Structural Investigation of Cell Wall Xylan Polysaccharides from the Leaves of Algerian Argania spinosa
Abstract
:1. Introduction
2. Results and Discussion
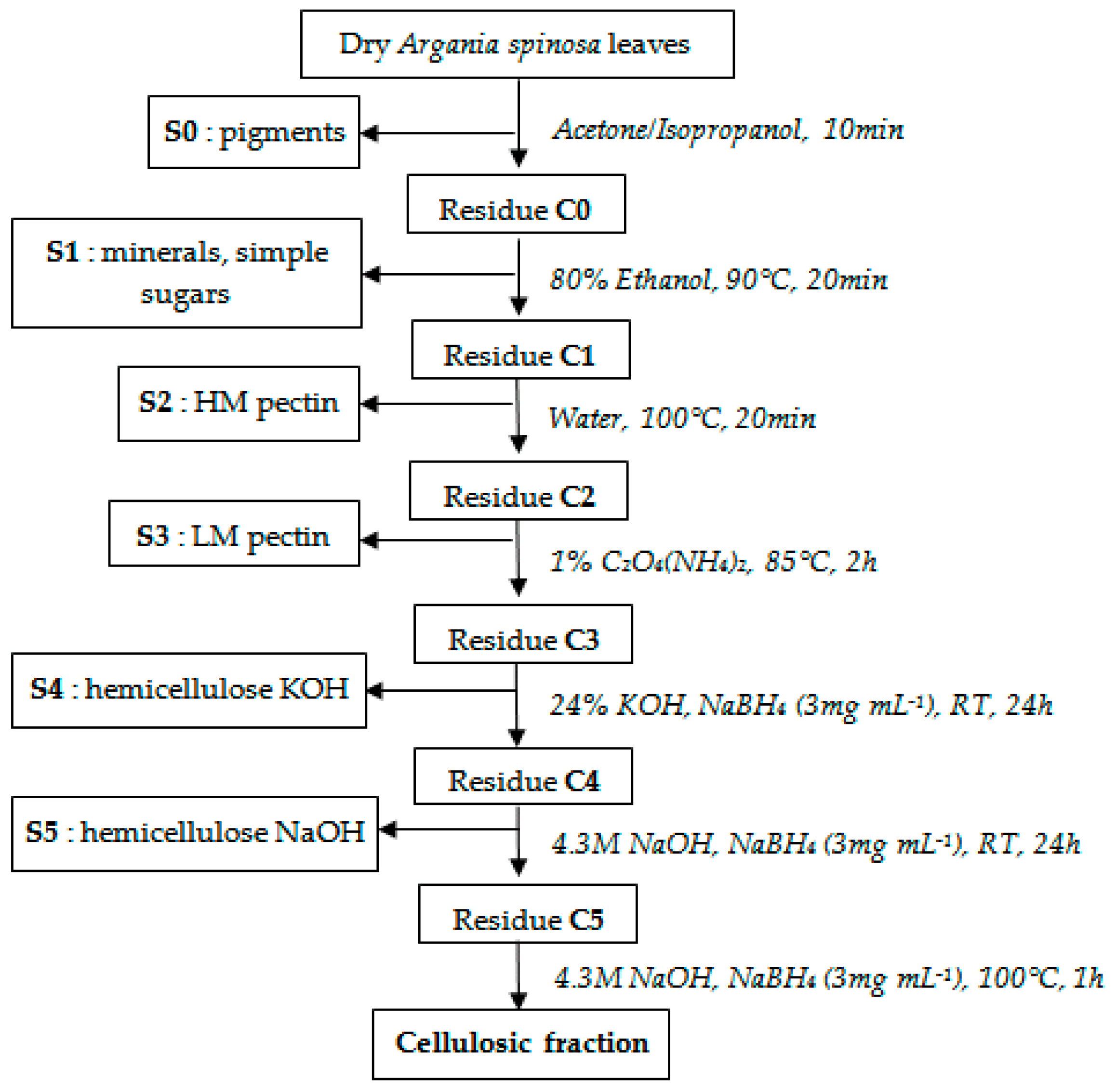
2.1. Extraction and Isolation of Structural Polysaccharides
2.2. Monosaccharide Composition of the Hemicellulosic Extracts
2.3. Infrared Analysis of the Hemicellulosic Extracts
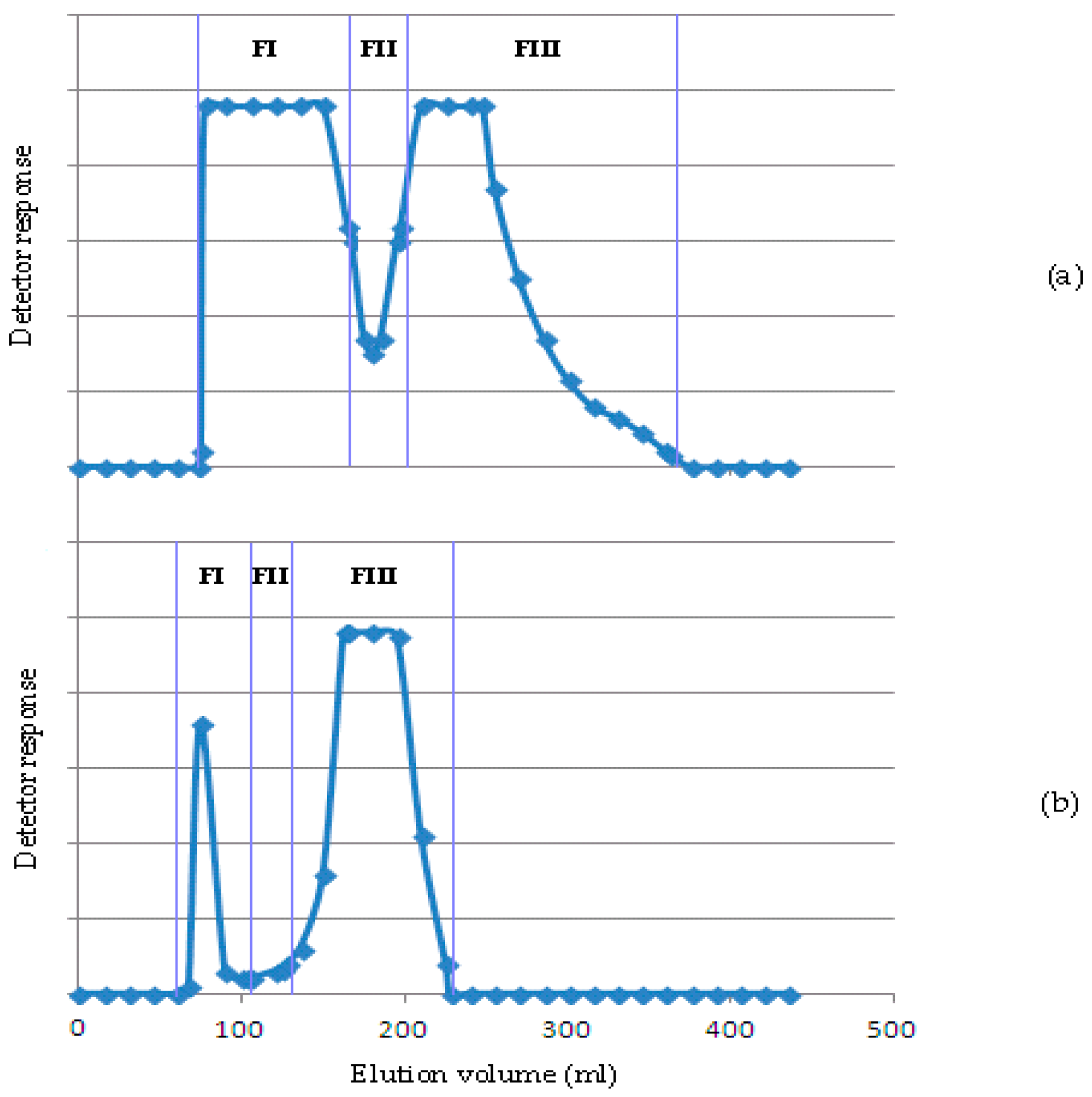
2.4. Purification and Characterization of KOH and NaOH Hemicellulosic Fractions
2.4.1. Purification and Monosaccharide Composition
2.4.2. Oligosaccharide Fractions Obtained by Enzymatic Hydrolysis
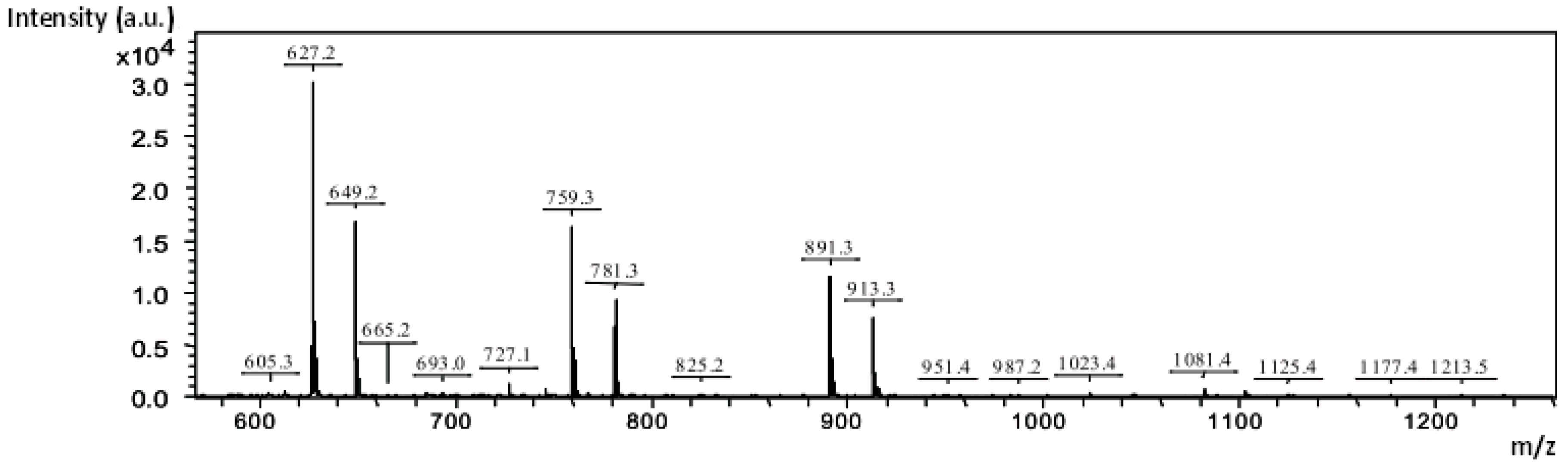
2.4.3. Mass Spectrometry Analysis
3. Materials and Methods
3.1. Biological Material
3.2. Chemicals and Enzyme
3.3. Isolation of Hemicellulose Fractions
3.4. Purification of Hemicellulose Fractions
3.5. Enzymatic Hydrolysis
3.6. Chemical Characterizations
3.6.1. Chemical Composition of Extracts
3.6.2. Monosaccharide Composition
3.6.3. FT-IR Spectroscopy
3.6.4. Mass Spectrometry
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alén, R. Structure and chemical composition of wood. In Papermaking Science and Technology Book 3: Papermaking Part 1, Forest Products Chemistry, 1st ed.; Stenius, P., Ed.; Fapet Oy: Helsinki, Finland, 2000; pp. 11–57. [Google Scholar]
- Ebringerová, A.; Hromádková, Z.; Heinze, T. Hemicellulose. In Advances in Polymer Science; Heinze, T., Ed.; Springer: Cham, Switzerland, 2005; Volume 186. [Google Scholar]
- Joseleau, J.P.; Comtat, J.; Ruel, K. Chemical structure of xylans and their interaction in the plant cell walls. Prog. Biotechnol. 1992, 7, 1–15. [Google Scholar]
- Baumer, M.; Zeraïa, L. La plus continentale des stations de l’Arganier en Afrique du nord. Rev. For. Fr. 1999, 51, 446–452. [Google Scholar] [CrossRef]
