Isolation, Identification and Cytotoxicity of a New Noroleanane-Type Triterpene Saponin from Salicornia bigelovii Torr.
Abstract
:1. Introduction
2. Results and Discussion
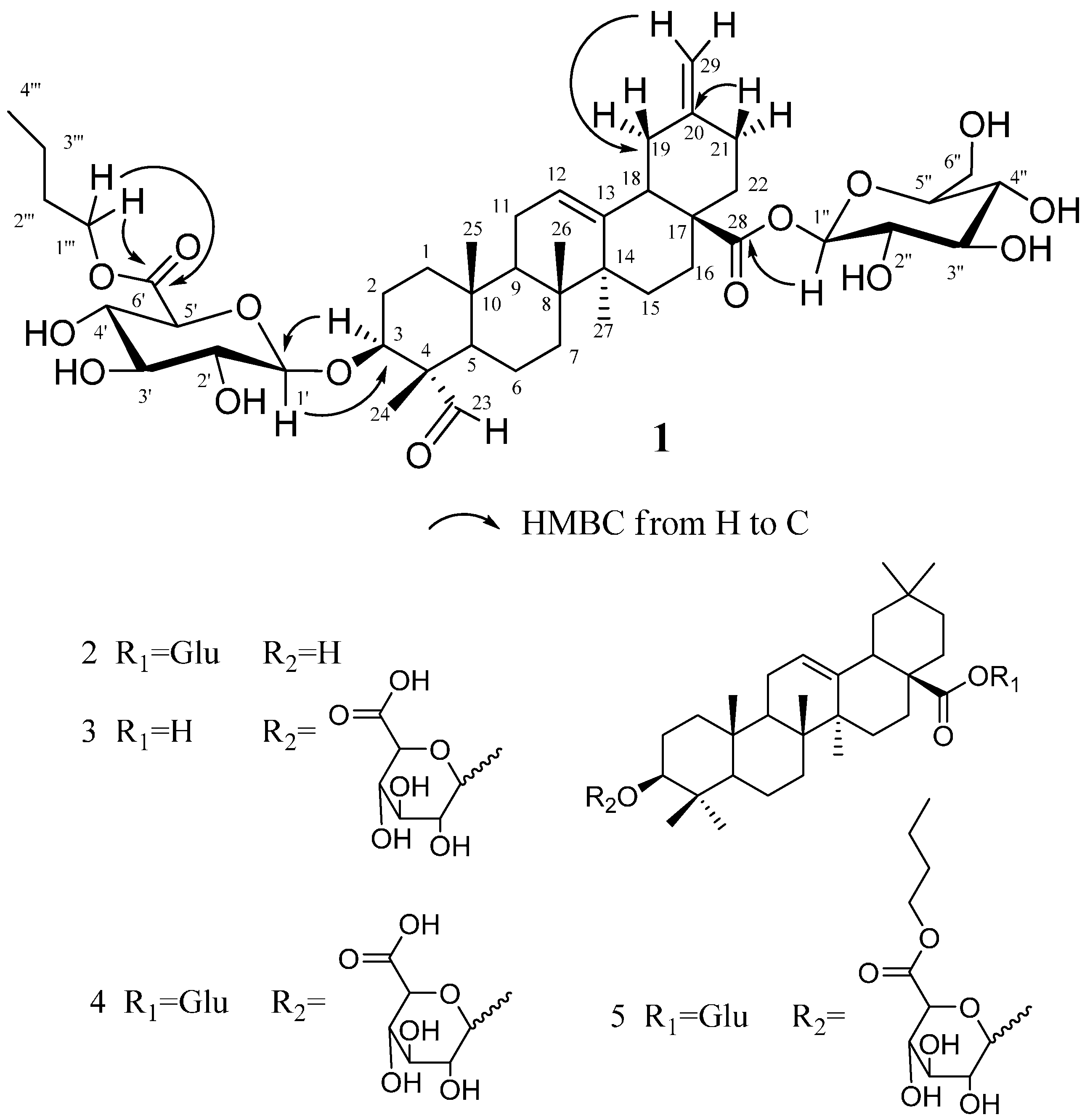
2.1. Structural Elucidation of the Isolated Compounds
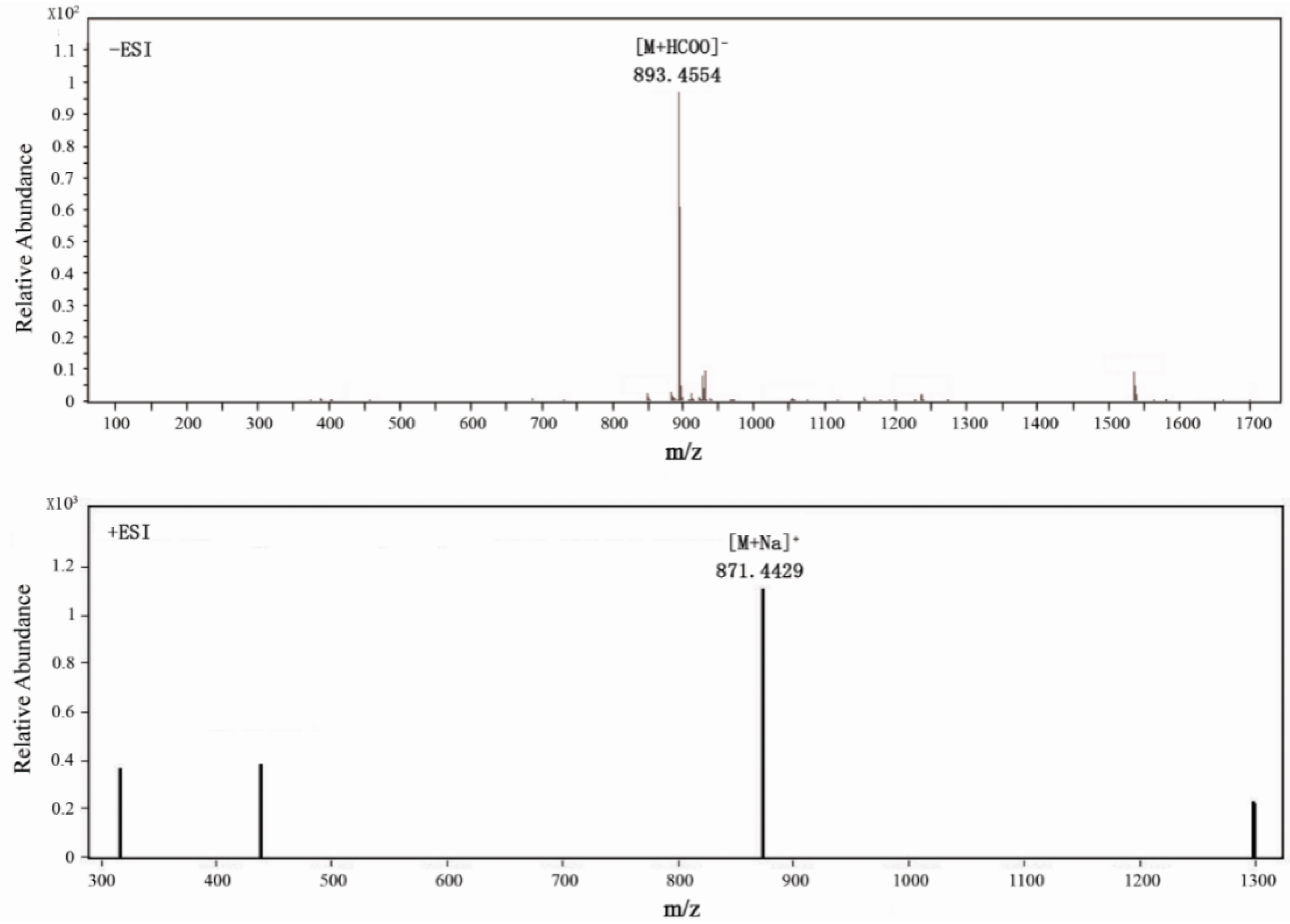
| No. | δC, Type | δH (J in Hz) | 1H-1HCOSY | ROESY | HMBC |
|---|---|---|---|---|---|
| 1 | 38.1, CH2 | 1.48, 0.93, m | 2 | 2 | |
| 2 | 25.2, CH2 | 2.16, 1.85, m | 1, 3 | 1,3 | |
| 3 | 82.2, CH | 4.14, dd (11.6, 4.5) | 2 | 2, 23, 1', 5 | 23, 1', 5, 24 |
| 4 | 55.4, C | ||||
| 5 | 47.9, CH | 1.35, dd (7.8, 7.2) | 6 | 3, 6, 23 | |
| 6 | 20.4, CH2 | 1.37, 0.98, m | 5, 7 | 5, 7 | |
| 7 | 32.4, CH2 | 1.40, 1.16, m | 6 | 6 | |
| 8 | 40.2, C | ||||
| 9 | 47.9, CH | 1.63, m | 11 | 11 | 10, 8, 11, 25, 26 |
| 10 | 36.1, C | ||||
| 11 | 23.6, CH2 | 1.84, m | 9, 12 | 9, 12 | |
| 12 | 123.3, CH | 5.40, t (3.5) | 11 | 11 | 14, 18 |
| 13 | 143.5, C | ||||
| 14 | 42.1, C | ||||
| 15 | 28.1, CH2 | 2.23, 1.11, m | 16 | 16 | |
| 16 | 23.5, CH2 | 2.14, 2.01, m | 15 | 15 | 18 |
| 17 | 47.3, C | ||||
| 18 | 47.6, CH | 3.08, dd (8.7, 4.65) | 19 | 19 | 20 |
