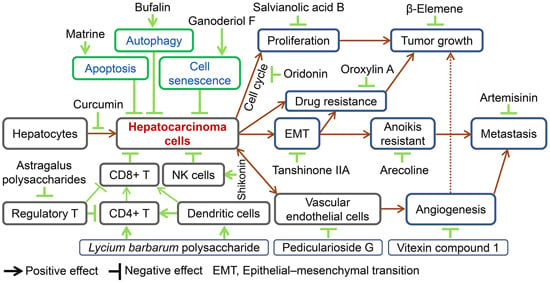
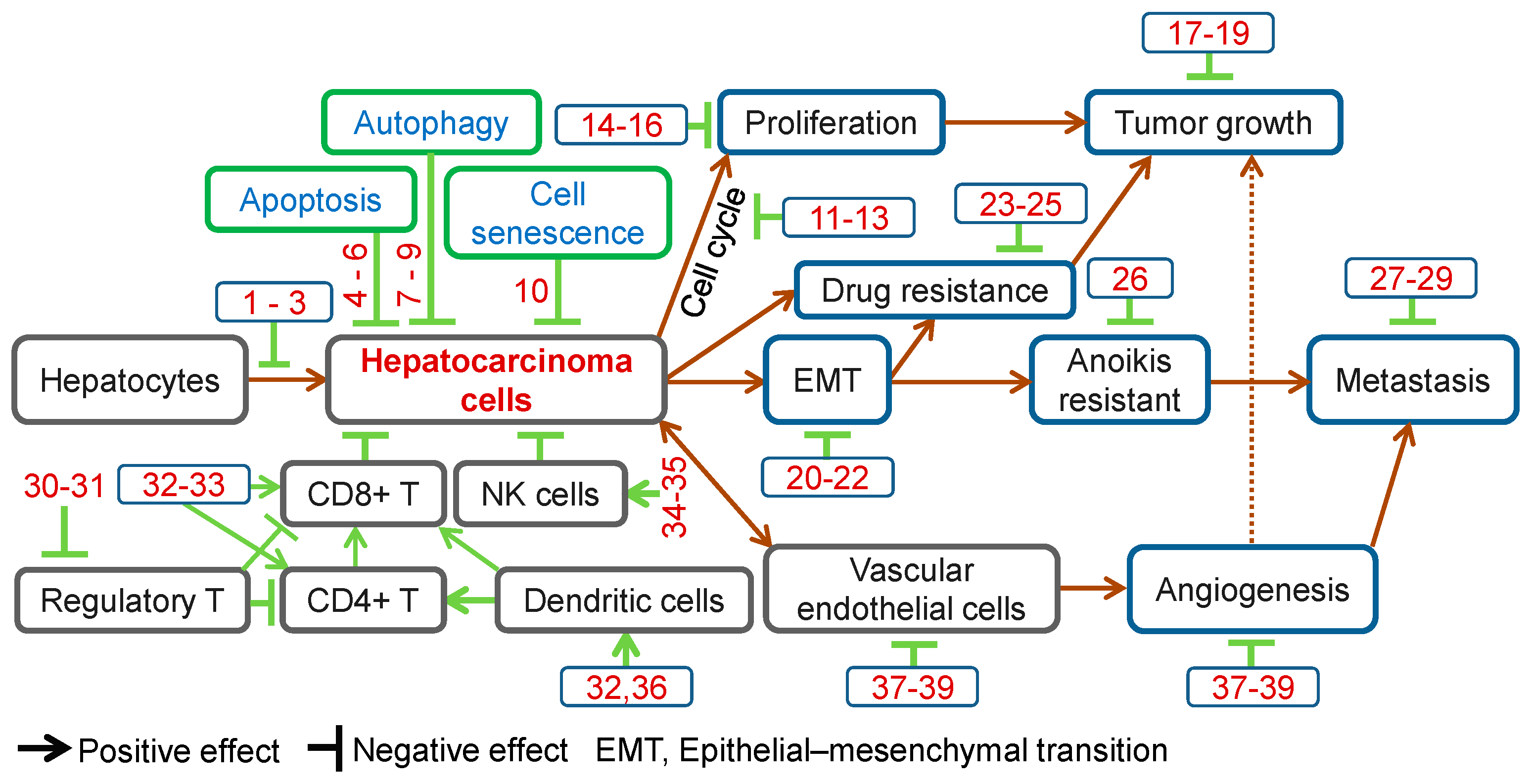
Preventive and Therapeutic Effects of Chinese Herbal Compounds against Hepatocellular Carcinoma
Abstract
:1. Introduction
2. Prevention of Hepatocarcinogenesis
| Compounds | Herbs | Effects | Targets/Molecular Events | Ref. |
|---|---|---|---|---|
| Ursolic acid | Gardenia jasminoides Ellis (Zhi-Zi), Prunella vulgaris (Xia-Ku-Cao), Hedyotis diffusa Willd. (Bai-Hua-She-She-Cao), etc. | ↓ DEN induced hepatocarcinogenesis | ↓ Oxidative stress | [5,6] |
| Penta-acetyl geniposide | Gardenia jasminoides Ellis (Zhi-Zi) | ↓ AFB1 induced hepatocarcinogenesis | ↓ GGT foci | [7] |
| Curcumin | Curcuma kwangsiensis (Yu-Jin or Er-Zhu), C. phaeocaulis (Yu-Jin or Er-Zhu), C. wenyujin (Yu-Jin or Er-Zhu), C. longa (Yu-Jin or Jiang-Huang), etc. | ↓ DEN induced hepatocarcinogenesis | ↓ p21(ras), PCNA and CDC2 | [8] |
| Berberine | Coptis chinensis Franch. (Huang-Lian), Phellodendron chinense Schnied. (Huang-Bai) | ↓ DEN-plus-PB induced hepatocyte proliferation | ↓ iNOS, cytochrome P450, CYP2E1 and CYP1A2 | [9] |
| Saikosaponin-d | Bupleurum chinense (Chai-Hu) | ↓ DEN induced hepatocarcinogenesis | ↓ COX-2 and C/EBPβ | [10] |
| Gomisin A | the fruits of Schisandra chinensis or Schisandra sphenanthera (Wu-Wei-Zi) | ↓ 3′-MeDAB induced hepatocarcinogenesis | Unknown | [11] |
| Tea polyphenols and tea pigments | Tea | ↓ DEN induced hepatocarcinogenesis | ↑ p21WAF1 and Bax, ↓ Bcl-2 | [12] |
| Astragalosides, Astragalus polysaccharide and salvianolic acids | Astragalus membranaceous (Huang-Qi), Salvia miltiorrhiza Bunge (Dan-shen) | ↓ DEN induced hepatocarcinogenesis | ↓ GST-P and α-SMA | [13] |
3. Inhibition of Cell Proliferation
4. Induction of Apoptosis
| Compounds | Herbs | Effects | Targets/Molecular Events | Ref. | ||
|---|---|---|---|---|---|---|
| Salvianolic acid B | Salvia miltiorrhiza Bunge (Dan-Shen) | ↓ HepG2 cell proliferation | ↓ CYP3A4 and CYP1A2, ↑ GST | [16] | ||
| Steroidal saponins | Dioscorea bulbifera (Huang-Du or Huang-Yao-Zi) | ↓ SMMC7721 and Bel-7402 cell proliferation | Unknown | [17] | ||
| Davidiin | Polygonum capitatum (Tou-Hua-Liao) | ↓ Hepatocellular tumor growth | ↓ EZH2 | [18] | ||
| β-Elemene | Curcuma kwangsiensis or C. phaeocaulis or C. wenyujin (E-Zhu) | ↓ H22 tumor growth | ↑ Histone H1 | [19] | ||
| Ardipusilloside-I | Ardisia pusilla (Jiu-Jie-Long) | ↓ SMMC-7721 tumor growth; ↓ invasion and metastasis in HCC | Unknown; ↓ MMP-9 and -2, ↑ Rac1 and E-cadherin | [20,30] | ||
| Raddeanin A | Anemone raddeana Regel (Liang-Tou-Jian) | ↓ H22 tumor growth | Unknown | [21] | ||
| Indole-3-acetonitrile-4-methoxy-2-C-β- d-glucopyranoside | Isatis indigotica (Song-Lan) | ↓ HepG2 cell proliferation | Unknown | [22] | ||
| Pinocembrin-7- O-[3-O-galloyl-4′′,6′′-hexahydroxydiphenoyl]-β-glucose and thonningianins A | Penthorum chinense Pursh (Che-Gen-Cai) | ↓ Hepatocarcinoma cell growth | Unknown | [23] | ||
| 20(R),22(xi),24(S)-dammar-25(26)-ene-3beta,6 alpha,12 beta,20,22,24-hexanol | Panax ginseng (Ren-Shen) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at the G1 phase | ↑ p53 phosphorylation, activate caspase-3 | [25] | ||
| 20(R)-ginsenoside Rg3 | Panax ginseng (Ren-Shen) | ↑ apoptosis, ↓ liver cancer growth | ↓ PCNA, ↑ TNF | [26] | ||
| Gypenoside | Gynostemma pentaphyllum (Jiao-Gu-Lan) | ↓ proliferation, ↑ apoptosis in Hep3B and HA22T cells | Unknown | [27] | ||
| Isorhamnetin | Hippophae rhamnoides (Sha-Ji) | ↓ proliferation, ↑ apoptosis in Bel-7402 cells | Unknown | [28] | ||
| Liquiritigenin | Glycyrrhiza uralensis (Gan-Cao) | ↑ apoptosis, ↓ H22 tumor growth | Unknown | [29] | ||
| N-butylidenephthalide | Angelica sinensis (Dang-Gui) | ↑ apoptosis in HepG2 and J5 cells, ↓ cell and tumor growth | ↑ Nurr1, NOR-1, Nur77, CREB, caspase-9 and caspase-3, ↓ phosphor-AKT | [31] | ||
| Lycium barbarum polysaccharide | Lycium barbarum (Gou-Qi) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at S phase in QGY7703 cells | ↑ Intracellular Ca2+ | [32] | ||
| Apigenin | Eclipta prostrate (Mo-Han-Lian), etc. | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2/M phase in Huh7 cells | ↑ 1336 genes, ↓ 428 genes | [33] | ||
| Icariin | Epimedium brevicornum Maxim. (Yin-Yang-Huo) | ↑ apoptosis in SMMC-7721 cells | ↑ ROS, JNK, Bax/Bcl-2 and caspase | [34] | ||
| Icaritin | Epimedium brevicornum Maxim. (Yin-Yang-Huo) | ↑ apoptosis in HepG2 cells | ↑ JNK1, Bax/Bcl-2 and caspase-3 | [35] | ||
| Oxymatrine | Sophora flavescens (Ku-Shen) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at S and G2/M phase in SMMC-7721 cells | ↓ Bcl-2, ↑ p53 | [36] | ||
| Scutellarin | Scutellaria baicalensis Georgi (Huang-Qin) | ↓ proliferation, ↑ apoptosis in HepG2 cells | ↓ ROS, STAT3, Bcl-XL and Mcl-1 | [37] | ||
| Sarsasapogenin | Anemarrhena asphodeloides (Zhi-Mu) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2/M phase in HepG2 cells | Unknown | [38] | ||
| Pheophorbide a | Scutellaria barbata (Ban-Zhi-Lian) | ↑ apoptosis in HepG2 and Hep3B cells | ↓ Bcl-2, ↑ pro-caspase 3 and pro-caspase 9 | [39] | ||
| Solamargine | Solanum nigrum (Long-Kui) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2/M phase in SMMC-7721 and HepG2 cells | ↑ caspase-3 | [40] | ||
| Ponicidin | Rabdosia rubescens (Dong-Ling-Cao) | ↓ proliferation, ↑ apoptosis in QGY-7701 and HepG-2 cells | ↓ Survivin and Bcl-2, ↑ Bax | [41] | ||
| Paeonol | Paeonia suffruticosa (Mu-Dan-Pi) | ↓ tumor growth, ↑ apoptosis in HepA-hepatoma bearing mice | ↓ Bcl-2, ↑ Bax, IL-2 and TNF-alpha | [42] | ||
| Cryptotanshinone, dihydrotanshinone, tanshinone I, tanshinone IIA | Salvia miltiorrhiza Bunge (Dan-shen) | ↑ apoptosis in HepG2 cells | ↑ ROS | [43] | ||
| Resveratrol-4-O-d-(2′-galloyl)-glucopyranoside | Polygonum cuspidatum (Hu-Zhang) | ↓ proliferation, ↑ apoptosis in SMMC-7721 cells | ↑ caspase-3 and -9, p-JNK, ↓ p-ERK | [44] | ||
| Tubeimoside I | Bolbostemma paniculatum (Tu-Bei-Mu) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2/M phase in HepG2 cells | ↑ caspase-3 and -9, Bax/Bcl-2 | [45] | ||
| Norcantharidin | Mylabris (Ban-Mao) | ↓ proliferation, ↑ apoptosis in HepG2 cells | ↑ ROS, caspase-3 and -9, and Bax, ↓ Bcl-2 | [46] | ||
| Resveratrol-4-O-d-(2′-galloyl)-glucopyranoside | Polygonum cuspidatum (Hu-Zhang) | ↓ proliferation, ↑ apoptosis in SMMC-7721 cells | ↑ caspase-3 and -9, p-JNK, ↓ p-ERK | [44] | ||
| Toosendanin | Melia toosendan (Chuan-Lian-Zi) | ↓ proliferation, ↑ apoptosis in SMMC-7721 and Hep3B cells | ↑ Bax, ↓ Bcl-2 | [47] | ||
| Honokiol | Magnolia officinalis (Hou-Po) | ↑ apoptosis in liver cancer cells | ↓ Bcl-X(L), Bcl-2, procaspase-3 and -9, ↑ MAPK and active caspase-3 | [48] | ||
| Magnolol | Magnolia officinalis (Hou-Po) | ↑ apoptosis in HepG2 cells | ↑ caspase-3, -8, and -9, ↓ Bcl-2 | [49] | ||
| Oleanolic acid and ursolic acid | The fruit of Ligustrum lucidum Ait. (Nü-zhen-zi), Salvia chinensis (Shi-Jian-Chuan), Hedyotis diffusa Willd. (Bai-Hua-She-She-Cao), etc. | ↓ proliferation and adhesion, ↑ apoptosis in liver cancer cells | ↑ caspase-3 and -8, ↓ Na(+)-K(+)-ATPase activity, VEGF and ICAM-1 | [50] | ||
| Chrysophanol | Rheum palmatum L. or R.tanguticum Maxim.ex Balf. or R.officinale Baill. (Da-Huang) | ↓ proliferation, ↑ necrosis in J5 cells | ↓ ATP level, ↑ ROS and lactate dehydrogenase activity | [51] | ||
| Rhein | Rheum palmatum L. or R.tanguticum Maxim.ex Balf. or R.officinale Baill. (Da-Huang) | ↑ apoptosis in HepG2 cells; ↓ proliferation, ↑ apoptosis, arrest cell cycle at S phase in Bel-7402 cells | ↑ caspase-3; ↑ caspase-3, ↓ c-Myc | [52,53] | ||
| Vitexin compound 1 | Vitex negundo(Huang-Jing) | ↓ proliferation, ↑ apoptosis in liver cancer cells | ↑ caspase-3, -8 and -9, FOXO3a, Bim, TRAIL, DR4 and DR5, ↓ phosphorylation of AKT and ERK1/2 | [54] | ||
| Quercetin | Bupleurum chinense (Chai-Hu), Euphorbia lunulata Bunge (Mao-Yan-Cao) and Taxillus chinensis (Sang-Ji-Sheng), etc. | ↓ proliferation, ↑ apoptosis in HA22T/VGH cells | ↑ ROS | [55] | ||