- Peltier, J.P. Les séries de l’arganeraie steppique dans le Souss (Maroc). Ecol. Mediterr. 1983, 9, 77–88. [Google Scholar]
- Sebaa, H.S.; Kaid-Harche, M. Anatomical structure and ultrastructure of the endocarp cell walls of Argania spinosa (L.) Skeels (Sapotaceae). Micron 2014, 67, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Kechairi, R.; Abdoun, F. État des lieux cartographiques de l’arganier Argania spinosa (L.) Skeels (Sapotaceae) en Afrique Nord-Occidentale (Algérie et Sahara Occidental). Int. J. Environ. Stud. 2016, 73, 286–293. [Google Scholar] [CrossRef]
- Ray, B.; Loutelier-Bourhis, C.; Lange, C.; Condamine, E.; Driouich, A.; Lerouge, P. Structural investigation of hemicellulosic polysaccharides from Argania spinosa: Characterisation of a novel xyloglucan motif. Carbohydr. Res. 2004, 339, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Hachem, K.; Labani, A.; Kaid-Harche, M. Isolation, Structural Characterization, and Valorization of Pectic Substances from Algerian Argan Tree Leaves (Argania spinosa (L.) Skeels). Int. J. Polym. Sci. 2015, 2015. [Google Scholar] [CrossRef]
- Hachem, K.; Benabdesslem, Y.; Ghomari, S.; Hasnaoui, O.; Kaid-Harche, M. Partial structural characterization of pectin cell wall from Argania spinosa leaves. Heliyon 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- Kačuráková, M.; Wellner, N.; Ebringerová, A.; Hromádková, Z.; Wilson, R.H.; Belton, P.S. Characterization of xylan-type polysaccharides and associated cell wall components by FT-IR and FT-Raman spectroscopies. Food Hydrocoll. 1999, 13, 35–41. [Google Scholar] [CrossRef]
- Sun, R.C.; Lawther, J.M.; Banks, W.B. Isolation and characterization of hemicellulose B and cellulose from pressure refined wheat straw. Ind. Crops Prod. 1998, 7, 121–128. [Google Scholar] [CrossRef]
- Brumell, D.A.; Schröder, R. Xylan Metabolism in Primary Cell Walls. N. Z. J. For. Sci. 2009, 39, 125–143. [Google Scholar]
