Role of the Red Ginseng in Defense against the Environmental Heat Stress in Sprague Dawley Rats
Abstract
:1. Introduction
2. Results
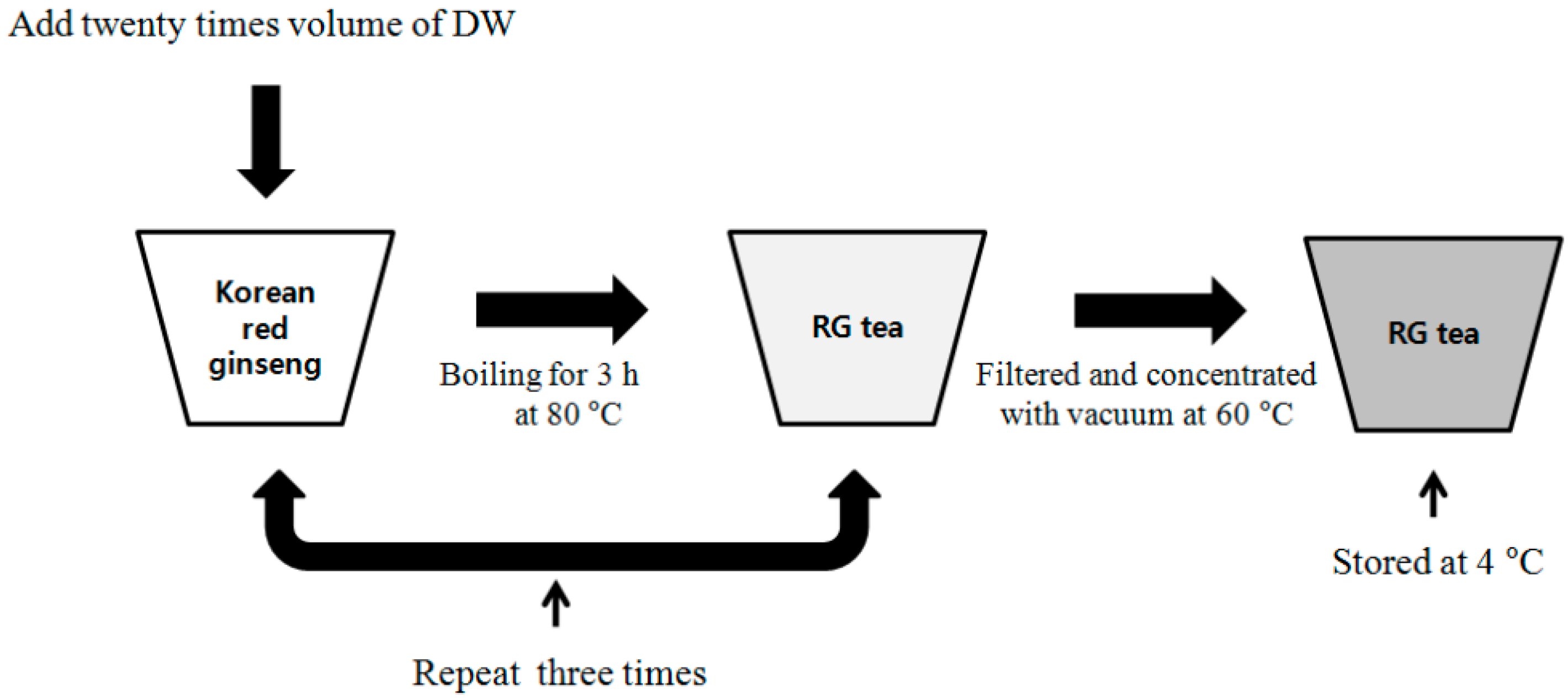
2.1. Extraction Process and Chemical Compositions of Red Ginseng
| Oligosaccharide | Acidic Polysaccharide | Phenolic Compounds | Saponin | Ginsenoside | |
|---|---|---|---|---|---|
| Rb1 | Rg1 | ||||
| 60.57 mg | 7.58 mg | 0.92 mg | 5.58 mg | 0.26 mg | 0.18 mg |
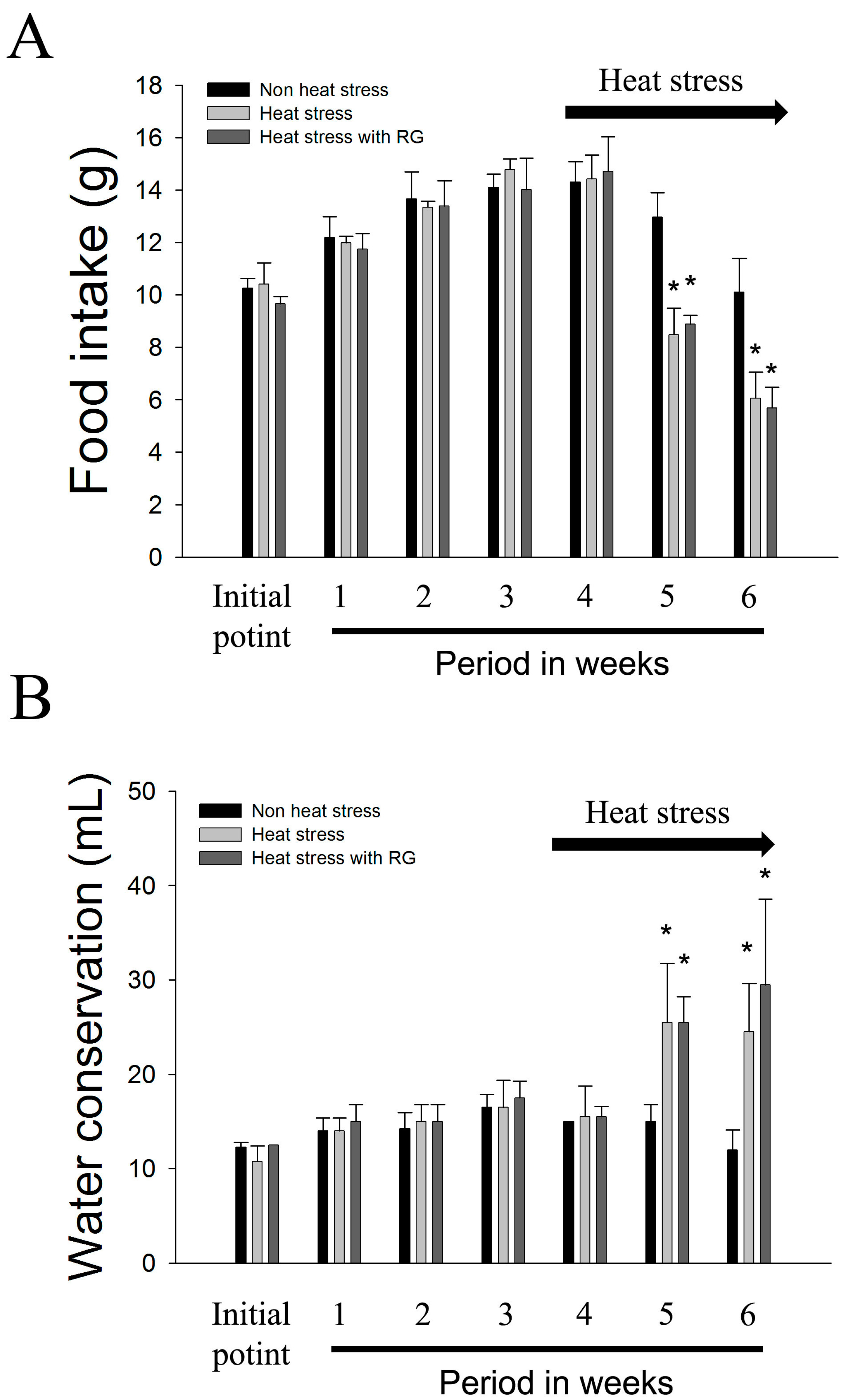
2.2. Body Weights, Food Intake, and Water Conservation

| Groups (n = 10) | Organ Weights (g) | ||||
|---|---|---|---|---|---|
| Liver | Kidney | Lung | Brain | Spleen | |
| Non heat stress | 10.08 ± 0.69 | 2.50 ± 0.15 | 1.48 ± 0.13 | 1.90 ± 0.04 | 0.75 ± 0.08 |
| Heat stress | 7.31 ± 0.87 * | 2.19 ± 0.21 * | 1.29 ± 0.14 * | 1.86 ± 0.06 | 0.53 ± 0.08 * |
| Heat stress with RG | 7.04 ± 0.47 * | 2.25 ± 0.13 * | 1.22 ± 0.15 * | 1.92 ± 0.08 | 0.55 ± 0.05 * |
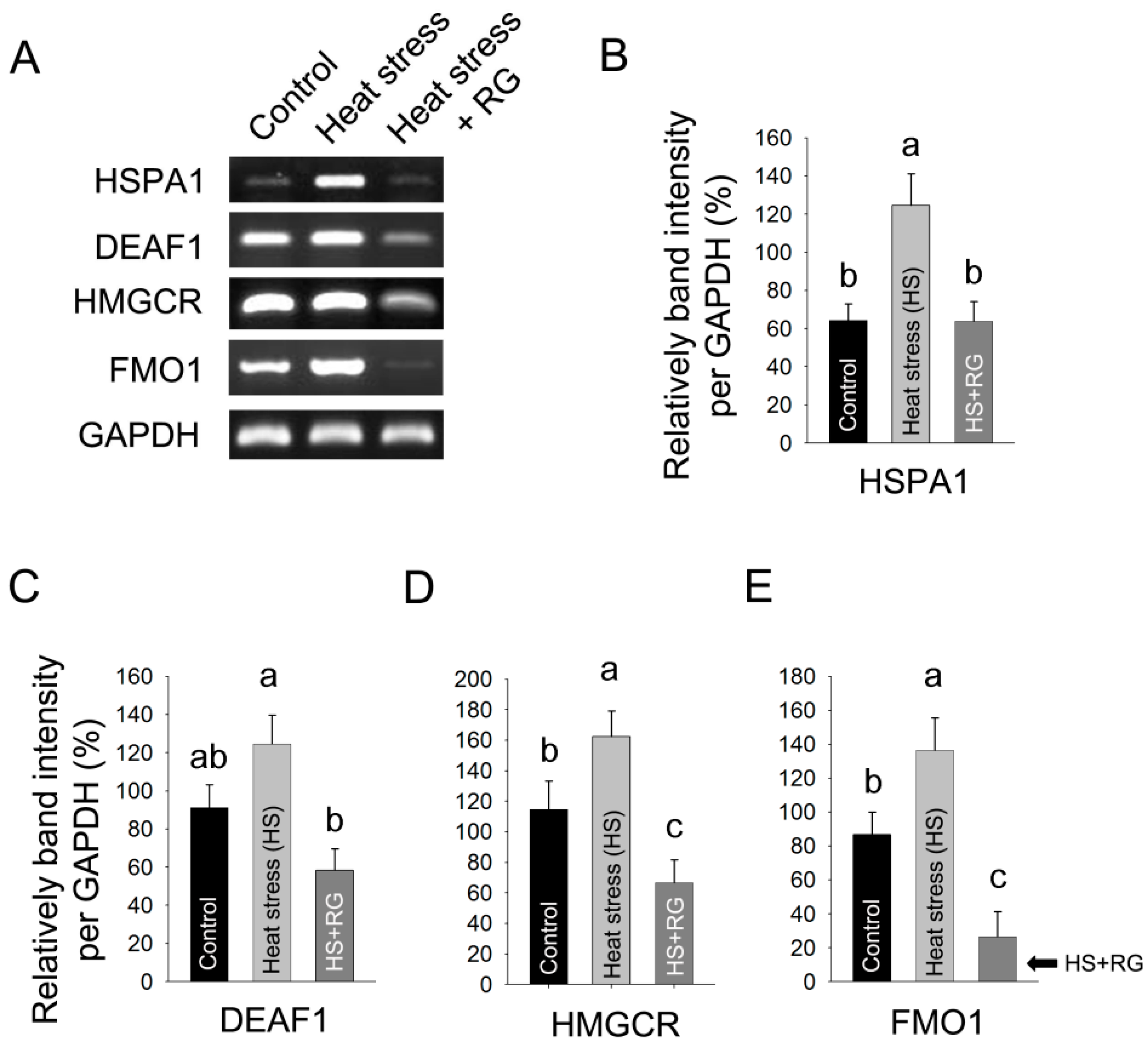
2.3. Red Ginseng Regulates Heat Stress Inducible Hepatic Gene Expression in Rat
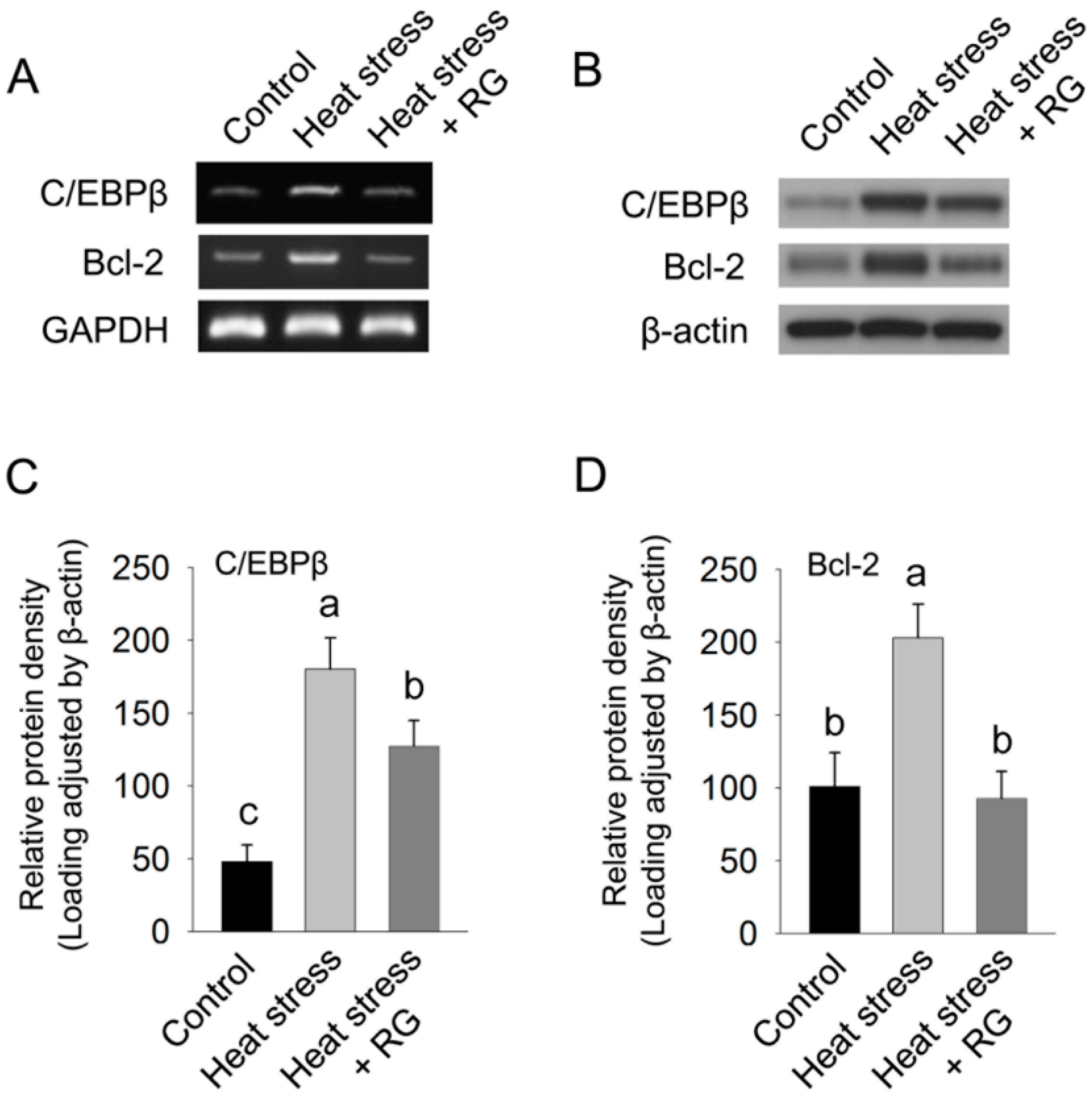
2.4. Red Ginseng Suppresses Heat Environment Stress Induced Lipid Accumulation and Lipid Peroxidation Related Gene Expression
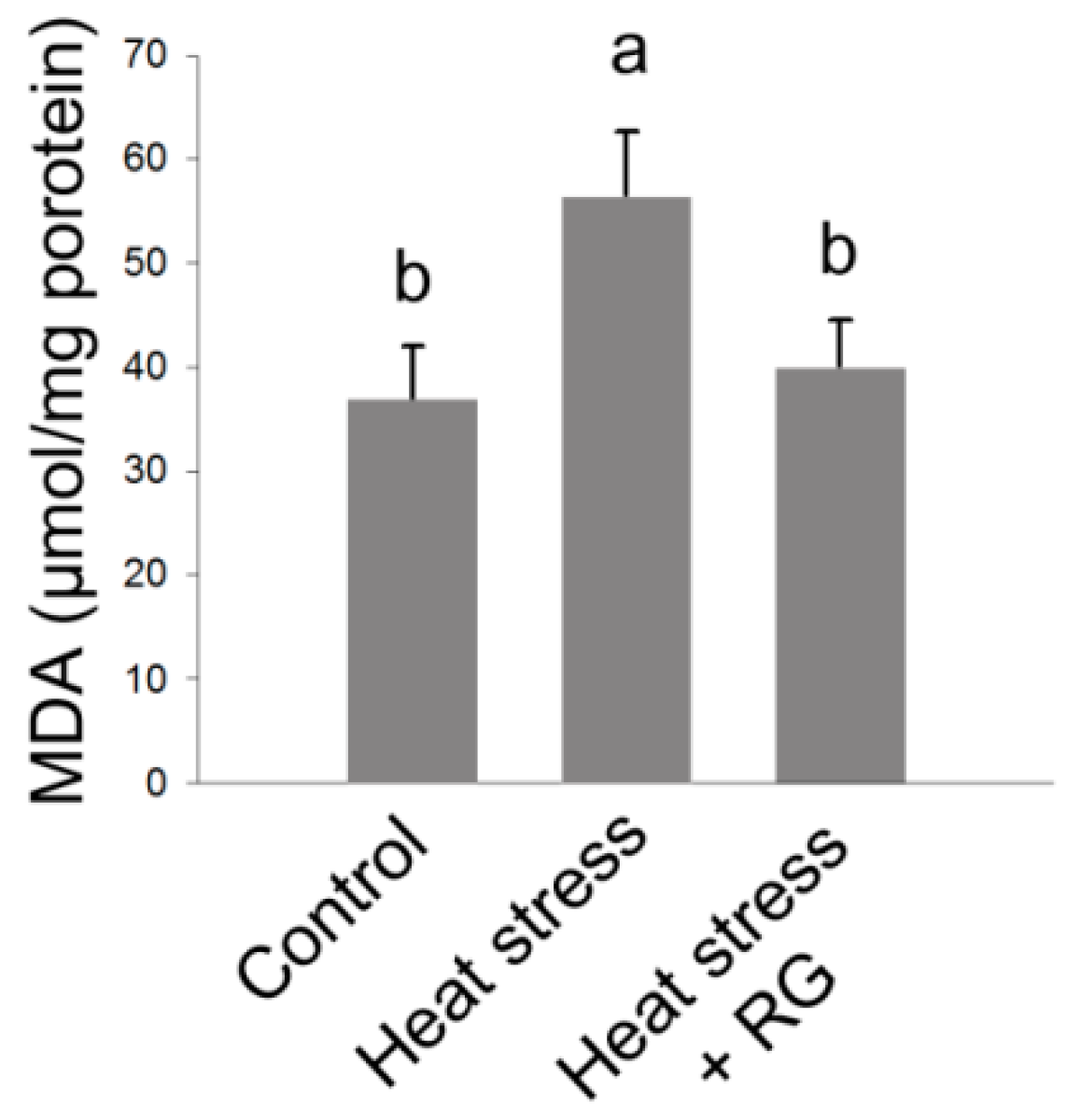
2.5. Effect of Red Ginseng on Hepatic Oxidative Stress in Environmental Heat Stress
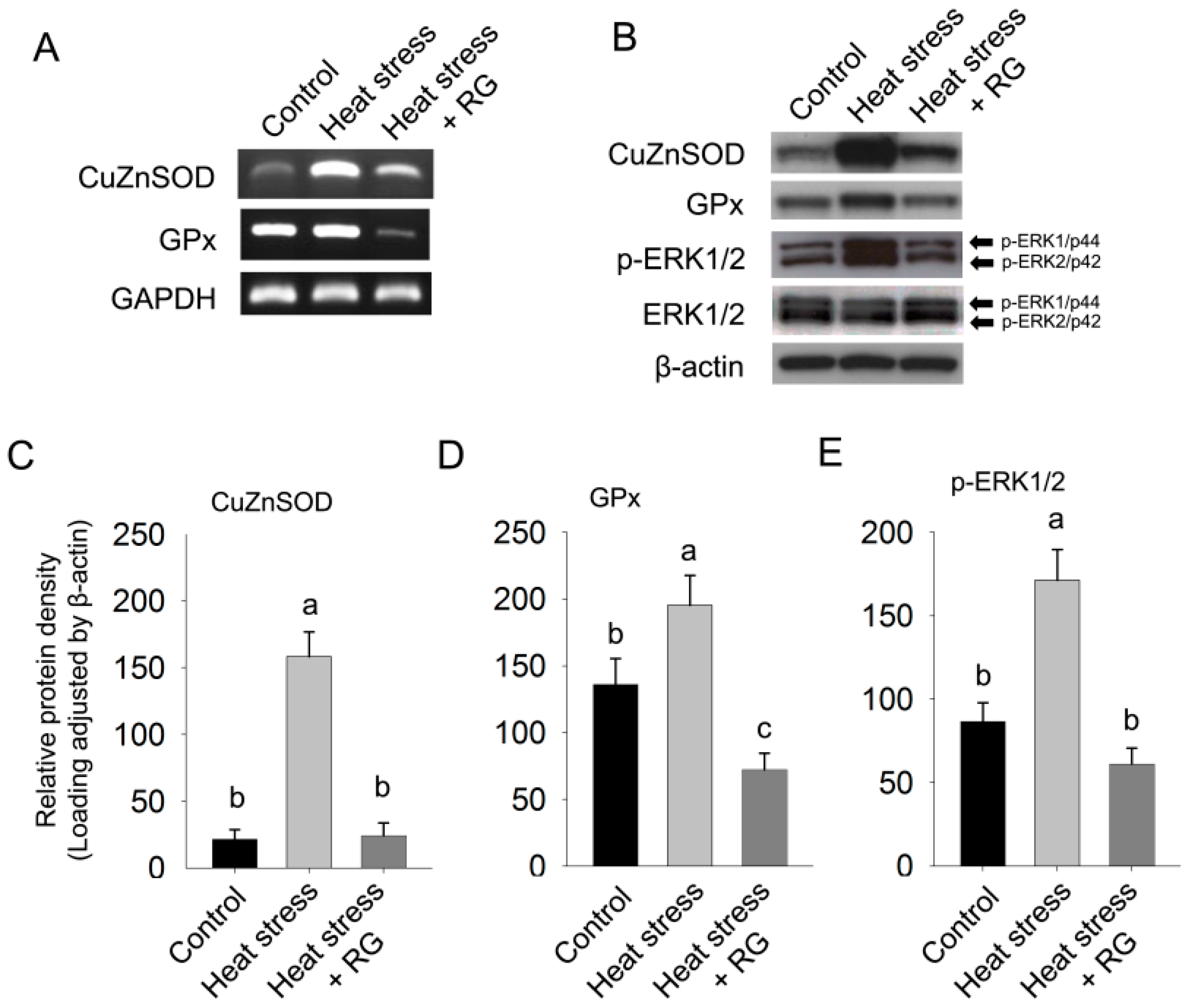
2.6. Discussion
3. Experimental Section
3.1. Materials
3.2. Animal Husbandry and Maintenance
3.3. Heat Environmental Stress Study Design
3.4. Preparation of Red Ginseng Extracts
3.5. Measurement of Total Phenolic Content in Red Ginseng
3.6. Determination of Acidic Polysaccharide in Red Ginseng
3.7. RNA Isolation and Semi-quantitative RT-PCR
| Gene | Sense Primer, 5′→3′ | Antisense Primer, 5′→3′ |
|---|---|---|
| GAPDH | TGATGACATCAAGAAGGTGG | TTTCTTACTCCTTGGAGGCC |
| HMGCR | GAGGCGCAACTGGAAACT | GCGGACGTCTGTGTAGAAGA |
| DEAF1 | AGAAGCCATCACAGGCTTAG | AGAAAGAGCACCGTGATAGG- |
| FMO1 | TGTCAAGGGAAGCAAAGC | CCTGAATCAAAGACTCGGC |
| HSPA1 | CTTACCTGGGCCAGAAGGT | ATGGTCAGGATGGACACGT |
| CuZnSOD | GTTCCGAGGCCGCCGCGCGT | GTCCCCATATTGATGGAC |
| GPx | CTCTCCGCGGTGGCACAGT | CCACCACCGGGTCGGACATAC |
