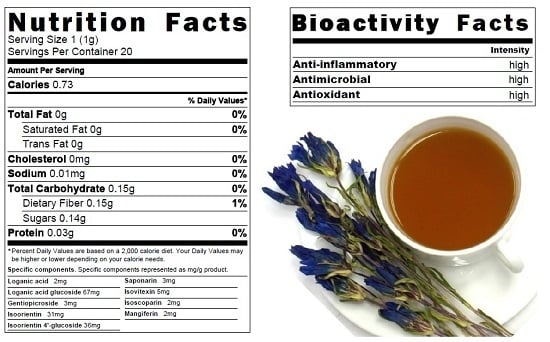
Bitter Gentian Teas: Nutritional and Phytochemical Profiles, Polysaccharide Characterisation and Bioactivity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Nutritional Profiles of Bitter Gentian Teas Decoctions
| Parameter | G. algida | G. decumbens | G. macrophylla | G. triflora |
|---|---|---|---|---|
| Extractives b | 272 ± 8 | 224 ± 8 | 263 ± 9 | 345 ± 12 |
| Organoleptic characteristics | ||||
| Color | Light-brown | Yellow | Yellow | Dark-yellow |
| Odor | Herbal | Herbal | Herbal | Herbal |
| Taste | Bitter, intensive | Bitter, light | Bitter, light | Bitter, medium |
| Bitter index c | 9300 (4200–12,000) | 500 (170–600) | 1000 (520–1200) | 3050 (1300–4800) |
| Nutritional characteristics | ||||
| Carbohydrates b | 98.39 ± 3.05 | 95.23 ± 2.76 | 107.11 ± 3.42 | 151.99 ± 4.71 |
| Protein b | 22.65 ± 0.77 | 22.75 ± 0.68 | 40.01 ± 1.12 | 30.91 ± 0.95 |
| Lipids b | <0.1 | <0.1 | <0.1 | <0.1 |
| Ash b | 8.63 ± 0.43 | 9.39 ± 0.49 | 5.18 ± 0.23 | 6.47 ± 0.32 |
| Energy d | 0.48 | 0.47 | 0.59 | 0.73 |
| Total free amino acids b | 2.39 ± 0.06 | 1.37 ± 0.03 | 3.84 ± 0.11 | 2.19 ± 0.06 |
| Free sugars | ||||
| Fructose b | 2.00 ± 0.05 | 8.18 ± 0.20 | 1.42 ± 0.04 | 7.42 ± 0.22 |
| Glucose b | 16.37 ± 0.39 | 32.64 ± 0.75 | 2.03 ± 0.05 | 51.57 ± 1.34 |
| Sucrose b | 26.35 ± 0.63 | 20.26 ±0.45 | 24.32 ± 0.58 | 25.28 ± 0.61 |
| Gentiobiose b | 4.16 ± 0.09 | 6.51 ± 0.15 | 9.20 ± 0.22 | 13.24 ± 0.30 |
| Gentianose b | 5.72 ± 0.15 | 21.80 ± 0.59 | 31.93 ± 0.83 | 37.76 ± 1.06 |
| Total free sugars b | 54.60 | 89.39 | 68.90 | 135.27 |
| Organic acids | ||||
| Malic acid b | 9.64 ± 0.29 | 15.63 ± 0.43 | 5.39 ± 0.15 | 4.87 ± 0.11 |
| Citric acid b | 4.16 ± 0.11 | 6.11 ± 0.15 | 4.83 ± 0.12 | 9.31 ± 0.22 |
| Tartaric acid b | 0.72 ± 0.02 | 1.24 ± 0.02 | 0.94 ± 0.02 | 0.52 ± 0.01 |
| Oxalic acid b | tr. | tr. | 0.11 ± 0.00 | 0.09 ± 0.00 |
| Succinic acid b | tr. | tr. | tr. | 0.12 ± 0.00 |
| Quinic acid b | tr. | tr. | tr. | tr. |
| Total organic acids b | 14.52 | 22.98 | 11.27 | 14.91 |
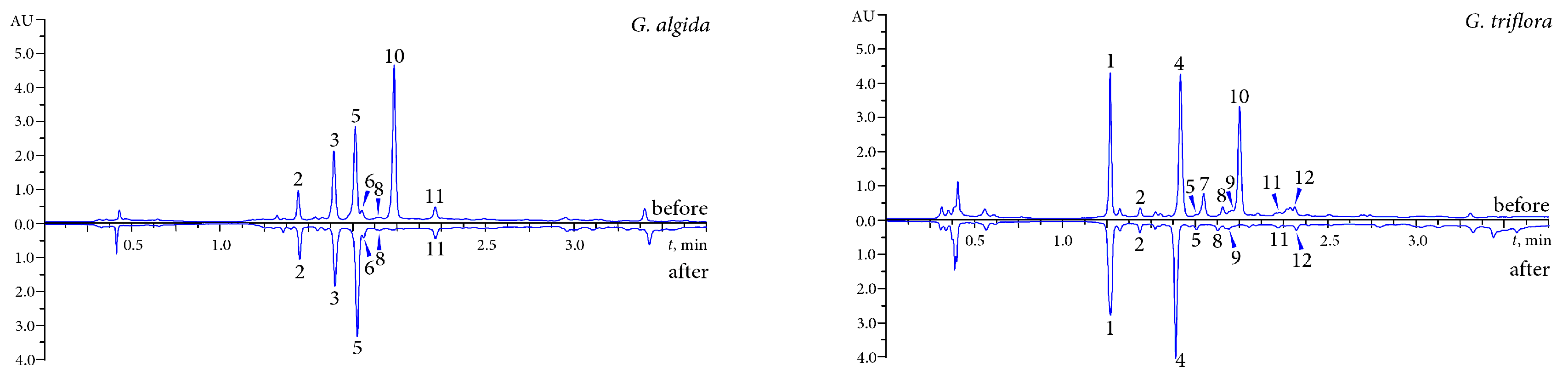
2.2. Phytochemical Profiles of Bitter Gentian Tea Decoctions
| Compound | G. algida | G. decumbens | G. macrophylla | G. triflora |
|---|---|---|---|---|
| Iridoids | ||||
| Loganic acid | 106.42 ± 1.91 | 32.69 ± 0.59 | 40.09 ± 0.72 | 33.04 ± 0.66 |
| Loganic acid-6′-O-β-d-glucoside | n.d. | 523.10 ± 10.99 | 123.08 ± 2.34 | 592.41 ± 13.03 |