| Gambogic acid | Garcinia hanburyi (Teng-Huang) | ↓ proliferation, ↑ apoptosis in SMMC-7721 cells | ↑ Bax, ↓ Bcl-2 | [56] | ||
| Flavonoids | Polygoni Orientalis Fructus (Shui-Hong-Hua-Zi ) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at S phase in SMMC-7721 cells | Unknown | [57] | ||
| TSP02 | Ardisia japonica (Zi-Jin-Niu) | ↓ proliferation, migration and invasiveness, ↑ apoptosis in HepG2 cells | ↓ CDK1, 2, 4, and TGF-beta1, ↑ Caspase-8 and E-cadherin | [58] | ||
| Bufothionine | Bufonis Venenum (Chan-Su) | ↓ proliferation, ↑ arrest cell cycle at G2/M phase in hepatocarcinoma cells | Unknown | [59] | ||
| Oridonin | Rabdosia rubescens (Dong-Ling-Cao) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2/M phase in HepG2 cells | ↑ p-JNK, p-p38, p-p53, p21, cyclin B1/p-Cdc2 (Tyr15), caspase-9 and -3, ↓ p-ERK | [60] | ||
| Curcumol | Curcuma kwangsiensis or C. phaeocaulis or C. wenyujin (E-Zhu) | ↓ proliferation, arrest cell cycle at G1 phase in HepG2 cells | ↑ pRB1, cyclin D1, CDK2, CDK8, p27KIP1, p53 and p21WAF1, ↓ cyclin A1 | [61] | ||
| Saikosaponin d | Bupleurum chinense (Chai-Hu) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G1 phase in HepG2 and Hep 3B cells | ↑ p53, p21/WAF1, Fas/APO-1, mFasL, sFasL, Bax and IkappaBalpha, ↓ NF-kappaB and Bcl-XL | [62] | ||
| Waltonitone | Gentiana waltonii (Chang-Geng-Qin-Jiao) | ↓ proliferation, arrest cell cycle at S phase in Bel-7402 cells | ↑ Akt and ERK1/2 phosporylation | [63] | ||
| Nobiletin | Citrus aurantium (Zhi-Shi) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2 phase in SMMC-7721 cells | ↑ Bax and caspase-3, ↓ Bcl-2 and COX-2 | [64] | ||
| Matrine | Sophora flavescens (Ku-Shen) | ↓ proliferation, ↑ apoptosis and autophagy, arrest cell cycle at G1 phase in HCC cells; ↓ invasion in SMMC-7721 cells | ↑ Bax/Bcl-2 and Beclin 1; ↓ MMP-9 and NF-κB | [65,66,67] | ||
| Berberine | Coptis chinensis Franch. (Huang-Lian) or Phellodendron chinense Schnied. (Huang-Bai) | ↑ apoptosis, arrest cell cycle at G1 phase in HuH7 cells; ↑ apoptosis and autophagy in HepG2 cells | ↓ PCNA, Bid and Bcl-2, ↑ caspase-3 and -7; ↑ AMPK, ↓ mTORC1 | [68,69] | ||
| Baicalein | Scutellaria baicalensis Georgi (Huang-Qin) | ↑ apoptosis, arrest cell cycle at G2/M phase in J5 cells; ↓ proliferation, ↑ apoptosis and autophagy in SMMC7721 cells | ↑ caspase-9 and -3, and Bax/Bcl-2 ratio; ↑ Βeclin 1, ↓ CD147 | [70,71] | ||
| Oroxylin-A | Scutellaria baicalensis Georgi (Huang-Qin) | ↓ proliferation, ↑ apoptosis and autophagy in HepG2 cells; reverse drug resistance and enhance apoptosis inducing effect of Paclitaxel in drug resistant HepG2 cells | Induction of Bax translocation, activation and oligomerization, ↑ Βeclin 1, ↓ PI3K-PTEN-Akt-mTOR signaling pathway; ↓ Integrinβ1 | [72,73,74] | ||
| Shikonin | Lithospermum erythrorhizon (Zi-Cao) | ↑ apoptosis in Huh7 and BEL7402 cells; ↑ autophagy in HCC cells; ↓ proliferation and migratory ability on HepJ5 and Mahlavu cells | ↑ ROS, ↓ Akt and RIP1/NF-κB; ↑ ROS and ERK, ↓ RIP pathway; ↓ MMP-2 and -9, vimnetin, AKT and IκB phosphorylation, NF-κB | [75,76,77] | ||
| Curcumin | Curcuma kwangsiensis (Yu-Jin or Er-Zhu), C. phaeocaulis (Yu-Jin or Er-Zhu), C. wenyujin (Yu-Jin or Er-Zhu), C. longa (Yu-Jin or Jiang-Huang), etc. | ↑ apoptosis in Huh7 cells; ↑ apoptosis and autophagy in HepG2 cells; ↓ proliferation, EMT and migration in hypoxic HepG2 cells | ↑ p38, FasL and caspase-3; ↑ caspase-3, ↓ Bcl-2/Bax; ↓ HIF-1alpha | [78,79,80] | ||
| Resveratrol | Polygonum cuspidatum (Hu-Zhang), etc. | ↓ proliferation, ↑ apoptosis in Hepa 1-6 cells; ↓ proliferation, ↑ apoptosis and autophagy, arrest cell cycle at S phase in HuH7 cells; ↓ invasion in HCC cells | ↑ROS and caspase-3; ↑ p21/WAF1, Atg5, Atg7, Atg9, and Atg12, ↓ cyclin E, cyclin A, CDK2, phospho-ERK and phospho-p38; ↓ MMP-9 | [81,82,83,84] | ||
| Bufalin and cinobufagin | Toad skin and venom | ↑ apoptosis in HepG2 cells (Bufalin and cinobufagin); ↑ apoptosis and autophagy in HepG2 cells (Bufalin) | ↑ Fas, Bax and Bid, caspase-3, -8, -9 and -10, ↓ Bcl-2 (Bufalin and cinobufagin; ↑ Βeclin 1 and AMPK phosphorylation, ↓ p62 and mTOR signaling (Bufalin) | [85,86] | ||
| Tetrandrine | Stephania tetrandra (Han-Fang-Ji) | ↓ proliferation, ↑ apoptosis and autophagy, arrest cell cycle at G2/m phase in liver cancer cells | ↑ ROS, ERK MAP kinase and ATG7, ↓ Akt | [87,88,89] | ||
| Arenobufagin | Toad venom | ↑ apoptosis and autophagy in HepG2 cells | ↑ Bax/Bcl-2, ↓ PI3K/Akt/mTOR pathway | [90] | ||
| Allicin | Garlic | ↓ proliferation, ↑ autophagy in HepG2 cells | ↑ AMPK/TSC2 and Beclin-1 signaling, ↓ p53, the PI3K/mTOR signaling and Bcl-2 | [91] | ||
| Galangin | Alpinia officinarum Hance (Gao-Liang-Jiang) | ↓ proliferation, ↑ apoptosis and autophagy in HepG2 cells | ↑ p53 | [92] | ||
| Kaempferol | Euphorbia lunulata Bunge. (Mao-Yan-Cao) | ↓ proliferation, ↑ autophagy, arrest cell cycle at G2/M phase in SK-HEP-1 cells | ↑ p-AMPK, LC3-II, Atg 5, Atg 7, Atg 12 and beclin 1, ↓CDK1, cyclin B, p-AKT and p-mTOR | [93] | ||
| EGCG | Tea | Inhibit autophagy to enhance anti-cancer effects of doxorubicin in Hep3B cells | ↓ Βeclin 1 and Atg5 | [94] | ||
| Elemene injection | Curcuma kwangsiensis or C. phaeocaulis or C. wenyujin (E-Zhu) | Induce autophagy and prevent HepG2 cells from undergoing apoptosis | ↓ Bcl-2/Bax and LC3 I/LC3 II ratio | [95] | ||
| Ganoderiol F | Ganoderma amboinense (Lu-Jiao-Ling-Zhi) | ↓ proliferation, ↑ cell senescence in HepG2 cells | ↑ EKR and p16 | [96] | ||
| Arecoline | Areca catechu L. (Bing-Lang) | ↑ anoikis in HA22T/VGH cells | ↑ Bax, caspase-3 and Rho/Rock activation, ↓ beta1-integrin, IL-6, STAT3 and p190RhoGAP phosphorylation, SHP2, Bcl-XL and Bcl-2 | [97] | ||
| Tanshinone II-A | Salvia miltiorrhiza Bunge (Dan-shen) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G0/G1 phase in SMMC-7721 cells; ↓ EMT and metastasis in HCC; ↓ migration and invasion in HCC cells | ↓ Bcl-2 and c-myc, ↑ Fas, Bax and p53; ↑ VEGFR1/PDGFR; ↓ MMP-2 and -9, NF-κB | [98,99,100] | ||
| Dihydroartemisinin | Artemisia annua L. (Qing-Hao) | ↓ proliferation, ↑ apoptosis, arrest cell cycle at G2/M phase in HCC cells; ↓ invasion and metastasis in HCC cells | ↑ p21, caspase-9 and -3 , Noxa and active Bak, ↓ cyclin B, CDC25C and Mcl-1; ↓ MMP2, ↑ TIMP2, Cdc42 and E-cadherin | [101,102] | ||
| Cordycepin | Cordyceps sinensis (Dong-Chong-Xia-Cao) | ↓ proliferation, EMT and migration/invasion | ↓ integrin α3, integrin α6, integrin β1 and phosphorylated FAK | [103] | ||
| Polysaccharides | Huaier | ↓ proliferation, EMT, adhesion, migration and invasion in MHCC97-H cells | ↓ AEG-1 | [104] | ||
| Platycodin D | Platycodon grandiflorum (Jie-Geng) | ↓ proliferation, adhesion, migration and invasion in HCC cells | ↑ Bax, ↓ survivin | [105] | ||
| Isofraxidin | Acanthopanax senticosus (Ci-Wu-Jia) | ↓ invasion in HCC cells | ↓ MMP-7 and ERK1/2 | [106] | ||
| β-Ionone | Aucklandia lappa Decne or Vladimiria souliei (Franch.) Ling (Mu-Xiang) | ↓ invasion, migration and adhesion in SK-Hep-1 cells | ↓ MMP-2 and -9, urokinase-type plasminogen activator activities, FAK, Rho, Rac1 and Cdc42, ↑ TIMP-1 and -2, plasminogen activator inhibitor-1 and nm23-H1 | [107] | ||
| Hesperidin | Citrus reticulata Blanco (Chen-Pi) | ↓ acetaldehyde-induced cell invasion in HepG2 cells | ↓ MMP-9, NF-kappaB, AP-1, JNK, and p38 signaling pathways | [108] | ||
| Astragalosides, astragalus polysaccharide and salvianolic acids | Astragalus membranaceous (Huang-Qi), Salvia miltiorrhiza Bunge (Dan-shen) | ↓ TGF-beta(1)-induced cell invasion in HepG2 cells | Modulating TGF-beta/Smad signaling | [109] | ||
| Astragalus polysaccharides | Astragalus membranaceous (Huang-Qi) | ↑anti-tumor effect of Adriamycin in H22 hepatocarcinoma | ↑ IL-1α, IL-2, IL-6 and TNF-α, ↓ IL-10 and MDR1 | [110] | ||
| Tetramethylpyrazine | Ligusticum chuanxiong Hort (Chuan-Xiong) | Reverse multidrug resistance in BEL-7402/ADM cells | ↓ MDR1, MRP2, MRP3 and MRP5 | [111] | ||