3.8. Preparation of Liver Homogenate for Malondiadehyde
3.9. Western Blot Analysis
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Post, E.; Forchhammer, M.C.; Bret-Harte, M.S.; Callaghan, T.V.; Christensen, T.R.; Elberling, B.; Fox, A.D.; Gilg, O.; Hik, D.S.; Hoye, T.T.; et al. Ecological dynamics across the arctic associated with recent climate change. Science 2009, 325, 1355–1358. [Google Scholar] [CrossRef] [PubMed]
- Myers, S.S.; Bernstein, A. The coming health crisis: Indirect health effects of global climate change. F1000 Biol. Rep. 2011, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- McGeehin, M.A.; Mirabelli, M. The potential impacts of climate variability and change on temperature-related morbidity and mortality in the United States. Environ. Health Perspect. 2001, 109 (Suppl 2), 185–189. [Google Scholar] [CrossRef] [PubMed]
- Baker, T.A.; Romero, J.; Bach, H.H.T.; Strom, J.A.; Gamelli, R.L.; Majetschak, M. Systemic release of cytokines and heat shock proteins in porcine models of polytrauma and hemorrhage*. Crit. Care Med. 2012, 40, 876–885. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.G. Heat shock factors and the control of the stress response. Biochem. Pharmacol. 2000, 59, 55–63. [Google Scholar] [CrossRef]
- Ando, M.; Katagiri, K.; Yamamoto, S.; Wakamatsu, K.; Kawahara, I.; Asanuma, S.; Usuda, M.; Sasaki, K. Age-related effects of heat stress on protective enzymes for peroxides and microsomal monooxygenase in rat liver. Environ. Health Perspect. 1997, 105, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Bao, E.; Yu, J. Heat shock protein 60 expression in heart, liver and kidney of broilers exposed to high temperature. Res. Vet. Sci. 2009, 86, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Jian, B.; Hsieh, C.H.; Chen, J.; Choudhry, M.; Bland, K.; Chaudry, I.; Raju, R. Activation of endoplasmic reticulum stress response following trauma-hemorrhage. Biochim. Biophys. Acta 2008, 1782, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Hong, H.D.; Lee, O.H.; Lee, B.Y. The effects of acanthopanax senticosus on global hepatic gene expression in rats subjected to heat environmental stress. Toxicology 2010, 278, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Yoon, K.Y.; Hong, H.D.; Lee, B.Y. Schisandra chinensis prevents hepatic lipid peroxidation and oxidative stress in rats subjected to heat environmental stress. Phytother. Res. 2012, 26, 1674–1680. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.B.; Ha, J.H.; Hawer, W.D.; Nahmgung, B.; Lee, B.-Y. Ginsenoside contents of Korean white ginseng and taegeuk ginseng with various sizes and cultivation years. Korean J. Food Sci. Technol. 2005, 37, 508–512. [Google Scholar]
- Lee, B.Y.; Kim, E.J.; Park, D.J.; Hong, S.I.; Chun, H.S. Composition of saponin and free sugar of some white ginsengs with processing conditions. Korean J. Food Sci. Technol. 2005, 28, 922–927. [Google Scholar]
- Suong-Nuen Kim, S.-J.K. Effects of black ginseng (9 times-steaming ginseng) on hypoglycemic action and changes in the composition of ginsenosides on the steaming process. Korean J. Food Sci. Technol. 2009, 41, 77–81. [Google Scholar]
- Shah, Z.A.; Gilani, R.A.; Sharma, P.; Vohora, S.B. Cerebroprotective effect of Korean ginseng tea against global and focal models of ischemia in rats. J. Ethnopharmacol. 2005, 101, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Achike, F.I.; Kwan, C.Y. Nitric oxide, human diseases and the herbal products that affect the nitric oxide signalling pathway. Clin. Exp. Pharmacol. Physiol. 2003, 30, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Oh, S. Administration of red ginseng ameliorates memory decline in aged mice. J. Ginseng Res. 2015, 39, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.T.; Luong, T.T.; Kim, G.L.; Pyo, S.; Rhee, D.K. Korean red ginseng inhibits apoptosis in neuroblastoma cells via estrogen receptor β-mediated phosphatidylinositol-3 kinase/akt signaling. J. Ginseng Res. 2015, 39, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Lim, Y.S.; Lee, H.J.; Eum, W.S.; Park, J.; Han, K.H.; Choi, S.Y.; Lee, K.S. Anti-oxidative effects of Phellinus linteus and red ginseng extracts on oxidative stress-induced DNA damage. BMB Rep. 2009, 42, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Bak, M.J.; Hong, S.G.; Lee, J.W.; Jeong, W.S. Red ginseng marc oil inhibits iNOS and COX-2 via NFκB and p38 pathways in LPS-stimulated RAW 264.7 macrophages. Molecules 2012, 17, 13769–13786. [Google Scholar] [PubMed]
