2.1. Chemical Characterization
The qualitative phytochemical analysis of the seed ethanolic extracts from
L. tomentosa (LtEE) and
L. rigida (LrEE) showed similar overall profile and revealed tannins, flavonoids and saponin classes. The data are presented in
Table 1.
The phytochemical analysis can be a first step to guide the subsequent tests, but the assays used for this analysis do not provide much information about the quantity or the proportion of the constituent compounds. For this purpose, we ran three further analyses—determinations of total phenolics, tannins and total flavonoids, as shown in
Table 2—which, in turn, confirmed some of the results of the previous analysis and gave us a more tangible perspective on the amount of each type of phenolic compound. In general,
L. rigida was shown to be richer in tannins than
L. tomentosa, the aqueous fraction of both species containing the highest levels (0.294 and 0.182 tannic acid equivalent in mg per g of the sample, respectively).The tannin content of the ethyl acetate fractions from both plants was below the detection limit of the method. The total phenolic contents of extracts and fractions of both
Licania seeds ranged from 96.95 to 206.98 mg of gallic acid equivalent/g of extract which are much higher than those described for some fruits, such as apple (1.91 mg/g), watermelon (1 mg/g), grape (0.93 mg/g), orange (1.95 mg/g) and kiwifruit (3.18 mg/g) [
14]. Similarly, the total flavonoids content of LtEAF (90.61 mg of quercetin/g of extract) showed values much higher than those described to apple (0.45 mg/g), mangosteen (0.41 mg/g), pear (0.32 mg/g) and tomato fruits (0.38 mg/g).
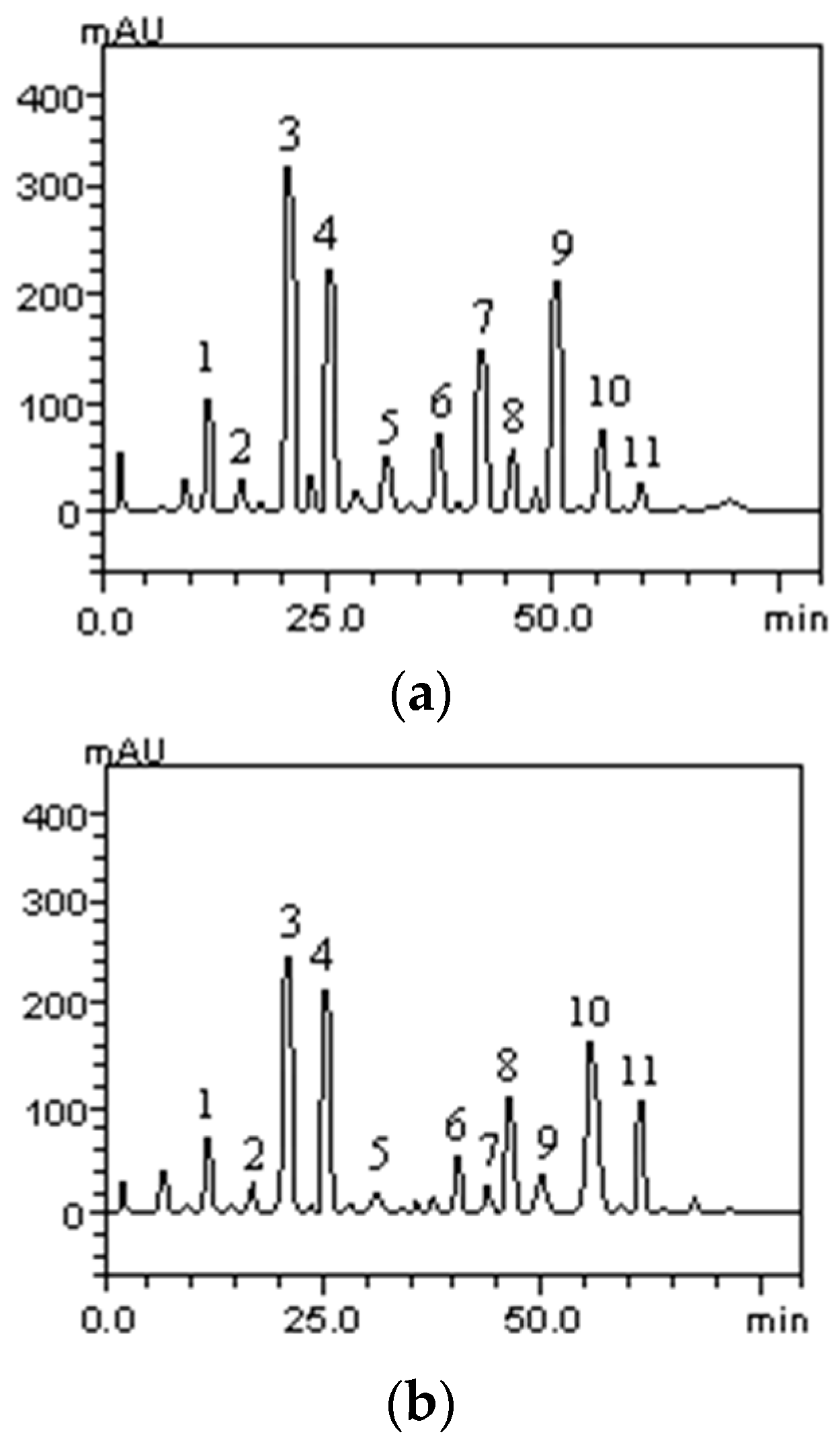
High Performance Liquid Chromatography (HPLC) analysis of the seed ethanolic extracts and their respective fractions indicated that the fractionation procedure used did not generate fractions with different compositions. Two representative chromatograms are shown in
Figure 1.