| Epicatechin gallate and epigallocatechin gallate | Tea | Increase intracellular DOX accumulation and enhance DOX-induced cell killing activities against BEL-7404/DOX cells | ↓ MDR1 | [112] | ||
| Hedyotiscone A | Hedyotis corymbosa (San-Fang-Hua-Er-Cao) | ↑ apoptosis in multidrug-resistant hepatocellular carcinoma cells | ↑ caspases-3, -7 and -9 | [113] | ||
| Polyphyllin D | Paris polyphylla Sm. (Chong-Lou) | ↑ apoptosis in multi-drug resistant HepG2 cells | Mitochondrial dysfunction | [114] | ||
| Ursolic acid | Gardenia jasminoides Ellis (Zhi-Zi ), Prunella vulgaris (Xia-Ku-Cao), Hedyotis diffusa Willd. (Bai-Hua-She-She-Cao), etc. | ↑ apoptosis in doxorubicin-resistant human hepatoma cells | ↑ Bak and apoptosis-inducing factor | [115] | ||
| Pseudolaric acid B | Pseudolarix kaempferi (Tu-Jin-Pi) | ↑ apoptosis and arrest cell cycle at G2/M phase in conventional and P-gp-overexpressing hepatocarcinoma cells | Disrupts cellular microtubule networks and inhibits the formation of mitotic spindles | [116] | ||
| Imperatorin | Angelica dahurica (Bai-Zhi) | ↑ apoptosis in multidrug-resistant liver cancer cells | ↑ proteosome-dependent Mcl-1 degradation to release Bak and Bax | [117] | ||
5. Cell Cycle Arrest
6. Induction of Autophagy
7. Induction of Cell Senescence
8. Induction of Anoikis
9. Inhibition of Epithelial-Mesenchymal Transition (EMT)
10. Inhibition of Metastasis
11. Targeting Drug Resistance
12. Regulation of Immune Function
| Compounds | Herbs | Effects | Targets/Molecular Events | Ref. |
|---|---|---|---|---|
| Lycium barbarum polysaccharide | Lycium barbarum (Gou-Qi) | ↑ CD4+ and CD8+ T cells in H22 hepatoma; promote dendritic cells to stimulate allogeneic lymphocyte proliferation, produce IL-12p70 and IFN-γ | Unknown; NF-κB | [138,144] |
| Polysaccharides | Artemisia annua L. (Huang-Hua-Hao) | ↑ CD4+ and CD8+ T cells, IFN-γ and IL-4 secretion, and induce cancer cell apoptosis in human hepatoma 7402 bearing mice | Unknown | [139] |
| Gastrodin | Gastrodia elata Blume (Tian-Ma) | ↑ cytotoxic activities of NK and CD8+ T cells against H22 cells | ↑ NF-κB, IL-2 and Bcl-2 in CD4+ T cells | [140] |
| Shikonin | Lithospermum erythrorhizon (Zi-Cao) | ↑ CD3+ and CD19+ lymphocytes, NK activities and IL-2 in HepA22 bearing mice | Unknown | [141] |
| Proteins extract | Omphalia lapidesces (Lei-Wan) | ↑ spleen mass and IFN-γ production in H22 hepatocarcinoma bearing mice | Unknown | [142] |
| Gekko sulfated polysaccharide-protein complex | Gekko swinhonis Guenther (Tian-Long) | Restore the defective biorheological characteristics of dendritic cells mediated by SMMC-7721 cells | Unknown | [145] |
| Astragalus polysaccharides | Astragalus membranaceous (Huang-Qi) | ↓ proliferation and migration in CD4+ CD25+ Treg cells | ↓ Foxp3, SDF-1 or its receptor through the CXCR4/CXCL12 pathway | [146] |
| Polysaccharide | Radix Glycyrrhizae (Gan-Cao) | ↓ Tregs cells | ↓ Foxp3 | [147] |
| Gekko-sulfated glycopeptide | Gekko swinhonis Guenther (Tian-Long) | ↓ bFGF stimulated proliferation and migration of endothelial cells, angiogenesis and tumor growth in liver cancer | ↓ bFGF secretion and binding to heparin/heparan sulfate | [148] |
| Pedicularioside G | Pedicularis striata (Ma-Xian-Hao) | ↓ proliferation and migration in HUVEC cells, and angiogenesis in chicken embryo chorioallantoic membrane and hepatoma | ↓ reactive oxygen species | [149] |
| Vitexin compound 1 | Vitex negundo (Huang-Jing) | ↓ proliferation and cell cycle arrest at G1/G0 in hepatocellular carcinoma cells, and HUVEC tube formation | ↓ VEGF | [150] |
| Resveratrol | Polygonum cuspidatum (Hu-Zhang), etc. | ↓ proliferation in liver cancer cells | ↓ hypoxia-induced activation of ERK1/2 and Akt, HIF-1α and VEGF expression | [151,152] |
| Cinobufotalin, Panax notoginseng saponins, Ginsenosides Rg3 and Lentinan | Bufonis Venenum (Chan-Su), Panax notoginseng (San-Qi), P. ginseng (Ren-Shen), Lentinula edodes (Xiang-Gu) | ↓ angiogenesis and tumor growth in H22 hepatocellular carcinoma | ↓ VEGF, EGFR and MMP-2 expression | [153] |
13. Inhibition of Angiogenesis
14. Herbal Compound-Based Combinational Treatment
| Compounds | Herbs | Effects | Targets/Molecular Events | Ref. |
|---|---|---|---|---|
| Astragalosides, Astragalus polysaccharide and salvianolic acids | Astragalus membranaceous (Huang-Qi), Salvia miltiorrhiza Bunge (Dan-shen) | ↓ DEN induced hepatocarcinogenesis; ↓ TGF-β1-induced cell invasion in HepG2 cells | ↓ GST-P and α-SMA; modulating TGF-β/Smad signaling | [13,109] |
| Cinobufotalin, Panax notoginseng saponins, Ginsenosides Rg3 and Lentinan | Bufonis Venenum (Chan-Su), Panax notoginseng (San-Qi), Panax ginseng (Ren-Shen), Lentinula edodes (Xiang-Gu) | ↓ angiogenesis and tumor growth in H22 hepatocellular carcinoma | ↓ VEGF, EGFR and MMP-2 expression | [153] |
| Curcumin and resveratrol | Curcuma kwangsiensis (Yu-Jin or Er-Zhu) or C. phaeocaulis (Yu-Jin or Er-Zhu) or C. wenyujin (Yu-Jin or Er-Zhu) or C. longa (Yu-Jin or Jiang-Huang), and Polygonum cuspidatum (Hu-Zhang), etc. | ↓ proliferation, ↑ apoptosis in Hepa1-6 cells | ↓ XIAP and Survivin, ↑ ROS production, caspase-3, -8 and -9 | [158] |
| Cinobufotalin, Cantharidin, Panax notoginseng saponins, Tanshinone, Ginsenosides Rg3 and Lentinan | Bufonis Venenum (Chan-Su), Mylabris phalerata Pallas or M. cichorii Linnaeus (Ban-Mao), Panax notoginseng (San-Qi), Salvia miltiorrhiza Bunge (Dan-Shen), Panax ginseng (Ren-Shen), Lentinula edodes (Xiang-Gu) | Inhibit tumor growth, prolong survival time, enhance anticancer effects and reduce toxicity of cisplatin in hepatocellular carcinoma bearing mice | Unknown | [159] |
| Acetylshikonin and β,β-dimethylacrylshikonin | Lithospermum erythrorhizon (Zi-Cao) | ↓ proliferation, ↑ apoptosis, arrest cell cycle in G2/M phase in SMMC-7721 cells | ↓ Bcl-2 and Bcl-2/Bax ratio | [162] |
15. Conclusions and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Forner, A.; Llovet, J.M.; Bruix, J. Hepatocellular carcinoma. Lancet 2012, 379, 1245–1255. [Google Scholar] [CrossRef]
- Tang, Z.Y. Combination of traditional Chinese medicine and western medicine in the treatment of liver cancer. J. Clin. Hepatol. 2011, 27, 449–450. (In Chinese) [Google Scholar]
- Wu, M.C. Traditional Chinese medicine in prevention and treatment of liver cancer: Function, status and existed problems. J. Chin. Integr. Med. 2003, 1, 163–164. (In Chinese) [Google Scholar] [CrossRef]
- Hu, B.; Wang, S.S.; Du, Q. Traditional Chinese Medicine for the prevention and treatment of hepatocarcinoma: From bench to bedside. World J. Hepatol. 2015, 7, 1209–1232. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.C.; Song, Y.J.; Zhang, J.; Jiao, Y.B.; Feng, L.L.; Bai, H.F.; Wang, X.P.; Cai, D.Y.; Wang, Y.Q. Effect of ursolic acid on DEN-induced hepatic precancerous lesions in mice. Chin. J. Integr. Tradit. West. Med. Liver Dis. 2012, 22, 287–292. (In Chinese) [Google Scholar]
- Gayathri, R.; Priya, D.K.; Gunassekaran, G.R.; Sakthisekaran, D. Ursolic acid attenuates oxidative stress-mediated hepatocellular carcinoma induction by diethylnitrosamine in male Wistar rats. Asian Pac. J. Cancer Prev. 2009, 10, 933–938. [Google Scholar] [PubMed]
- Lin, Y.L.; Hsu, J.D.; Chou, F.P.; Lee, M.J.; Shiow, S.J.; Wang, C.J. Suppressive effect of penta-acetyl geniposide on the development of γ-glutamyl transpeptidase foci-induced by aflatoxin B1 in rats. Chem. Biol. Interact. 2000, 128, 115–126. [Google Scholar] [CrossRef]
- Chuang, S.E.; Kuo, M.L.; Hsu, C.H.; Chen, C.R.; Lin, J.K.; Lai, G.M.; Hsieh, C.Y.; Cheng, A.L. Curcumin-containing diet inhibits diethylnitrosamine-induced murine hepatocarcinogenesis. Carcinogenesis 2000, 21, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, J.J.; Wang, X.; Bu, X.Y.; Lou, Y.Q.; Zhang, G.L. Effect of berberine on hepatocyte proliferation, inducible nitric oxide synthase expression, cytochrome P450 2E1 and 1A2 activities in diethylnitrosamine- and phenobarbital-treated rats. Biomed. Pharmacother. 2008, 62, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.L.; He, S.X.; Ren, M.D.; Wang, Y.L.; Zhang, Y.X.; Liu, E.Q. Chemopreventive effect of saikosaponin-d on diethylinitrosamine-induced hepatocarcinogenesis: Involvement of CCAAT/enhancer binding protein β and cyclooxygenase-2. Mol. Med. Rep. 2012, 5, 637–644. [Google Scholar] [PubMed]