- Lee, S.E.; Park, Y.S. Korean red ginseng water extract inhibits COX-2 expression by suppressing p38 in acrolein-treated human endothelial cells. J. Ginseng Res. 2014, 38, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Gambino, R.; Cassader, M. Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (nafld). Prog. Lipid Res. 2009, 48, 1–26. [Google Scholar] [CrossRef] [PubMed]
- MacDougald, O.A.; Lane, M.D. Transcriptional regulation of gene expression during adipocyte differentiation. Annu. Rev. Biochem. 1995, 64, 345–373. [Google Scholar] [CrossRef] [PubMed]
- Malhi, H.; Gores, G.J. Molecular mechanisms of lipotoxicity in nonalcoholic fatty liver disease. Semin. Liver Dis. 2008, 28, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.R.; Dunigan, D.D.; Dickman, M.B. Bcl-2 family members inhibit oxidative stress-induced programmed cell death in saccharomyces cerevisiae. Free Radic. Biol. Med. 2003, 34, 1315–1325. [Google Scholar] [CrossRef]
- Altan, O.; Pabuccuoglu, A.; Altan, A.; Konyalioglu, S.; Bayraktar, H. Effect of heat stress on oxidative stress, lipid peroxidation and some stress parameters in broilers. Br. Poult. Sci. 2003, 44, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Mager, W.H.; de Kruijff, A.J. Stress-induced transcriptional activation. Microbiol. Rev. 1995, 59, 506–531. [Google Scholar] [PubMed]
- Zhang, Y.; Mian, M.A.; Chekhovskiy, K.; So, S.; Kupfer, D.; Lai, H.; Roe, B.A. Differential gene expression in festuca under heat stress conditions. J. Exp. Bot. 2005, 56, 897–907. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, N.; Ogura, H.; Tanaka, H.; Koh, T.; Nakamori, Y.; Noborio, M.; Shiozaki, T.; Nishino, M.; Kuwagata, Y.; Shimazu, T.; et al. Enhanced expression of heat shock proteins in activated polymorphonuclear leukocytes in patients with sepsis. J. Trauma 2001, 51, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Didelot, C.; Schmitt, E.; Brunet, M.; Maingret, L.; Parcellier, A.; Garrido, C. Heat shock proteins: Endogenous modulators of apoptotic cell death. Handb. Exp. Pharmacol. 2006, 172, 171–198. [Google Scholar] [PubMed]
