Isolation and Characterization of an α-Glucosidase Inhibitor from Musa spp. (Baxijiao) Flowers
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of α-Glucosidase Inhibitors
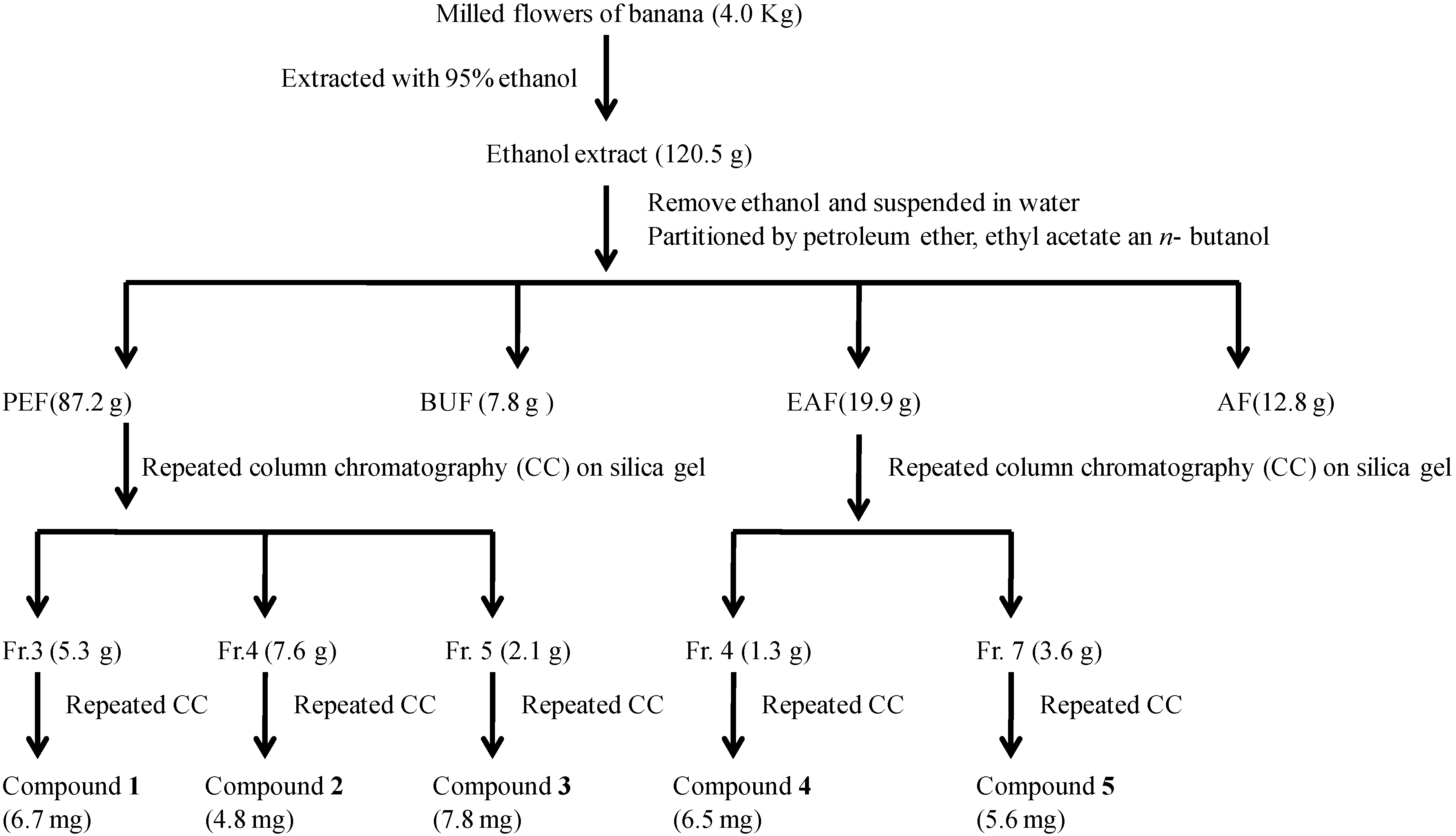
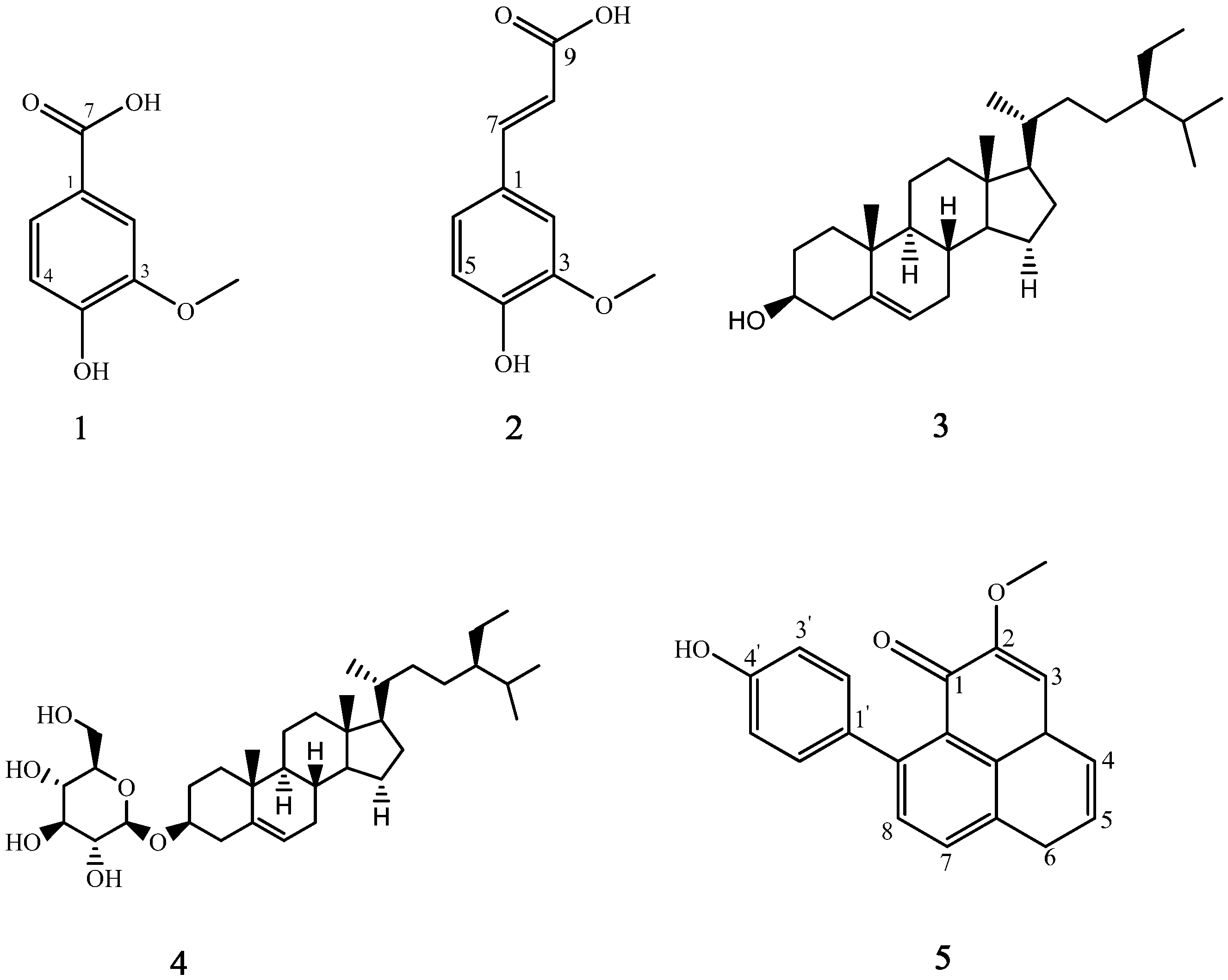
2.2. Structural Elucidation of Isolated Compounds
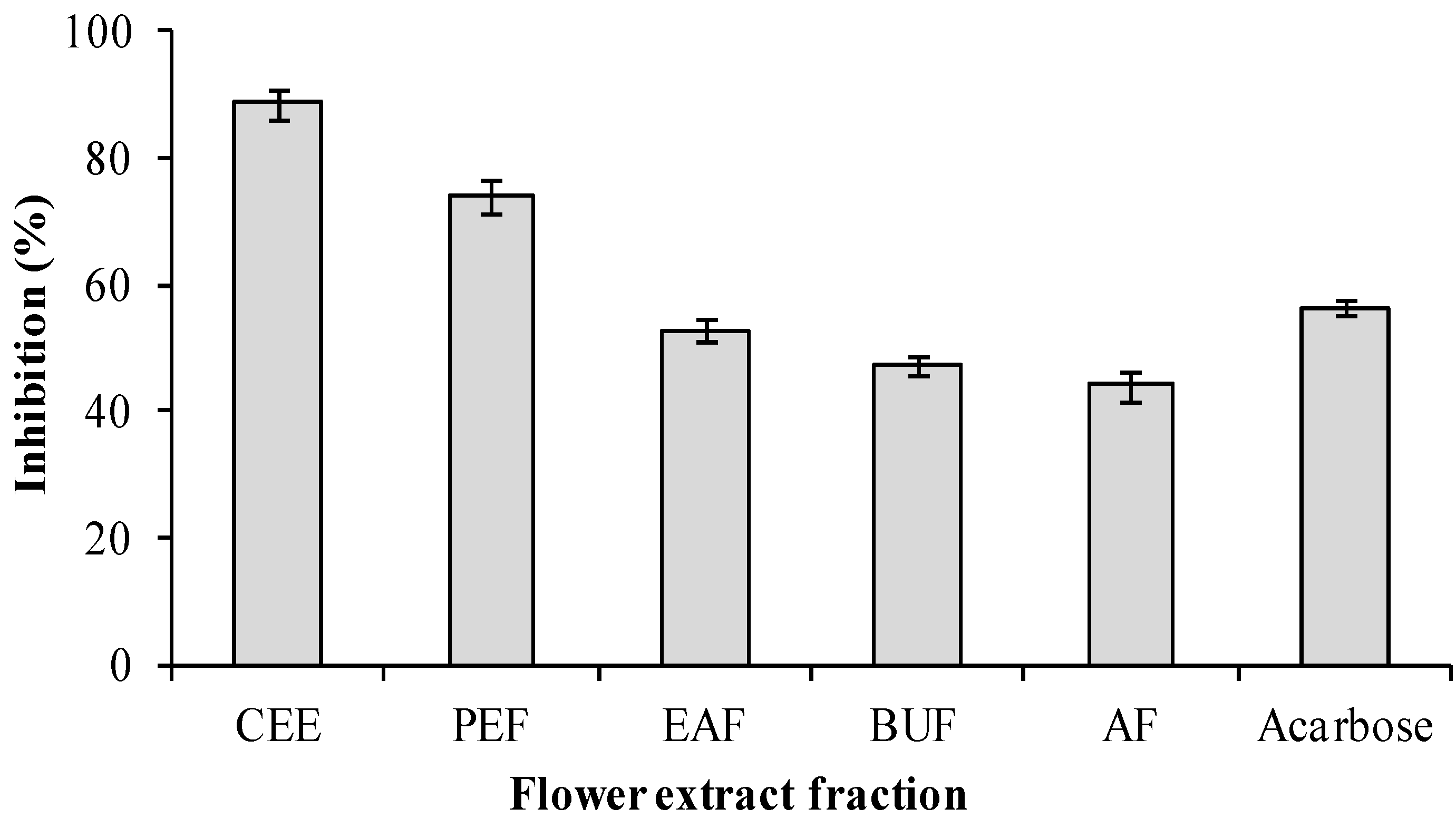
2.3. α-glucosidase Inhibitory Activity of Extracts Fractionated from Flowers of Musa spp. (Baxijiao)
2.4. Evaluation of α-Glucosidase Inhibitory Activity
| Test Sample | Content (mg/L) | Maximum Inhibition (%) | IC50 (mg/L) |
|---|---|---|---|
| 1 | 1500 | 18.29 | 2004.58 |
| 2 | 1500 | 42.38 | 1258.35 |
| 3 | 1600 | 98.67 | 283.67 |
| 4 | 1000 | 99.18 | 247.35 |
| 5 | 400 | 101.24 | 3.86 |
| Acarbose | 1500 | 56.45 | 999.31 |
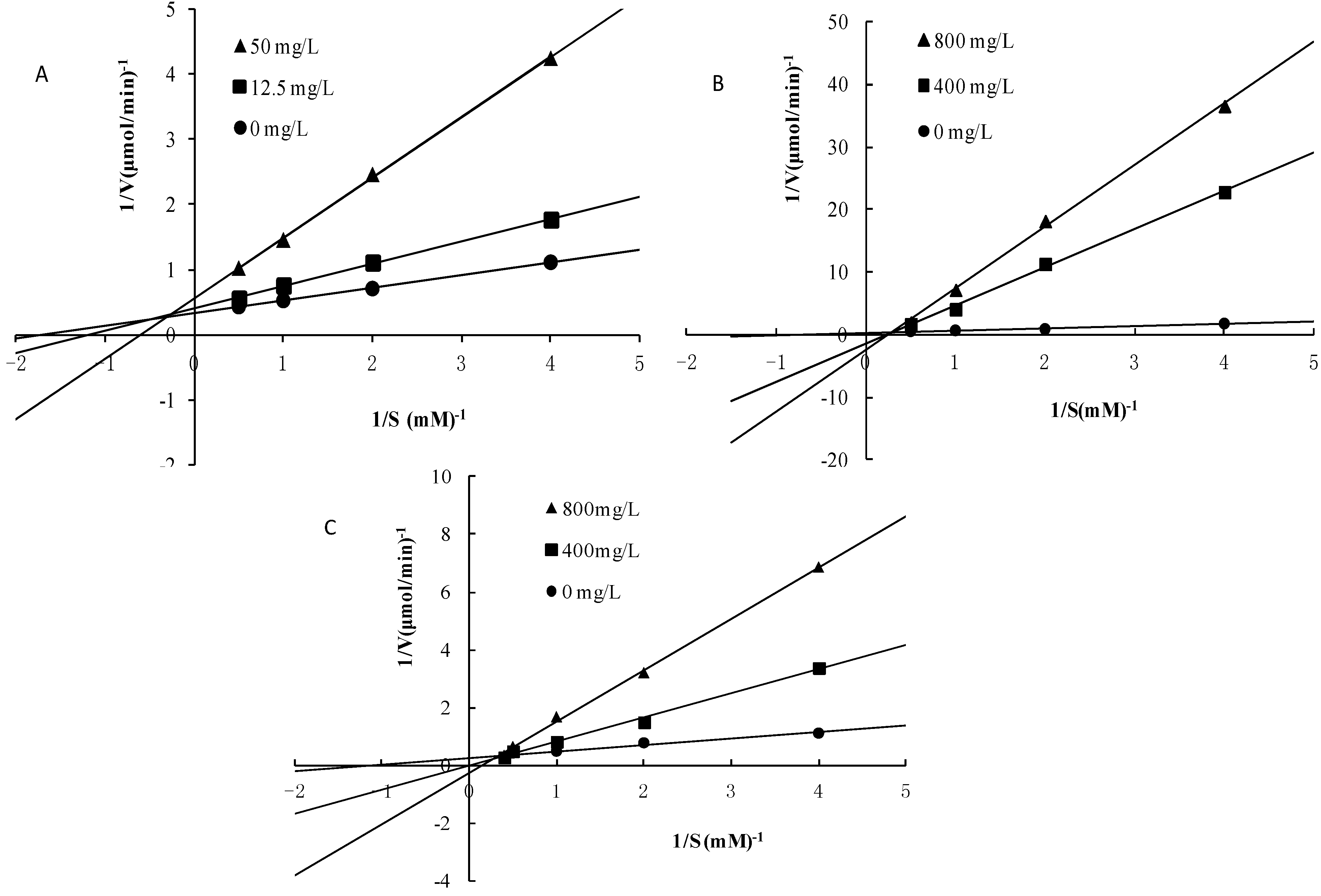
2.5. Mode of Inhibition of α-Glucosidase for compounds 3, 4 and 5
3. Experimental
3.1. General Procedures and Reagents
