Results and Discussion
Reaction of 3-furancarbaldehyde with methyl azidoacetate in the presence of sodium methoxide proceeded smoothly to give the azide
1, thermolysis of which was carried out in boiling toluene leading to the compound
2a. Phase transfer catalysis was found to be successful for methylation and benzylation of
2a giving the derivatives
2b and
2c (
Scheme 1). The compounds
2a-
2c gave under Vilsmeier conditions 2-formylated products
3a-
3c. By refluxing the compounds
2b and
2c with hydrazine in ethanol the corresponding hydrazides
4b and
4c were formed. Our experiments to synthesize 6
H- furo[2,3-
b]pyrrole-5-carboxyhydrazide (
4a) under conditions which were used for preparation of
4b and
4c were unsuccessful.
The reaction of 3a-3c with hydroxylammonium chloride in acetic anhydride in the presence of pyridine at 90 °C gave the corresponding cyano-substituted compounds 5a-5c. The reaction of the compounds 5a-5c with sodium azide and ammonium chloride in dimethylformamide led to the tetrazoles 6a-6c. N,N-Dimethylhydrazones 7a-7c were prepared from the aldehydes (3a-3c) and unsym-dimethylhydrazine in refluxing toluene, using a catalytic amount of 4-methylbenzenesulfonic acid.
During the synthesis and reaction studies of both systems we found out that the 1,4 system (
8) is more stable than its 1,6 positional isomer (
2). This empirical conclusion is in agreement with the results of AM1 semiempirical MO calculations that we have carried out for the parent furopyrroles and the ester derivatives
2a and
8a.
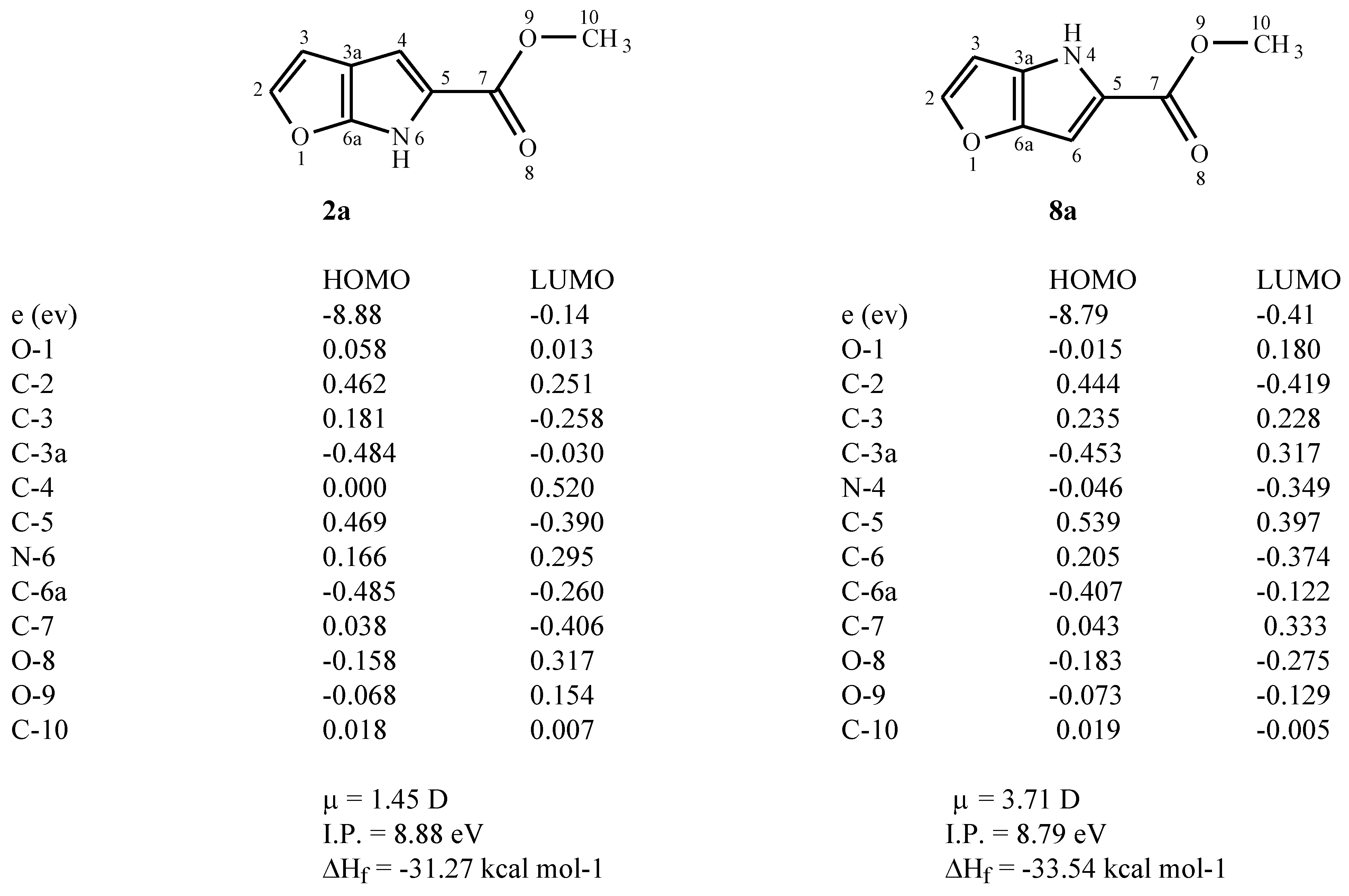
Figure 1 shows the calculated properties for the methyl esters
2a and
8a including their heats of formation (ΔH
f). The 1,4 system (
8a) is calculated to be thermodynamically more stable than the 1,6-isomer (
2a). The 1,4-system (
8a) is also calculated to have a significantly larger dipole moment (μ) (
Figure 1), which may result in greater solvent stabilisation. Comparable results were obtained for the unsubstituted heterocycles (
Figure 2). Calculated net atomic charges and molecular geometries are given in
Table 4,
Table 5,
Table 6 and
Table 7. The calculated ionisation potentials (using Koopmaan' theorem) (
Figure 1) are consistent with the classification of these heterocycles as electron-rich.
