Chemical Composition and Bioactivities of the Essential Oil from Etlingera yunnanensis against Two Stored Product Insects
Abstract
:1. Introduction
2. Results and Discussion
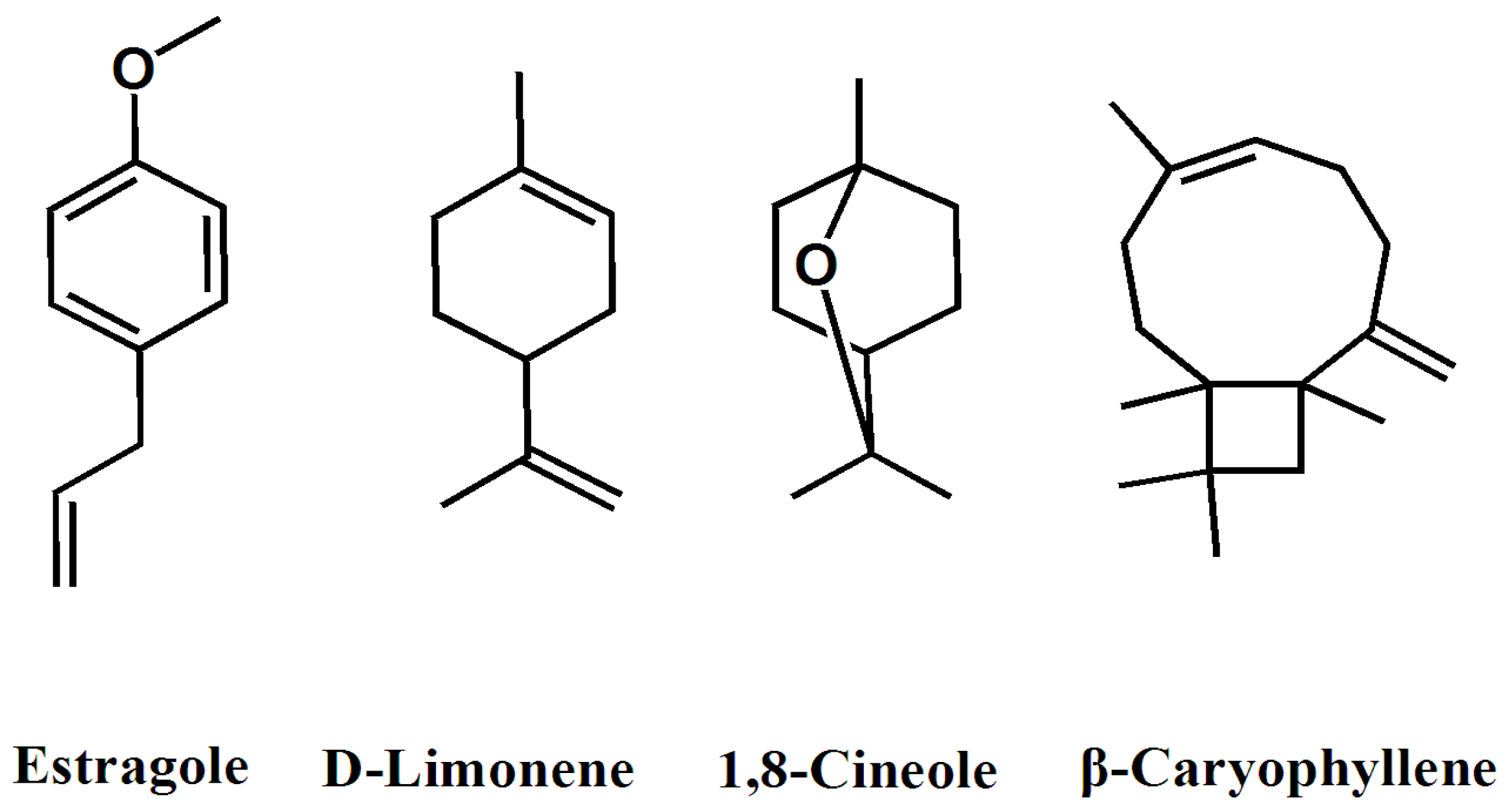
2.1. Chemical Composition of the Essential Oil
| Peak No. | Compound | RI * | Area % |
|---|---|---|---|
| 1 | α-Pinene | 932 | 2.4 |
| 2 | Camphene | 943 | 0.1 |
| 3 | Limonene | 1029 | 5.2 |
| 4 | 1,8-Cineole | 1032 | 6.4 |
| 5 | Camphor | 1120 | 1.0 |
| 6 | α-Terpineol | 1189 | 0.6 |
| 7 | Estragole | 1197 | 65.2 |
| 8 | Dodecane | 1203 | 0.1 |
| 9 | Isobornyl acetate | 1286 | 2.7 |
| 10 | β-Caryophyllene | 1420 | 6.4 |
| 11 | α-Caryophyllene | 1454 | 0.7 |
| 12 | α-Farnesene | 1489 | 0.1 |
| Monoterpenoids | 15.7 | ||
| Sesquiterpenes | 7.2 | ||
| Phenylpropanoid | 65.2 | ||
| Others | 2.8 | ||
| Total | 90.9 |
2.2. Contact Toxicity
| Insects | Treatments *** | LD50 (µg/adult; µg/cm2) | 95% FL (µg/adult; µg/cm2) | Slope ± SE | Chi Square (χ2) | p-Value |
|---|---|---|---|---|---|---|
| TC | Essential oil | 23.33 | 20.09–25.27 | 5.07 ± 0.57 | 13.29 | 0.946 |
| Estragole | 20.41 | 18.95–22.36 | 5.18 ± 0.53 | 15.64 | 0.87 | |
| β-Caryophyllene | 41.72 | 37.94–45.52 | 5.28 ± 0.54 | 18.58 | 0.725 | |
| 1,8-Cineole | 18.86 | 17.51–20.65 | 5.04 ± 0.51 | 18.00 | 0.757 | |
| Limonene | 13.40 | 11.72–15.49 | 3.23 ± 0.41 | 9.96 | 0.992 | |
| Pyrethrins * | 0.26 | 0.22–0.30 | 3.34 ± 0.32 | 13.11 | 0.950 | |
| LB | Essential oil | 47.38 | 45.66–49.11 | 14.09 ± 1.51 | 10.02 | 0.991 |
| Estragole | 30.22 | 28.13–32.49 | 8.04 ± 0.96 | 6.25 | 0.995 | |
| β-Caryophyllene | 74.11 | 71.26–77.75 | 10.40 ± 1.08 | 20.11 | 0.635 | |
| 1,8-Cineole | 321.20 | 302.38–333.90 | 12.49 ± 1.31 | 11.37 | 0.979 | |
| Limonene | 259.62 | 238.13–283.68 | 5.56 ± 0.57 | 16.10 | 0.851 | |
| Pyrethrins ** | 18.72 | 17.60–19.92 | 2.98 ± 0.40 | 10.56 | 0.987 |

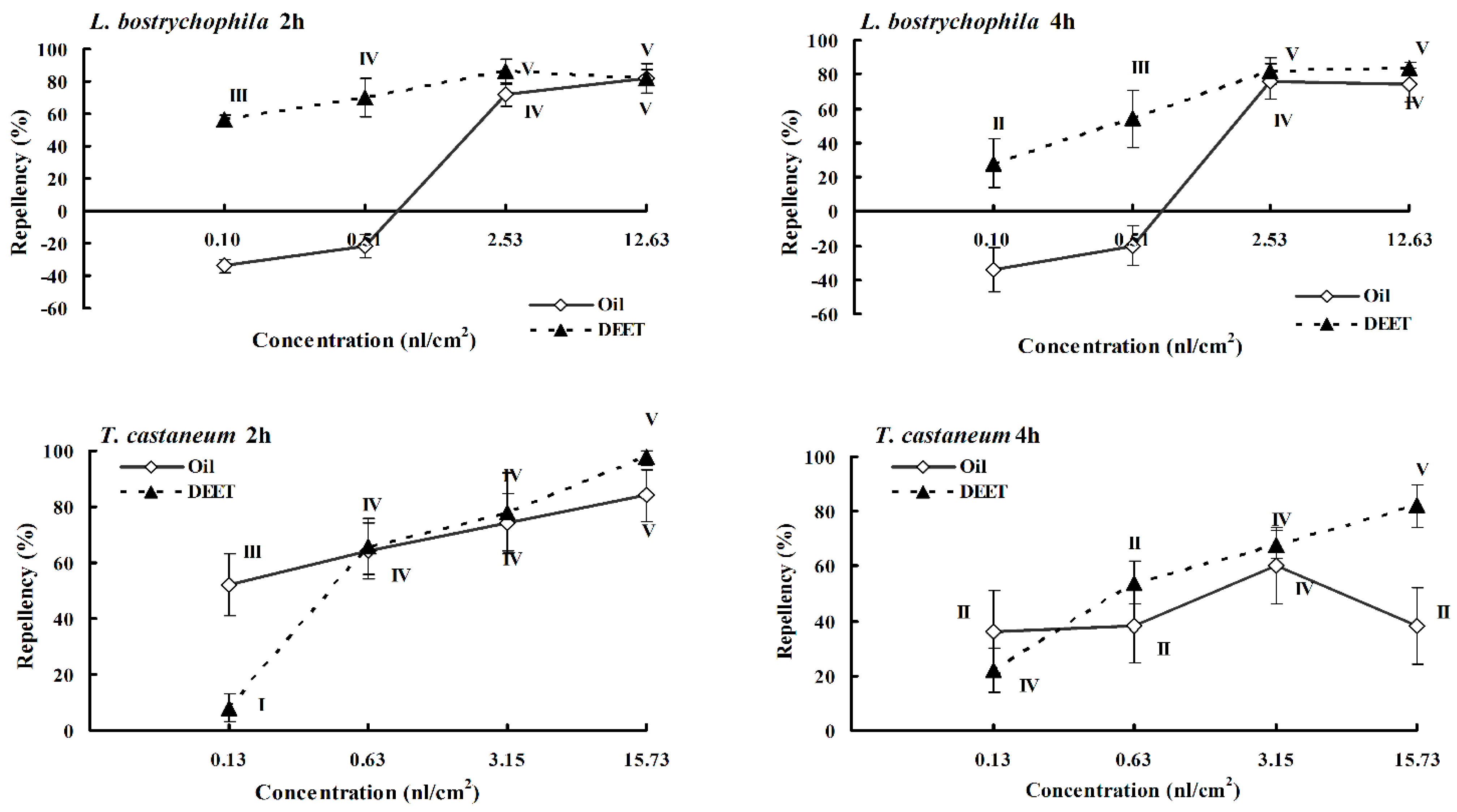
2.3. Repellency
| Insect | Treatment | 2 h/4 h (nL/cm2) | |||
|---|---|---|---|---|---|
| 15.73 | 3.15 | 0.63 | 0.13 | ||
| TC | Essential oil | 84 ± 9ab; 38 ± 14a | 74 ± 11b; 60 ± 14a | 64 ± 10a; 38 ± 13a | 52 ± 11bc; 36 ± 15ab |