| 19 | 41.6, CH2 | 2.57, m 2.18, t (13.3, 8.7) | 18 | 18 | |
| 20 | 148.4, C | ||||
| 21 | 30.1, CH2 | 2.19, 2.03, m | 22 | 22 | |
| 22 | 37.6, CH2 | 1.98, 1.68, m | 21 | 21 | 20 |
| 23 | 206.8, CH | 9.74, s | 3, 5 | ||
| 24 | 10.4, CH3 | 1.28, s | 25 | 4, 3, 23 | |
| 25 | 15.6, CH3 | 0.83, s | 24 | 10, 1 | |
| 26 | 17.4, CH3 | 1.01, s | 7, 8 | ||
| 27 | 26.0, CH3 | 1.20, s | 15, 8 | ||
| 28 | 175.6, C | ||||
| 29 | 107.2, CH2 | 4.74, 4.67, s | 19, 21 | ||
| 3-O-GlcA | |||||
| GlcA-1' | 105.4, CH | 4.87, d (7.75) | 2' | 2', 3 | 3 |
| GlcA-2' | 75.0, CH | 3.94, d (8.5) | 3', 1' | 3', 1' | 1', 3' |
| GlcA-3' | 77.8, CH | 4.16, t (8.5, 7.2) | 2', 4' | 2', 4' | 1' |
| GlcA-4' | 72.9, CH | 4.43, t (7.2, 9.75) | 3', 5' | 3', 5' | 6' |
| GlcA-5' | 77.3, CH | 4.50, d (9.75) | 4' | 4' | 6', 4', 1' |
| GlcA-6' | 170.1, C | ||||
| 28-O-Glc | |||||
| Glc-1'' | 95.8, CH | 6.23, d (8.1) | 2'' | 2'' | 28 |
| Glc-2'' | 74.0, CH | 4.12, d (8.55) | 3'', 1'' | 3'', 1'' | 1'', 3'' |
| Glc-3'' | 78.8, CH | 4.22, t (8.55, 7.85) | 2'', 4'' | 2'', 4'' | 4'', 2'' |
| Glc-4'' | 71.2, CH | 4.27, t (7.85, 9.95) | 5'', 3'' | 5'', 3'' | 5'', 6'' |