Despite the similar chromatographic profile of the fractions, the constituents were present at different concentrations. For example, in the ethyl acetate fraction of both plant species all the identified phenolic acids and flavonoid compounds were present in much higher concentrations than in the other samples, as described in
Table 3 and
Table 4. In
L. tomentosa, chlorogenic acid, rutin and quercetin were the compounds with the highest concentrations. The last two are widely known to be strong antioxidant substances. On the other hand,
L. rigida showed as main compounds in all the fractions, except for LrEAF, chlorogenic acid, kaempferol and caffeic acid. In this instance, only the latter has been scientifically recognized as having remarkable antioxidant potential [
15,
16,
17,
18]. There are reports of other plants of the same genus, such as Licania licaniaeflora, which has been described as having strong antioxidant activity due to the presence of quercetin derivatives whereas for the kaempferol derivatives the opposite pattern is observed [
19]. As a matter of fact, the flavonoids quercetin and kaempferol have been used as chemotaxonomic markers in the Chrysobalanaceae family and it has been noted that they are of wide-spread occurrence in the genus
Licania [
20].
2.2. Total Antioxidant Capacity
In general, the results showed a wide range of values for antioxidant activities when considering both methods (DPPH and TBARS, the latter with or without iron stress), as described in
Table 5. In addition, the LrEAF had an outstanding performance in the DPPH test with values similar to those of ascorbic acid (SC
50 9.15 and 10.17 µg/mL, respectively). It is noteworthy that some of these samples could inhibit lipid peroxidation as well as scavenge directly free radicals. If the results were analyzed by considering only the TBARS assay, it could be said that LrAF and LtMF had the best results, without and with iron, respectively (IC
50 60.80 and 8.55 µg/mL). As a matter of fact, is the result shown by LrAF is remarkable considering that in most studies, water is rarely used as solvent for extraction or fractionation. Focusing on the DPPH results, the LtEE, LtMF, LtAF, LrEAF and LrAF showed excellent results (SC
50 ranging from 9.15 to 42.68 µg/mL) compared to ascorbic acid (SC
50 = 10.17 µg/mL) and the SC
50 observed for these samples were much lower than that observed in leaves of
L. tomentosa described elsewhere (SC
50 73.33 µg/mL) [
21]. The “total antioxidant capacity” (TAC) of a particular sample cannot be accurately measured by any isolated assay because of the chemical complexity of the antioxidant compounds. For instance, the methodologies should be able to evaluate both lipophilic and hydrophilic nature of the phenolic compounds, and distinguish hydrogen atom transfer, electron transfer, as well as transition metal chelation.
The antioxidant activity of these high phenolic-content samples (LtEAF and LrEAF) was disappointing. The former had a bad performance in boths tests whereas the latter had a bad result only in the DPPH assay. This strongly contradicts the literature that normally states that phenolic compounds correlate well with antioxidant activity [
22]. It is, however, possible that ethyl acetate solvent must have extracted other compounds that could act antagonistically on antioxidant activity [
23]. It is also important to stress that some phenolic antioxidants can auto-oxidize and behave like prooxidants under certain circumstances. Instead of breaking the free radical chain reaction, the phenoxy radical may also interact with oxygen and produce quinones and superoxide anion (
) [
24]. Simple phenolics which are easily oxidized, such as quercetin and gallic acid, possess prooxidant activity [
25]. These arguments could partially explain why the samples with high concentration of simple phenolic compounds had poor antioxidant activity.