| Estragole | 94 ± 6b; 82 ± 5b | 88 ± 5b; 68 ± 15a | 78 ± 13a; 64 ± 15a | 58 ± 13bc; 48 ± 16ab | |
| β-Caryophyllene | 80 ± 8ab; 98 ± 3b | 68 ± 8ab; 90 ± 5a | 38 ± 12a; 36 ± 13a | 36 ± 14ab; 34 ± 13ab | |
| 1,8-Cineole | 50 ± 13a; 74 ± 16ab | 28 ± 14a; 54 ± 15a | 42 ± 16a; 38 ± 12a | 28 ± 14ab; 40 ± 10ab | |
| Limonene | 88 ± 14b; 94 ± 8b | 70 ± 9b; 56 ± 16a | 76 ± 11a; 56 ± 14a | 78 ± 10c; 70 ± 12b | |
| DEET | 98 ± 3b; 82 ± 8b | 78 ± 14b; 68 ± 5a | 66 ± 10a; 54 ± 8a | 8 ± 5a; 22 ± 8a | |
| Insect | Treatment | 2 h/4 h nL/cm2 | |||
|---|---|---|---|---|---|
| 12.63 | 2.53 | 0.51 | 0.10 | ||
| LB | Essential oil | 82 ± 9ab; 74 ± 10a | 72 ± 7a; 76 ± 10a | −22 ± 7a; −20 ± 12a | -34 ± 4bc; −34 ± 13ab |
| Estragole | −54 ± 9b; −40 ± 3b | 76 ± 6a; 38 ± 16a | 0 ± 16a; 20 ± 15a | 22 ± 11bc; −30 ± 12ab | |
| β-Caryophyllene | 36 ± 8ab; 24 ± 18b | −24 ± 8ab; 32 ± 14a | 24 ± 16a; 42 ± 14a | 24 ± 15ab; 32 ± 16ab | |
| 1,8-Cineole | 38 ± 14a; 28 ± 12ab | 12 ± 8a; 6 ± 14a | −10 ± 10a; 10 ± 14a | −16 ± 11ab; 14 ± 11ab | |
| Limonene | 64 ± 7b; 52 ± 10b | 30 ± 10b; 16 ± 11a | 24 ± 15a; 20 ± 6a | 36 ± 13c; 18 ± 12b | |
| DEET | 82 ± 5b; 84 ± 3b | 86 ± 8b; 82 ± 8a | 70 ± 12a; 54 ± 17a | 56 ± 3a; 28 ± 14a | |
3. Experimental Section
3.1. Plant Material and Extractions
3.2. Insects
3.3. Gas Chromatography and Mass Spectrometry (GC-MS)
3.4. Contact Toxicity
3.5. Repellency
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Copping, L.G.; Menn, J.J. Biopesticides: A review of their action, applications and efficacy. Pest Manag. Sci. 2000, 8, 651–676. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef] [PubMed]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.I.; Roh, J.Y.; Kim, D.H.; Lee, H.S.; Ahn, Y.J. Insecticidal activities of aromatic plant extracts and essential oils against Sitophilus oryzae and Callosobruchus chinensis. J. Stored Prod. Res. 2003, 39, 293–303. [Google Scholar] [CrossRef]
- Zhao, N.N.; Zhou, L.G.; Liu, Z.L.; Du, S.S.; Deng, Z.W. Evaluation of the toxicity of the essential oils of some common Chinese spices against Liposcelis bostrychophila. Food Control. 2012, 26, 486–490. [Google Scholar] [CrossRef]