- Nomura, M.; Nakachiyama, M.; Hida, T.; Ohtaki, Y.; Sudo, K.; Aizawa, T.; Aburada, M.; Miyamoto, K.I. Gomisin A, a lignan component of Schizandora fruits, inhibits development of preneoplastic lesions in rat liver by 3′-methyl-4-dimethylamino-azobenzene. Cancer Lett. 1994, 76, 11–18. [Google Scholar] [CrossRef]
- Jia, X.; Han, C.; Chen, J. Studies on the inhibitory effects of tea polyphenols and tea pigments on liver precancerous lesion in rats. J. Hyg. Res. 2001, 30, 168–169. (In Chinese) [Google Scholar]
- Rui, W.; Xie, L.; Liu, X.; He, S.; Wu, C.; Zhang, X.; Zhang, L.; Yang, Y. Compound Astragalus and Salvia miltiorrhiza extract suppresses hepatocellular carcinoma progression by inhibiting fibrosis and PAI-1 mRNA transcription. J. Ethnopharmacol. 2014, 151, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.Q.; Chen, Z.Y.; Qin, G.Z.; Qin, L.L. Effects of twelve herbs on Aflatoxin B1 induced hepatocarcinogensis in rats. J. Guangxi Med. Coll. 1986, 3, 21–23. (In Chinese) [Google Scholar]
- Ruan, C.C.; Liang, Y.; Liu, Z.H. Inhibition of 12 Chinese Traditional Medicinal herbs on mutagenic effects induced by Aflatoxin B1. Chin. J. Cancer 1987, 8, 29–31. (In Chinese) [Google Scholar]
- Wang, Q.L.; Wu, Q.; Tao, Y.Y.; Liu, C.H.; El-Nezami, H. Salvianolic acid B modulates the expression of drug-metabolizing enzymes in HepG2 cells. Hepatobiliary Pancreat. Dis. Int. 2011, 10, 502–508. [Google Scholar] [CrossRef]
- Liu, H.; Chou, G.X.; Wang, J.M.; Ji, L.L.; Wang, Z.T. Steroidal saponins from the rhizomes of Dioscorea bulbifera and their cytotoxic activity. Planta Med. 2011, 77, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ma, J.; Chow, S.C.; Li, C.H.; Xiao, Z.; Feng, R.; Fu, J.; Chen, Y. A potential antitumor ellagitannin, davidiin, inhibited hepatocellular tumor growth by targeting EZH2. Tumour. Biol. 2014, 35, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Bao, F.; Qiu, J.; Zhang, H. Potential role of β-elemene on histone H1 in the H22 ascites hepatoma cell line. Mol. Med. Rep. 2012, 6, 185–190. [Google Scholar] [PubMed]
- Tao, X.; Wang, P.; Yang, X.; Yao, H.; Liu, J.; Cao, Y. Inhibitory effect of ardipusilloside-I on Lewis pulmonary carcinoma and hepatocarcinoma SMMC-7721. J. Chin. Med. Mater. 2005, 28, 574–577. (In Chinese) [Google Scholar]
- Wang, M.K.; Ding, L.S.; Wu, F.E. Antitumor effects of raddeanin A on S180, H22 and U14 cell xenografts in mice. Chin. J. Cancer 2008, 27, 910–913. (In Chinese) [Google Scholar]
- Wu, Y.; Zhang, Z.X.; Hu, H.; Li, D.; Qiu, G.; Hu, X.; He, X. Novel indole C-glycosides from Isatis indigotica and their potential cytotoxic activity. Fitoterapia 2011, 82, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Jiang, M.H.; Jiang, J.G.; Zhang, R.F.; Zhang, M.W. Isolation and identification of compounds from Penthorum chinense Pursh with antioxidant and antihepatocarcinoma properties. J. Agric. Food Chem. 2012, 60, 11097–11103. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xu, S.S.; Chen, J.W.; Wang, Y.; Xu, H.Q.; Fan, N.B.; Li, X. Anti-tumor activity of Annona squamosa seeds extract containing annonaceous acetogenin compounds. J. Ethnopharmacol. 2012, 142, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Tang, X.H.; Ikejima, T.; Sun, X.J.; Wang, X.B.; Xi, R.G.; Wu, L.J. A new triterpenoid from Panax ginseng exhibits cytotoxicity through p53 and the caspase signaling pathway in the HepG2 cell line. Arch. Pharm. Res. 2008, 31, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Guan, Y.; Zhou, X.; Sun, L.; Liu, Y.; He, Q.; Fu, L.; Mao, Y.Q. Anticarcinogenic effect of 20(R)-ginsenoside Rg3 on induced hepatocellular carcinoma in rats. J. Sichuan Univ. Med. Sci. Edit. 2005, 36, 217–220. (In Chinese) [Google Scholar]
- Chen, J.C.; Chung, J.G.; Chen, L.D. Gypenoside induces apoptosis in human Hep3B and HA22T tumour cells. Cytobios. 1999, 100, 37–48. [Google Scholar] [PubMed]
- Teng, B.S.; Lu, Y.H.; Wang, Z.T.; Tao, X.Y.; Wei, D.Z. In vitro anti-tumor activity of isorhamnetin isolated from Hippophae rhamnoides L. against BEL-7402 cells. Pharmacol. Res. 2006, 54, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Higo, H.; Cai, Y. Inhibition of hepatoma 22 tumor by Liquiritigenin. Phytother. Res. 2010, 24, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Lou, L.; Ye, W.; Chen, Y.; Wu, S.; Jin, L.; He, J.; Tao, X.; Zhu, J.; Chen, X.; Deng, A.; Wang, J. Ardipusilloside inhibits survival, invasion and metastasis of human hepatocellular carcinoma cells. Phytomedicine 2012, 19, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Jian, M.H.; Lin, C.C.; Kang, J.C.; Chen, S.P.; Lin, P.C.; Hung, P.J.; Chen, J.R.; Chang, W.L.; Lin, S.Z.; et al. The induction of orphan nuclear receptor Nur77 expression by n-butylenephthalide as pharmaceuticals on hepatocellular carcinoma cell therapy. Mol. Pharmacol. 2008, 74, 1046–1058. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chen, H.; Huang, J.; Li, Z.; Zhu, C.; Zhang, S. Effect of lycium barbarum polysaccharide on human hepatoma QGY7703 cells: Inhibition of proliferation and induction of apoptosis. Life Sci. 2005, 76, 2115–2124. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Zhao, X.L.; Liu, A.W.; Nian, H.; Zhang, S.H. Apigenin inhibits hepatoma cell growth through alteration of gene expression patterns. Phytomedicine 2011, 18, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Dong, P.; Wang, J.; Zhang, J.; Gu, J.; Wu, X.; Wu, W.; Fei, X.; Zhang, Z.; Wang, Y.; et al. Icariin, a natural flavonol glycoside, induces apoptosis in human hepatoma SMMC-7721 cells via a ROS/JNK-dependent mitochondrial pathway. Cancer Lett. 2010, 298, 222–230. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Wang, Y.; Duan, F.; Jiang, H.; Chen, M.F.; Tang, S.Y. Icaritin induces apoptosis of HepG2 cells via the JNK1 signaling pathway independent of the estrogen receptor. Planta Med. 2010, 76, 1834–1839. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Luo, Q.; Qin, J.; Wang, L.; Shi, Y.; Sun, C. Effects of oxymatrine on proliferation and apoptosis in human hepatoma cells. Colloids Surf. B Biointerfaces 2006, 48, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zhang, S. Scutellarin-induced apoptosis in HepG2 hepatocellular carcinoma cells via a STAT3 pathway. Phytother. Res. 2013, 27, 1524–1528. [Google Scholar] [CrossRef] [PubMed]
- Bao, W.; Pan, H.; Lu, M.; Ni, Y.; Zhang, R.; Gong, X. The apoptotic effect of sarsasapogenin from Anemarrhena asphodeloides on HepG2 human hepatoma cells. Cell Biol. Int. 2007, 31, 887–892. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.Y.; Tang, P.M.; Hon, P.M.; Au, S.W.; Tsui, S.K.; Waye, M.M.; Kong, S.K.; Mak, T.C.; Fung, K.P. Pheophorbide a, a major antitumor component purified from Scutellaria barbata, induces apoptosis in human hepatocellular carcinoma cells. Planta Med. 2006, 72, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Zhu, F.S.; Li, M.; Gao, S.G. Induction of apoptosis in human hepatoma SMMC-7721 cells by solamargine from Solanum nigrum L. J. Ethnopharmacol. 2012, 139, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.F.; Liu, P.Q.; Chen, G.H.; Lu, M.Q.; Cai, C.J.; Yang, Y.; Li, H. Ponicidin inhibits cell growth on hepatocellular carcinoma cells by induction of apoptosis. Dig. Liver Dis. 2007, 39, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.P.; Wang, H.; Xu, S.P.; Shen, Y.X.; Wu, Q.; Chen, Z.D.; Wei, W. Anti-tumor effects of paeonol in a HepA-hepatoma bearing mouse model via induction of tumor cell apoptosis and stimulation of IL-2 and TNF-alpha production. Eur. J. Pharmacol. 