In the present study, the results on antioxidant activities of the different samples were crossed with the phenolic compounds composition data, so that a correlation could be established. The antioxidant activity measured by the DPPH correlated very well with quercetin (r = 0.788) and quercitrin (r = 0.744). As for the results of TBARS assay without iron-induced stress, catechin (r = 0.690) and quercetin (r = 0.514) presented the best correlations. In general, flavonoids showed better correlation values than the phenolic acids in the results of these two assays. The data are presented in
Table 6.
In fact, the flavonoids are recognized for exceptional antioxidant activity [
26], and quercetin is very frequently chosen as a positive control in antioxidant assays [
27]. Catechin has an analogous chemical structure, but it lacks the oxo group in the carbon 4 and a double bond in the carbons 2 and 3, which confers a less potent antioxidant activity when used alone [
28]. When the TBARS was performed with iron-induced stress, a different outcome was observed: the highest correlation values were those of chlorogenic acid (r = 0.465) and rutin (r = 0.346), a flavonoid derived from quercetin. Phenolic acids showed a low positive correlation in this assay (r = 0.246) and flavonoids no correlation at all. In the case of phenolic acids, the interaction with the egg phospholipids and the ability to form stable complexes with iron may have been more important than the mere ability to neutralize free radicals. For the iron chelating property, it is of utmost importance to have one or both two groups: catechol and galloyl. In addition, to give greater protection to the phospholipids, polyphenols must have hydrophilic groups in their chemical structure, such as hydroxyls and carbohydrates that can interact with the polar part of the micelles formed by them [
29]. These characteristics are presented by rutin and chlorogenic acid chemical structure [
30].
2.3. Chelating Capacity
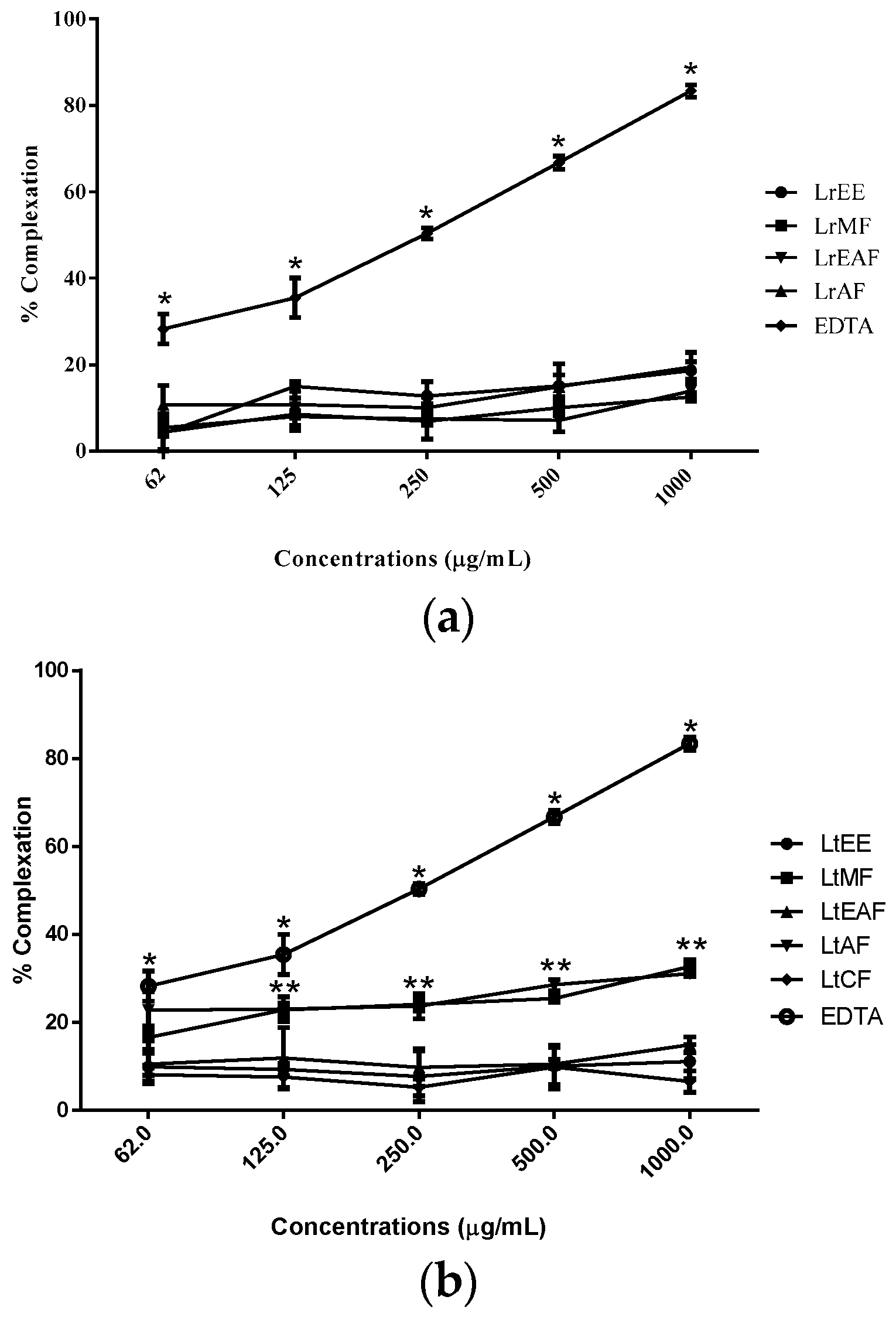
As for the
