Headspace Solid-Phase Microextraction Analysis of Volatile Components in Phalaenopsis Nobby’s Pacific Sunset
Abstract
:1. Introduction
2. Results and Discussion
2.1. Comparison Optimal of SPME Fiber Coating
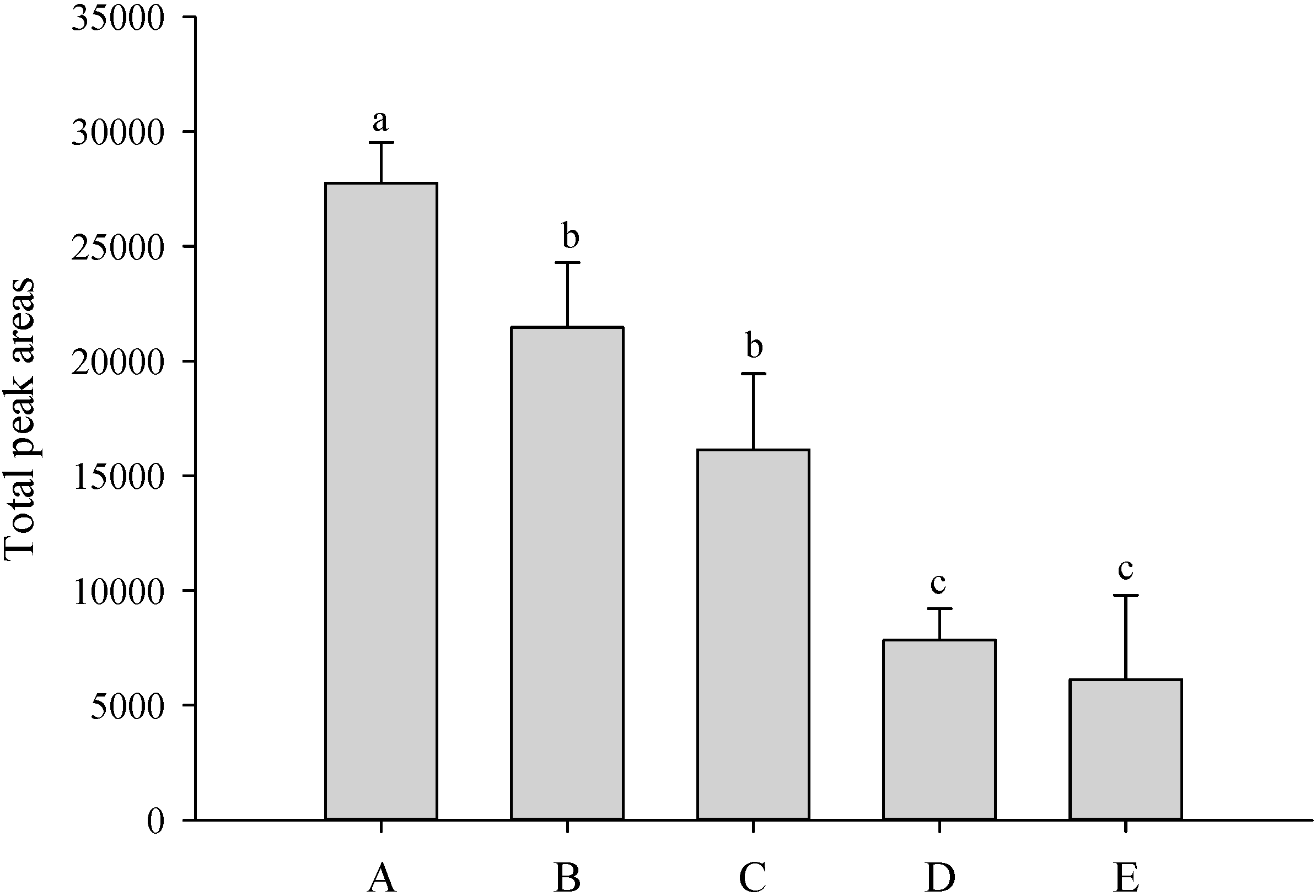
2.2. Analysis of the Volatiles in P. Nobby’s Pacific Sunset Flowers
| RI a | Compound | Content (%) |
|---|---|---|
| 641 | 2-methylbutanal | 0.60 ± 0.15 |
| 882 | styrene | 0.02 ± 0.00 |
| 933 | benzaldehyde | 0.08 ± 0.02 |
| 964 | 6-methyl-5-hepten-2-one | 0.69 ± 0.55 |
| 981 | β-myrcene | 3.87 ± 0.18 |
| 1021 | limonene | 1.30 ± 0.04 |
| 1023 | cis-β-ocimene | 2.17 ± 0.29 |
| 1035 | trans-β-ocimene | 0.07 ± 0.02 |
| 1050 | α-ocimene | 0.10 ± 0.01 |
| 1060 | 3-methylphenol | 0.07 ± 0.02 |
| 1065 | 2-methylphenol | 0.22 ± 0.10 |
| 1080 | linalool | 28.30 ± 16.78 |
| 1099 | (E)-4,8-dimethyl-1,3,7-nonatriene | 0.82 ± 0.08 |
| 1189 | methyl salicylate | 0.07 ± 0.02 |
| 1201 | nerol | 4.54 ± 1.05 |
| 1231 | geraniol | 28.31 ± 8.97 |
| 1233 | geranial | 2.04 ± 0.17 |
| 1357 | neryl acetate | 0.91 ± 0.57 |
| 1360 | geranyl acetate | 0.52 ± 0.26 |
| 1444 | trans-β-farnesene | 1.08 ± 0.87 |
| 1469 | α-humulene | 0.17 ± 0.13 |
| 1477 | α-curcumene | 0.29 ± 0.19 |
| 1481 | (Z,E)-α-farnesene | 0.47 ± 0.07 |
| 1489 | α-zingiberene | 0.20 ± 0.10 |
| 1490 | cis-γ-bisabolene | <0.01 |
| 1495 | (E,E)-α-farnesene | 11.10 ± 4.49 |
| 1544 | nerolidol | 0.31 ± 0.25 |
| 1556 | trans-γ-bisabolene | 0.17 ± 0.04 |
| 1686 | cis-11-hexadecenal | 0.10 ± 0.07 |
| 1705 | (Z,E)-farnesol | 1.18 ± 0.82 |
| 1721 | (E,E)-farnesal | 0.17 ± 0.15 |
| Total | 89.91 ± 1.19 |
2.2.1. Volatile Compounds of P. Nobby’s Pacific Sunset Flowers over a 24 h Period

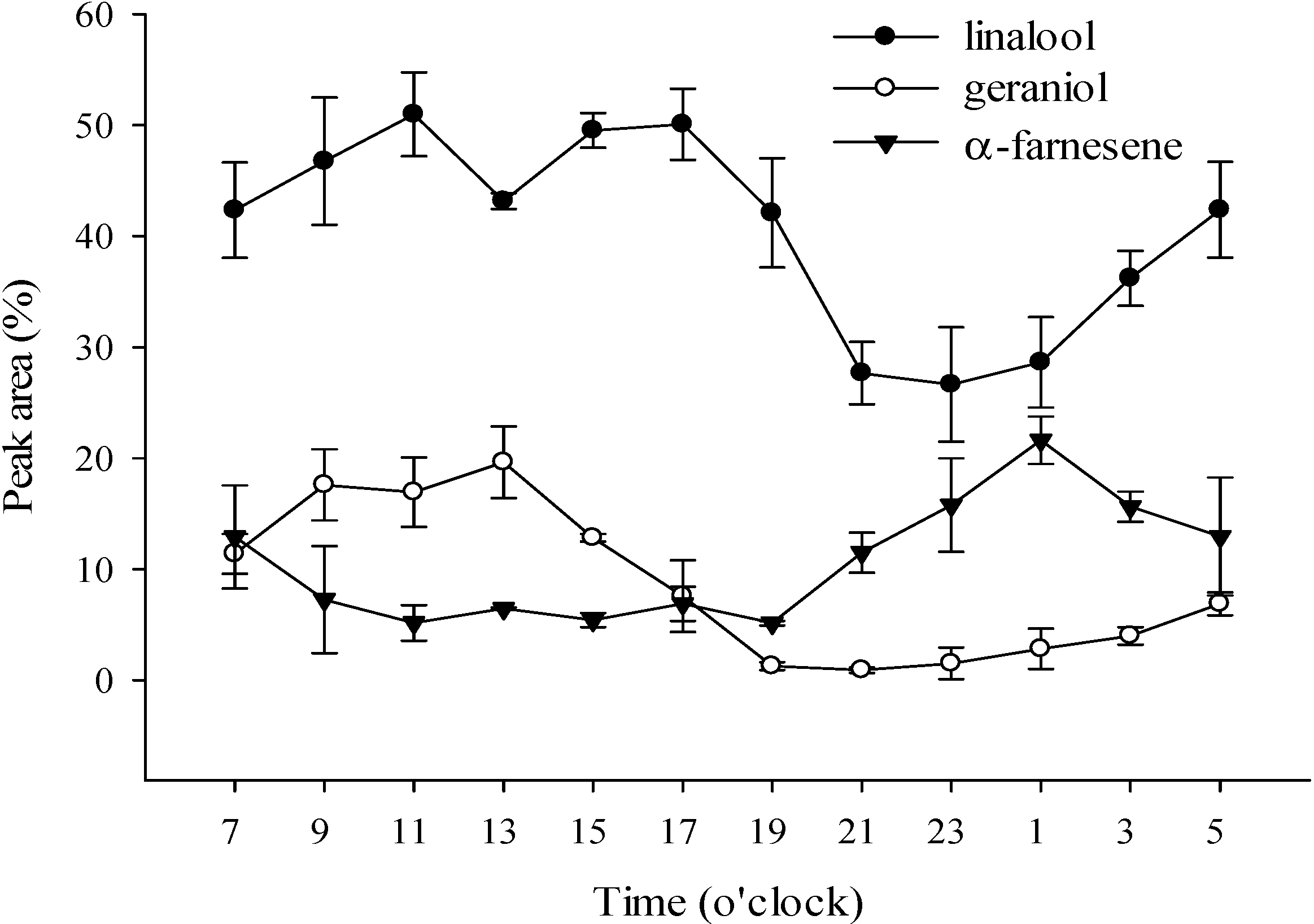