| Glc-5'' | 79.2, CH | 3.97, d (9.95) | 4'', 6'' | 4'', 6'' | |
| Glc-6'' | 62.3, CH2 | 4.41, brd (11.8) 4.33, dd (11.8, 4.55) | 5'' | 5'' | |
| 6'-O-butyl | |||||
| 1''' | 65.0, CH2 | 4.26, t (7.35) | 2''' | 2''' | 6', 2''', 3''' |
| 2''' | 30.8, CH2 | 1.57, m | 1''', 3''' | 1''', 3''' | 4''', 3''' |
| 3''' | 19.2, CH2 | 1.33, m | 2''', 4''' | 2''', 4''' | 1''', 4''' |
| 4''' | 13.7, CH3 | 0.76, t (7.4) | 3''' | 3''' | 2''', 3''' |
2.2. Three of the Isolated Compounds, Including Bigelovii C, Had Potent Antiproliferative Activity in Vitro
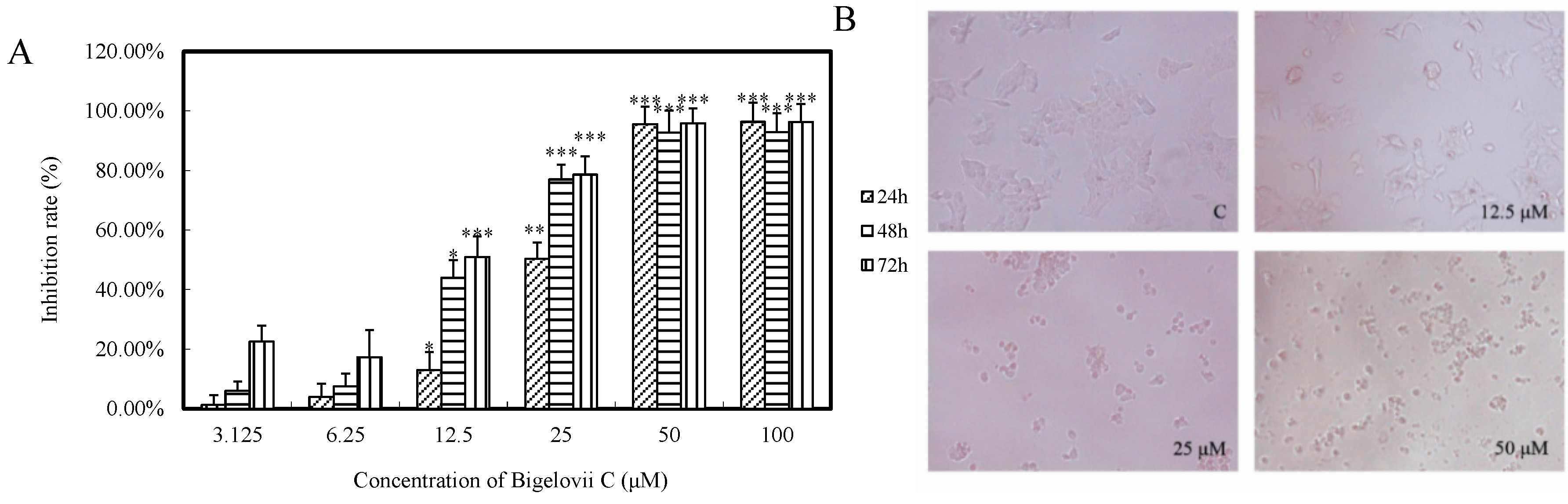
| Compounds | MCF7 | Lovo | LN229 |
|---|---|---|---|
| 1 | 12.6 ± 2.49 | 21.35 ± 2.55 | 29.66 ± 6.99 |
| 2 | >100 | >100 | >100 |
| 3 | 13.42 ± 1.22 | 22.15 ± 3.61 | 31.18 ± 4.72 |
| 4 | >100 | >100 | >100 |
| 5 | 3.52 ± 1.19 | 11.45 ± 2.58 | 10.71 ± 2.26 |
| Cis-platinum | 14.73 ± 5.30 | 51.90 ± 7.15 | 55.24 ± 7.82 |
2.3. Effects of Bigelovii C on Cell Apoptosis and Mitochondrial Membrane Potential
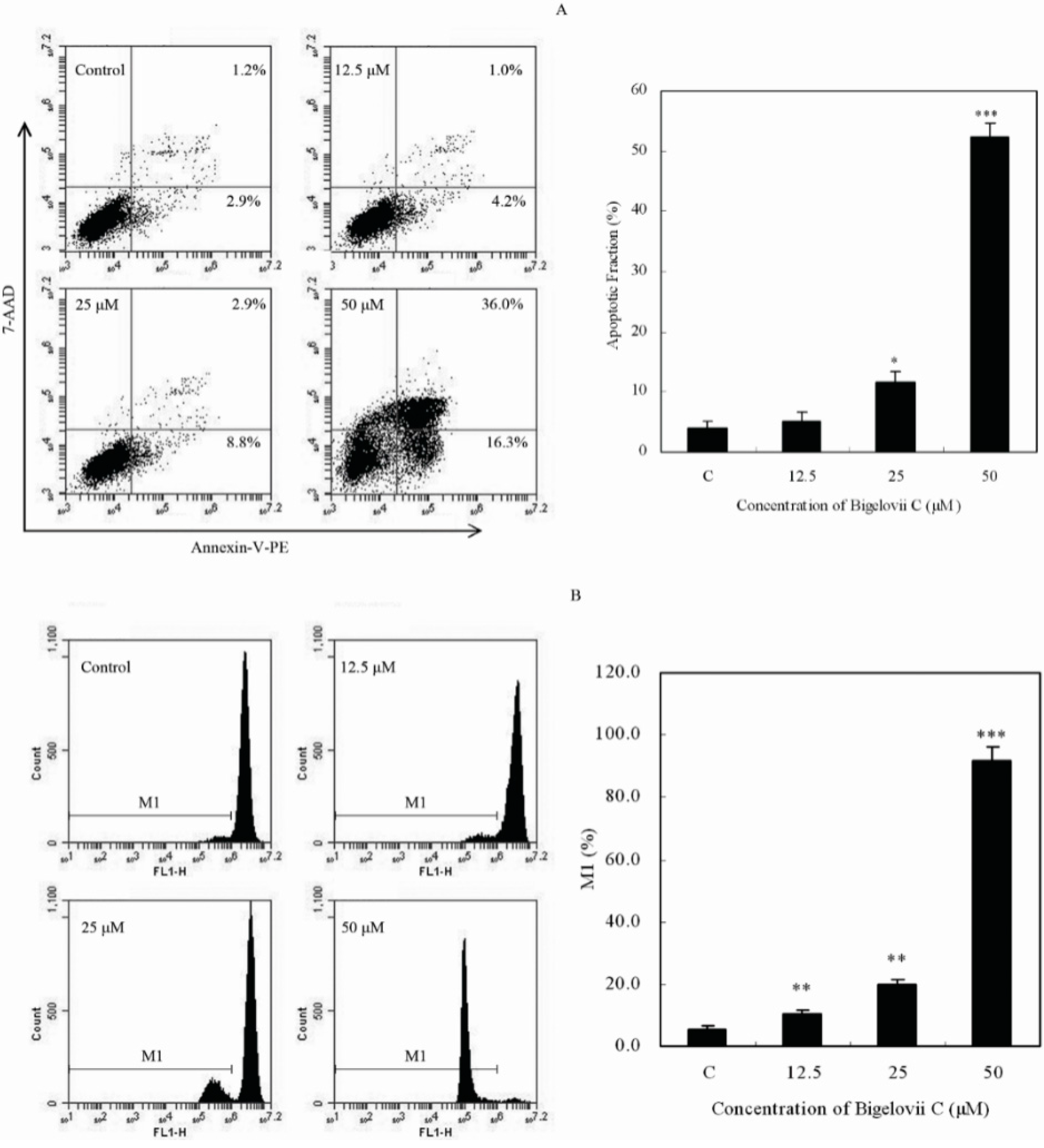
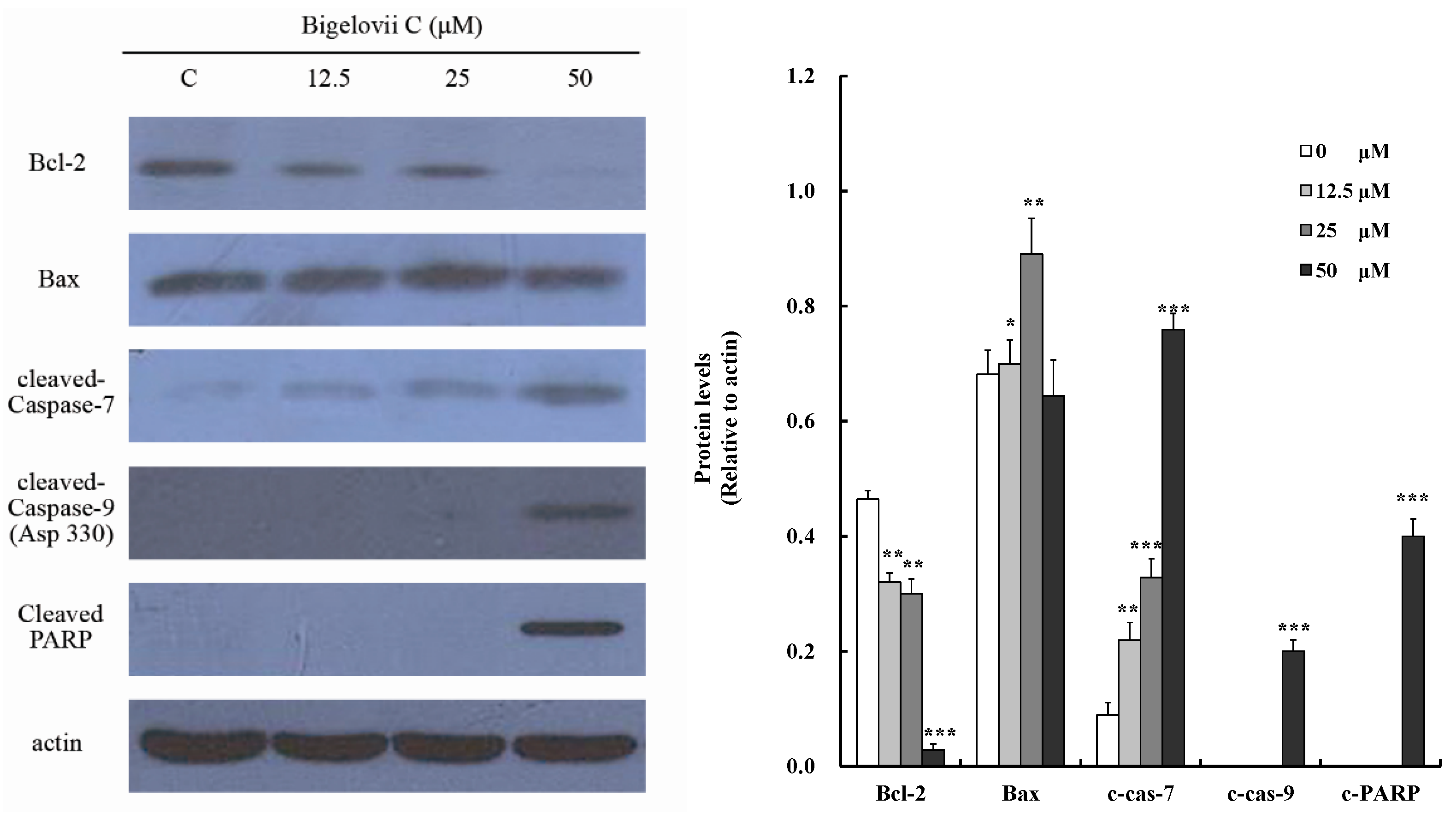
2.4. Effects of Bigelovii C on Apoptosis-Related Protein Levels in MCF7 Cells