- Zhu, W.X.; Zhao, K.; Chu, S.S.; Liu, Z.L. Evaluation of essential oil and its three main active ingredients of Chinese Chenopodium ambrosioides (Family: Chenopodiaceae) against Blattella germanica. J. Arthropod Borne Dis. 2012, 6, 90–97. [Google Scholar] [PubMed]
- Liang, Y.; Li, J.L.; Xu, S.; Zhao, N.N.; Zhou, L.G.; Cheng, J.; Liu, Z.L. Evaluation of repellency of some Chinese medicinal herbs essential oils against Liposcelis bostrychophila (Psocoptera: Liposcelidae) and Tribolium castaneum (Coleoptera: Tenebrionidae). J. Econ. Entomol. 2013, 106, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.I.; Abdelgaleil, S.A.M. Chemical composition and insecticidal potential of essential oils from Egyptian plants against Sitophilus oryzae (L.) (Coleoptera: Curculionidae) and Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Appl. Entomol. Zool. 2008, 43, 599–607. [Google Scholar] [CrossRef]
- Liu, Z.L.; Yu, M.; Li, X.M.; Wan, T.; Chu, S.S. Repellent activity of eight essential oils of Chinese medicinal herbs to Blattella germanica L. Rec. Nat. Prod. 2011, 5, 176–183. [Google Scholar]
- Ding, W.; Liu, H.; Long-Shu, L.I. The main stratagems and technology for stored product pest control in ancient China. J. Southwest Agric. Univ. 2000, 22, 335–338. [Google Scholar]
- Yang, K.; You, C.X.; Wang, C.F.; Guo, S.S.; Li, Y.P.; Wu, Y.; Geng, Z.F.; Deng, Z.W.; Du, S.S. Composition and repellency of the essential oils of Evodia calcicola Chun ex Huang and Evodia trichotoma (Lour.) Pierre against three stored product insects. J. Oleo Sci. 2014, 63, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.H. Flora Reipublicae Popularis Sinicae; Science Press: Beijing, China, 1981; Volume 16, p. 137. [Google Scholar]
- Liu, Z.L.; Ho, S.H. Bioactivity of the essential oil extracted from Evodia rutaecarpa Hook f. et Thomas against the grain storage insects, Sitophilus zeamais Motsch. and Tribolium castaneum (Herbst). J. Stored Prod. Res. 1999, 35, 317–328. [Google Scholar] [CrossRef]
- Magan, N.; Hope, R.; Cairns, V.; Aldred, D. Postharvest fungal ecology: Impact of fungal growth and mycotoxin accumulation in stored grain. Eur. J. Plant. Pathol. 2003, 109, 723–730. [Google Scholar] [CrossRef]