2.2.2. Volatile Compounds of P. Nobby’s Pacific Sunset Flowers in a Blooming Cycle

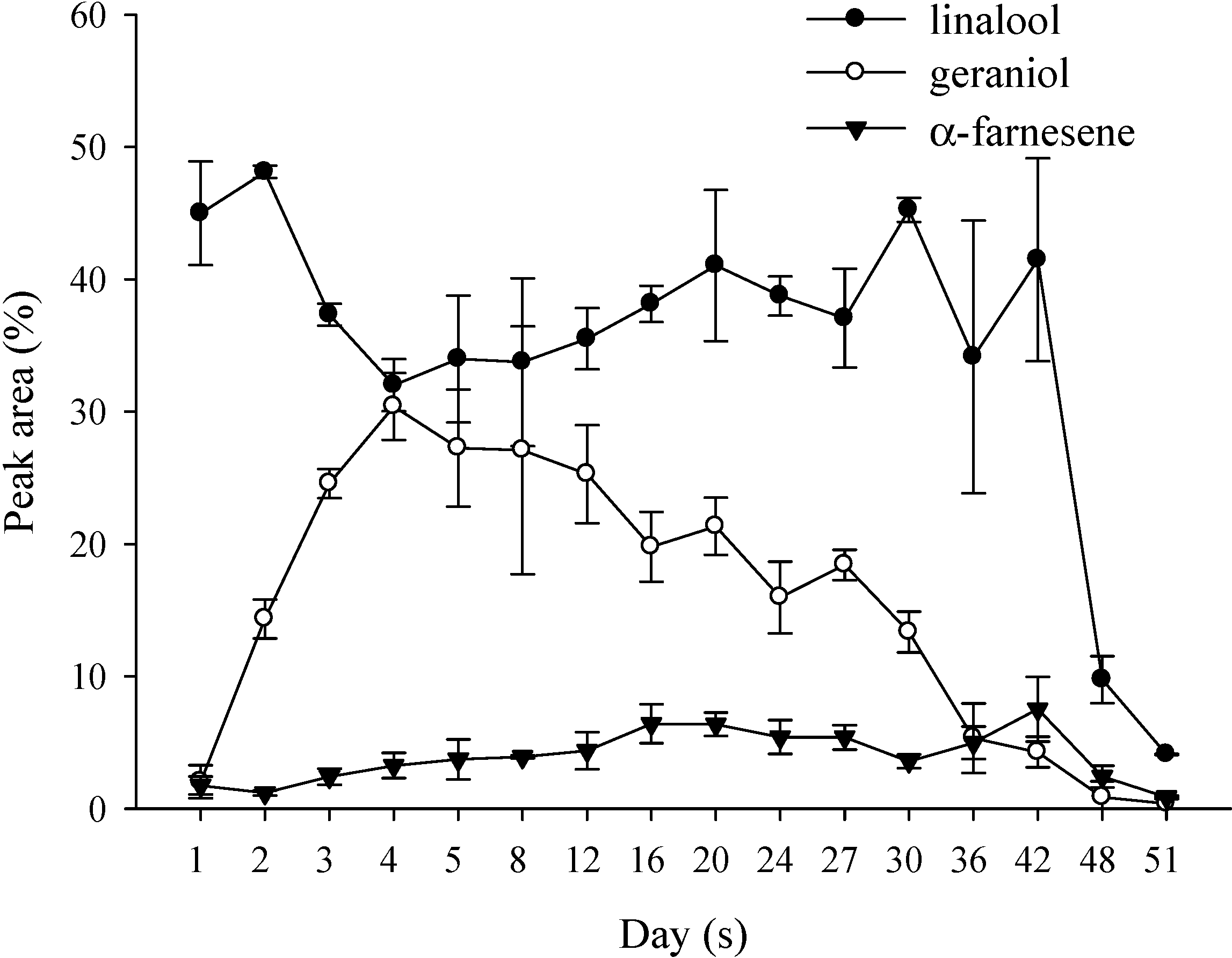
2.3. Volatile Compounds of Different Parts of the P. Nobby’s Pacific Sunset Flowers
| RI a | Compound | Whole Flowers | Petals | Lateral Sepals | Dorsal Sepals | Lip | Column |
|---|---|---|---|---|---|---|---|
| 641 | 2-methyl butanal | 0.60 ± 0.15 | 0.26 ± 0.24 | - b | - | 0.17 ± 0.12 | - |
| 882 | styrene | 0.02 ± 0.00 | 1.28 ± 1.16 | 0.86 ± 0.62 | 2.80 ± 1.78 | - | - |
| 933 | benzaldehyde | 0.08 ± 0.02 | - | 0.07 ± 0.00 | 0.07 ± 0.00 | 0.05 ± 0.02 | - |
| 964 | 6-methyl-5-hepten-2-one | 0.69 ± 0.55 | - | 0.48 ± 0.24 | - | - | - |
| 981 | β-myrcene | 3.87 ± 0.18 | 6.83 ± 0.05 | 6.35 ± 0.46 | 5.93 ± 0.45 | 5.62 ± 1.33 | 4.89 ± 0.21 |
| 1021 | limonene | 1.30 ± 0.04 | 2.27 ± 0.02 | 2.12 ± 0.34 | - | 1.95 ± 0.39 | - |
| 1023 | cis-β-ocimene | <0.01 | - | - | - | - | 2.39 ± 0.32 |
| 1035 | trans-β-ocimene | 2.16 ± 0.28 | 3.60 ± 1.03 | 2.98 ± 0.22 | 3.21 ± 0.65 | 2.53 ± 0.68 | 6.44 ± 1.62 |
| 1050 | α-ocimene | 0.07 ± 0.02 | 0.10 ± 0.01 | 0.10 ± 0.03 | 0.08 ± 0.02 | 0.12 ± 0.01 | 0.35 ± 0.02 |
| 1060 | 3-methylphenol | 0.07 ± 0.02 | 0.10 ± 0.01 | - | - | - | - |
| 1065 | 2-methylphenol | 0.22 ± 0.10 | 0.33 ± 0.02 | 0.32 ± 0.13 | 0.29 ± 0.06 | 0.53 ± 0.06 | 0.84 ± 0.24 |
| 1080 | linalool | 28.30 ± 16.78 | 40.24 ± 2.77 | 39.53 ± 7.40 | 42.04 ± 3.08 | 67.77 ± 4.98 | 63.53 ± 6.57 |
| 1099 | (E)-4,8-dimethyl-1,3,7-nonatriene | 0.51 ± 0.06 | 1.33 ± 0.15 | 1.20 ± 0.13 | 1.90 ± 0.29 | 0.80 ± 0.05 | - |
| 1189 | methyl salicylate | 0.11 ± 0.05 | - | - | - | - | - |
| 1201 | nerol | 4.54 ± 1.05 | 2.40 ± 0.77 | - | - | - | - |
| 1231 | geraniol | 28.31 ± 8.97 | 15.68 ± 4.39 | 17.77 ± 6.43 | 9.88 ± 2.07 | 6.50 ± 1.92 | - |