2.5. Discussion
3. Experimental Section
3.1. General Methods
3.2. Materials and Chemicals
3.3. Extraction and Isolation
3.4. Cell Culture
3.5. Cytotoxicity Assay
3.6. Annexin-V-PE/7-AAD Analysis
3.7. Analysis of the Mitochondrial Membrane Potential
3.8. Western Blotting
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lu, Z.J.; Hodges, R.M.; Mota-Urbina, C.J.; Gallawa, P.L.; Chaturvedi, R.; Hodges, C.N. Nutrient constituents of Salicornia (Salicornia bigelovii Torr.): A seawater-irrigated new green vegetable. Hortscience 2001, 36, 484. [Google Scholar]
- Feng, L.T.; Weng, L. The development and utilization of Salicornia Bigelovii. J. Salt Chem. Ind. 2009, 38, 38–42. [Google Scholar]
- Glenn, E.P.; O’leary, J.W.; Watson, M.C.; Thompson, T.L.; Kuehl, R.O. Salicornia bigelovii Torr.: An oilseed halophyte for seawater irrigation. Science 1991, 251, 1065–1067. [Google Scholar] [CrossRef] [PubMed]
- Guan, F.Q.; Shan, Y.; Zhao, Y.Y.; Wang, Q.Z.; Wang, M.; Sun, H.; Chen, Y.; Yin, M.; Feng, X. Research development on pharmacological activities and mechanism of Salicornia plants. Chin. Pharm. Bull. 2013, 29, 1188–1191. [Google Scholar]