| Swertiamarin | 26.38 ± 0.47 | tr. | 4.02 ± 0.06 | tr. |
| Sweroside | 23.03 ± 0.46 | n.d. | 2.91 ± 0.05 | n.d. |
| Gentiopicroside | 259.57 ± 4.67 | n.d. | 7.08 ± 0.13 | 15.59 ± 0.34 |
| Subtotal | 415.40 | 555.79 | 177.18 | 641.04 |
| Flavonoids | ||||
| Isoorientin | 173.60 ± 2.77 | 14.83 ± 0.27 | 27.83 ± 0.53 | 120.22 ± 1.92 |
| Isoorientin-4′-O-β-d-glucoside | tr. | 40.71 ± 0.89 | 111.10 ± 2.22 | 204.43 ± 3.88 |
| Isovitexin | 37.96 ± 0.75 | 18.11 ± 0.34 | 40.04 ± 0.92 | 9.70 ± 0.19 |
| Saponarin | 25.62 ± 0.46 | 15.50 ± 0.25 | 6.12 ± 0.13 | 29.31 ± 0.59 |
| Isosaponarin | n.d. | tr. | tr. | 21.22 ± 0.45 |
| Isoscoparin | n.d. | tr. | 12.04 ± 0.26 | 20.83 ± 0.48 |
| Subtotal | 237.18 | 89.15 | 197.13 | 405.71 |
| Xanthones | ||||
| Mangiferin | n.d. | n.d. | n.d. | 19.89 ± 0.46 |
| Subtotal | n.d. | n.d. | n.d. | 19.89 |
| Total | 652.58 | 644.94 | 374.31 | 1066.64 |
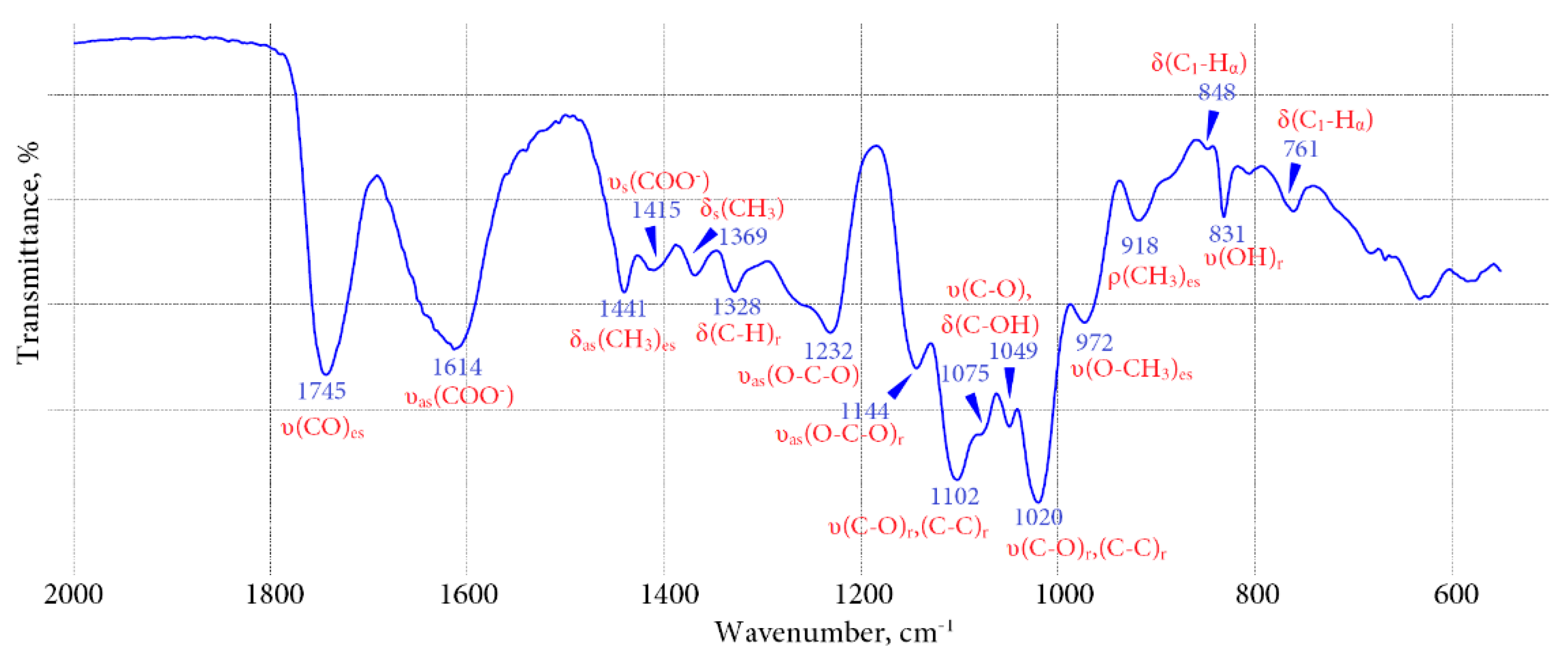
2.3. Polysaccharide Characterisation of Bitter Gentian Teas
| Parameter | G. algida | G. decumbens | G. macrophylla | G. triflora |
|---|---|---|---|---|
| Yield, mg/g | 19.34 | 6.27 | 22.18 | 15.31 |
| Total carbohydrate content, mg/g | 946.14 ± 24.59 | 931.63 ± 24.15 | 932.07 ± 22.37 | 954.91 ± 24.83 |
| Uronic acid content, mg/g | 802.37 ± 20.86 | 732.64 ± 18.31 | 824.63 ± 22.26 | 797.68 ± 20.74 |
| Protein content, mg/g | 16.27 ± 0.41 | 22.86 ± 0.48 | 21.15 ± 0.51 | 18.37 ± 0.38 |
| , ° | +116 | +101 | +124 | +110 |
| Reaction with I2 | negative | negative | negative | negative |
| Reaction with resorcinol | negative | negative | negative | negative |
| Reaction with Yariv reagent | positive | positive | positive | positive |
| Monosaccharide composition, mol % | ||||
| Ara | 9.4 | 8.0 | 4.2 | 10.6 |
| Gal | 8.4 | 12.9 | 7.3 | 7.7 |
| Glc | 2.7 | 7.4 | 1.8 | 1.6 |
| Man | 0.9 | 2.1 | 1.2 | 1.1 |
| Rha | 5.6 | 4.8 | 6.3 | 2.8 |
| Xyl | 1.4 | 0.9 | 0.8 | 1.1 |
| GalA | 71.6 | 63.9 | 78.4 | 75.1 |
| GlcA | tr. | tr. | tr. | tr. |
2.4. Biological Activity of Bitter Gentian Teas
2.4.1. Anti-Inflammatory Activity

2.4.2. Antimicrobial Activity
| Microorganism | G. algida | G. decumbens | G. macrophylla | G. triflora | G | LG | PC b |
|---|---|---|---|---|---|---|---|
| B. subtillis | 100 | 400 | 400 | 200 | 200 | 400 | 4.0 |
| S. aureus | 200 | 800 | 400 | 400 | 200 | >800 | 4.0 |
| E. faecalis | 100 | 200 | 200 | 200 | 100 | 400 | 32.0 |
| E. coli | 100 | 400 | 400 | 200 | 100 | 400 | 2.0 |