- Nayak, M.K.; Daglish, G.J.; Byrne, V.S. Effectiveness of spinosad as a grain protectant against resistant beetle and psocid pests of stored grain in Australia. J. Stored Prod. Res. 2005, 41, 455–467. [Google Scholar] [CrossRef]
- Zhou, T. Studies on Reproductive Strategy of Two Species in Etlingera. Master’s Thesis, South China Normal University, Guangzhou, China, 2012. [Google Scholar]
- State administration of traditional Chinese medicine. Chinese Materia Medica (No. 34); Shanghai Scientific and Technical Publishers: Shanghai, China, 2005; pp. 173–174. [Google Scholar]
- Fahlbusch, K.G.; Hammerschmidt, F.J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. Flavors and fragrances. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Misztal, P.K.; Owen, S.M.; Guenther, A.B.; Rasmussen, R.; Geron, C.; Harley, P.; Phillips, G.J.; Ryan, A.; Edwards, D.P.; Hewitt, C.N. Large estragole fluxes from oil palms in Borneo. Atmos. Chem. Phys. 2010, 10, 4343–4358. [Google Scholar] [CrossRef] [Green Version]
- Zeller, A.; Rychlik, M. Impact of estragole and other odorants on the flavour of anise and tarragon. Flavour Fragr. J. 2007, 22, 105–113. [Google Scholar] [CrossRef]
- Chang, C.L.; Cho, I.K.; Li, Q.X. Insecticidal activity of basil oil, trans-anethole, estragole, and linalool to adult fruit flies of Ceratitis capitata, Bactrocera dorsalis, and Bactrocera cucurbitae. J. Econ. Entomol. 2009, 102, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, D. Toxicity of basil and orange essential oils and their components against two coleopteran stored products insect pests. J. Asia-Pac. Entomol. 2014, 17, 13–17. [Google Scholar] [CrossRef]
- Albuquerque, A.A.; Sorenson, A.L.; Lealcardoso, J.H. Effects of essential oil of Croton zehntneri, and of anethole and estragole on skeletal muscles. J. Ethnopharmacol. 1995, 49, 41–49. [Google Scholar] [CrossRef]