- Kim, J.Y.; Cho, J.; Ma, Y.; Park, K.Y.; Lee, S.; Ham, K.; Lee, H.J.; Park, K.; Moon, J. Dicaffeoylquinic acid derivatives and flavonoid glucosides from glasswort (Salicornia herbacea L.) and their antioxidative activity. Food Chem. 2011, 125, 55–62. [Google Scholar] [CrossRef]
- Wang, Q.Z.; Liu, X.F.; Shan, Y.; Guan, F.Q.; Chen, Y.; Wang, X.Y.; Wang, M.; Feng, X. Two new nortriterpenoid saponins from Salicornia bigelovii Torr. and their cytotoxic activity. Fitoterapia 2012, 83, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.A.; Kong, C.S.; Lee, J.I.; Kim, H.; Park, H.Y.; Lee, H.S.; Lee, C.; Seo, Y. Evaluation of novel antioxidant triterpenoid saponins from the halophyte Salicornia herbacea. Bioorg. Med. Chem. Lett. 2012, 22, 4318–4322. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.H.; Wang, X.M.; Wang, H.; Liu, T.X.; Xin, Z.H. Two new noroleanane-type triterpene saponins from the methanol extract of Salicornia herbacea. Food Chem. 2014, 151, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Guan, F.Q.; Wang, H.T.; Shan, Y.; Zhang, D.M.; Zhao, Y.Y.; Chen, Y.; Wang, Q.Z.; Wang, M.; Feng, X. Bigelovii