| P. aeruginosa | 100 | 800 | 800 | 400 | 400 | >800 | 16.0 |
| C. albicans | 200 | >800 | >800 | 800 | 400 | >800 | 8.0 |
2.4.3. Antioxidant Activity
| Method b | G. algida | G. decumbens | G. macrophylla | G. triflora | iO | Man |
|---|---|---|---|---|---|---|
| DPPH• | 89.22 ± 3.83 ii | 287.91 ± 10.36 iii | 278.82 ± 9.87 iii | 104.86 ± 3.56 ii | 18.67 ± 0.63 i | 16.94 ± 0.52 i |
| ABTS•+ | 101.67 ± 3.15 v | 217.03 ± 7.41 vi | 187.93 ± 6.76 vi | 103.76 ± 3.32 v | 14.20 ± 0.42 iv | 12.80 ± 0.34 iv |
| O2•− | 58.63 ± 2.11 viii | > 300 | > 300 | 189.39 ± 6.81 viii | 27.69 ± 1.14 vii | 21.09 ± 0.97 vii |
| CBA | 32.04 ± 0.89 x | 84.15 ± 2.52 | 55.61 ± 1.56 x | 49.37 ± 1.43 x | 12.32 ± 0.40 ix | 9.62 ± 0.37 ix |
3. Experimental Section
3.1. General Information
3.1.1. Chemicals, Sorbents
3.1.2. Equipment
3.2. Plant Material
3.3. Oranoleptic and Nutritional Analysis
3.3.1. Decoction Preparation
3.3.2. Crude Composition
3.3.3. Free Sugars Composition
3.3.4. Organic Acids Composition
3.4. MC-RP-HPLC-UV Quantification of Phytochemicals in Bitter Gentian Teas
3.5. Polysaccharide Analysis
3.6. Biological Activity Assays
3.6.1. Carrageenan-Induced Paw Edema in Male Rats
3.6.2. Antimicrobial Activity
3.6.3. Antioxidant Activity
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anonymous. Buryats; Abaeva, L.L., Zhukovskaya, N.L., Eds.; Nauka: Moscow, Russia, 2004; p. 633. [Google Scholar]
- Anonymous. Traditions of the Buryat Cuisine; Mikhailova, V.T., Ed.; Printhouse L.T.: Ulan-Ude, Bria, 2010; p. 93. [Google Scholar]
- Makarov, A.A. Plant Remedies of the Traditional Yakutian Medicine; Yakutian Scientific Center: Yakutsk, Russia, 1974; p. 64. [Google Scholar]
- Anonymous. Traditions and Innovations in Life and Culture of the Siberian Peoples; Rusakova, L.M., Ed.; SD RAS: Novosibirsk, Russia, 1983; p. 138. [Google Scholar]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Tankhaeva, L.M. Iridoids and flavonoids of four Siberian gentians: Chemical profile and gastric stimulatory effect. Molecules 2015, 20, 19172–19188. [Google Scholar] [CrossRef] [PubMed]
- Hostettmann-Kaldas, M.; Hostettmann, K.; Sticher, O. Xanthones, flavones and secoiridoids of American Gentiana species. Phytochemistry 1981, 20, 443–446. [Google Scholar] [CrossRef]
- Tan, R.X.; Wolfender, J.-L.; Ma, W.G.; Zhang, L.X.; Hostettmann, K. Secoiridoids and antifungal aromatic acids from Gentiana algida. Phytochemistry 1996, 41, 111–116. [Google Scholar] [CrossRef]