2008, 584, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.Y.; Liu, K.W.; Yeung, J.H. Reactive oxygen species-mediated kinase activation by dihydrotanshinone in tanshinones-induced apoptosis in HepG2 cells. Cancer Lett. 2009, 28, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Yang, Y.; Wang, Z.; Chen, F.; Zhang, A.; Liu, C. Resveratrol-4-O-d-(2′-galloyl)-glucopyranoside isolated from Polygonum cuspidatum exhibits anti-hepatocellular carcinoma viability by inducing apoptosis via the JNK and ERK pathway. Molecules 2014, 19, 1592–1602. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Deng, L.; Zhong, H.; Wang, Y.; Jiang, X.; Chen, J. Natural plant extract tubeimoside I promotes apoptosis-mediated cell death in cultured human hepatoma (HepG2) cells. Biol. Pharm. Bull. 2011, 34, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Zhu, Y.Q.; Mei, J.J.; Liu, S.Q.; Luo, J. Involvement of mitochondrial pathway in NCTD-induced cytotoxicity in human hepG2 cells. J. Exp. Clin. Cancer Res. 2010, 29, 145. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, J.; Liu, X.; Zhang, L.; Yi, G.; Li, C.; He, X.; Wang, P.; Jiang, H. Toosendanin inhibits hepatocellular carcinoma cells by inducing mitochondria-dependent apoptosis. Planta Med. 2010, 76, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Qian, Y.; Geng, L.; Chen, J.; Wang, X.; Xie, H.; Yan, S.; Jiang, G.; Zhou, L.; Zheng, S. Involvement of p38 mitogen-activated protein kinase pathway in honokiol-induced apoptosis in a human hepatoma cell line (hepG2). Liver Int. 2008, 28, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.Y.; Chang, Y.T.; Liu, J.D.; Yu, C.H.; Ho, Y.S.; Lee, Y.H.; Lee, W.S. Molecular mechanisms of apoptosis induced by magnolol in colon and liver cancer cells. Mol. Carcinog. 2001, 32, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.L.; Huang, C.Y.; Wu, S.T.; Yin, M.C. Oleanolic acid and ursolic acid induce apoptosis in four human liver cancer cell lines. Toxicol. in Vitro 2010, 24, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.C.; Yang, J.S.; Huang, A.C.; Hsia, T.C.; Chou, S.T.; Kuo, C.L.; Lu, H.F.; Lee, T.H.; Wood, W.G.; Chung, J.G. Chrysophanol induces necrosis through the production of ROS and alteration of ATP levels in J5 human liver cancer cells. Mol. Nutr. Food Res. 2010, 54, 967–976. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Bian, X.L.; Xu, X.L.; Zhu, B.; Yu, B.; Zhai, Q. Role of mitochondrial permeability transition in human hepatocellular carcinoma Hep-G2 cell death induced by rhein. Fitoterapia 2013, 91, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Huang, Z.; Chen, G. Rhein induces apoptosis and cell cycle arrest in human hepatocellular carcinoma BEL-7402 cells. Am. J. Chin. Med. 2008, 36, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.G.; Zheng, X.X.; Zeng, G.Y.; Zhou, Y.J.; Yuan, H. Purified vitexin compound 1 induces apoptosis through activation of FOXO3a in hepatocellular carcinoma. Oncol. Rep. 2014, 31, 488–496. [Google Scholar] [PubMed]
- Chang, Y.F.; Chi, C.W.; Wang, J.J. Reactive oxygen species production is involved in quercetin-induced apoptosis in human hepatoma cells. Nutr. Cancer 2006, 55, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, L.; You, Q.D.; Nie, F.F.; Gu, H.Y.; Zhao, L.; Wang, X.T.; Guo, Q.L. Differential apoptotic induction of gambogic acid, a novel anticancer natural product, on hepatoma cells and normal hepatocytes. Cancer Lett. 2007, 256, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.R.; Wang, S.; Meng, X.S.; Chou, J.; Yang, X.X. Effect of flavonoids of polygoni orientails fructus on human hepatoma cell line SMMC-7721. J. Chin. Med. Mater. 2013, 36, 255–259. (In Chinese) [Google Scholar]
- Zhao, C.Y.; Hui, L.P.; He, L.; Li, Q. Study on inhibitory effect of triterpenoid saponin from Ardisia japonica TSP02 on proliferation and metastasis of human hepatocellular carcinoma cells and its mechanism. Chin. J. Chin. Mater. Med. 2013, 38, 861–865. (In Chinese) [Google Scholar]
- Xie, R.F.; Li, Z.C.; Gao, B.; Shi, Z.N.; Zhou, X. Bufothionine, a possible effective component in cinobufocini injection for hepatocellular carcinoma. J. Ethnopharmacol. 2012, 141, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ye, Y.; Chui, J.H.; Zhu, G.Y.; Li, Y.W.; Fong, D.W.; Yu, Z.L. Oridonin induces G2/M cell cycle arrest and apoptosis through MAPK and p53 signaling pathways in HepG2 cells. Oncol. Rep. 2010, 24, 647–651. [Google Scholar] [PubMed]
- Huang, L.Z.; Wang, J.; Lu, F.T.; Yang, F.C.; Chen, X.; Hong, X.; Jiang, X.S. Mechanism study on anti-proliferative effects of curcumol in human hepatocarcinoma HepG2 cells. Chin. J. Chin. Mater. Med. 2013, 38, 1812–1815. (In Chinese) [Google Scholar]
- Hsu, Y.L.; Kuo, P.L.; Chiang, L.C.; Lin, C.C. Involvement of p53, nuclear factor kappaB and Fas/Fas ligand in induction of apoptosis and cell cycle arrest by saikosaponin d in human hepatoma cell lines. Cancer Lett. 2004, 213, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Duan, C.; Ding, K.; Wang, Z. WT inhibit human hepatocellular carcinoma BEL-7402 cells growth by modulating Akt and ERK1/2 phosphorylation. Chin. J. Chin. Mater. Med. 2009, 34, 3277–3280. (In Chinese) [Google Scholar]
- Ma, X.; Jin, S.; Zhang, Y.; Wan, L.; Zhao, Y.; Zhou, L. Inhibitory effects of nobiletin on hepatocellular carcinoma in vitro and in vivo. Phytother. Res. 2014, 28, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Wen, S.; Zhan, Y.; He, Y.; Liu, X.; Jiang, J. Anticancer effects of the Chinese medicine matrine on murine hepatocellular carcinoma cells. Planta Med. 2008, 74, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Q.; Li, Y.M.; Liu, T.; He, W.T.; Chen, Y.T.; Chen, X.H.; Li, X.; Zhou, W.C.; Yi, J.F.; Ren, Z.J. Antitumor effect of matrine in human hepatoma G2 cells by inducing apoptosis and autophagy. World J. Gastroenterol. 2010, 16, 4281–4290. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.B.; Zhang, H.F.; Li, D.Y.; Zhang, X.; Xue, H.Z.; Zhao, S.H. Matrine inhibits matrix metalloproteinase-9 expression and invasion of human hepatocellular carcinoma cells. J. Asian Nat. Prod. Res. 2011, 13, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Yip, N.K.; Ho, W.S. Berberine induces apoptosis via the mitochondrial pathway in liver cancer cells. Oncol. Rep. 2013, 30, 1107–1112. [Google Scholar] [PubMed]
- Yu, R.; Zhang, Z.Q.; Wang, B.; Jiang, H.X.; Cheng, L.; Shen, L.M. Berberine-induced apoptotic and autophagic death of HepG2 cells requires AMPK activation. Cancer Cell Int. 2014, 14, 49. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.M.; Tsai, H.C.; Lin, Y.L.; Yang, J.S.; Huang, A.C.; Yang, M.D.; Hsu, S.C.; Chung, M.C.; Gibson Wood, W.; Chung, J.G. Mitochondrial-dependent caspase activation pathway is involved in baicalein-induced apoptosis in human hepatoma J5 cells. Int. J. Oncol. 2009, 35, 717–724. [Google Scholar] [PubMed]
- Zhang, X.; Tang, X.; Liu, H.; Li, L.; Hou, Q.; Gao, J. Autophagy induced by baicalin involves downregulation of CD147 in SMMC-7721 cells in vitro. Oncol. Rep. 2012, 27, 1128–1134. [Google Scholar] [PubMed]
- Liu, W.; Mu, R.; Nie, F.F.; Yang, Y.; Wang, J.; Dai, Q.S.; Lu, N.; Qi, Q.; Rong, J.J.; Hu, R.; et al. MAC-related mitochondrial pathway in oroxylin-A-induced apoptosis in human hepatocellular carcinoma HepG2 cells. Cancer Lett. 2009, 284, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Lu, N.; Hu, C.; Liu, W.; Sun, Y.; Wang, X.; You, Q.; Gu, C.; Xi, T.; Guo, Q. Beclin 1-mediated autophagy in hepatocellular carcinoma cells: Implication in anticancer efficiency of oroxylin A via inhibition of mTOR signaling. Cell Signal. 2012, 24, 1722–1732. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Zhao, L.; Zhu, L.; Wang, H.; Sha, Y.; Yao, J.; Li, Z.; You, Q.; Guo, Q. Oroxylin A reverses CAM-DR of HepG2 cells by suppressing Integrinβ1 and its related pathway. Toxicol. Appl. Pharmacol. 2012, 259, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Gong, K.; Li, W. Shikonin, a Chinese plant-derived naphthoquinone, induces apoptosis in hepatocellular carcinoma cells through reactive oxygen species: A potential new treatment for hepatocellular carcinoma. Free Radic. Biol. Med. 2011, 51, 2259–2271. [Google Scholar] [CrossRef] [PubMed]