o-phenanthroline assay, no sample was able to chelate free iron as the positive control EDTA, at least when tested in vitro (
Figure 2). However, LtMF and LtAF showed chelating activity higher than the other fractions of
L. tomentosa. This result is not necessarily negative in what concerns antioxidant potential since chelating capacity is just one among many mechanisms of action to help prevent oxidative stress in living beings [
31,
32].
In fact, many studies with plant extracts have shown that beyond the capacity to neutralize free radicals, plant compounds show also the ability to chelate transition metals. Indeed, the intracellular redox state is related to the iron (or copper) redox couple and the maintenance of its levels is under strict physiological control [
33]. It has been inferred that iron regulation guarantees no free iron is present in the cell. Nevertheless, under stress circumstances, iron levels may surpass the capacity of transferrin to bind it. The released Fe(II) can participate in the Fenton reaction, generating hydroxyl radical [Fe(II) + H
2O
2 → Fe(III) +
•OH + OH
−], which is extremely reactive and might impair various cellular components [
34]. Thus, it is useful to measure the iron chelating capacity of a sample. The results for this test helped us to abandon the hypothesis that lipid peroxidation protection was only due to the ability to trap iron, turning it less available for the Fenton reaction (
Figure 2). It is more plausible to believe that there is more than one mechanism involved in the lipid peroxidation inhibition. Antioxidants are compounds that can delay autoxidation by inhibiting formation or interrupting propagation of free radicals in different ways: (1) scavenging species that initiate peroxidation; (2) chelating metal ions so that they are unable to generate reactive species or decompose lipid peroxides; (3) quenching O
2− preventing formation of peroxides; (4) breaking the autoxidative chain reaction; and/or (5) reducing localized O
2 concentrations [
35].