- Gao, T.; Newton, A.C. The turn motif is a phosphorylation switch that regulates the binding of HSP70 to protein kinase C. J. Biol. Chem. 2002, 277, 31585–31592. [Google Scholar] [CrossRef] [PubMed]
- Pratt, W.B.; Toft, D.O. Steroid receptor interactions with heat shock protein and immunophilin chaperones. Endocr. Rev. 1997, 18, 306–360. [Google Scholar] [CrossRef] [PubMed]
- Chambon, M.; Rochefort, H.; Vial, H.J.; Chalbos, D. Progestins and androgens stimulate lipid accumulation in T47D breast cancer cells via their own receptors. J. Steroid Biochem. 1989, 33, 915–922. [Google Scholar] [CrossRef]
- Sharpe, L.J.; Brown, A.J. Controlling cholesterol synthesis beyond 3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR). J. Biol. Chem. 2013, 288, 18707–18715. [Google Scholar] [CrossRef] [PubMed]
- Beloor, J.; Kang, H.K.; Kim, Y.J.; Subramani, V.K.; Jang, I.S.; Sohn, S.H.; Moon, Y.S. The effect of stocking density on stress related genes and telomeric length in broiler chickens. Asian Australas. J. Anim. Sci. 2010, 23, 437–443. [Google Scholar] [CrossRef]
- Barker, H.E.; Smyth, G.K.; Wettenhall, J.; Ward, T.A.; Bath, M.L.; Lindeman, G.J.; Visvader, J.E. DEAF-1 regulates epithelial cell proliferation and side-branching in the mammary gland. BMC Dev. Biol. 2008, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Veraksa, A.; Kennison, J.; McGinnis, W. DEAF-1 function is essential for the early embryonic development of drosophila. Genesis 2002, 33, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Krueger, S.K.; Vandyke, J.E.; Williams, D.E.; Hines, R.N. The role of flavin-containing monooxygenase (FMO) in the metabolism of tamoxifen and other tertiary amines. Drug Metab. Rev. 2006, 38, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Cashman, J.R.; Zhang, J. Human flavin-containing monooxygenases. Annu. Rev. Pharmacol. Toxicol. 2006, 46, 65–100. [Google Scholar] [CrossRef] [PubMed]