3.2. Plant Materials
3.3. Extraction and Isolation of α-Glucosidase Inhibitors
3.4. Spectroscopic Data
3.5. α-Glucosidase Inhibitory Activity Assay
3.6. Determination of the Inhibitory Mode of Action of the Active Compounds
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tewari, N.; Tiwari, V.K.; Mishra, R.C.; Tripathi, R.P.; Srivastava, A.K.; Ahmad, R.; Srivastava, R.; Srivastava, B.S. Synthesis and bioevaluation of glycosyl ureas as α-glucosidase inhibitors and their effect on mycobacterium. Bioorg. Med. Chem. 2003, 11, 2911–2922. [Google Scholar] [CrossRef]
- Kimura, A.; Lee, J.-H.; Lee, I.-S.; Lee, H.-S.; Park, K.-H.; Chiba, S.; Kim, D. Two potent competitive inhibitors discriminating α-glucosidase family I from family II. Carbohyd. Res. 2004, 339, 1035–1040. [Google Scholar] [CrossRef]
- Baig, I. Phytochemical Studies on Ferula Mongolica and Other Mongolian Medicinal Plants. Ph.D. Thesis, University of Karachi, Karachi, Sindh, Pakistan, 2002. [Google Scholar]
- Wang, Z.B.; Jiang, H.; Xia, Y.G.; Yang, B.Y.; Kuang, H.X. α-Glucosidase Inhibitory Constituents from Acanthopanax senticosus Harm Leaves. Molecules 2012, 17, 6269–6276. [Google Scholar] [CrossRef]
- Chen, Y.G.; Li, P.; Li, P.; Yan, R.; Zhang, X.Q.; Wang, Y.; Zhang, X.T.; Ye, W.C.; Zhang, Q.W. α-Glucosidase Inhibitory Effect and Simultaneous Quantification of Three Major Flavonoid Glycosides in Microctis folium. Molecules 2013, 18, 4221–4232. [Google Scholar] [CrossRef]
- Gao, J.; Xu, P.; Wang, Y.; Wang, Y.; Hochstetter, D. Combined Effects of Green Tea Extracts, Green Tea Polyphenols or Epigallocatechin Gallate with Acarbose on Inhibition against α-Amylase and α-Glucosidase in vitro. Molecules 2013, 18, 11614–11623. [Google Scholar] [CrossRef]
- Bhaskar, J.J.; Shobha, M.S.; Sambaiah, K.; Salimath, P.V. Beneficial effects of banana (Musa sp. var. elakki bale) flower and pseudostem on hyperglycemia and advanced glycation end-products (AGEs) in streptozotocin-induced diabetic rats. J. Physiol. Biochem. 