| 1233 | geranial | - | 1.15 ± 0.12 | 1.17 ± 0.15 | - | - | - |
| 1357 | neryl acetate | 0.91 ± 0.57 | - | - | - | 0.59 ± 0.53 | - |
| 1360 | geranyl acetate | 0.52 ± 0.26 | - | - | - | - | - |
| 1444 | trans-β-farnesene | 1.08 ± 0.87 | - | - | - | - | - |
| 1469 | α-humulene | 0.27 ± 0.07 | - | - | - | - | - |
| 1477 | α-curcumene | 0.29 ± 0.19 | 0.48 ± 0.16 | 0.46 ± 0.08 | 0.57 ± 0.11 | 0.16 ± 0.03 | - |
| 1481 | (Z,E)-α-farnesene | 0.47 ± 0.07 | - | - | - | - | - |
| 1489 | α-zingiberene | 0.20 ± 0.10 | - | - | - | - | - |
| 1490 | cis-γ-bisabolene | <0.01 | - | - | - | - | - |
| 1495 | (E,E)-α-farnesene | 11.10 ± 4.49 | 10.33 ± 1.27 | 10.71 ± 3.10 | 13.73 ± 1.32 | 2.59 ± 0.47 | 4.83 ± 3.08 |
| 1544 | nerolidol | 0.31 ± 0.24 | - | - | - | - | - |
| 1556 | trans-γ-bisabolene | 0.17 ± 0.05 | - | - | - | - | - |
| 1686 | cis-11-hexadecenal | 0.19 ± 0.05 | - | 0.03 ± 0.03 | - | - | - |
| 1705 | (Z,E)-farnesol | 0.82 ± 0.85 | - | - | - | - | - |
| 1721 | (E,E)-farnesal | 0.17 ± 0.15 | - | - | - | - | - |

3. Experimental Section
3.1. Plant Materials
3.2. Methods
3.2.1. Comparison of SPME Fiber Coating
3.2.2. Volatile Components of P. Nobby’s Pacific Sunset
3.2.3. Analysis of Volatile Compounds
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Awano, K.; Honda, T.; Ogawa, T.; Suzuki, S.; Matsunaga, Y. Volatile components of Phalaenopsis schilleriana Rehb. f. Flavour Frag. J. 1997, 12, 341–344. [Google Scholar] [CrossRef]
- Christenson, E.A. Phalaenopsis: A Monograph; Timber Press: Portland, OR, USA, 2001; pp. 24–25. [Google Scholar]
- An, F.M.; Chan, M.T. Transcriptome-wide characterization of miRNA-directed and non-miRNA-directed endonucleolytic cleavage using Degradome analysis under low ambient temperature in Phalaenopsis aphrodite subsp. formosana. Plant Cell Physiol. 2012, 53, 1737–1750. [Google Scholar] [CrossRef]