- Cashman, J.R. Human and plant flavin-containing monooxygenase N-oxygenation of amines: Detoxication vs. Bioactivation. Drug Metab. Rev. 2002, 34, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Kohjima, M.; Enjoji, M.; Higuchi, N.; Kato, M.; Kotoh, K.; Yoshimoto, T.; Fujino, T.; Yada, M.; Yada, R.; Harada, N.; et al. Re-evaluation of fatty acid metabolism-related gene expression in nonalcoholic fatty liver disease. Int. J. Mol. Med. 2007, 20, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sakuma, T.; Asafu-Adjaye, E.; Shiu, G.K. Determination of ginsenosides in plant extracts from Panax ginseng and Panax quinquefolius L. by LC/MS/MS. Anal. Chem. 1999, 71, 1579–1584. [Google Scholar] [CrossRef] [PubMed]
- Chuang, W.C.; Wu, H.K.; Sheu, S.J.; Chiou, S.H.; Chang, H.C.; Chen, Y.P. A comparative study on commercial samples of ginseng radix. Planta Med. 1995, 61, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.Y.; Kim, J.M.; Han, S.B.; Lee, S.K.; Kim, N.D.; Park, M.K.; Kim, C.K.; Park, J.H. Steaming of ginseng at high temperature enhances biological activity. J. Nat. Prod. 2000, 63, 1702–1704. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Jang, J.H.; Chen, C.Y.; Na, H.K.; Surh, Y.J. A formulated red ginseng extract rescues PC12 cells from PCB-induced oxidative cell death through Nrf2-mediated upregulation of heme oxygenase-1 and glutamate cysteine ligase. Toxicology 2010, 278, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Korivi, M.; Hou, C.W.; Huang, C.Y.; Lee, S.D.; Hsu, M.F.; Yu, S.H.; Chen, C.Y.; Liu, Y.Y.; Kuo, C.H. Ginsenoside-rg1 protects the liver against exhaustive exercise-induced oxidative stress in rats. Evid. Based Complement. Altern. Med. 2012, 2012, 932165. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, Y.Z.; Wang, X.R.; Lu, Z.R.; Shi, D.Z.; Liu, X.H. Panax quinquefolium saponins reduce myocardial hypoxia-reoxygenation injury by inhibiting excessive endoplasmic reticulum stress. Shock 2012, 37, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Hahm, D.H.; Yang, D.C.; Lee, H.J.; Shim, I. Effect of crude saponin of Korean red ginseng on high-fat diet-induced obesity in the rat. J. Pharmacol. Sci. 2005, 97, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, K.; Sakata, T.; Ishimaru, T.; Etou, H.; Ookuma, K.; Kurokawa, M.; Machidori, H. Attenuation of anorexia induced by heat or surgery during sustained administration of ginsenoside Rg1 into rat third ventricle. Psychopharmacology 1989, 99, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kang, S.A.; Han, S.M.; Shim, I. Comparison of the antiobesity effects of the protopanaxadiol- and protopanaxatriol-type saponins of red ginseng. Phytother. Res. 2009, 23, 78–85. [Google Scholar] [PubMed]
