Coumarin Antifungal Lead Compounds from Millettia thonningii and Their Predicted Mechanism of Action
Abstract
:1. Introduction
2. Results and Discussion
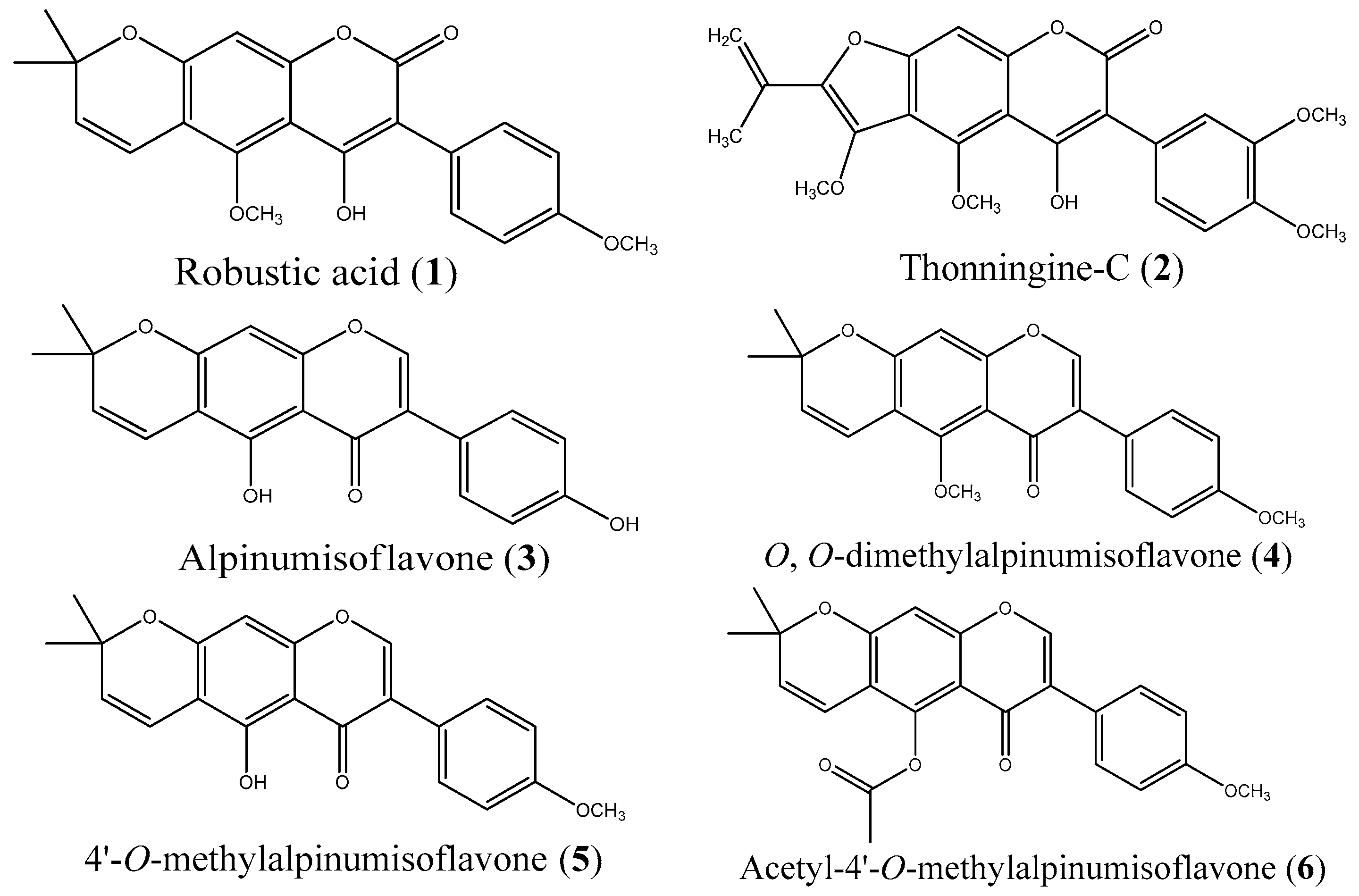
2.1. Isolation and Synthesis
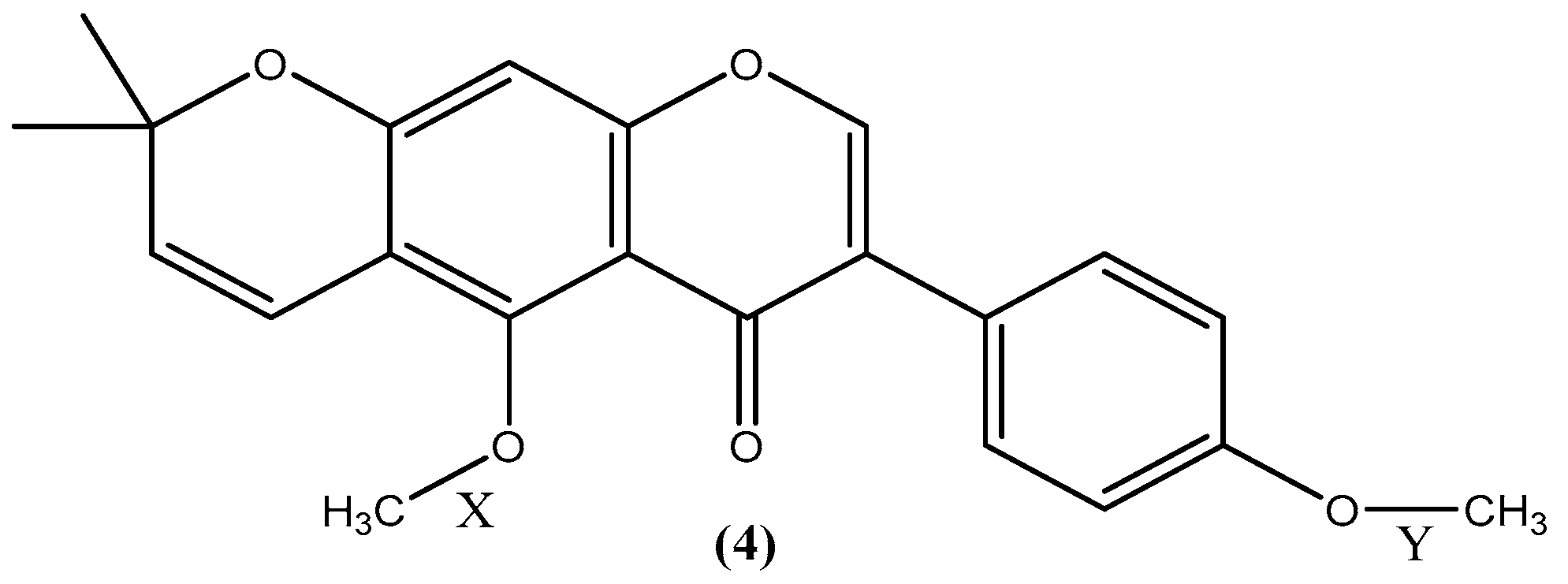
2.2. Investigation into the Selective Demethylation of Isoflavone 4
2.3. Biological Testing
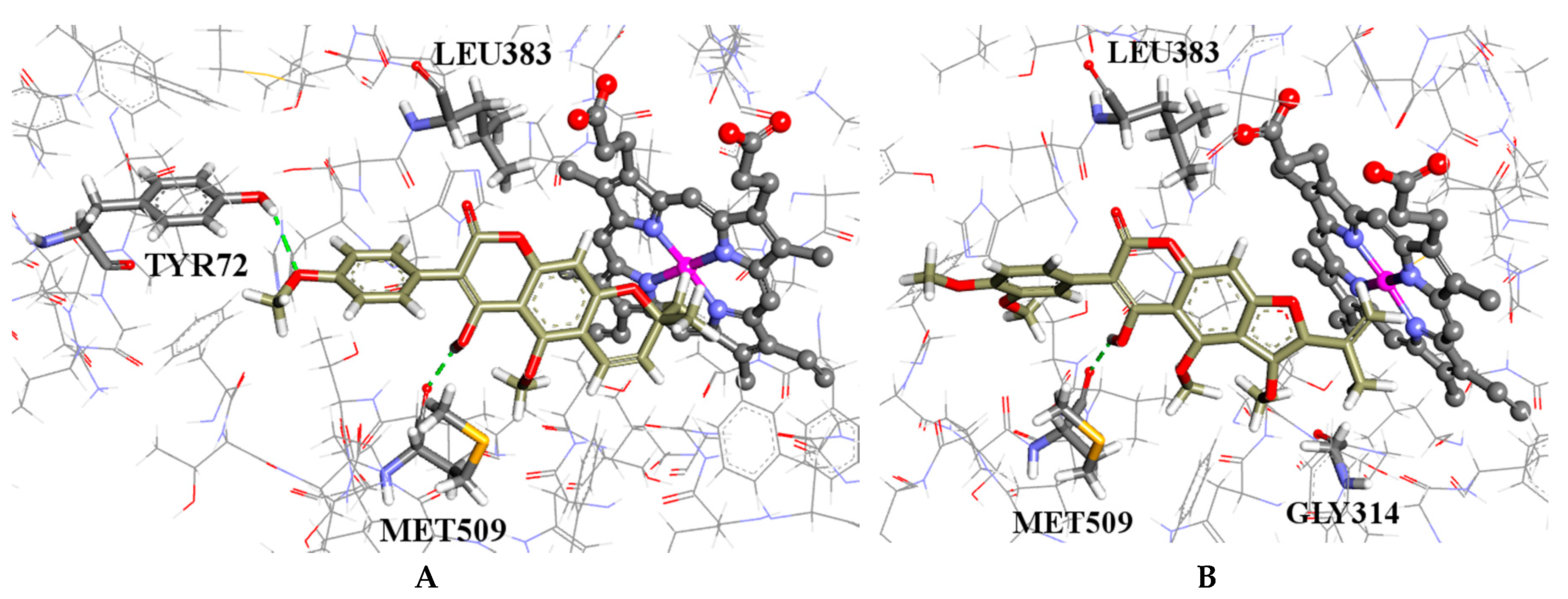
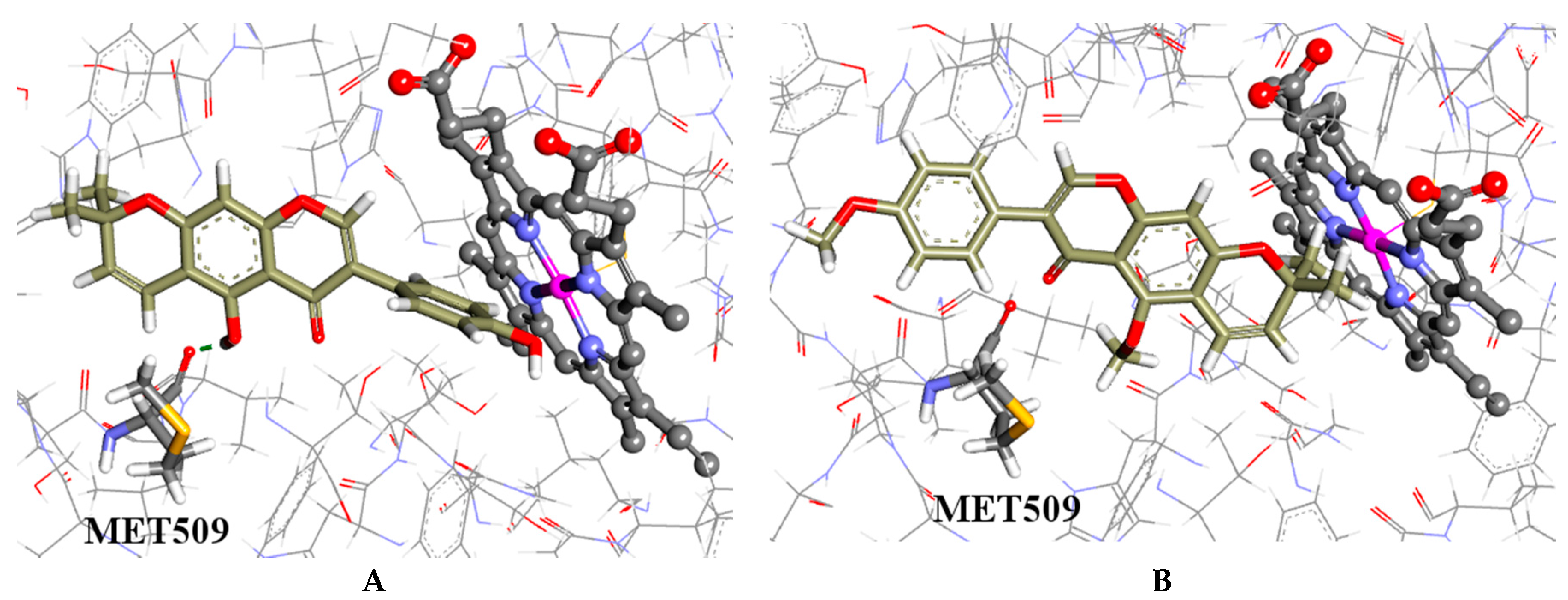
2.4. Molecular Modelling
2.5. Chemical Space
3. Materials and Methods
3.1. Isolation
3.1.1. Plant Material
3.1.2. Extraction and Isolation
3.2. Synthesis
3.2.1. Synthesis of 4′-O-methylalpinumisoflavone (5)
3.2.2. Synthesis of Acetyl-4′-methylalpinumisoflavone (6)
3.3. Bioactivity Studies
3.3.1. Preparation of Solutions and Media
3.3.2. Determination of the Potency of the Compounds
3.3.3. Determination of Minimum Inhibitory Concentration and the Fungicidal or Fungistatic Effect of the Active Compounds
3.4. Thermochemical Calculations
3.5. Molecular Modelling
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ellis, M. Invasive fungal infections: Evolving challenges for diagnosis and therapeutics. Mol. Immunol. 2002, 38, 947–957. [Google Scholar] [CrossRef]
- Huang, W.; Na, L.; Fidel, P.L.; Schwarzenberger, P. Requirement of interleukin-17A for systemic anti-Candida albicans host defense in mice. J. Infect. Dis. 2004, 190, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Eggimann, P.; Garbino, J.; Pittet, D. Epidemiology of Candida species infections in critically ill non-immunosuppressed patients. Lancet Infect. Dis. 2003, 3, 685–702. [Google Scholar] [CrossRef]
- McNeil, M.M.; Nash, S.L.; Hajjeh, R.A.; Phelan, M.A.; Conn, L.A.; Plikaytis, B.D.; Warnock, D.W. Trends in mortality due to invasive mycotic diseases in the United States, 1980–1997. Clin. Infect. Dis. 2001, 33, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Bodey, G.P.; Mardani, M.; Hanna, H.A.; Boktour, M.; Abbas, J.; Girgawy, E.; Hachem, R.Y.; Kontoyiannis, D.P.; Raad, I.I. The epidemiology of Candida glabrata and Candida albicans fungemia in immunocompromised patients with cancer. Am. J. Med. 2002, 112, 380–385. [Google Scholar] [CrossRef]