- Kaiser, R. The Scent of Orchids-Olfactory and Chemical Investigations; Elsevier: Amsterdam, The Netherlands, 1993; pp. 239–240. [Google Scholar]
- Hsiao, Y.Y.; Tsai, W.C.; Kuoh, C.S.; Huang, T.H.; Wang, H.C.; Wu, T.S.; Leu, Y.L.; Chen, W.H.; Chen, H.H. Comparison of transcripts in Phalaenopsis bellina and Phalaenopsis equestris (Orchidaceae) flowers to deduce monoterpene biosynthesis pathway. BMC Plant Biol. 2006, 6, 14. [Google Scholar]
- Schlossman, M.L. The Chemistry and Manufacture of Cosmetics; Allured Publishing Corporation: Carol Stream, IL, USA, 2009; Volume 2, p. 851. [Google Scholar]
- Zhu, F.; Xu, J.; Ke, Y.; Huang, S.; Zeng, F.; Luan, T.; Ouyang, G. Applications of in vivo and in vitro solid-phase microextraction techniques in plant analysis: A review. Anal. Chim. Acta 2013, 794, 11–14. [Google Scholar]
- Ouyang, G.; Vuckovic, D.; Pawliszyn, J. Nondestructive sampling of living systems using in vivo solid-phase microextraction. Chem. Rev. 2011, 111, 2784–2814. [Google Scholar] [CrossRef]
- Kataoka, H.; Lord, L.H.; Pawliszyn, J. Applications of solid-phase microextraction in food analysis. J. Chromatogr. A 2000, 880, 35–62. [Google Scholar]
- Alpendurada, M.F. Solid-phase microextraction: A promising technique for sample preparation in environmental analysis. J. Chromatogr. A 2000, 889, 3–14. [Google Scholar]
- Stashenko, E.E.; Martinez, J.R.; Cardenas-Vargas, S.; Saavedra-Barrera, R.; Duran, D.C. GC-MS study of compounds isolated from Coffea arabica flowers by different extraction techniques. J. Sep. Sci. 2013, 36, 2901–2914. [Google Scholar]