- Gong, K.; Zhang, Z.; Chen, Y.; Shu, H.B.; Li, W. Extracellular signal-regulated kinase, receptor interacting protein, and reactive oxygen species regulate shikonin-induced autophagy in human hepatocellular carcinoma. Eur. J. Pharmacol. 2014, 738C, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Wei, P.L.; Tu, C.C.; Chen, C.H.; Ho, Y.S.; Wu, C.T.; Su, H.Y.; Chen, W.Y.; Liu, J.J.; Chang, Y.J. Shikonin suppresses the migratory ability of hepatocellular carcinoma cells. J. Agric. Food Chem. 2013, 61, 8191–8197. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Z.; Li, L.; Liu, M.Y.; Jin, X.B.; Mao, J.W.; Pu, Q.H.; Meng, M.J.; Chen, X.G.; Zhu, J.Y. Curcumin induces FasL-related apoptosis through p38 activation in human hepatocellular carcinoma Huh7 cells. Life Sci. 2013, 92, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Yang, Y.; Wang, X. Curcumin enhanced adriamycin-induced human liver-derived Hepatoma G2 cell death through activation of mitochondria-mediated apoptosis and autophagy. Eur. J. Pharm. Sci. 2011, 43, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.H.; Jiang, M.; Liu, K.G.; Li, X.Q. Curcumin inhibited hypoxia induced epithelial-mesenchymal transition in hepatic carcinoma cell line HepG2 in vitro. Chin. J. Integr. Tradit. West. Med. 2013, 33, 1102–1106. (In Chinese) [Google Scholar]
- Du, Q.; Shen, K.P.; Hu, B.; Deng, S. Effects of resveratrol on apoptosis and ROS production in Hepa 1–6 hepatocarcinoma cells. J. Chin. Med. Mater. 2012, 35, 443–448. (In Chinese) [Google Scholar]
- Liao, P.C.; Ng, L.T.; Lin, L.T.; Richardson, C.D.; Wang, G.H.; Lin, C.C. Resveratrol arrests cell cycle and induces apoptosis in human hepatocellular carcinoma Huh-7 cells. J. Med. Food. 2010, 13, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Chen, Y.L. The effects of Resveratrol on proliferation, apoptosis and invasion of Bel-7404 hepatocarcinoma cell line. J. Fujian Med. Univ. 2007, 41, 509–513. (In Chinese) [Google Scholar]
- Yu, H.B.; Pan, C.E.; Wu, W.J.; Zhao, S.H.; Zhang, H.F. Effects of resveratrol on matrix metalloproteinase-9 expression in hepatoma cells. J. Chin. Integr. Med. 2008, 6, 270–273. (In Chinese) [Google Scholar] [CrossRef]
- Qi, F.; Inagaki, Y.; Gao, B.; Cui, X.; Xu, H.; Kokudo, N.; Li, A.; Tang, W. Bufalin and cinobufagin induce apoptosis of human hepatocellular carcinoma cells via Fas- and mitochondria-mediated pathways. Cancer Sci. 2011, 102, 951–958. [Google Scholar] [CrossRef] [PubMed]
- Miao, Q.; Bi, L.L.; Li, X.; Miao, S.; Zhang, J.; Zhang, S.; Yang, Q.; Xie, Y.H.; Zhang, J.; Wang, S.W. Anticancer effects of Bufalin on human hepatocellular carcinoma HepG2 cells: Roles of apoptosis and autophagy. Int. J. Mol. Sci. 2013, 14, 1370–1382. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Gong, K.; Mao, X.; Li, W. Tetrandrine induces apoptosis by activating reactive oxygen species and repressing Akt activity in human hepatocellular carcinoma. Int. J. Cancer 2011, 129, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.T.; Chiang, L.C.; Lin, Y.T.; Lin, C.C. Antiproliferative and apoptotic effects of tetrandrine on different human hepatoma cell lines. Am. J. Chin. Med. 2006, 34, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Gong, K.; Chen, C.; Zhan, Y.; Chen, Y.; Huang, Z.; Li, W. Autophagy-related gene 7 (ATG7) and reactive oxygen species/extracellular signal-regulated kinase regulate tetrandrine-induced autophagy in human hepatocellular carcinoma. J. Biol. Chem. 2012, 287, 35576–35588. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.M.; Liu, J.S.; Deng, L.J.; Chen, M.F.; Yiu, A.; Cao, H.H.; Tian, H.Y.; Fung, K.P.; Kurihara, H.; Pan, J.X.; et al. Arenobufagin, a natural bufadienolide from toad venom, induces apoptosis and autophagy in human hepatocellular carcinoma cells through inhibition of PI3K/Akt/mTOR pathway. Carcinogenesis 2013, 34, 1331–1342. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.L.; Ho, C.T.; Chung, J.G.; Rajasekaran, R.; Sheen, L.Y. Allicin induces p53-mediated autophagy in Hep G2 human liver cancer cells. J. Agric. Food Chem. 2012, 60, 8363–8371. [Google Scholar] [CrossRef] [PubMed]
- Wen, M.; Wu, J.; Luo, H.; Zhang, H. Galangin induces autophagy through upregulation of p53 in HepG2 cells. Pharmacology 2012, 89, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.W.; Tsai, S.C.; Peng, S.F.; Lin, M.W.; Chiang, J.H.; Chiu, Y.J.; Fushiya, S.; Tseng, M.T.; Yang, J.S. Kaempferol induces autophagy through AMPK and AKT signaling molecules and causes G2/M arrest via downregulation of CDK1/cyclin B in SK-HEP-1 human hepatic cancer cells. Int. J. Oncol. 2013, 42, 2069–2077. [Google Scholar] [PubMed]
- Chen, L.; Ye, H.L.; Zhang, G.; Yao, W.M.; Chen, X.Z.; Zhang, F.C.; Liang, G. Autophagy inhibition contributes to the synergistic interaction between EGCG and doxorubicin to kill the hepatoma Hep3B cells. PLoS ONE 2014, 9, e85771. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wang, K.; Hu, C.; Lin, L.; Qin, S.; Cai, X. Elemene injection induced autophagy protects human hepatoma cancer cells from starvation and undergoing apoptosis. Evid. Based Complement. Altern. Med. 2014, 2014, 637528. [Google Scholar] [CrossRef] [PubMed]
- Chang, U.M.; Li, C.H.; Lin, L.I.; Huang, C.P.; Kan, L.S.; Lin, S.B. Ganoderiol F, a ganoderma triterpene, induces senescence in hepatoma HepG2 cells. Life Sci. 2006, 79, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.L.; Su, S.J.; Huang, L.W.; Hsieh, B.S.; Hu, Y.C.; Hung, T.C.; Chang, K.L. Arecoline induces HA22T/VGH hepatoma cells to undergo anoikis - involvement of STAT3 and RhoA activation. Mol. Cancer 2010, 9, 126. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.L.; Wei, Y.Q.; Wang, X.J.; Xiao, F.; Li, S.F.; Zhang, J. Growth inhibition and apoptosis induction of tanshinone II-A on human hepatocellular carcinoma cells. World J. Gastroenterol. 2004, 10, 2024–2028. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Q.; Liu, L.; Sun, H.C.; Fu, Y.L.; Xu, H.X.; Chai, Z.T.; Zhang, Q.B.; Kong, L.Q.; Zhu, X.D.; Lu, L.; et al. Tanshinone IIA inhibits metastasis after palliative resection of hepatocellular carcinoma and prolongs survival in part via vascular normalization. J. Hematol. Oncol. 2012, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.X.; Feng, T.; Ren, L.; Zheng, C.L. Tanshinone II-A inhibits invasion and metastasis of human hepatocellular carcinoma cells in vitro and in vivo. Tumori. 2009, 95, 789–795. [Google Scholar]
- Zhang, C.Z.; Zhang, H.; Yun, J.; Chen, G.G.; Lai, P.B. Dihydroartemisinin exhibits antitumor activity toward hepatocellular carcinoma in vitro and in vivo. Biochem. Pharmacol. 2012, 83, 1278–1289. [Google Scholar] [CrossRef] [PubMed]