- Martino, P.; Girmenia, C.; Micozzi, A.; De Bernardis, F.; Boccanera, M.; Cassone, A. Prospective study of Candida colonization, use of empiric amphotericin B and development of invasive mycosis in neutropenic patients. Eur. J. Clin. Microbiol. Infect. Dis. 1994, 13, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Calderone, R.A.; Fonzi, W.A. Virulence factors of Candida albicans. Trends Microbiol. 2001, 9, 327–335. [Google Scholar] [CrossRef]
- Mann, P.A.; McLellan, C.A.; Koseoglu, S.; Si, Q.; Kuzmin, E.; Flattery, A.; Harris, G.; Sher, X.; Murgolo, N.; Wang, H. Chemical Genomics-Based Antifungal Drug Discovery: Targeting Glycosylphosphatidylinositol (GPI) Precursor Biosynthesis. ACS Infect. Dis. 2014, 1, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.; Diekema, D. Epidemiology of invasive candidiasis: A persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Gauwerky, K.; Borelli, C.; Korting, H.C. Targeting virulence: A new paradigm for antifungals. Drug Discov. Today 2009, 14, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Sanglard, D.; Kuchler, K.; Ischer, F.; Pagani, J.; Monod, M.; Bille, J. Mechanisms of resistance to azole antifungal agents in Candida albicans isolates from AIDS patients involve specific multidrug transporters. Antimicrob. Agents Chemother. 1995, 39, 2378–2386. [Google Scholar] [CrossRef] [PubMed]
- White, T.C.; Holleman, S.; Dy, F.; Mirels, L.F.; Stevens, D.A. Resistance mechanisms in clinical isolates of Candida albicans. Antimicrob. Agents Chemother. 2002, 46, 1704–1713. [Google Scholar] [CrossRef] [PubMed]
- Hong Nguyen, M.; Peacock, J.E., Jr.; Morris, A.J.; Tanner, D.C.; Nguyen, M.L.; Snydman, D.R.; Wagener, M.M.; Rinaldi, M.G.; Yu, V.L. The changing face of candidemia: Emergence of non-Candida albicans species and antifungal resistance. Am. J. Med. 1996, 100, 617–623. [Google Scholar] [CrossRef]
- Cowen, L.E.; Sanglard, D.; Calabrese, D.; Sirjusingh, C.; Anderson, J.B.; Kohn, L.M. Evolution of drug resistance in experimental populations of Candida albicans. J. Bacteriol. 2000, 182, 1515–1522. [Google Scholar] [CrossRef] [PubMed]