The
1H and
13C chemical shifts were assigned after comparison with model compounds [
10,
11] and H, C-COSY [
12] was used to correlate carbons with directly bonded protons.
Figure 1.
Calculated (AM1) heats of formation (ΔHf), dipole moments (μ) and frontier orbital coefficient and energies (ε) for methyl esters 2a and 8a.
Figure 1.
Calculated (AM1) heats of formation (ΔHf), dipole moments (μ) and frontier orbital coefficient and energies (ε) for methyl esters 2a and 8a.
Figure 2.
Calculated (AM1) heats of formation (ΔHf) and dipole moments (μ) for parent furopyrroles.
Figure 2.
Calculated (AM1) heats of formation (ΔHf) and dipole moments (μ) for parent furopyrroles.
Table 1.
13C Chemical shifts (δ, ppm) of compounds 2a-2c and 8a-8c in CDCl3
Table 1.
13C Chemical shifts (δ, ppm) of compounds 2a-2c and 8a-8c in CDCl3
| Carbon | 2a | 2b | 2c | 8a | 8b | 8c |
|---|
| C-2 | 143.65 | 143.48 | 143.72 | 148.69 | 148.29 | 148.30 |
| C-3 | 105.62 | 105.71 | 105.67 | 98.89 | 97.83 | 98.64 |
| C-3a | 110.56 | 107.57 | 108.05 | 128.86 | 133.34 | 132.99 |
| C-4 | 106.36 | 106.86 | 107.76 | - | - | - |
| C-5 | 120.70 | 120.76 | 120.16 | 123.77 | 123.40 | 123.19 |
| C-6 | - | - | - | 96.93 | 97.83 | 98.80 |
| C-6a | 151.60 | 153.56 | 153.52 | 147.93 | 145.34 | 145.80 |
| CO | 162.75 | 162.33 | 162.12 | 162.61 | 162.40 | 162.28 |
| OCH3 | 51.60 | 50.37 | 50.90 | 51.58 | 50.89 | 51.04 |
| R | - | 32.00a | 48.10b | - | 34.59a | 50.62c |
Table 2.
Difference of 13C chemical shifts (Δ δ, ppm) a of 2a and 8a carbons relative to carbons of furan and methyl 2-pyrrolecarboxylate.
Table 2.
Difference of 13C chemical shifts (Δ δ, ppm) a of 2a and 8a carbons relative to carbons of furan and methyl 2-pyrrolecarboxylate.