- Okunade, A.L.; Olaifa, J.I. Estragole: An acute toxic principle from the volatile oil of the leaves of Clausena anisata. J. Nat. Prod. 1987, 50, 990–991. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.F.; Yang, K.; Zhang, H.M.; Cao, J.; Fang, R.; Liu, Z.L.; Du, S.S.; Wang, Y.Y.; Deng, Z.W.; Zhou, L.G. Components and insecticidal activity against the maize weevils of Zanthoxylum schinifolium fruits and leaves. Molecules 2011, 16, 3077–3088. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.F. Chemical Constituents and their Bioactivities of Zanthoxylum Plants against Stored-Product Insects in Chinese Medicinal Materials. Ph.D. Thesis, Beijing Normal University, Beijing, 2013. [Google Scholar]
- Zhou, H.Y.; Zhao, N.N.; Du, S.S.; Yang, K.; Wang, C.F.; Liu, Z.L.; Qiao, Y.J. Insecticidal activity of the essential oil of Lonicera japonica flower buds and its main constituent compounds against two grain storage insects. J. Med. Plants Res. 2012, 6, 912–917. [Google Scholar]
- Chau, D.T.; Hoi, T.M.; Thai, T.H.; Thang, T.D.; Ogunwande, I.A. Essential oil constituents of Etlingera yunnanensis and Hornstedtia sanhan grown in Vietnam. Nat. Prod. Commun. 2015, 10, 365–366. [Google Scholar] [PubMed]
- Wong, K.C.; Sivasothy, Y.; Boey, P.L.; Osman, H.; Sulaiman, B. Essential oils of Etlingera elatior (Jack) R.M. Smith and Etlingera littoralis (Koenig) Giseke. J. Essent. Oil Res. 2010, 22, 461–466. [Google Scholar] [CrossRef]
- Wang, C.F.; Yang, K.; You, C.X.; Zhang, W.J.; Guo, S.S.; Geng, Z.F.; Du, S.S.; Wang, Y.Y. Chemical composition and insecticidal activity of essential oils from Zanthoxylum dissitum leaves and roots against three species of storage pests. Molecules 2015, 20, 7990–7999. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Wang, C.F.; You, C.X.; Geng, Z.F.; Sun, R.Q.; Guo, S.S.; Du, S.S.; Liu, Z.L.; Deng, Z.W. Bioactivity of essential oil of Litsea cubeba from China and its main compounds against two stored product insects. J. Asia-Pac. Entomol. 2014, 17, 459–466. [Google Scholar] [CrossRef]
- Liu, X.C.; Zhou, L.G.; Liu, Z.L.; Du, S.S. Identification of insecticidal constituents of the essential oil of Acorus calamus rhizomes against Liposcelis bostrychophila Badonnel. Molecules 2013, 8, 5684–5696. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.L.; Zhao, N.N.; Liu, C.M.; Zhou, L.G.; Du, S.S. Identification of insecticidal constituents of the essential oil of Curcuma wenyujin rhizomes active against Liposcelis bostrychophila Badonnel. Molecules 2012, 17, 12049–12060. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.C.; Li, Y.P.; Li, H.Q.; Deng, Z.W.; Zhou, L.G.; Liu, Z.G.; Du, S.S. Identification of repellent and insecticidal constituents of the essential oil of Artemisia rupestris L. aerial parts against Liposcelis bostrychophila Badonnel. Molecules 2013, 18, 10733–10746. [Google Scholar] [CrossRef] [PubMed]
- You, C.X.; Yang, K.; Wu, Y.; Zhang, W.J.; Wang, Y.; Geng, Z.F.; Chen, H.P.; Jiang, H.Y.; Du, S.S.; Deng, Z.W. Chemical composition and insecticidal activities of the essential oil of Perilla frutescens (L.) Britt aerial parts against two stored product insects. Eur. Food Res. Technol. 2014, 239, 481–490. [Google Scholar] [CrossRef]
- Zhang, W.J.; You, C.X.; Yang, K.; Wang, Y.; Su, Y.; Geng, Z.F.; Du, S.S.; Wang, C.F.; Deng, Z.W.; Wang, Y.Y. Bioactivity and chemical constituents of the essential oil from Dendranthema indicum (L.) Des Moul against two stored insects. J. Oleo Sci. 2015, 64, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Ahn, Y.J. Contact and fumigant activities of constituents of Foeniculum vulgare fruit against three coleopteran stored-product insects. Pest. Manag. Sci. 2001, 57, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Huignard, J.; Lapied, B.; Dugravot, S.; Magnin-Robert, M.; Ketoh, G.K. Modes d’action neurotoxiques des dérivés Soufrés et de certaines huiles essentielles et risques liés à leur utilisation. In Biopesticides D’origine Végétale, 2nd ed.; Lavoisier: Paris, France, 2008; pp. 219–231. [Google Scholar]
- López, M.D.; Pascual-Villalobosa, M.J. Mode of inhibition of acetylcholinesterase by monoterpenoids and implications for pest control. Ind. Crops Prod. 2010, 31, 284–288. [Google Scholar] [CrossRef]
- Opinion of the Scientific Committee on Food on Estragole (1-Allyl-4-methoxybenzene). Available online: http://ec.europa.eu/food/fs/sc/scf/out104_en.pdf (accessed on 5 October 2001).