- Weifeng, T.; Feng, S.; Xiangji, L.; Changqing, S.; Zhiquan, Q.; Huazhong, Z.; Peining, Y.; Yong, Y.; Mengchao, W.; Xiaoqing, J.; et al. Artemisinin inhibits in vitro and in vivo invasion and metastasis of human hepatocellular carcinoma cells. Phytomedicine 2011, 18, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.L.; Ko, B.S.; Liu, T.A.; Liang, S.M.; Liu, C.C.; Lu, Y.J.; Tzean, S.S.; Shen, T.L.; Liou, J.Y. Cordycepin suppresses integrin/FAK signaling and epithelial-mesenchymal transition in hepatocellular carcinoma. Anticancer Agents Med. Chem. 2014, 14, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Li, C.; Wu, X.; Liu, M.; Sun, X.; Yang, Y.; Hao, M.; Sheng, S.; Sun, Y.; Zhang, H.; et al. Huaier polysaccharides suppresses hepatocarcinoma MHCC97-H cell metastasis via inactivation of EMT and AEG-1 pathway. Int. J. Biol. Macromol. 2014, 64, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Xu, W.S.; Wu, G.S.; Chen, X.P.; Wang, Y.T.; Lu, J.J. Platycodin D induces apoptosis, and inhibits adhesion, migration and invasion in HepG2 hepatocellular carcinoma cells. Asian Pac. J. Cancer Prev. 2014, 15, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, T.; Tokiwa, T. Isofraxidin, a coumarin component from Acanthopanax senticosus, inhibits matrix metalloproteinase-7 expression and cell invasion of human hepatoma cells. Biol. Pharm. Bull. 2010, 33, 1716–1722. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.S.; Lyu, S.C.; Chen, J.Y.; Hu, M.L. The anti-metastatic efficacy of β-ionone and the possible mechanisms of action in human hepatocarcinoma SK-Hep-1 cells. Br. J. Nutr. 2012, 107, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Yeh, M.H.; Kao, S.T.; Hung, C.M.; Liu, C.J.; Lee, K.H.; Yeh, C.C. Hesperidin inhibited acetaldehyde-induced matrix metalloproteinase-9 gene expression in human hepatocellular carcinoma cells. Toxicol. Lett. 2009, 184, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, Y.; Zhang, X.; Xu, S.; He, S.; Huang, W.; Roberts, M.S. Compound Astragalus and Salvia miltiorrhiza extract inhibits cell invasion by modulating transforming growth factor-β/SMAD in HepG2 cell. J. Gastroenterol. Hepatol. 2010, 25, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.E.; Li, H.D.; Yan, M.; Cai, H.L.; Tan, Q.Y.; Zhang, W.Y. Astragalus polysaccharides can regulate cytokine and P-glycoprotein expression in H22 tumor-bearing mice. World J. Gastroenterol. 2012, 18, 7079–7086. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.B.; Wang, S.S.; Zhang, Q.F.; Liu, M.; Li, H.L.; Liu, Y.; Wang, J.N.; Zheng, F.; Guo, L.Y.; Xiang, J.Z. Inhibition of tetramethylpyrazine on P-gp, MRP2, MRP3 and MRP5 in multidrug resistant human hepatocellular carcinoma cells. Oncol. Rep. 2010, 23, 211–215. [Google Scholar] [PubMed]
- Liang, G.; Tang, A.; Lin, X.; Li, L.; Zhang, S.; Huang, Z.; Tang, H.; Li, Q.Q. Green tea catechins augment the antitumor activity of doxorubicin in an in vivo mouse model for chemoresistant liver cancer. Int. J. Oncol. 2010, 37, 111–123. [Google Scholar] [PubMed]
- Yue, G.G.; Kin-Ming Lee, J.; Cheng, L.; Chung-Lap Chan, B.; Jiang, L.; Fung, K.P.; Leung, P.C.; Bik-San Lau, C. Reversal of P-glycoprotein-mediated multidrug resistance in human hepatoma cells by hedyotiscone A, a compound isolated from Hedyotis corymbosa. Xenobiotica 2012, 42, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Cheung, J.Y.; Ong, R.C.; Suen, Y.K.; Ooi, V.; Wong, H.N.; Mak, T.C.; Fung, K.P.; Yu, B.; Kong, S.K. Polyphyllin D is a potent apoptosis inducer in drug-resistant HepG2 cells. Cancer Lett. 2005, 217, 203–211. [Google Scholar] [CrossRef]
- Yang, L.; Liu, X.; Lu, Z.; Yuet-Wa Chan, J.; Zhou, L.; Fung, K.P.; Wu, P.; Wu, S. Ursolic acid induces doxorubicin-resistant HepG2 cell death via the release of apoptosis-inducing factor. Cancer Lett. 2010, 298, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.K.; Chiu, P.; Chung, S.S.; Chow, L.M.; Zhao, Y.Z.; Yang, B.B.; Ko, B.C. Pseudolaric acid B, a novel microtubule-destabilizing agent that circumvents multidrug resistance phenotype and exhibits antitumor activity in vivo. Clin. Cancer Res. 2005, 11, 6002–6011. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zeng, X.; Sun, J.; Li, H.; Wu, P.; Fung, K.P.; Liu, F. Imperatorin induces Mcl-1 degradation to cooperatively trigger Bax translocation and Bak activation to suppress drug-resistant human hepatoma. Cancer Lett. 2014, 348, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.Y.; Su, C.C. Tanshinone IIA inhibits Hep-J5 cells by increasing calreticulin, caspase 12 and GADD153 protein expression. Int. J. Mol. Med. 2010, 26, 379–385. [Google Scholar] [PubMed]
- Wang, H.C.; Chung, P.J.; Wu, C.H.; Lan, K.P.; Yang, M.Y.; Wang, C.J. Solanum nigrum L. polyphenolic extract inhibits hepatocarcinoma cell growth by inducing G2/M phase arrest and apoptosis. J. Sci. Food Agric. 2011, 91, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.B.; Li, C.H.; Lee, S.S.; Kan, L.S. Triterpene-enriched extracts from Ganoderma lucidum inhibit growth of hepatoma cells via suppressing protein kinase C, activating mitogen-activated protein kinases and G2-phase cell cycle arrest. Life Sci. 2003, 72, 2381–2390. [Google Scholar] [CrossRef]
- Deng, S.; Hu, B.; An, H.M. Traditional Chinese Medicinal Syndromes and treatment in colorectal cancer. J. Cancer Ther. 2012, 3, 888–897. [Google Scholar] [CrossRef]
- Wang, Z.; Han, W.; Sui, X.; Fang, Y.; Pan, H. Autophagy: A novel therapeutic target for hepatocarcinoma (Review). Oncol. Lett. 2014, 7, 1345–1351. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.; Chen, R.; Wang, Z.; Huang, Z.; Kong, N.; Zhang, M.; Han, W.; Lou, F.; Yang, J.; Zhang, Q.; et al. Autophagy and chemotherapy resistance: A promising therapeutic target for cancer treatment. Cell Death Dis. 2013, 4, e838. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.S.; Zhao, H.C.; Yao, S.K.; Zhuo, D.X.; Jin, B.; Lv, D.C.; Wu, C.L.; Ma, D.L.; Gao, C.; Shu, X.M.; et al. Autophagy inhibition enhances etoposide-induced cell death in human hepatoma G2 cells. Int. J. Mol. Med. 2011, 27, 599–606. [Google Scholar] [PubMed]
- Shi, Y.H.; Ding, Z.B.; Zhou, J.; Hui, B.; Shi, G.M.; Ke, A.W.; Wang, X.Y.; Dai, Z.; Peng, Y.F.; Gu, C.Y.; et al. Targeting autophagy enhances sorafenib lethality for hepatocellular carcinoma via ER stress-related apoptosis. Autophagy 2011, 7, 1159–1172. [Google Scholar] [CrossRef] [PubMed]
- Muehlich, S.; Gudermann, T. Pro-senescence therapy for hepatocellular carcinoma. Aging (Albany NY) 2013, 5, 639–640. [Google Scholar]
- Hu, B.; Du, Q.; Deng, S.; An, H.M.; Pan, C.F.; Shen, K.P.; Xu, L.; Wei, M.M.; Wang, S.S. Ligustrum lucidum Ait. fruit extract induces apoptosis and cell senescence in human hepatocellular carcinoma cells through upregulation of p21. Oncol. Rep. 2014, 32, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; An, H.M.; Shen, K.P.; Xu, L.; Du, Q.; Deng, S.; Wu, Y. Liver Yin deficiency tonifying herbal extract induces apoptosis and cell senescence in Bel-7402 human hepatocarcinoma cells. Exp. Ther. Med. 2012, 3, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Han, L.; Zhang, Z.; Li, J.; Qu, Z.; Du, J.; Liang, X.; Liu, Y.; Liu, H.; Shi, Y.; et al. Involvement of anoikis-resistance in the metastasis of hepatoma cells. Exp. Cell Res. 2009, 315, 1148–1156. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; An, H.M.; Shen, K.P.; Song, H.Y.; Deng, S. Polygonum cuspidatum extract induces Anoikis in hepatocarcinoma cells associated with generation of reactive oxygen species and downregulation of focal adhesion kinase. Evid. Based Complement. Altern. Med. 2012, 2012, 607675. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; An, H.M.; Shen, K.P.; Xu, L.; Du, Q.; Deng, S.; Wu, Y. Modified Yi Guan Jian, a Chinese herbal formula, induces anoikis in Bel-7402 human hepatocarcinoma cells in vitro. Oncol. Rep. 2011, 26, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- van Zijl, F.; Zulehner, G.; Petz, M.; Schneller, D.; Kornauth, C.; Hau, M.; Machat, G.; Grubinger, M.; Huber, H.; Mikulits, W. Epithelial-mesenchymal transition in hepatocellular carcinoma. Future Oncol. 2009, 5, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.; Cai, X.; Fan, D. Roles of epithelial-mesenchymal transition in cancer drug resistance. Curr. Cancer Drug Targets 2013, 13, 915–929. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Ren, Z.G.; Qiu, S.J.; Sun, H.C.; Wang, L.; Liu, B.B.; Li, Q.S.; Zhang, W.; Zhu, X.D.; Liu, L.; et al. Residual hepatocellular carcinoma after oxaliplatin treatment has increased metastatic potential in a nude mouse model and is attenuated by Songyou Yin. BMC Cancer 2010, 10, 219. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Yao, W.J.; Zhang, X.L.; Han, X.Q.; Qu, X.Y.; Ka, W.B.; Sun, D.G.; Wu, X.Z.; Wen, Z.Y. Effects of Gekko sulfated polysaccharide-protein complex on human hepatoma SMMC-7721 cells: Inhibition of proliferation and migration. J. Ethnopharmacol. 2010, 127, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Yau, T.; Chan, P.; Epstein, R.; Poon, R.T. Evolution of systemic therapy of advanced hepatocellular carcinoma. World J. Gastroenterol. 2008, 14, 6437–6441. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Xu, D.; Xia, Q.; Wang, P.; Rong, C.; Su, Y. Reversal of P-glycoprotein-mediated multidrug resistance of human hepatic cancer cells by Astragaloside II. J. Pharm. Pharmacol. 2012, 64, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- He, Y.L.; Ying, Y.; Xu, Y.L.; Su, J.F.; Luo, H.; Wang, H.F. Effects of Lycium barbarum polysaccharide on tumor microenvironment T-lymphocyte subsets and dendritic cells in H22-bearing mice. J. Chin. Integr. Med. 2005, 3, 374–377. (In Chinese) [Google Scholar] [CrossRef]