| Compound | Δ (Cδ-2) | Δ (Cδ-3) | Δ (Cδ-4) | Δ (Cδ-5) | Δ (Cδ-6) |
|---|
| 2a | 0.05 | -4.78 | -7.74 | -1.30 | - |
| 8a | 5.09 | -11.51 | - | 1.77 | -18.17 |
The
13C chemical shifts of
2a-2c and
8a-
8c are reported in
Table 1,
1H and
13C NMR data of other compounds are in the experimental part. The different
13C chemical shift values of the corresponding carbon positions for compounds
2a and
8a relative to the carbons of furan [
13] and methyl 2-pyrrolecarboxylate [
14] (
Table 2) show, that in the 1,4-isomer
8a the differences are greater than in 2a. In
8a carbon C-2 shows a downfield shift Δδ= 5.09 ppm, C-3 an upfield shift Δδ= -11.51 ppm as well as for C-6 Δδ= -18.17 ppm. This demonstrates that the electron density of both compared systems changes due to the annelated ring interaction, but the effect of the annelated ring is greater in the case of the 1,4 system. An analogous upfield shift was observed in
1H,4H-pyrrolo[3,2-
b]pyrrole [
15]. In order to make a direct comparison of both types of furopyrroles we carried out the correlation of the
13C and
15N chemical shifts (
Table 1 and
Table 3) with net atomic charges, calculated using the AM1 method (
Table 4 and
Table 6). In compounds
2a-2c signals C-2 and C-5 appear at higher magnetic field and C-3 and C-4 at lower field in comparison with corresponding carbons in
8a-
8c (
Table 1). The relative values of the calculated net atomic charges for the parent systems (
Table 4) and the esters
2a and
8a (
Table 6) are in good agreement with these experimental data. The comparison of
13C chemical shifts of substituted furo[2,3-
b]pyrroles shows that the greatest effect of substituents in the 2-position was observed at C-2 and C-3, analogous to the 2-substituted furans [
13] and the 1,4-
O,N-system [
16].
Table 3.
15N Chemical shifts (δ, ppm and nJ(15N,H) coupling constants (Hz) of compounds 2a-2c and 8a-8c in CDCl3.
Table 3.
15N Chemical shifts (δ, ppm and nJ(15N,H) coupling constants (Hz) of compounds 2a-2c and 8a-8c in CDCl3.
| Compound | δ(15N) | 1J(15N,H) | 3J(15N,H)a | 2J(15N,H) |
|---|
| 2a | -262.1 | 100.8 | 3.4 | - |
| 2b | -260.4 | - | 3.3 | 1.6 |
| 2c | -248.3 | - | 3.3 | 1.7 |
| 8a | -259.2 | 100.9 | 3.8 | - |
| 8b | -259.0 | - | 3.6 | 1.7 |
| 8c | -247.6 | - | 3.5 | 1.7 |
Table 4.
Calculated (AM1) net atomic charges of parent furopyrroles.
Table 4.
Calculated (AM1) net atomic charges of parent furopyrroles.
| Position |
|---|
| Isomer | 1 | 2 | 3 | 4 | Position5 | 6 | 3a | 6a |
|---|
| [2,3-b] | -0.092 | -0.116 | -0.122 | -0.130 | -0.141 | -0.167[N] | -0.164 | -0.001 |
| [3,2-b] | -0.074 | -0.083 | -0.164 | -0.153[N] | -0.114 | -0.165 | -0.121 | -0.058 |
The
15N NMR spectra of the 1,4- and 1,6-
O,N-diheteropentalenes have not previously been published. We measured the
15N NMR spectra of
2a-2c and
8a-8c in order to compare both types of
O,N-diheteropentalenes (
Table 3). Coupling constants
1J(
15N,H) and
3J(
15N,H) were obtained directly from the spectra. Selective excitation was applied to prove that the
3J(
15N,H) coupling constants are due to the proton on the pyrrole ring; 60 and 100 ms evolution times were used for other compounds and spectral patterns measured were compared with simulated ones using the SIMEPT programme [
17]. It was assumed, taking the data for compounds
2a and
8a into account, that the greater coupling constants were due to interaction with the proton on the pyrrole ring. The slightly larger negative values of the
15N chemical shifts in
2a-2c compared to
8a-8c agree with the relative values of the calculated (AM1) negative charges on nitrogen (
Table 4 and
Table 6).
The configurational assignment of the substituents on the double bond of the hydrazone
7a has been determined by
15N NMR spectra using the stereospecific coupling constants
2J(
15N,H-7). The orientation of the lone-pair of the nitrogen and the corresponding proton has a marked effect on the value of the respective coupling constant. The comparison of the coupling constants
2J(
15N,H-7) = 6.5 Hz with those of model compounds in ref. [
18,
19] confirms the
E-isomer of
7a. The same configuration was determined for some hydrazones in our previous paper [
20].
Table 5.
Calculated (AM1) geometries of parent furopyrroles.
Table 5.
Calculated (AM1) geometries of parent furopyrroles.
| Bond lengths (Å) |
|---|
| Isomer | a | b | c | d | e | f | g | h | i | | | |
|---|
| [2,3-b] | 1.384 | 1.420 | 1.381 | 1.435 | 1.443 | 1.428 | 1.405 | 1.413 | 1.371 | | | |
|---|
| [3,2-b] | 1.380 | 1.411 | 1.383 | 1.436 | 1.446 | 1.380 | 1.399 | 1.412 | 1.418 | | | |
| Bond angles (deg.) |
| | | bc | | cd | | de | | ef | | fg | | gh |
| [2,3-b] | | 113.2 | | 106.5 | | 104.7 | | 105.3 | | 106.8 | | 111.0 |
| [3,2-b] | | 113.1 | | 105.4 | | 106.5 | | 107.6 | | 107.7 | | 111.1 |
Table 6.