- Redding, S.; Smith, J.; Farinacci, G.; Rinaldi, M.; Fothergill, A.; Rhine-Chalberg, J.; Pfaller, M. Resistance of Candida albicans to fluconazole during treatment of oropharyngeal candidiasis in a patient with AIDS: Documentation by in vitro susceptibility testing and DNA subtype analysis. Clin. Infect. Dis. 1994, 18, 240–242. [Google Scholar] [CrossRef] [PubMed]
- Abbiw, D.K. Useful Plants of Ghana: West African Uses of Wild and Cultivated Plants; Intermediate Technology Publications and The Royal Botanic Gardens, Kew: London, UK, 1990. [Google Scholar]
- Hutchinson, J.; Dalziel, J.M. Flora of West Tropical Africa; Crown Agent London: London, UK, 1958; Volume 1, p. 1. [Google Scholar]
- Irvine, F.R. Woody Plants of Ghana; Oxford University Press: London, UK, 1961; p. 2. [Google Scholar]
- Borokini, T.I.; Omotayo, F.O. Phytochemical and ethnobotanical study of some selected medicinal plants from Nigeria. J. Med. Plants Res. 2012, 6, 1106–1118. [Google Scholar]
- Asomaning, W.A.; Amoako, C.; Oppong, I.V.; Phillips, W.R.; Addae-Mensah, I.; Osei-Twum, E.Y.; Waibel, R.; Achenbach, H. Pyrano- and dihydrofurano-isoflavones from Milletia thonningii. Phytochemistry 1995, 39, 1215–1218. [Google Scholar] [CrossRef]
- Kingsford-Adaboh, R.; Dittrich, B.; Hübschle, C.B.; Gbewonyo, W.S.; Okamoto, H.; Kimura, M.; Ishida, H. Invariom structure refinement, electrostatic potential and toxicity of 4-O-methylalpinumisoflavone, O,O-dimethylalpinumisoflavone and 5-O-methyl-4-O-(3-methylbut-2-en-1-yl) alpinumisoflavone. Acta Crystallogr. Sect. B Struct. Sci. 2006, 62, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Veena, C.K.; Morgan, J.B.; Mohammed, K.A.; Jekabsons, M.B.; Nagle, D.G.; Zhou, Y.-D. Methylalpinumisoflavone inhibits hypoxia-inducible factor-1 (HIF-1) activation by simultaneously targeting multiple pathways. J. Biol. Chem. 2009, 284, 5859–5868. [Google Scholar] [CrossRef] [PubMed]
- Sardari, S.; Nishibe, S.; Daneshtalab, M. Coumarins, the bioactive structures with antifungal property. Stud. Nat. Prod. Chem. 2000, 23, 335–393. [Google Scholar]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. BioMed. Res. Int. 2013, 963248. [Google Scholar] [CrossRef] [PubMed]