- Adam, M.; Juklová, M.; Bajer, T.; Eisner, A.; Ventura, K. Comparison of three different solid-phase microextraction fibres for analysis of essential oils in yacon (Smallanthus sonchifolius) leaves. J. Chromatogr. A 2005, 1084, 2–6. [Google Scholar]
- Lee, S.N.; Kim, N.S.; Lee, D.S. Comparative study of extraction techniques for determination of garlic flavor components by gas chromatography-mass spectrometry. Anal. Bioanal. Chem. 2003, 377, 749–756. [Google Scholar]
- Rega, B.; Fournier, N.; Guichard, E. Solid phase microextraction (SPME) of orange juice flavor- odor representativeness by direct gas chromatography olfactometry (D-GC-O). J. Agric. Food Chem. 2003, 51, 7092–7099. [Google Scholar]
- Eyres, G.T.; Marriott, P.J.; Dofour, J.-P. Comparison of odor-active compounds in the spicy fraction of hop (Humulus lupulus L.) essential oil from four different varieties. J. Agric. Food Chem. 2007, 55, 6252–6261. [Google Scholar] [CrossRef]
- Högnadóttir, Á.; Rouseff, R.L. Identification of aroma active compounds in orange essence oil using gas chromatography-olfactometry and gas chromatography-mass spectrometry. J. Chromatogr. A 2003, 998, 201–211. [Google Scholar] [CrossRef]
- Fernando, L.N.; Grun, I.U. Headspace-SPME analysis of volatiles of the ridge gourd (Luffa acutangula) and bitter gourd (Momordica charantia) flowers. Flavour Frag. J. 2001, 16, 289–293. [Google Scholar] [CrossRef]
- Shang, C.; Hu, Y.; Deng, C.; Hu, K. Rapid determination of volatile constituents of Michelia alba flowers by gas chromatography-mass spectrometry with solid-phase microextraction. J. Chromatogr. A 2002, 942, 283–288. [Google Scholar] [CrossRef]
- Deng, C.; Song, G.; Hu, Y. Rapid determination of volatile compounds emitted from Chimonanthus praecox flowers by HS-SPME-GC-MS. Z. Naturforsch C 2004, 59, 636–640. [Google Scholar]