- Ebringerová, A.; Heinze, T. Xylan and xylan derivatives—Biopolymers with valuable properties, 1. Naturally occuring xylans structures, isolation procedures and properties. Macromolecul. Rapid Commun. 2000, 21, 542–556. [Google Scholar] [CrossRef]
- Wilkie, K.C.B. The hemicelluloses of grasses and cereals. Adv. Carbohydr. Chem. Biochem. 1979, 36, 215–264. [Google Scholar]
- Jacobs, A.; Larsson, P.T.; Dahlman, O. Distribution of uronic acids in xylans from various species of soft- and hardwood as determined by MALDI mass spectrometry. Biomacromolecules 2001, 2, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Habibi, Y.; Mahrouz, M.; Vignon, M.R. Isolation and structure of d-xylans from pericarp seeds of Opuntia ficus-indica prickly pear fruits. Carbohydr. Res. 2002, 337, 1593–1598. [Google Scholar] [CrossRef]
- Biely, P.; Vršanská, M.; Tenkanen, M.; Kluepfel, D. Endo-β-1,4-xylanase families: Differences in catalytic properties. J. Biotechnol. 1997, 57, 151–166. [Google Scholar] [CrossRef]
- Takenishi, S.; Tsujisaka, Y. Structures of the oligosaccharides from the enzymic hydrolysate of rice-straw arabinoxylan by a xylanase of Aspergillus niger. Agric. Biol. Chem. 1973, 37, 1385–1391. [Google Scholar] [CrossRef]
- Kolenová, K.; Vršanská, M.; Biely, P. Mode of action of endo-β-1,4-xylanases of families 10 and 11 on acidic xylooligosaccharides. J. Biotechnol. 2006, 121, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, S.V.; Diaz, P.; Pastor, F.I.J. Modular glucuronoxylan-specific xylanase with a family CBM35 carbohydrate-binding module. Appl. Environ. Microbiol. 2012, 78, 3923–3931. [Google Scholar] [CrossRef] [PubMed]