- Tan, R.X.; Wolfender, J.-L.; Zhang, L.X.; Ma, W.G.; Fuzzati, N.; Marston, A.; Hostettmann, K. Acyl secoiridoids and antifungal constituents from Gentiana macrophylla. Phytochemistry 1996, 42, 1305–1313. [Google Scholar] [CrossRef]
- Wang, S.; Xu, Y.; Jiang, W.; Zhang, Y. Isolation and identification of constituents with activity of inhibiting nitric oxide production in RAW 264.7 macrophages from Gentiana triflora. Planta Med. 2013, 79, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, J.Y.; Yang, Q.W.; Chen, Y.; Piao, Y.A.; Li, H.Y. Inhibition activities of polysaccharide (RG4–1) from Gentiana rigescens against RSV. J. Asian Nat. Prod. Res. 2011, 13, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, C.; Su, T.; Zhang, J. Antioxidant and immunological activities of polysaccharides from Gentiana scabra Bunge roots. Carbohydr. Polym. 2014, 112, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Massias, M.; Carbonnier, J.; Mohlo, D. Implications chimiotaxonomiques de la répartition des substances osidiques dans le genre Gentiana L. Bull. Mus. Nat. Hist. Natur. Sci. Phys. Chim. N. 1977, 13, 41–54. [Google Scholar]
- Eswaranandam, S.; Hettiarachchy, N.S.; Johnson, M.G. Antimicrobial activity of citric, lactic, malic, or tartaric acids and nisin-incorporated soy protein film against Listeria monocytogenes, Escherichia coli O157:H7, and Salmonella gaminara. J. Food Sci. 2004, 69, FMS79–FMS84. [Google Scholar] [CrossRef]
- Tang, X.; Liu, J.; Dong, W. The cardioprotective effects of citric Acid and l-malic acid on myocardial ischemia/reperfusion injury. Evid.-Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.C.; Zhao, Y.; Bian, H.M. Antiplatelet activity of a novel formula composed of malic acid, succinic acid and citric acid from Cornus officinalis fruit. Phytother. Res. 2013, 27, 1894–1896. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, R.; Barros, L.; Dueñas, M.; Calhelha, R.C.; Carvalho, A.M.; Santos-Buelga, C.; Queiroz, M.J.; Ferreira, I.C. Nutrients, phytochemicals and bioactivity of wild Roman chamomile: A comparison between the herb and its preparations. Food Chem. 2013, 15, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Olennikov, D.N. Chemical investigation of Anagallidium dichotomum and anticholinesterase activity of its constituents. Chem. Nat. Comp. 2013, 49, 1137–1139. [Google Scholar] [CrossRef]