- Kishimoto, T.; Wanikawa, A.; Kono, K.; Shibata, K. Comparison of the odor-active compounds in unhopped beer and beers hopped with different hop varieties. J. Agric. Food Chem. 2006, 54, 8855–8861. [Google Scholar]
- Lin, S.Y.; Roan, S.F.; Lee, C.L.; Chen, I.Z. Volatile organic components of fresh leaves as indicators of indigenous and cultivated Citrus Species in Taiwan. Biosci. Biotechnol. Biochem. 2010, 74, 806–811. [Google Scholar]
- Joshi, R.; Saini, R.; Guleria, S.; Babu, G.D.; Kumari, M.; Gulati, A. Characterization of volatile components of tea flowers (Camellia sinensis) growing in Kangra by GC/MS. Nat. Prod. Commun. 2011, 6, 1155–1158. [Google Scholar]
- Tibaldi, G.; Fontana, E.; Nicola, S. Growing conditions and postharvest management can affect the essential oil of Origanum vulgare L. ssp. hirtum (Link) Ietswaart. Ind. Crops Prod. 2011, 34, 1516–1522. [Google Scholar] [CrossRef]
- Seo, W.H.; Baek, H.H. Identification of characteristic aroma-active compounds from water dropwort (Oenanthe javanica DC). J. Agric. Food Chem. 2005, 53, 6766–6770. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Stoilova, I. Chemical composition and antioxidant properties of clove leaf. J. Agric. Food Chem. 2006, 54, 6303–6307. [Google Scholar]
- Zhao, Y.P.; Wang, L.; Li, J.M.; Pei, G.R.; Liu, Q.S. Comparison of volatile compounds in two brandies using HS-SPME coupled with GC-O, GC-MS and sensory evaluation. S. Afr. J. Enol. Vitic. 2011, 32, 9–20. [Google Scholar]
- Tamura, H.; Boonbumrung, S.; Yoshizawa, T.; Varanyanond, W. The volatile constituents in the peel and pulp of a green thai mango, khieo sawoei cultivar (Mangifera indica L.). Food Sci. Technol. Res. 2001, 7, 72–77. [Google Scholar] [CrossRef]
- Arctander, S. Perfume and Flavor Chemicals; Arctander, S.: Elizabeth, NJ, USA, 1969; pp. 1369–1370. [Google Scholar]