- Angone, S.A.; Bardor, M.; Nguema-Ona, E.; Rihouey, C.; Ishii, T.; Lerouge, P.; Driouich, A. Structural characterization of cell wall polysaccharides from two plant species endemic to central Africa, Fleurya aestuans and Phragmenthera capitata. Carbohydr. Polym. 2009, 75, 104–109. [Google Scholar] [CrossRef]
- Dinand, E.; Vignon, M.R. Isolation and NMR characterisation of a (4-O-methyl-d-glucurono) d-xylan from sugar beet pulp. Carbohydr. Res. 2001, 330, 285–288. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
| Fraction | Yield (Mass %) | UA (%) | NS (%) |
|---|---|---|---|
| S0: pigments | 45.6 * | - | - |
| S1: minerals, simple sugars | 3.5 * | 25.1 | 74.9 |
| S2: HM pectin | 2.9 ** | 48.6 | 51.4 |
| S3: LM pectin | 13.2 ** | 67.9 | 32.1 |
| S4: Hemicellulose KOH | 11.8 ** | 18.3 | 81.7 |
| S5: Hemicellulose NaOH | 6.7 ** | 18.6 | 81.4 |
| C6: Cellulose | 21.1 ** | - | - |
| Glycosyl Residues | S4: Hemicelluloses KOH | S5: Hemicelluloses NaOH |
|---|---|---|
| Rha | 7.2 | 5.8 |
| Fuc | 1.6 | 2.3 |
| Ara | 39.4 | 31.9 |
| Xyl | 24.9 | 33.3 |
| Man | 1.3 | 1.0 |
| Gal | 8.2 | 8.0 |
| Glc | 6.3 | 6.8 |
| Gal A | 4.9 | 6.4 |
| Glc A | 0.6 | 0.5 |
| 4-O-Me Glc A | 5.6 | 4.0 |
| Glycosyl Residues (Molar %) | S4: Hemicelluloses KOH | S5: Hemicelluloses NaOH | ||
|---|---|---|---|---|
| FI | FIII | FI | FIII | |
| UA (%) | 17.1 | 20.6 | 30.8 | 17.1 |
| NS (%) | 82.9 | 79.4 | 69.2 | 82.9 |
| Rha | 6.6 | 8.3 | 6.7 | 7.1 |
| Fuc | 1.2 | 2.2 | 2.5 | 4.3 |
| Ara | 35.8 | 43.0 | 35.4 | 36.1 |
| Xyl | 29.8 | 18.2 | 30.8 | 22.2 |
| Man | 0.9 | 0.8 | 0.7 | 1.7 |
| Gal | 6.3 | 10.8 | 6.2 | 8.6 |
| Glc | 8.4 | 6.0 | 7.4 | 8.3 |
| Gal A | 7.6 | 7.4 | 7.1 | 8.2 |
| Glc A | 0.5 | 1.2 | 0.5 | 0.6 |
| 4-O-Me Glc A | 2.9 | 2.1 | 2.8 | 2.7 |
| Glycosyl Residues | Oligosaccharides KOH Obtained by Enzymatic Hydrolysis | Oligosaccharides NaOH Obtained by Enzymatic Hydrolysis | ||
|---|---|---|---|---|
| FI | FIII | FI | FIII | |
| Rha | 4.3 | 3.3 | 4.3 | 5.2 |