Calculated (AM1) net atomic charges of methyl esters
2a and
8a.
Table 6.
Calculated (AM1) net atomic charges of methyl esters 2a and 8a.
| Isomer | 1 | 2 | 3 | 4 | 5 | 6 | 3a | 6a | 7 | 8 | 9 | 10 |
|---|
| [2,3-b] | -0.093 | -0.104 | -0.116 | -0.031 | -0.148 | -0.131[N] | -0.184 | +0.04 | +0.372 | -0.373 | -0.265 | -0.061 |
| [3,2-b] | -0.071 | -0.053 | -0.180 | -0.115[N] | -0.122 | -0.069 | -0.073 | -0.083 | +0.371 | -0.343 | -0.295 | -0.058 |
Table 7.
Calculated (AM1) geometries of methyl esters
2a and
8a.
Table 7.
Calculated (AM1) geometries of methyl esters 2a and 8a.
| Bond lengths (Å) |
|---|
| Isomer | a | b | c | d | e | f | g | h | i | j | k | l | m |
|---|
| [2,3-b] | 1.383 | 1.425 | 1.378 | 1.440 | 1.450 | 1.418 | 1.415 | 1.422 | 1.363 | 1.452 | 1.237 | 1.370 | 1.430 |
| [3,2-b] | 1.384 | 1.409 | 1.382 | 1.440 | 1.450 | 1.369 | 1.410 | 1.423 | 1.410 | 1.454 | 1.233 | 1.374 | 1.429 |
| Bond angles (deg.) |
| | | | bc | cd | de | ef | fg | gh | gj | jl | jk | lm | |
| | | 112.9 | 106.9 | 104.4 | 105.5 | 106.7 | 110.9 | 128.9 | 113.8 | 127.8 | 116.0 | |
| [3,2-b] | | | 113.5 | 105.1 | 106.5 | 108.2 | 107.4 | 110.7 | 124.2 | 114.2 | 127.9 | 116.5 | |
Experimental Part
The 1H (360.13 MHz), 13C (90.56) and 15N NMR (36.50 MHz) spectra were recorded on a Bruker AMX 360 spectrometer equipped with a 5 mm broadband probe and a X32 computer using the UX NMR software. 1H and 13C chemical shifts were referred to internal TMS (δ= 0.00).
15N NMR spectra were measured using non-refocused INEPT [
21]. The evolution time used was 2.6 ms for compounds
2a and
8a and 60 and 100 ms for other compounds.
15N chemical shifts were referred to external nitromethane (δ= 0.0) placed in a coaxial capillary. Negative values of chemical shifts denote upfield shifts with respect to standards.
Melting points were determined on a Kofler hot plate apparatus and are uncorrected. UV spectra were measured on a M-40 (Carl Zeiss, Jena) spectrophotometer in methanol [λmax (log ε); λmax in nm, εin m2mol-1]. The IR spectra were taken on a FTIR PU 9802/25 (Philips) spectrophotometer using KBr technique (0.5 mg in 300 mg KBr, νin cm-1).
Molecular orbital calculations were carried out using the AM1 semiempirical method [
22]. The geometry of each molecule studied was found by minimising the energy with respect to all geometrical variables.
Methyl 2-azido-3-(3-furyl)propenoate (1a)
A solution of 3-furancarbaldehyde (1.92 g, 20 mmol) and methyl azidoacetate (9.2 g, 80 mmol) was added at 0 °C during 30 min to sodium metal (1.84 g, 0.08 gat.) in methanol (60 ml). Stirring was continued for an additional 60 min at a temperature not exceeding 5 °C. The reaction mixture was then cooled to 0 oC, a solution of ammonium chloride (4.4 g, 80 mmol) in water (10 ml) was added and then the mixture was poured into ice water. The separated precipitate was filtered off and crystallized. Yield 2.55 g (66%); m.p. 40-41 °C (methanol). 1H NMR (CDCl3): 7.91 (dd, 1H, J(2,5) = 1.7, J(2,4) = 0.9, H-2), 7.42 (t, 1H, J(5,2) = J(5,4) = 1.7, H-5), 6.78 (2H, H-4, H-6); 13C NMR (CDCl3): 163.56 (C=O), 145.04 (C-2), 143.25 (C-5), 124.57 (C-7), 119.74 (C-3), 116.78 (C-6), 110.71 (C-4), 52.67 (O-CH3); IR: 1713 (C=O), 2130 (N3); UV: 299 (3.32), 219 (3.02). Anal. Calcd for C8H7N3O3: C, 49.74; H, 3,65; N, 21.75. Found: C, 49.62; H, 3,55; N, 21.65.