A induced apoptosis of human acute promyelocytic leukaemia HL-60 cells. Mol. Med. Rep. 2013, 7, 1585–1590. [Google Scholar] [PubMed]
- Rabi, T.; Banerjee, S. Novel Synthetic triterpenoid methyl 25-hydroxy-3-oxoolean-12-en-28-oate induces apoptosis through JNK and p38 MAPK pathways in human breast adenocarcinoma MCF-7 cells. Mol. Carcinog. 2008, 47, 415–423. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, C.; Siegel, R.; Bandi, P.; Jemal, A. Breast cancer statistics. CA Cancer J. Clin. 2011, 61, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A.; Ahmed, S.; Brankov, N.; Perloff, M. Triterpenoids as potential agents for the chemoprevention and therapy of breast cancer. Front. Biosci. 2011, 16, 980–996. [Google Scholar] [CrossRef]
- Bang, S.C.; Lee, J.H.; Song, G.Y.; Kim, D.H.; Yoon, M.Y.; Ahn, B.Z. Antitumor activity of Pulsatilla koreana saponins and their structure-activity relationship. Chem. Pharm. Bull. 2005, 53, 1451–1454. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.R.; Fang, W.S. Pentacyclic triterpenoids and their saponins with apoptosis-inducing activity. Curr. Top. Med. Chem. 2009, 9, 1581–1596. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; O’Rourke, K.; Chinnaiyan, A.M.; Gentz, R.; Ebner, R.; Ni, J.; Dixit, V.M. The receptor for the cytotoxic ligand TRAIL. Science 1997, 276, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Zapata, J.M.; Pawlowski, K.; Haas, E.; Ware, C.F.; Godzik, A.; Reed, J.C. A diverse family of proteins containing tumor necrosis factor receptor-associated factor domains. J. Biol. Chem. 2001, 276, 24242–24252. [Google Scholar] [CrossRef] [PubMed]
- Yip, K.W.; Reed, J.C. Bcl-2 family proteins and cancer. Oncogene 2008, 27, 6398–6406. [Google Scholar] [CrossRef] [PubMed]
- Ashe, P.C.; Berry, M.D. Apoptotic signaling cascades. Prog. Neuropsychopharmacol. Biol. Psychiatry 2003, 27, 199–214. [Google Scholar] [CrossRef] [PubMed]
- Gambi, N.; Tramontano, F.; Quesada, P. Poly(ADPR)polymerase inhibition and apoptosis induction in cDDP-treated human carcinoma cell lines. Biochem. Pharmacol. 2008, 75, 2356–2363. [Google Scholar] [CrossRef] [PubMed]
- Scovassi, A.I.; Poirier, G.G. Poly(ADP-ribosylation) and apoptosis. Mol. Cell. Biochem. 1999, 199, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, J.; Degraff, W.G.; Gazdar, A.F. Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–941. [Google Scholar] [PubMed]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guan, F.; Wang, Q.; Wang, M.; Shan, Y.; Chen, Y.; Yin, M.; Zhao, Y.; Feng, X.; Liu, F.; Zhang, J. Isolation, Identification and Cytotoxicity of a New Noroleanane-Type Triterpene Saponin from Salicornia bigelovii Torr. Molecules 2015, 20, 6419-6431. https://doi.org/10.3390/molecules20046419
Guan F, Wang Q, Wang M, Shan Y, Chen Y, Yin M, Zhao Y, Feng X, Liu F, Zhang J. Isolation, Identification and Cytotoxicity of a New Noroleanane-Type Triterpene Saponin from Salicornia bigelovii Torr. Molecules. 2015; 20(4):6419-6431. https://doi.org/10.3390/molecules20046419
Chicago/Turabian StyleGuan, Fuqin, Qizhi Wang, Ming Wang, Yu Shan, Yu Chen, Min Yin, Youyi Zhao, Xu Feng, Fei Liu, and Jianhua Zhang. 2015. "Isolation, Identification and Cytotoxicity of a New Noroleanane-Type Triterpene Saponin from Salicornia bigelovii Torr." Molecules 20, no. 4: 6419-6431. https://doi.org/10.3390/molecules20046419