- Paulsen, B.S.; Barsett, H. Bioactive pectic polysaccharides. Adv. Polym. Sci. 2005, 186, 69–101. [Google Scholar]
- Kačuráková, M.; Wilson, R.H. Development in mid-infrared FT-IR spectroscopy of selected carbohydrates. Carbohydr. Polym. 2001, 44, 291–303. [Google Scholar] [CrossRef]
- Čopíková, J.; Synytsyá, A.; Černá, M.; Kaasová, J.; Novotná, M. Application of FT-IR spectroscopy in detection of food hydrocolloids in confectionery jellies and food supplements. Czech J. Food Sci. 2001, 19, 51–56. [Google Scholar]
- Synytsyá, A.; Čopíková, J.; Matějka, P.; Machovič, V. Fourier transform Raman and infrared spectroscopy of pectins. Carbohydr. Polym. 2003, 54, 97–106. [Google Scholar] [CrossRef]
- Kačuráková, M.; Capek, P.; Sasinková, V.; Wellner, N.; Ebringerová, A. FT-IR study of plant cell wall model compounds: Pectic polysaccharides and hemicelluloses. Carbohydr. Polym. 2000, 43, 195–203. [Google Scholar] [CrossRef]
- Winiyard, P.G.; Willoughby, D.A. Inflammation Protocols; Humana Press: New York City, NY, USA, 2003; p. 365. [Google Scholar]
- Wang, C.-H.; Cheng, X.-M.; Bligh, S.W.A.; White, K.N.; Branford-White, C.-J.; Wang, Z.-T. Pharmacokinetics and bioavailability of gentiopicroside from decoctions of Gentianae and Longdan Xiegan Tang after oral administration in rats—Comparison with gentiopicroside alone. J. Pharm. Biomed. Anal. 2007, 44, 1113–1117. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Lin, X.; Yao, H. Metabolism and plasma pharmacokinetics of isoorientin, a natural active ingredient, in Sprague-Dawley male rats after oral and intravenous administration. Xenobiotica 2015, 45, 999–1008. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-J.; Niu, H.-S.; Lin, M.-H.; Cheng, J.-T.; Hsu, F.-L. Antihyperglycemic effect of catalpol in streptozotocin-induced diabetic rats. J. Nat. Prod. 2010, 73, 1170–1172. [Google Scholar] [CrossRef] [PubMed]
- Recio, D.L.M.; Giner, R.M.; Manez, S.; Rios, J.H. Structural consideration on the iridoids as anti-inflammatory agents. Planta Med. 1994, 60, 232–234. [Google Scholar] [CrossRef] [PubMed]
- Küpeli, E.; Aslan, M.; Gürbüz, I.; Yesilada, E. Evaluation of in vivo biological activity profile of isoorientin. Z. Naturforsch. 2004, 59c, 787–790. [Google Scholar] [CrossRef]