- Rahman, S.M.; Schroeder-Gloeckler, J.M.; Janssen, R.C.; Jiang, H.; Qadri, I.; Maclean, K.N.; Friedman, J.E. CCAAT/enhancing binding protein β deletion in mice attenuates inflammation, endoplasmic reticulum stress, and lipid accumulation in diet-induced nonalcoholic steatohepatitis. Hepatology 2007, 45, 1108–1117. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Lee, M.S.; Kim, H.J.; Sung, M.J.; Kim, H.Y.; Kim, M.S.; Kwon, D.Y. Antiobesity effect of ginsenoside Rg3 involves the AMPK and PPAR-γ signal pathways. Phytother. Res. 2009, 23, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Day, C.P.; James, O.F. Steatohepatitis: A tale of two “hits”? Gastroenterology 1998, 114, 842–845. [Google Scholar] [CrossRef]
- Leclercq, I.A.; Farrell, G.C.; Field, J.; Bell, D.R.; Gonzalez, F.J.; Robertson, G.R. CYP2E1 and CYP4A as microsomal catalysts of lipid peroxides in murine nonalcoholic steatohepatitis. J Clin Investig. 2000, 105, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Yacoub, L.K.; Fogt, F.; Griniuviene, B.; Nanji, A.A. Apoptosis and Bcl-2 protein expression in experimental alcoholic liver disease in the rat. Alcohol Clin. Exp. Res. 1995, 19, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.Y.; Li, Y.W.; Tse, A.K.; Hau, D.K.; Leung, C.H.; Yu, Z.L.; Fong, W.F. 20(S)-Protopanaxadiol, a metabolite of ginsenosides, induced cell apoptosis through endoplasmic reticulum stress in human hepatocarcinoma HepG2 cells. Eur. J. Pharmacol. 2011, 668, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, M.; Zheng, Y.N.; Li, J.; Wang, Y.P.; Wang, Y.J.; Gu, J.; Jin, Y.; Wang, H.; Chen, L. Snailase preparation of ginsenoside M1 from protopanaxadiol-type ginsenoside and their protective effects against CCl4-induced chronic hepatotoxicity in mice. Molecules 2011, 16, 10093–10103. [Google Scholar] [CrossRef] [PubMed]