- Borg-Karlson, A.-K.; Tengö, J.; Valterová, I.; Unelius, C.-R.; Taghizadeh, T.; Tolasch, T.; Francke, W. (S)-(+)-linalool, a mate attractant pheromone component in the bee Colletes cunicularius. J. Chem. Ecol. 2003, 29, 1–14. [Google Scholar]
- Thies, N. Fragrance of canada thistle (Cirsium arvense) attracts both floral herbivores and pollinators. J. Chem. Ecol. 2006, 32, 917–927. [Google Scholar] [CrossRef]
- Dötterl, S.; Jahreiß, K.; Jhumur, U.S.; Jürgens, A. Temporal variation of flower scent in Silene otites (Caryophyllaceae) a species with a mixed pollination system. Bot. J. Linn. Soc. 2012, 169, 447–460. [Google Scholar]
- Kolosova, N.; Sherman, D.; Karlson, D.; Dudareva, N. Cellular and subcellular localization of S-adenosyl-l-methionine-benzoic acid carboxyl methyltransferase, the enzyme responsible for biosynthesis of the volatile ester methylbenzoate in snapdragon flowers. Plant Physiol. 2001, 126, 956–964. [Google Scholar]
- Dudareva, N.; Pichersky, E. Biochemical and molecular genetic aspects of floral scents. Plant Physiol. 2000, 122, 627–633. [Google Scholar]
- Hsiao, Y.Y.; Jeng, M.F.; Tsai, W.C.; Chuang, Y.C.; Li, C.Y.; Wu, T.S.; Kuoh, C.S.; Chen, W.H.; Chen, H.H. A novel homodimeric geranyl diphosphate synthase from the orchid Phalaenopsis bellina lacking a DD(X)2-4D motif. Plant J. 2008, 55, 719–733. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X.-L.; Wang, Y.; Tian, M.; Fan, M.-H. Changes of aroma components in oncidium sharry baby in different florescence and flower parts. Sci. Agric. Sin. 2011, 44, 110–117. [Google Scholar]
- Rusanov, K.; Kovacheva, N.; Rusanova, M.; Atanassov, I. Traditional Rosa damascena flower harvesting practices evaluated through GC/MS metabolite profiling of flower volatiles. Food Chem. 2011, 129, 1851–1859. [Google Scholar] [CrossRef]