Methyl 6H-furo[2,3-b]pyrrole-5-carboxylate (2a)
Methyl 2-azido-3-(3-furyl)propenoate (1) (1 g, 5.2 mmol) was dissolved in toluene (100 ml). The mixture was refluxed under stirring for 1h. The solvent was evaporated in vacuo and the product was crystallized. Yield 0.5 g (61%); m.p. 162-166 °C (toluene-isohexane). 1H NMR (CDCl3): 9.80 (bs, 1H, NH), 7.25 (d, 1H, J = 2.2, H-2), 6.83 (d, J=1.8, 1H, H-4), 6.50 (d, 1H, J = 2.2, H-3), 3.88 (s, 3H, O-CH3); IR: 1682, 1644 (C=O), 3252 (NH); UV: 292 (3.35), 247 (2.81). Anal. Calcd for C8H7NO3: C, 58.18; H, 4.27; N, 8.48. Found: C, 58.24; H, 4.36; N, 8.54.
Methyl 6-methylfuro[2,3-b]pyrrole-5-carboxylate (2b)
A solution of sodium hydroxide (50%, 30 ml), methyl iodide (1.56 g, 11 mmol) and triethylbenzylammonium chloride (0.4 g) was added to a stirred solution of 2a (1.65 g, 10 mmol) in toluene (100 ml). The temperature was then raised to 65 °C, the mixture stirred for 4 h, diluted with water and the organic layer separated. The aqueous layer was extracted with ether and combined with the toluene solution, dried with sodium sulphate and the solvent removed. The residue was crystallized. Yield 0.82 g (45.7%); m.p. 33-35 °C (isohexane). 1H NMR (CDCl3): 7.25 (d, 1H, J = 2.2, H-2), 6.84 (s, 1H, H-4), 6.47 (d, 1H, J = 2.2, H-3), 3.93 (s, 3H, N-CH3), 3.80 (s, 3H, O-CH3); IR: 1701 (C=O) ; UV: 292 (3.30), 253 (2.85). Anal. Calcd for C9H9NO3: C, 58.18; H, 4.27; N, 8.48. Found: C, 58.24; H, 4.36; N, 8.54.
Methyl 6-benzylfuro[2,3-b]pyrrole-5-carboxylate (2c)
This was prepared according to the above procedure. Yield 38%; m.p. 52-54 °C (isohexane). 1H NMR (CDCl3): 7.25 (d, 1H, J = 2.3, H-2), 6.98 (s, 1H, H-4), 7.34 - 7.18 (m, 5H, H-arom), 6.53 (d, 1H, J = 2.3, H-3), 5.69 (s, 2H, N-CH2), 3.82 (s, 3H, O-CH3); IR: 1698 (C=O); UV: 293 (3.31), 250 (2.93). Anal. Calcd for C15H13NO3: C, 70.58; H, 5.13; N, 5.43. Found: C, 70.65; H, 5.19; N, 5.37.
Methyl 2-formyl-6H-furo[2,3-b]pyrrole-5-carboxylate (3a)
A mixture of dimethylformamide (6 g, 80 mmol) and phosphorus oxychloride (3.4 g, 20 mmol) was stirred at 0 oC for 20 min. Methyl 6H-furo[2,3-b]pyrrole-5-carboxylate (2a) (3.30 g, 20 mmol) dissolved in dimethylformamide (6g) was added at a temperature not exceeding 10 oC. The mixture was stirred at 60 oC for 2h, poured into ice cold water, neutralized with sodium hydrogen carbonate, allowed to stand and the separated substance was filtered off and crystallized from methanol. Yield 2.7 g (70%); m.p. 203-207 °C (methanol). 1H NMR (DMSO-d6): 12.96 (bs, 1H, NH), 9.49 (s, 1H, CH=O), 7.74 (s, 1H, H-3), 6.97 (s, 1H, H-4), 3.82 (s, 3H, O-CH3); 13C NMR (DMSO-d6): 177.56 (CH=O), 161.08 (C=O), 154.75 (C-2), 153.55 (C-6a), 124.82 (C-5), 120.63 (C-3), 111.21 (C-3a), 107. 01 (C-4), 51.71 (O-CH3); IR: 1698 (CO), 1644 (CH=O), 3120 (NH); UV: 334 (3.50), 251 (2.74), 228 (3.04). Anal. Calcd for C9H7NO4: C, 55.96; H, 3.65; N, 7.25. Found: C, 55.86; H, 3.71; N, 7.45.
According to this procedure the following compounds were prepared:
Methyl 2-formyl-6-methylfuro[2,3-b]pyrrole-5-carboxylate (3b).