- Al-Amiery, A.A.; Kadhum, A.A.H.; Mohamad, A.B. Antifungal activities of new coumarins. Molecules 2012, 17, 5713–5723. [Google Scholar] [CrossRef] [PubMed]
- Curir, P.; Galeotti, F.; Dolci, M.; Barile, E.; Lanzotti, V. Pavietin, a Coumarin from Aesculus pavia with Antifungal Activity. J. Nat. Prod. 2007, 70, 1668–1671. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-K.; Leung, S.S.; Guilbert, C.; Jacobson, M.P.; McKerrow, J.H.; Podust, L.M. Structural characterization of CYP51 from Trypanosoma cruzi and Trypanosoma brucei bound to the antifungal drugs posaconazole and fluconazole. PLoS Negl. Trop. Dis. 2010, 4, e651–e651. [Google Scholar] [CrossRef] [PubMed]
- Rupp, B.; Raub, S.; Marian, C.; Höltje, H.-D. Molecular design of two sterol 14α-demethylase homology models and their interactions with the azole antifungals ketoconazole and bifonazole. J. Comput. Aided Mol. Des. 2005, 19, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Sanati, H.; Belanger, P.; Fratti, R.; Ghannoum, M. A new triazole, voriconazole (UK-109,496), blocks sterol biosynthesis in Candida albicans and Candida krusei. Antimicrob. Agents Chemother. 1997, 41, 2492–2496. [Google Scholar] [PubMed]
- Olivares, E.M.; Lwande, W.; Monache, F.D.; Marini Bettolo, G.B. A pyrano-isoflavone from seeds of Milletia thonningii. Phytochemistry 1982, 21, 1763–1765. [Google Scholar] [CrossRef]
- Khalid, S.A.; Waterman, P.G. Thonningine-A and thonningine-B: Two 3-phenylcoumarins from the seeds of Millettia thonningii. Phytochemistry 1983, 22, 1001–1003. [Google Scholar] [CrossRef]
- Asomaning, W.A.; Otoo, E.; Akoto, O.; Oppong, I.V.; Addae-Mensah, I.; Waibel, R.; Achenbach, H. Isoflavones and coumarins from Milletia thonningii. Phytochemistry 1999, 51, 937–941. [Google Scholar] [CrossRef]
- Mustafa, A.; Sidky, M.; Mahran, M. Untersuchungen an substituierten 7-Methyl-furochromonen; Khellin und Visnagin. Liebigs Ann. 1967, 704, 182–187. [Google Scholar] [CrossRef]
- Ahluwalia, V.K.; Singh, D.; Singh, R.P. A facile synthesis of 4-aryl-2H-1-benzopyran-2-ones. Monatsh. Chem. 1985, 116, 869–872. [Google Scholar] [CrossRef]