2.4. Cytotoxicity to Cancer Cell Lines
Since several studies have demonstrated the cytotoxic effects of flavonoids [
36,
37,
38] and phenolic acids [
39,
40] to human cancer cells, in the present study LrEE and LtEE were tested for possible cytotoxicity towards the human MCF-7 breast cancer and Caco-2 colorectal adenocarcinoma cell lines. Also the flavonoid quercetin and phenolic acid galic acid, which both are constituents of the ethanolic extracts, were included in the cytotoxicity experiments for comparison. After treatment of both cell lines for 24 h with increasing concentrations of LrEE, LtEE, quercetin and gallic acid, cell viability was determined using the ATPlite assay. It was not possible to test the ethanol extracts at concentrations exceeding 250 µg/mL, due to the formation of precipitates in the culture medium. The same was true for quercetin at concentrations higher than 80 µg/mL medium. Flavonoids are known to easily precipitate in aqueous medium [
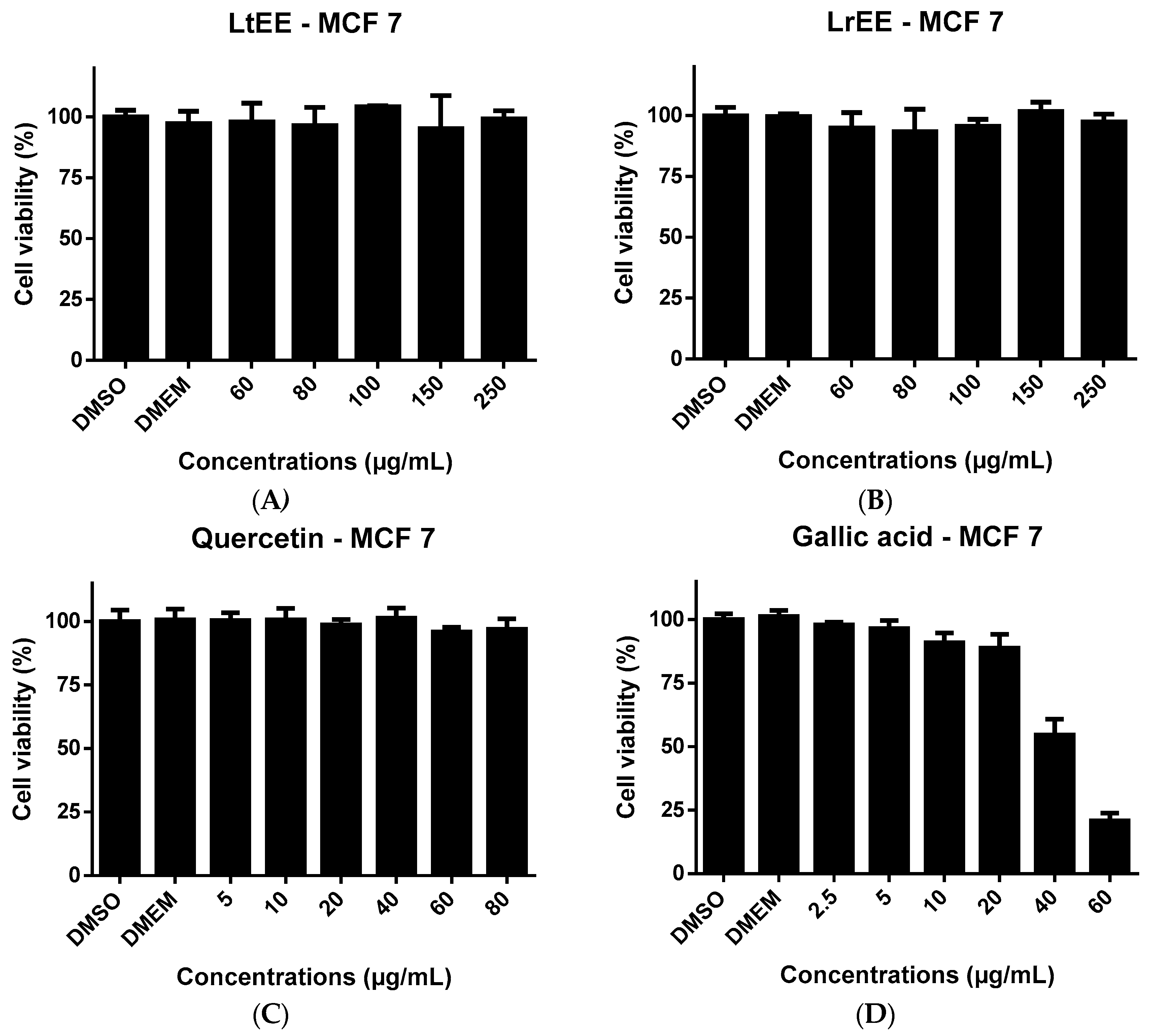
41]. LrEE and LtEE concentrations up to 250 µg/mL culture medium did not show any toxicity to MCF-7 cells (
Figure 3) and Caco-2 cells (
Figure 4). The higher concentrations of quercetin were slightly cytotoxic to Caco-2 (
Figure 4) but not to MCF-7 (
Figure 3).
Gallic acid at 40 and 60 µg/mL resulted in clear cytotoxicity to both cell lines. The lack of cytotoxicity of LrEE and LtEE is in contrast with previous finding by ohers. Extracts of several plants from the Licania genius have been reported to exert in vitro cytotoxic effects towards cancer cell lines, for example extracts of L. heteromorpha [
42] and L. michauxii in human hepatoma HepG2 cells and Caco-2 cells [
43]. This discrepancy might be related to differences in extract preparation and type of viability assay used, e.g., ATPlite versus MTT assays.