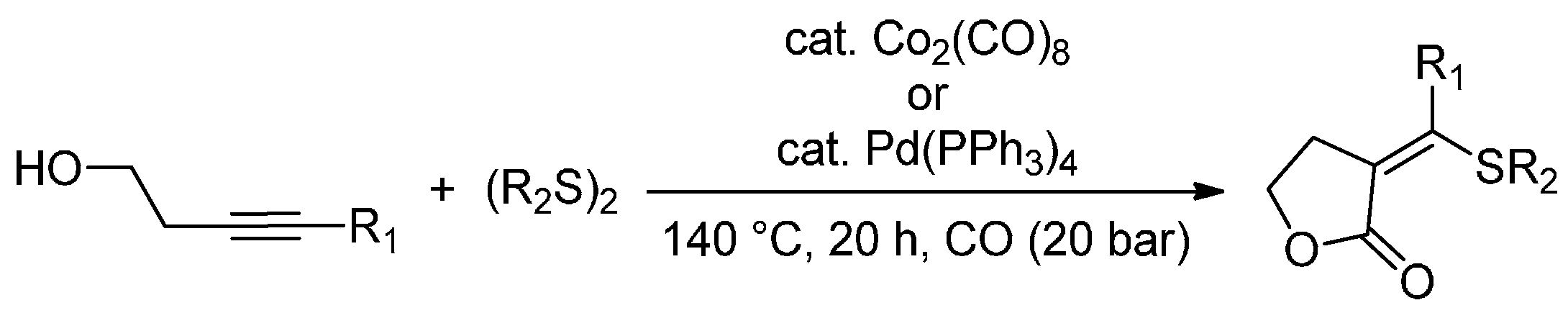


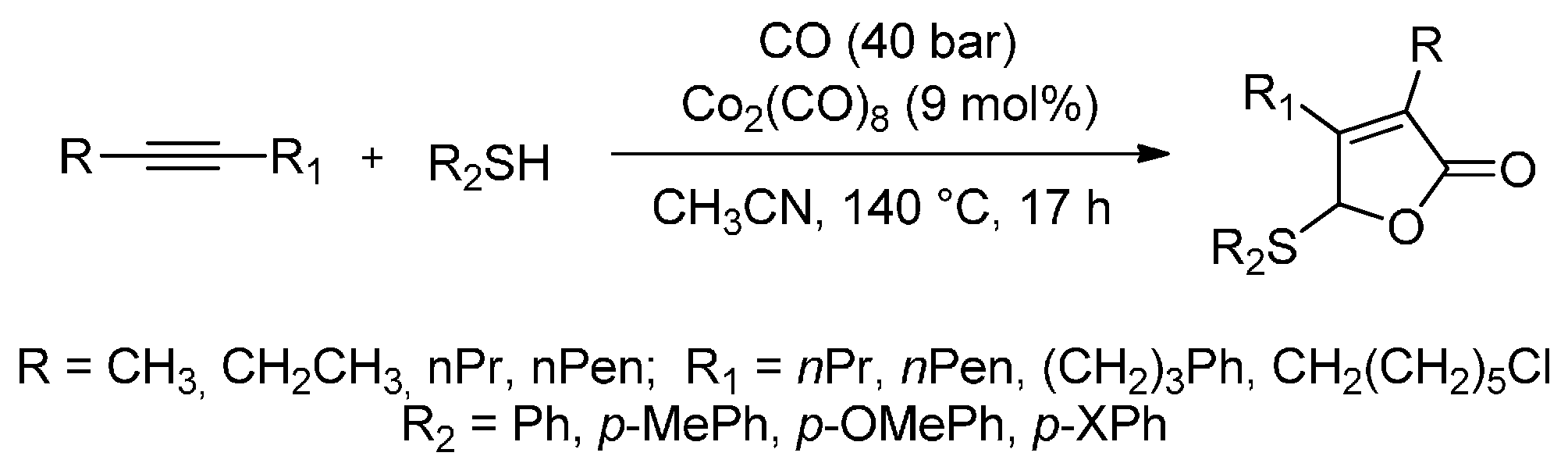

| Entry | Catalytic System | MW (Watt) | Temp. (°C) | CO2 (bar) | Time (min) | Yield (%) | Conv. (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | [C6MIm]Br (0.05 mol %) | / | 140 | 10 | 60 | 86 | 97 | [56] |
| 2 | ZnPO (0.043 mol %), C4NBr (2.174 mol %) | 300 | 120 | 30 | 60 | 97 | / | [58] |
| 3 | TABC (0.027 mol %) | 200 | 120–160 | 9.7 | 30 | 91.4 | / | [59] |
| 4 | SiO2/C4ImBr (4 wt %) | 100 | 120–160 | 9.7 | 15 | 87.9 | / | [59] |
| 5 | HCOOH (0.145 mol %), KI (0.016 mol %) | 200 | / | 9.6 | 15 | 59 | 60 | [62] |
| 6 | C4PyBr (0.054 mol %) | 200 | 0-180 | 9.6 | 0.5 | 67.2 | 70 | [63] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calcio Gaudino, E.; Rinaldi, L.; Rotolo, L.; Carnaroglio, D.; Pirola, C.; Cravotto, G. Heterogeneous Phase Microwave-Assisted Reactions under CO2 or CO Pressure. Molecules 2016, 21, 253. https://doi.org/10.3390/molecules21030253
Calcio Gaudino E, Rinaldi L, Rotolo L, Carnaroglio D, Pirola C, Cravotto G. Heterogeneous Phase Microwave-Assisted Reactions under CO2 or CO Pressure. Molecules. 2016; 21(3):253. https://doi.org/10.3390/molecules21030253
Chicago/Turabian StyleCalcio Gaudino, Emanuela, Laura Rinaldi, Laura Rotolo, Diego Carnaroglio, Camillo Pirola, and Giancarlo Cravotto. 2016. "Heterogeneous Phase Microwave-Assisted Reactions under CO2 or CO Pressure" Molecules 21, no. 3: 253. https://doi.org/10.3390/molecules21030253