- Yu, B.; Reynisson, J. Bond stability of the “undesirable” heteroatom–heteroatom molecular moieties for high-throughput screening libraries. Eur. J. Med. Chem. 2011, 46, 5833–5837. [Google Scholar] [CrossRef] [PubMed]
- Drew, K.L.; Reynisson, J. The impact of carbon–hydrogen bond dissociation energies on the prediction of the cytochrome P450 mediated major metabolic site of drug-like compounds. Eur. J. Med. Chem. 2012, 56, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, A.; Sims, L.A.; Snead, R.; Gronert, S.; Maclagan, R.G.A.R.; Meot-Ner, M. Protonation Energies of 1–5-Ring Polycyclic Aromatic Nitrogen Heterocyclics: Comparing Experiment and Theory. J. Phys. Chem. A 2014, 119, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Safi, Z.S.; Omar, S. Proton affinity and molecular basicity of m- and p-substituted benzamides in gas phase and in solution: A theoretical study. Chem. Phys. Lett. 2014, 610–611, 321–330. [Google Scholar] [CrossRef]
- Soares, B.M.; da Silva, D.L.; Sousa, G.R.; Amorim, J.C.F.; de Resende, M.A.; Pinotti, M.; Cisalpino, P.S. In vitro photodynamic inactivation of Candida spp. growth and adhesion to buccal epithelial cells. J. Photochem. Photobiol. B 2009, 94, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Rybalchenko, N.P.; Prykhodko, V.A.; Nagorna, S.S.; Volynets, N.N.; Ostapchuk, A.N.; Klochko, V.V.; Rybalchenko, T.V.; Avdeeva, L.V. In vitro antifungal activity of phenylheptatriyne from Bidens cernua L. against yeasts. Fitoterapia 2010, 81, 336–338. [Google Scholar] [CrossRef] [PubMed]
- Mimee, B.; Labbé, C.; Pelletier, R.; Bélanger, R.R. Antifungal activity of flocculosin, a novel glycolipid isolated from Pseudozyma flocculosa. Antimicrob. Agents Chemother. 2005, 49, 1597–1599. [Google Scholar] [CrossRef] [PubMed]
- de Souza, W.; Fernandes Rodrigues, J.C. Sterol biosynthesis pathway as target for anti-trypanosomatid drugs. Interdis. Perspect. Infect. Dis. 2009, 642502. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.; McLeod, R.; Rice, D.; Ginger, M.; Chance, M.L.; Goad, L.J. Fatty acid and sterol metabolism: Potential antimicrobial targets in apicomplexan and trypanosomatid parasitic protozoa. Mol. Biochem. Parasitol. 2003, 126, 129–142. [Google Scholar] [CrossRef]