- Chen, H.C.; Chi, H.S.; Lin, L.Y. Headspace solid-phase microextraction analysis of volatile components in Narcissus tazetta var. chinensis Roem. Molecules 2013, 18, 13723–13734. [Google Scholar] [CrossRef]
- Kilic, A.; Hafizoglu, H.; Kollmannsberger, H.; Nitz, S. Volatile constituents and key odorants in leaves, buds, flowers, and fruits of Laurus nobilis L. J. Agric. Food Chem. 2004, 52, 1601–1606. [Google Scholar] [CrossRef]
- Dobson, H.E.M.; Bergström, G.; Groth, I. Differences in fragrance chemistry between flower parts of Rosa rugosa Thunb. (Rosaceae). Isr. J. Bot. 1990, 39, 143–156. [Google Scholar]
- Schomburg, G.; Dielmann, G. Identification by means of retention parameters. J. Chromatogr. Sci. 1973, 11, 151–159. [Google Scholar]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yeh, C.-H.; Tsai, W.-Y.; Chiang, H.-M.; Wu, C.-S.; Lee, Y.-I.; Lin, L.-Y.; Chen, H.-C. Headspace Solid-Phase Microextraction Analysis of Volatile Components in Phalaenopsis Nobby’s Pacific Sunset. Molecules 2014, 19, 14080-14093. https://doi.org/10.3390/molecules190914080
Yeh C-H, Tsai W-Y, Chiang H-M, Wu C-S, Lee Y-I, Lin L-Y, Chen H-C. Headspace Solid-Phase Microextraction Analysis of Volatile Components in Phalaenopsis Nobby’s Pacific Sunset. Molecules. 2014; 19(9):14080-14093. https://doi.org/10.3390/molecules190914080
Chicago/Turabian StyleYeh, Chih-Hsin, Wan-Yu Tsai, Hsiu-Mei Chiang, Chin-Sheng Wu, Yung-I Lee, Li-Yun Lin, and Hsin-Chun Chen. 2014. "Headspace Solid-Phase Microextraction Analysis of Volatile Components in Phalaenopsis Nobby’s Pacific Sunset" Molecules 19, no. 9: 14080-14093. https://doi.org/10.3390/molecules190914080