Yield 79.6%; m.p. 157-159 °C (methanol). 1H NMR (DMSO-d6): 9.50 (s, 1H, CH=O), 7.78 (s, 1H, H-3), 7.05 (s, 1H, H-4), 3.81 (s, 3H, O-CH3), 3.91 (s, 3H, N-CH3); 13C NMR (DMSO-d6): 177.39 (CH=O), 160.86 (C=O), 155.24 (C-2), 153.48 (C-6a), 124.57 (C-5), 120.96 (C-3), 108.76 (C-3a), 108.20 (C-4), 51.47 (O-CH3), 32.24 (N-CH3); IR: 1709 (C=O), 1655 (CH=O); UV: 334 (3.51), 250 (2.74), 231 (3.06). Anal. Calcd for C10H9NO4: C, 57.97; H, 4.38; N, 6.76. Found: C, 57.83; H, 4.35; N, 6.92.
6-Methylfuro[2,3-b]pyrrole-5-carboxyhydrazide (4b)
To a solution of 2b (1.79 g, 10 mmol) in ethanol (30 ml) was added hydrazine hydrate (100%, 3 g), the mixture was refluxed for 40 h and after cooling the separated crystals were filtered off. Yield 0.896 g (50%); m.p. 163-167 °C (methanol). 1H NMR (DMSO-d6): 9.28 (bs, 1H, NH), 7.54 (d, 1H, J = 2.0, H-2), 6.74 (s, 1H, H-4), 6.63 (d, 1H, J = 2.0, H-3), 4.28 (bs, 2H, NH2), 3.88 (s, 3H, N-CH3); 13C NMR (DMSO-d6): 162.34 (C=O), 152.09 (C-6a), 143.34 (C-2), 123.31 (C-5), 106.28 (C-3a), 105.86 (C-3), 100.91 (C-4), 31.84 (N-CH3); IR: 1618 (C=O), 3281, 3227 (NH); UV: 289 (3.28). Anal. Calcd for C8H9N3O2: C, 53.63; H, 5.06; N, 23.45. Found: C, 53.88; H, 5.22; N, 23.37.
6-Benzylfuro[2,3-b]pyrrole-5-carboxyhydrazide (4c)
This compound was obtained analogously. Yield 71.4%; m.p. 140-143 °C (methanol). 1H NMR (DMSO-d6): 9.37 (bs, 1H, NH), 7.53 (d, 1H, J = 2.0, H-2), 7.30 - 7.00 (m, 5H, H-arom), 6.82 (s, 1H, H-4), 6.64 (d, 1H, J = 2.0, H-3), 5.70 (s, 2H, CH2), 4.32 (bs, 2H, NH2); 13C NMR (DMSO-d6): 162.45 (C=O), 152.11 (C-6a), 143.85 (C-2), 122.91 (C-5), 107.05 (C-3a), 105.97 (C-4), 101.81 (C-3), 47.49 (N-CH2), C-arom: 138.26 (C-1'), 128.63 (C-3', C-5'), 127.47 (C-4'), 127.07 (C-2', C-6'); IR: 1686, 1609 (C=O), 3316, 3265 (NH); UV: 289 (3.14).
Anal. Calcd for C14H13N3O2: C, 65.87; H, 5.13; N, 16.46. Found: C, 66.07; H,5.19; N, 16.32.
Methyl 2-cyano-6H-furo[2,3-b]pyrrole-5-carboxylate (5a)
To the mixture of 3a (1.93 g, 10 mmol), pyridine (8 ml) and hydroxylammonium chloride (1.2 g, 17 mmol) in acetic anhydride (5.5 ml) were added under stirring at 95 °C. The reaction mixture was kept at 85-95 °C for 2 h, cooled and poured onto ice. The separated precipitate was filtered off and crystallized. Yield 1.738 g (91.4%); m.p. 212-214 °C (methanol). 1H NMR (DMSO-d6): 13.12 (bs, 1H, NH), 7.82 (s, 1H, H-3), 6.91 (s, 1H, H-4), 3.82 (s, 3H, O-CH3); 13C NMR (DMSO-d6): 160.99 (C=O), 152.42 (C-6a), 124.94 (C-2), 124.81 (C-5), 119.66 (C-3), 112.51 (CN), 109.28 (C-3a), 105.90 (C-4), 51.63 (O-CH3); IR: 1690 (C=O), 2222 (CN), 3235 (NH); UV: 303 (3.49), 222 (3.10). Anal. Calcd for C9H6N2O3: C, 56.85; H, 3.18; N, 14,73. Found: C, 56.75; H, 3.24; N, 14,65.