- Urbina, J. Lipid biosynthesis pathways as chemotherapeutic targets in kinetoplastid parasites. Parasitology 1997, 114, 91–99. [Google Scholar]
- Podust, L.M.; von Kries, J.P.; Eddine, A.N.; Kim, Y.; Yermalitskaya, L.V.; Kuehne, R.; Ouellet, H.; Warrier, T.; Alteköster, M.; Lee, J.-S. Small-molecule scaffolds for CYP51 inhibitors identified by high-throughput screening and defined by X-ray crystallography. Antimicrob. Agents Chemother. 2007, 51, 3915–3923. [Google Scholar] [CrossRef] [PubMed]
- Warrilow, A.; Hull, C.; Parker, J.; Garvey, E.; Hoekstra, W.; Moore, W.; Schotzinger, R.; Kelly, D.; Kelly, S. The clinical candidate VT-1161 is a highly potent inhibitor of Candida albicans CYP51 but fails to bind the human enzyme. Antimicrob. Agents Chemother. 2014, 58, 7121–7127. [Google Scholar] [CrossRef] [PubMed]
- Villaseñor-Granados, T.; García, S.; Vazquez, M.A.; Robles, J. Molecular docking-based screening of newly designed coumarin derivatives with potential antifungal activity against lanosterol 14α-demethylase. Theor. Chem. Acc. 2016, 135, 210. [Google Scholar] [CrossRef]
- Gidaro, M.C.; Alcaro, S.; Secci, D.; Rivanera, D.; Mollica, A.; Agamennone, M.; Giampietro, L.; Carradori, S. Identification of new anti-Candida compounds by ligand-based pharmacophore virtual screening. J. Enz. Inhib. Med. Chem. 2016, 31, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sagatova, A.A.; Keniya, M.V.; Wilson, R.K.; Sabherwal, M.; Tyndall, J.D.; Monk, B.C. Triazole resistance mediated by mutations of a conserved active site tyrosine in fungal lanosterol 14α-demethylase. Sci. Rep. 2016, 6, 26213. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Logan, G.; Reynisson, J. Wine Compounds as a Source for HTS Screening Collections. A Feasibility Study. Mol. Inf. 2012, 31, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.; Owen, P.; Scheinmann, F. Extractives from poisonous British plants. Part I. The structure of alpinumisoflavone, a new pyranoisoflavone from Laburnum alpinum J. Presl. J. Chem. Soc. C 1971, 3389–3392. [Google Scholar] [CrossRef]