| Fuc | 0.0 | 0.0 | 0.0 | 0.0 |
| Ara | 17.1 | 20.4 | 16.1 | 15.8 |
| Xyl | 58.1 | 45.8 | 24.0 | 31.6 |
| Man | 2.2 | 8.1 | 24.1 | 19.3 |
| Gal | 1.5 | 14.9 | 10.2 | 10.0 |
| Glc | 2.1 | 2.3 | 6.8 | 10.9 |
| Gal A | 5.4 | 0.3 | 9.1 | 3.9 |
| Glc A | 0.6 | 1.9 | 2.6 | 1.2 |
| 4-O-Me Glc A | 8.7 | 3.0 | 2.8 | 2.1 |
| Subfraction Obtained by Enzymatic Hydrolysis | Compound | [M + Na]+ |
|---|---|---|
| FI KOH | 3(pentose) + 1(uronic acid methylated) | 627.2 |
| 4(pentose) + 1(uronic acid methylated) | 759.3 | |
| 5(pentose) + 1(uronic acid methylated) | 891.3 | |
| 6(pentose) + 1(uronic acid methylated) | 1023.4 | |
| 5(pentose) + 2(uronic acid methylated) | 1081.4 | |
| FIII KOH | 3(pentose) + 1(uronic acid methylated) | 627.2 |
| 4(pentose) + 1(uronic acid methylated) | 759.3 | |
| 5(pentose) + 1(uronic acid methylated) | 891.3 | |
| FI NaOH | 3(pentose) + 1(uronic acid methylated) | 627.2 |
| 4(pentose) + 1(uronic acid methylated) | 759.3 | |
| FIII NaOH | 3(pentose) + 1(uronic acid methylated) | 627.2 |
| 4(pentose) + 1(uronic acid methylated) | 759.3 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hachem, K.; Faugeron, C.; Kaid-Harche, M.; Gloaguen, V. Structural Investigation of Cell Wall Xylan Polysaccharides from the Leaves of Algerian Argania spinosa. Molecules 2016, 21, 1587. https://doi.org/10.3390/molecules21111587
Hachem K, Faugeron C, Kaid-Harche M, Gloaguen V. Structural Investigation of Cell Wall Xylan Polysaccharides from the Leaves of Algerian Argania spinosa. Molecules. 2016; 21(11):1587. https://doi.org/10.3390/molecules21111587
Chicago/Turabian StyleHachem, Kadda, Céline Faugeron, Meriem Kaid-Harche, and Vincent Gloaguen. 2016. "Structural Investigation of Cell Wall Xylan Polysaccharides from the Leaves of Algerian Argania spinosa" Molecules 21, no. 11: 1587. https://doi.org/10.3390/molecules21111587