- Odontuya, G.; Hoult, J.R.S.; Houghton, P.J. Structure-activity relationship for anti-inflammatory effect of luteolin and its derived glucosides. Phytother. Res. 2005, 19, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Wedler, J.; Daubitz, T.; Schlotterbeck, G.; Butterweck, V. In vivo anti-inflammatory and wound-healing potential of a Phyllostachys edulis leaf extract—Identification of isoorientin as an active compound. Planta Med. 2014, 80, 1678–1684. [Google Scholar] [PubMed]
- Ovodova, R.G.; Golovchenko, V.V.; Popov, S.V.; Popova, G.Y.; Paderin, N.M.; Shashkov, A.S.; Ovodov, Y.S. Chemical composition and anti-inflammatory activity of pectic polysaccharide isolated from celery stalks. Food Chem. 2009, 114, 610–615. [Google Scholar] [CrossRef]
- Chen, L.; Liu, J.; Zhang, Y.; Dai, B.; An, Y.; Yu, L. Structural, thermal, and anti-inflammatory properties of a novel pectic polysaccharide from Alfalfa (Medicago sativa L.) stem. J. Agric. Food Chem. 2015, 63, 3219–3228. [Google Scholar] [CrossRef] [PubMed]
- Popov, S.V.; Popova, G.Y.; Ovodova, R.G.; Ovodov, Y.S. Antiinflammatory activity of the pectic polysaccharide from Comarum palustre. Fitoterapia 2005, 76, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Savikin, K.; Menković, N.; Zdunić, G.; Stević, T.; Radanović, D.; Janković, T. Antimicrobial activity of Gentiana lutea L. extracts. Z. Naturforsch. 2009, 64c, 339–342. [Google Scholar] [CrossRef]
- Mihailović, V.; Vuković, N.; Nićiforović, N.; Solujić, S.; Mladenović, M.; Mašković, P.; Stanković, M.S. Studies on the antimicrobial activity and chemical composition of the essential oils and alcoholic extracts of Gentiana asclepiadea L. J. Med. Plants Res. 2011, 5, 1164–1174. [Google Scholar]
- Graikou, K.; Aligianis, N.; Chinou, I.B.; Harvala, C. Cantleyoside-dimethyl-acetal and other iridoid glucosides from Pterocephalus perrenis—Antimicrobial activities. Z. Naturforsch. C 2002, 57, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Cottiglia, F.; Loy, G.; Garau, D.; Floris, C.; Casu, M.; Pompei, R.; Bonsignore, L. Antimicrobial evaluation of coumarins and flavonoids from the stems of Daphne gnidium L. Phytomedicine 2001, 8, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.Q.; Song, J.; Ye, S.P.; Yuan, K. Separation, identification and its antibacterial activity of glycosylflavones in Lophantherum gracile Brong. West China J. Pharm. Sci. 2009, 24, 218–220. [Google Scholar]
- Mihailović, V.; Mišić, D.; Matić, S.; Mihailović, M.; Stanić, S.; Vrvić, M.M.; Katanić, J.; Mladenović, M.; Stanković, N.; Boroja, T.; et al. Comparative phytochemical analysis of Gentiana cruciata L. roots and aerial parts, and their biological activities. Ind. Crops Prod. 2015, 73, 49–62. [Google Scholar] [CrossRef]