- Kwok, H.H.; Ng, W.Y.; Yang, M.S.; Mak, N.K.; Wong, R.N.; Yue, P.Y. The ginsenoside protopanaxatriol protects endothelial cells from hydrogen peroxide-induced cell injury and cell death by modulating intracellular redox status. Free Radic. Biol. Med. 2010, 48, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Sorn, S.; Baek, S.; Jang, S.; Kim, S. Antioxidant and apoptotic effects of Korean white ginseng extracted with the same ratio of protopanaxadiol and protopanaxatriol saponins in human hepatoma HepG2 cells. Ann. N. Y. Acad. Sci. 2009, 1171, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, S. [therapeutic drug monitoring of lithium carbonate]. Nihon Rinsho 1990, 48 (Supp 1), 1124–1126. [Google Scholar] [PubMed]
- Lee, O.H.; Lee, B.Y.; Lee, J.; Lee, H.B.; Son, J.Y.; Park, C.S.; Shetty, K.; Kim, Y.C. Assessment of phenolics-enriched extract and fractions of olive leaves and their antioxidant activities. Bioresour. Technol. 2009, 100, 6107–6113. [Google Scholar] [CrossRef] [PubMed]
- Do, J.H.; Lee, H.O.; Lee, S.K.; Jang, J.K.; Lee, S.D.; Sung, H.S. Colorimetric determination of acidic polysaccharide from Panax ginseng, its extraction condition and stability. Korean J. Ginseng Sci. 1993, 17, 139–144. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Kim, K.J.; Lee, O.H.; Lee, B.Y. Low-molecular-weight fucoidan regulates myogenic differentiation through the mitogen-activated protein kinase pathway in C2C12 cells. Br. J. Nutr. 2011, 106, 1836–11844. [Google Scholar] [CrossRef]
- Sample Availability: Samples are not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.-J.; Yoon, K.-Y.; Hong, H.-D.; Lee, B.-Y. Role of the Red Ginseng in Defense against the Environmental Heat Stress in Sprague Dawley Rats. Molecules 2015, 20, 20240-20253. https://doi.org/10.3390/molecules201119692
Kim K-J, Yoon K-Y, Hong H-D, Lee B-Y. Role of the Red Ginseng in Defense against the Environmental Heat Stress in Sprague Dawley Rats. Molecules. 2015; 20(11):20240-20253. https://doi.org/10.3390/molecules201119692
Chicago/Turabian StyleKim, Kui-Jin, Kye-Yoon Yoon, Hee-Do Hong, and Boo-Yong Lee. 2015. "Role of the Red Ginseng in Defense against the Environmental Heat Stress in Sprague Dawley Rats" Molecules 20, no. 11: 20240-20253. https://doi.org/10.3390/molecules201119692