According to this procedure the following compounds were prepared:
Methyl 2-cyano-6-methylfuro[2,3-b]pyrrole-5-carboxylate (5b)
Yield 98.5%; m.p. 181-183 °C (methanol). 1H NMR (DMSO-d6): 7.82 (s, 1H, H-3), 6.98 (s, 1H, H-4), 3.90 (s, 3H, N-CH 3), 3.80 (s, 3H, O-CH3); 13C NMR (DMSO-d6): 160.91 (C=O), 153.07 (C-6a), 125.24 (C-2), 124.65 (C-5), 119.98 (C-3), 112.34 (CN), 107.01 (C-3a), 107.13 (C-4), 51.47 (O-CH3), 32.32 (N-CH3); IR: 1694 (C=O), 2216 (CN); UV: 303 (3.50), 225 (3.10). Anal. Calcd for C10H8N2O3: C, 58.82; H, 3.95; N, 13.72. Found: C, 58.63; H, 3.85; N, 13.68.
Methyl 2-cyano-6-benzylfuro[2,3-b]pyrrole-5-carboxylate (5c)
Yield 91%; m.p. 153-156 °C (methanol). 1H NMR (DMSO-d6): 7.87 (s, 1H, H-3), 7.40 - 7.10 (m, 5H, H-arom), 7.09 (s, 1H, H-4), 5.67 (s, 2H, N-CH2), 3.78 (s, 3H, O-CH3); 13C NMR ( DMSO-d6): 161.08 (C=O), 153.19 (C-6a), 125.79 (C-2), 124.23 (C-5), 120.13 (C-3), 112.36 (CN), 108.34 (C-4), 107.78 (C-3a), 51.75 (O-CH3), 48.16 (N-CH2), C-arom: 136.77 (C-1'), 128.89 (C3', C-5'), 127.94 (C-4'), 127.07 (C-2', C-6'); IR: 1717 (C=O), 2220 (CN); UV: 303 (3.49), 226 (3.14). Anal. Calcd for C16H12N2O3: C, 68.57; H, 4.32; N, 9.99. Found: C, 68.44; H, 4.52; N, 10.02.
Methyl 2-(5'-tetrazolyl)-6H-furo[2,3-b]pyrrole-5-carboxylate (6a)
A stirred mixture of 5a (0.95 g; 5 mmol), sodium azide (0.36 g, 6 mmol), ammonium chloride (0.32 g, 6 mmol) and dimethylformamide (7 ml) was heated at 100 °C for 4 h. The solvent was distilled off in vacuo, the residue was dissolved in water, the solution was acidified with hydrochloric acid and the precipitate was filtered off and crystallized. Yield 0.855 g (73.3%); m.p. 270-274 °C (methanol). 1H NMR (DMSO-d6): 13.00 (bs, 1H, NH), 7.44 (s, 1H, H-3), 6.90 (d, 1H, J = 1.7, H-4), 3.77 (s, 3H, O-CH3); 13C NMR ( DMSO-d6): 161.18 (C=O), 152.60 (C-6a), 148.60 (C-5'), 140.39 (C-2), 122.47 (C-5), 110.80 (C-3a), 108.45 (C-3), 105.84 (C-4), 51.43 (O-CH3); IR: 1684 (C=O), 3247 (NH); UV: 313 (3.53), 253 (2.84), 228 (3.07). Anal. Calcd for C9H7N5O3: C, 46.36; H, 3.03; N, 30.03. Found: C, 46.28; H, 3.17; N, 30.12.
According to this procedure the following compounds were prepared:
Methyl 2-(5'-tetrazolyl)-6-methylfuro[2,3-b]pyrrole-5-car-boxylate (6b)
Yield 69.5%; m.p. 249-251 °C (methanol). 1H NMR (DMSO-d6): 7.44 (s, 1H, H-3), 6.95 (s, 1H, H-4), 3.97 (s, 3H, N-CH 3), 3.79 (s, 3H, O-CH3); 13C NMR (DMSO-d6): 161.16 (C=O), 153.54 (C-6a), 149.01 (C-5'), 141.05 (C-2), 122.45 (C-5), 108.59 (C-3a), 108.49 (C-3), 107.07 (C-4), 51.15 (O-CH 3) 32.19 (N-CH3); IR: 1705 (C=O); UV: 314 (3.54), 253 (2.86), 228 (3.07). Anal. Calcd for C10H9N5O3: C, 48.59; H, 3.67; N, 28.33. Found: C, 48.68; H, 3.55; N, 28.22.
Methyl 2-(5'-tetrazolyl)-6-benzylfuro[2,3-b]pyrrole-5-car-boxylate (6c)
Yield 66.5%; m.p. 239-244 °C (methanol). 1H NMR (DMSO-d6): 7.49 (s, 1H, H-3), 7.40 - 7.16 (m, 5H, H-arom), 7.06 (s, 1H, H-4), 5.72 (s, 2H, N-CH2), 3.77 (s, 3H, O-CH3); 13C NMR ( DMSO-d6): 161.20 (C=O), 153.56 (C-6a), 148.98 (C-5'), 141.51 (C-2), 121.81 (C-5), 109.26 (C-3a), 108.74 (C-3), 108.25 (C-4), 51.49 (O-CH3), 48.06 (N-CH2), C-arom: 137.19 (C-1'), 128.86 (C-3', C-5') 127.79 (C-4'), 126.84 (C-2', C-6'); IR: 1707 (C=O); UV: 313 (3.52), 257 (2.96), 229 (3.10). Anal. Calcd for C16H13N5O3: C, 59.44; H, 4.05; N, 21.66. Found: C, 59.47; H, 3.97; N, 21.82.