- Senol, F.S.; Tuzun, C.Y.; Toker, G.; Orhan, I.E. An in vitro perspective to cholinesterase inhibitory and antioxidant activity of five Gentiana species and Gentianella caucasea. Int. J. Food Sci. Nutr. 2012, 63, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Organoleptic Analysis of Herbal Ingredients; American Herbal Products Association: Silver Spring, MD, USA, 2013; p. 37. [Google Scholar]
- Anonymous. Quality Control Methods for Herbal Materials; World Health Organization: Geneva, Switzerland, 2011; p. 173. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 76, 248–254. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Sevag, M.G.; Lackman, D.B.; Smolens, J. The isolation of the components of Streptococcal nucleoproteins in serologically active form. J. Biol. Chem. 1938, 124, 425–436. [Google Scholar]
- Usov, A.T.; Bilan, M.I.; Klochkova, N.G. Polysaccharides of algae. 48. Polysaccharide composition of several calcareous red algae: Isolation of alginate from Corallina pilulitara P. et R. (Rhodophyta, Corallinaceae). Bot. Mar. 1995, 35, 43–51. [Google Scholar]
- Olennikov, D.N.; Stolbikova, A.V.; Rokhin, A.V.; Khobrakova, V.B.; Tankhaeva, L.M. Polysaccharides from Fabaceae. V. α-Glucan from Sophora flavescens roots. Chem. Nat. Comp. 2011, 47, 1–6. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Tankhaeva, L.M. A quantitative assay for total fructans in burdock (Arctium spp.) roots. Russ. J. Bioorg. Chem. 2011, 37, 893–898. [Google Scholar] [CrossRef]
- Togola, A.; Inngjerdingen, M.; Diallo, D.; Barsett, H.; Rolstad, B.; Michaelsen, T.E.; Paulsen, B.S. Polysaccharides with complement fixing and macrophage stimulation activity from Opilia celtidifolia, isolation and partial characterization. J. Ethnopharmacol. 2007, 115, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Olennikov, D.N.; Rokhin, A.V. Water-soluble glucans from true cardamom (Elettaria cardamomum White at Maton) seeds. Appl. Biochem. Microbiol. 2013, 49, 182–187. [Google Scholar] [CrossRef]
- Yesilada, E.; Kupeli, E. Berberis crataegina root exhibits potent anti-inflammatory, analgesic and febrifuge effects in mice and rats. J. Ethnopharmacol. 2002, 79, 237–248. [Google Scholar] [CrossRef]