- National Center for Infectious Disease, Center for Disease Control and Prevention, World Health Organisation. Laboratory Methods for the Diagnosis of Epidemic Dysentery and Cholera, Center for Disease Control and Prevention; Atlanta, GA, USA, 1999; pp. 71–72. [Google Scholar]
- Holder, I.A.; Boyce, S.T. Agar well diffusion assay testing of bacterial susceptibility to various antimicrobials in concentrations non-toxic for human cells in culture. Burns 1994, 20, 426–429. [Google Scholar] [CrossRef]
- Eloff, J. A sensitive and quick microplate method to determine the minimal inhibitory concentration of plant extracts for bacteria. Planta Med. 1998, 64, 711–713. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision D. 01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Becke, A. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098. [Google Scholar] [CrossRef]
- Becke, A. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785. [Google Scholar] [CrossRef]
- Hariharan, P.; Pople, J. The influence of polarization functions on molecular orbital hydrogenation energies. Theoret. chim. Acta 1973, 28, 213–222. [Google Scholar] [CrossRef]
- Frisch, M.J.; Pople, J.A.; Binkley, J.S. Self-consistent molecular orbital methods 25. Supplementary functions for Gaussian basis sets. J. Chem. Phys. 1984, 80, 3265–3269. [Google Scholar] [CrossRef]
- Wong, M. Vibrational frequency prediction using density functional theory. Chem. Phys. Lett. 1996, 256, 391–399. [Google Scholar] [CrossRef]
- Foresman, J.; Frisch, A.E. Exploring Chemistry with Electronic Structure Methods; Gaussian Inc.: Pittsburgh, PA, USA, 1996. [Google Scholar]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The protein data bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Berman, H.; Henrick, K.; Nakamura, H. Announcing the worldwide Protein Data Bank. Nat. Struct. Biol. 2003, 10, 980–980. [Google Scholar] [CrossRef] [PubMed]
- Scigress: Version FJ 2.6 (EU 3.1.7). In Scigress: Version FJ 2.6 (EU 3.1.7); Fijitsu Limited: Kanagawa, Japan, 2008–2016.