5-Methoxycarbonyl-6H-furo[2,3-b]pyrrole-2-carbaldehyde dimethylhydrazone (7a)
A stirred solution of 3a (0.96 g, 5 mmol) in toluene (5 ml) containing a catalytic amount of 4-methylbenzene sulphonic acid (3 mg) was treated carefully with N,N-dimethylhydrazine (0.30 g, 5 mmol) in toluene (5 ml). The solution was then refluxed for 2 h and the water formed during the reaction was removed in a Dean-Stark trap. The solvent was removed under reduced pressure and the residue was crystallized. Yield 0.882 g (75%); m.p. 191-194 °C (toluene-isohexane). 1H NMR [(CD 3)2CO]: 11.18 (bs, 1H, NH), 7.17 (s, 1H, H-7), 6.75 (s, 1H, H-4), 6.51 (s, 1H, H-3), 3.79 (s, 3H, O-CH3), 2.92 [s, 6H, N(CH3)2]; 13C NMR [(CD3)2CO]: 162.20 (C=O), 155.23 (C-2), 152.60 (C-6a), 123.48 (C-7), 121.63 (C-5), 112.46 (C-3a), 106.18 (C-4), 102.46 (C-3), 51.35 (O-CH3), 42.73 [(N(CH3)2)]; 5N NMR (DMSO-d6): -275.5 [(CH3)2N, 3J(15N, H-7) = 5.2], -251.8 (N-6, bs), -34.3[N=CH, 2J(15N, H-7) = 6.5]; IR: 1667 (C=O), 3260 (NH); UV: 341 (3.63), 274 (2.81), 235 (3.03). Anal. Calcd for C11H13N3O3: C, 56.16; H, 5.57; N, 17.86. Found: C, 56.37; H, 5.47; N, 17.72.
According to this procedure the following compounds were prepared:
5-Methoxycarbonyl-6-methylfuro[2,3-b]pyrrole-2-carbaldehyde dimethylhydrazone (7b)
Yield 73%; m.p. 114-118 °C (toluene-isohexane). 1H NMR (DMSO-d6): 7.21 (bs, 1H, H-7), 6.82 (s, 1H, H-4), 6.45 (bs, 1H, H-3), 3.97 (s, 3H, N-CH3), 3.81 (s, 3H, O-CH3), 2.97 [s, 6H, N(CH3)2]; 13C NMR (DMSO-d6): 162.26 (C=O), 153.88 (C-2), 153.42 (C-6a), C-7 not found, 121.37 (C-5), 109.35 (C-3a), 107.09 (C-4), 103.91 (C-3) 50.92 (O-CH3), 43.01 [N(CH3)2], 32.36 (N-CH3); IR: 1697 (C=O); UV: 341 (3.59), 274 (2.81), 235 (3.00). Anal. Calcd for C12H15N3O3: C, 57.82; H, 6.07; N, 16.86. Found: C, 57.67; H, 5.97; N, 16.72.
5-Methoxycarbonyl-6-benzylfuro[2,3-b]pyrrole-2-carbaldehyde dimethylhydrazone (7c)
Yield 72%; m.p. 129-132 °C (toluene-isohexane). 1H NMR [(CD3)2CO]: 7.26 (m, 5H, H-arom), 7.15 (s, 1H, H-7), 6.88 (s, 1H, H-4), 6.53 (s, 1H, H-3), 5.73 (s, 2H, N-CH2), 3.75 (s, 3H, O-CH3), 2.92 [s, 6H, N(CH3)2]; 13C NMR [(CD3)2CO]: 162.41 (C=O), 155.87 (C-2), 154.05 (C-6a), 123.13 (C-7), 120.94 (C-5), 110.59 (C-3a), 108.25 (C-4), 102.46 (C-3), 51.20 (O-CH3), 48.62 (N-CH 2), 42.69 [N(CH3)2], C-arom: 138.78 (C-1'), 129.38 (C-3', C-5'), 128.29 (C-4'), 127.68 (C-2', C-6'); IR: 1705 (C=O); UV: 342 (3.58), 270 (2.81), 236 (3.00). Anal. Calcd for C18H19N3O3): C, 66.45; H, 5.89; N, 12.91. Found: C, 66.54; H, 6.00; N, 12.83.