- Olajide, A.A.; Olubusayo, A.S.; Modupe, M.J. Studies on the anti-inflammatory, antipyretic and analgesic properties of Alstonia boonei stem bark. J. Ethnopharmacol. 2000, 71, 179–186. [Google Scholar] [CrossRef]
- Sarker, S.D.; Nahar, L.; Kumarasamy, Y. Microtitre plate-based antibacterial assay incorporating resazurin as indicator of cell growth, and its application in the in vitro antibacterial screening of phytochemicals. Methods 2007, 42, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K. A novel HPLC-assisted method for investigation of the Fe2+-chelating activity of flavonoids and plant extracts. Molecules 2014, 19, 18296–18316. [Google Scholar] [CrossRef] [PubMed]
- Asker, M.M.S.; Shawky, B.T. Structural characterization and antioxidant activity of an extracellular polysaccharide isolated from Brevibacterium otitidis BTS 44. Food Chem. 2010, 123, 315–320. [Google Scholar] [CrossRef]
- Ding, H.; Chou, T.; Liang, C. Antioxidant and antimelanogenic properties of rosmarinic acid methyl ester from Origanum vulgare. Food Chem. 2010, 123, 254–262. [Google Scholar] [CrossRef]
- Ozen, T.; Demirtas, I.; Aksit, H. Determination of antioxidant activities of various extracts and essential oil compositions of Thymus praecox subsp. skorpilii var. skorpilii. Food Chem. 2011, 124, 58–64. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Tankhaeva, L.M.; Agafonova, S.V. Antioxidant components of Laetiporus sulphureus (Bull.: Fr.) Murr. fruit bodies. Appl. Biochem. Microbiol. 2011, 47, 419–425. [Google Scholar] [CrossRef]
- Sample Availability: Samples of G. algida, G. decumbens, G. macrophylla, G. triflora plants and extracts are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Koryakina, L.P.; Vladimirov, L.N. Bitter Gentian Teas: Nutritional and Phytochemical Profiles, Polysaccharide Characterisation and Bioactivity. Molecules 2015, 20, 20014-20030. https://doi.org/10.3390/molecules201119674
Olennikov DN, Kashchenko NI, Chirikova NK, Koryakina LP, Vladimirov LN. Bitter Gentian Teas: Nutritional and Phytochemical Profiles, Polysaccharide Characterisation and Bioactivity. Molecules. 2015; 20(11):20014-20030. https://doi.org/10.3390/molecules201119674
Chicago/Turabian StyleOlennikov, Daniil N., Nina I. Kashchenko, Nadezhda K. Chirikova, Lena P. Koryakina, and Leonid N. Vladimirov. 2015. "Bitter Gentian Teas: Nutritional and Phytochemical Profiles, Polysaccharide Characterisation and Bioactivity" Molecules 20, no. 11: 20014-20030. https://doi.org/10.3390/molecules201119674