- Allinger, N.L. Conformational analysis. 130. MM2. A hydrocarbon force field utilizing V1 and V2 torsional terms. J. Am. Chem. Soc. 1977, 99, 8127–8134. [Google Scholar] [CrossRef]
- Jones, G.; Willett, P.; Glen, R.C.; Leach, A.R.; Taylor, R. Development and validation of a genetic algorithm for flexible docking. J. Mol. Biol. 1997, 267, 727–748. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, M.D.; Murray, C.W.; Auton, T.R.; Paolini, G.V.; Mee, R.P. Empirical scoring functions: I. The development of a fast empirical scoring function to estimate the binding affinity of ligands in receptor complexes. J. Comput. Aided Mol. Des. 1997, 11, 425–445. [Google Scholar] [CrossRef] [PubMed]
- Verdonk, M.L.; Cole, J.C.; Hartshorn, M.J.; Murray, C.W.; Taylor, R.D. Improved protein–ligand docking using GOLD. Proteins 2003, 52, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Kirton, S.B.; Murray, C.W.; Verdonk, M.L.; Taylor, R.D. Prediction of binding modes for ligands in the cytochromes P450 and other heme-containing proteins. Proteins 2005, 58, 836–844. [Google Scholar] [CrossRef] [PubMed]
- QikProp Version 3.2, Schrödinger: New York, NY, USA, 2009.
- Ioakimidis, L.; Thoukydidis, L.; Mirza, A.; Naeem, S.; Reynisson, J. Benchmarking the reliability of QikProp. Correlation between experimental and predicted values. QSAR Comb. Sci. 2008, 27, 445–456. [Google Scholar] [CrossRef]
- Sample Availability: Samples of all the compounds are available from the authors.
| Bond Dissociation | X | Y | Difference |
|---|---|---|---|
| Homolytic | 51.2 | 54.0 | 2.8 |
| Heterolytic | 216.0 | 227.8 | 11.8 |
| Homolytic-protonated | 42.5 | 62.7 | 20.2 |
| Heterolytic-protonated | 111.4 | 167.6 | 56.2 |
| Compound | ZOI (mm) | MIC (mg/mL) | MFC (mg/mL) | |||
|---|---|---|---|---|---|---|
| WILD | 18804 | WILD | 18804 | WILD | 18804 | |
| Robustic acid (1) | 10.3 | 14.7 | 0.25 | 1.00 | 1.0 | 1.0 |
| Thonningine-C (2) | 10.3 | 14.7 | 0.13 | 0.25 | 0.5 | 0.5 |
| Alpinumisoflavone (3) | 10.7 | 15.0 | 0.25 | 0.50 | F | F |
| O,O-dimethylalpinumisoflavone (4) | 0 | 0 | X | X | X | X |
| 4-O-methylalpinumisoflavone (5) | 0 | 0 | X | X | X | X |
| Acetyl-4-O-methylalpinumisoflavone (6) | 0 | 0 | X | X | X | X |
| Clotrimazole | 14.4 | 17.6 | X | X | X | X |
| DMSO (20%) | 0 | 0 | X | X | X | X |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayine-Tora, D.M.; Kingsford-Adaboh, R.; Asomaning, W.A.; Harrison, J.J.E.K.; Mills-Robertson, F.C.; Bukari, Y.; Sakyi, P.O.; Kaminta, S.; Reynisson, J. Coumarin Antifungal Lead Compounds from Millettia thonningii and Their Predicted Mechanism of Action. Molecules 2016, 21, 1369. https://doi.org/10.3390/molecules21101369
Ayine-Tora DM, Kingsford-Adaboh R, Asomaning WA, Harrison JJEK, Mills-Robertson FC, Bukari Y, Sakyi PO, Kaminta S, Reynisson J. Coumarin Antifungal Lead Compounds from Millettia thonningii and Their Predicted Mechanism of Action. Molecules. 2016; 21(10):1369. https://doi.org/10.3390/molecules21101369
Chicago/Turabian StyleAyine-Tora, Daniel M., Robert Kingsford-Adaboh, William A. Asomaning, Jerry J.E.K. Harrison, Felix C. Mills-Robertson, Yahaya Bukari, Patrick O. Sakyi, Sylvester Kaminta, and Jóhannes Reynisson. 2016. "Coumarin Antifungal Lead Compounds from Millettia thonningii and Their Predicted Mechanism of Action" Molecules 21, no. 10: 1369. https://doi.org/10.3390/molecules21101369
APA StyleAyine-Tora, D. M., Kingsford-Adaboh, R., Asomaning, W. A., Harrison, J. J. E. K., Mills-Robertson, F. C., Bukari, Y., Sakyi, P. O., Kaminta, S., & Reynisson, J. (2016). Coumarin Antifungal Lead Compounds from Millettia thonningii and Their Predicted Mechanism of Action. Molecules, 21(10), 1369. https://doi.org/10.3390/molecules21101369