- Chen, J.; Chen, J.; Wang, X.; Liu, C. Anti-tumour effects of polysaccharides isolated from Artemisia annua L by inducing cell apoptosis and immunomodulatory anti-hepatoma effects of polysaccharides. Afr. J. Tradit. Complement. Altern. Med. 2013, 11, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Shu, G.; Yang, T.; Wang, C.; Su, H.; Xiang, M. Gastrodin stimulates anticancer immune response and represses transplanted H22 hepatic ascitic tumor cell growth: Involvement of NF-κB signaling activation in CD4+ T cells. Toxicol. Appl. Pharmacol. 2013, 269, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Long, S.; GuangZhi, Y.; BaoJie, G.; Wei, X.; YanYong, H.; YingLi, W.; Yang, Z.; LiHua, L. Shikonin derivatives protect immune organs from damage and promote immune responses in vivo in tumour-bearing mice. Phytother. Res. 2012, 26, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.T.; Lin, M.A.; Cheng, D.Q.; Shi, Z.J.; Zhu, J.L.; Wu, J. Effect of proteins extracted from mycelia of Omphalia lapidescens on inhibiting H, liver cancer in mice and regulating immune function. J. Chin. Med. Mater. 2009, 32, 1870–1874. (In Chinese) [Google Scholar]
- Ge, G.F.; Yu, C.H.; Yu, B.; Shen, Z.H.; Zhang, D.L.; Wu, Q.F. Antitumor effects and chemical compositions of Eupolyphaga sinensis Walker ethanol extract. J. Ethnopharmacol. 2012, 141, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.R.; Li, E.Q.; Dai, C.Q.; Yu, B.; Wu, X.L.; Huang, C.R.; Chen, X.Y. The inducible effect of LBP on maturation of dendritic cells and the related immune signaling pathways in hepatocellular carcinoma (HCC). Curr. Drug Deliv. 2012, 9, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Zhang, X.; Du, Y.; Jia, B.; Ka, W.; Sun, D.; Yao, W.; Wen, Z. Effects of Gekko sulfated polysaccharide-protein complex on the defective biorheological characters of dendritic cells under tumor microenvironment. Cell Biochem. Biophys. 2012, 62, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Bao, J.M.; Li, X.L.; Zhang, T.; Shen, X.H. Inhibiting effect of Astragalus polysaccharides on the functions of CD4+CD25 high Treg cells in the tumor microenvironment of human hepatocellular carcinoma. Chin. Med. J. (Engl.) 2012, 125, 786–793. [Google Scholar] [PubMed]
- He, X.; Li, X.; Liu, B.; Xu, L.; Zhao, H.; Lu, A. Down-regulation of Treg cells and up-regulation of TH1/TH2 cytokine ratio were induced by polysaccharide from Radix Glycyrrhizae in H22 hepatocarcinoma bearing mice. Molecules 2011, 16, 8343–8352. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.X.; Zhu, C.; Ba, Y.; Chen, D.; Zhou, X.L.; Cao, R.; Wang, L.P.; Ren, Y.; Wu, X.Z. Gekko-sulfated glycopeptide inhibits tumor angiogenesis by targeting basic fibroblast growth factor. J. Biol. Chem. 2012, 287, 13206–13215. [Google Scholar] [CrossRef] [PubMed]
- Mu, P.; Gao, X.; Jia, Z.J.; Zheng, R.L. Natural antioxidant pedicularioside G inhibits angiogenesis and tumourigenesis in vitro and in vivo. Basic Clin. Pharmacol. Toxicol. 2008, 102, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zheng, X.; Zeng, G.; Zhou, Y.; Yuan, H. Purified vitexin compound 1 inhibits growth and angiogenesis through activation of FOXO3a by inactivation of Akt in hepatocellular carcinoma. Int. J. Mol. Med. 2014, 33, 441–448. [Google Scholar] [PubMed]
- Zhang, H.; Yang, R. Resveratrol inhibits VEGF gene expression and proliferation of hepatocarcinoma cells. Hepatogastroenterology 2014, 61, 410–412. [Google Scholar] [PubMed]
- Zhang, Q.; Tang, X.; Lu, Q.Y.; Zhang, Z.F.; Brown, J.; le, A.D. Resveratrol inhibits hypoxia-induced accumulation of hypoxia-inducible factor-1α and VEGF expression in human tongue squamous cell carcinoma and hepatoma cells. Mol. Cancer Ther. 2005, 4, 1465–1474. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Fu, Y.L.; Gong, Z.P.; Deng, L.R.; Hu, Y.Q. Studies on the anti-angiogenic mechanism of the formula of Chinese medicine active ingredients combined with small dose cisplatin in mice of hepatocellular carcinoma. Chin. J. Exp. Tradit. Med. Formul. 2010, 1, 157–160. (In Chinese) [Google Scholar]
- Deng, S.; Hu, B.; An, H.M.; Du, Q.; Xu, L.; Shen, K.P.; Shi, X.F.; Wei, M.M.; Wu, Y. Teng-Long-Bu-Zhong-Tang, a Chinese herbal formula, enhances anticancer effects of 5-Fluorouracil in CT26 colon carcinoma. BMC Complement. Altern. Med. 2013, 13, 128. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A.; Darvesh, A.S. Angiogenesis in hepatocellular carcinoma: A potential target for chemoprevention and therapy. Curr. Cancer Drug Targets 2012, 12, 1095–1118. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Wang, X.M.; Xie, S. Experimental study on mechanisms of lyophilized powder of fresh gekko Chinenis in inhibiting H22 hepatocarcinoma angiogenesis. Chin. J. Integr. Tradit. West. Med. 2006, 26, 58–62. (In Chinese) [Google Scholar]
- Hu, B.; Du, Q.; Shen, K.P.; Xu, L. Principles and scientific basis of Traditional Chinese Medicine in cancer treatment. J. Bioanal. Biomed. 2012, S6, 005. [Google Scholar] [CrossRef]
- Du, Q.; Hu, B.; An, H.M.; Shen, K.P.; Xu, L.; Deng, S.; Wei, M.M. Synergistic anticancer effects of curcumin and resveratrol in Hepa1–6 hepatocellular carcinoma cells. Oncol. Rep. 2013, 29, 1851–1858. [Google Scholar] [PubMed]
- Chen, T.; Li, D.; Fu, Y.L.; Hu, W. Screening of QHF formula for effective ingredients from Chinese herbs and its anti-hepatic cell cancer effect in combination with chemotherapy. Chin. Med. J. (Engl.) 2008, 121, 363–368. [Google Scholar] [PubMed]
- Wu, Y.Y.; Wan, L.H.; Zheng, X.W.; Shao, Z.J.; Chen, J.; Chen, X.J.; Liu, L.T.; Kuang, W.J.; Tan, X.S.; Zhou, L.M. Inhibitory effects of β,β-dimethylacrylshikonin on hepatocellular carcinoma in vitro and in vivo. Phytother. Res. 2012, 26, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Luo, G.; Zhou, L.; Zeng, Y.; Yang, W. In vitro and in vivo antitumor effects of acetylshikonin isolated from Arnebia euchroma (Royle) Johnst (Ruanzicao) cell suspension cultures. Chin. Med. 2009, 4, 14. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.Y.; Zhu, L.; Ma, X.Y.; Shao, Z.J.; Chen, J.; Chen, X.J.; Wan, L.H.; Zhou, L.M. The anti-proliferation effect of Aikete injection on hepatocellular carcinoma in vitro and in vivo. Pharm. Biol. 2011, 49, 531–538. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, B.; An, H.-M.; Wang, S.-S.; Chen, J.-J.; Xu, L. Preventive and Therapeutic Effects of Chinese Herbal Compounds against Hepatocellular Carcinoma. Molecules 2016, 21, 142. https://doi.org/10.3390/molecules21020142
Hu B, An H-M, Wang S-S, Chen J-J, Xu L. Preventive and Therapeutic Effects of Chinese Herbal Compounds against Hepatocellular Carcinoma. Molecules. 2016; 21(2):142. https://doi.org/10.3390/molecules21020142
Chicago/Turabian StyleHu, Bing, Hong-Mei An, Shuang-Shuang Wang, Jin-Jun Chen, and Ling Xu. 2016. "Preventive and Therapeutic Effects of Chinese Herbal Compounds against Hepatocellular Carcinoma" Molecules 21, no. 2: 142. https://doi.org/10.3390/molecules21020142
APA StyleHu, B., An, H.-M., Wang, S.-S., Chen, J.-J., & Xu, L. (2016). Preventive and Therapeutic Effects of Chinese Herbal Compounds against Hepatocellular Carcinoma. Molecules, 21(2), 142. https://doi.org/10.3390/molecules21020142