- Regulation (EC) No 1334/2008 of the European Parliament and of the Council of 16 December 2008 on Flavourings and Certain Food Ingredients with Flavouring Properties for Use in and on Foods and Amending Council. Regulation (EEC) No 1601/91, Regulations (EC) No 2232/96 and (EC) No 110/2008 and Directive 2000/13/EC. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1440545390594&uri=CELEX:32008R1334 (accessed on 16 December 2008).
- Carratù, B.; Federici, E.; Gallo, F.R.; Geraci, A.; Guidotti, M.; Multari, G.; Palazzino, G.; Sanzini, E. Plants and parts of plants used in food supplements: An approach to their safety assessment. Ann. Ist. Super. Sanità 2010, 46, 370–388. [Google Scholar] [PubMed]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils: A review. Bioresour. Technol. 2010, 101, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential oil Components by Gas Chromatography/Quadrupole Mass Spectroscopy; Allured: Carol Stream, IL, USA, 2001. [Google Scholar]
- Sakuma, M. Probit analysis of preference data. Appl. Entomol. Zool. 1998, 33, 339–347. [Google Scholar]
- Zhang, J.S.; Zhao, N.N.; Liu, Q.Z.; Liu, Z.L.; Du, S.S.; Zhou, L.G.; Deng, Z.W. Repellent constituents of essential oil of Cymbopogon distans aerial parts against two stored-product insects. J. Agric. Food Chem. 2011, 59, 9910–9915. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, S.-S.; You, C.-X.; Liang, J.-Y.; Zhang, W.-J.; Geng, Z.-F.; Wang, C.-F.; Du, S.-S.; Lei, N. Chemical Composition and Bioactivities of the Essential Oil from Etlingera yunnanensis against Two Stored Product Insects. Molecules 2015, 20, 15735-15747. https://doi.org/10.3390/molecules200915735
Guo S-S, You C-X, Liang J-Y, Zhang W-J, Geng Z-F, Wang C-F, Du S-S, Lei N. Chemical Composition and Bioactivities of the Essential Oil from Etlingera yunnanensis against Two Stored Product Insects. Molecules. 2015; 20(9):15735-15747. https://doi.org/10.3390/molecules200915735
Chicago/Turabian StyleGuo, Shan-Shan, Chun-Xue You, Jun-Yu Liang, Wen-Juan Zhang, Zhu-Feng Geng, Cheng-Fang Wang, Shu-Shan Du, and Ning Lei. 2015. "Chemical Composition and Bioactivities of the Essential Oil from Etlingera yunnanensis against Two Stored Product Insects" Molecules 20, no. 9: 15735-15747. https://doi.org/10.3390/molecules200915735