2011, 67, 415–425. [Google Scholar] [CrossRef]
- Bagavan, A.; Rahuman, A.A.; Kaushik, N.K.; Sahal, D. In vitro antimalarial activity of medicinal plant extracts against plasmodium falciparum. Parasitol. Res. 2011, 108, 15–22. [Google Scholar] [CrossRef]
- Bhaskar, J.J.; Salimath, P.V. Effect of banana (Musa sp. var. elakki bale) flower and pseudostem on antioxidant and lysosomal enzyme activities in streptozotocin-induced diabetic rats. J. Pharm. Res. 2011, 3, 415–425. [Google Scholar]
- Calixtro, R.S., Jr.; Malalay, A.P.; Epino, P.B.; Avelino, L.E. Woound hearing potential to the ethanolic extract of banana flower (Musa sapientum, BBB'Saba', Family Musaceae). Int. J. Pharm. 2014, 4, 33–37. [Google Scholar]
- Sheng, Z.W.; Ma, W.H.; Jin, Z.Q.; Bi, Y.; Sun, Z.G.; Dou, H.T.; Li, J.Y.; Han, L.N. Investigation of dietary fiber, protein, vitamin E and other nutritional compounds of banana flower of two cultivars grown in China. Afr. J. Biotechnol. 2010, 9, 3888–3895. [Google Scholar]
- Sheng, Z.W.; Ma, W.H.; Gao, J.H.; Bi, Y.; Zhang, W.M.; Dou, H.T.; Jin, Z.Q. Antioxidant properties of banana flower of two cultivars in China using 2,2-diphenyl-1-picrylhydrazyl (DPPH,) reducing power, 2,2ꞌ-azinobis-(3-ethylbenzthiazoline-6-sulphonate (ABTS) and inhibition of lipid peroxidation assays. Afr. J. Biotechnol. 2011, 10, 4470–4477. [Google Scholar]
- Tsunashi, K.; Nagomi, K.; Nobuhiro, H.; Mitsuya, T.; Daie, F.; Hajime, O. Phenylphenalenone-type phytoalexins from unripe bungulan banana fruit. Biosci. Biotech. Bioch. 1998, 62, 95–101. [Google Scholar] [CrossRef]
- Tabussum, A.; Riaz, N.; Saleem, M.; Ashraf, M.; Ahmad, M.; Alam, U.; Jabeen, B.; Malik, A.; Jabbar, A. α-Glucosidase inhibitory constituents from Chrozophora plicata. Phytochem. Lett. 2013, 6, 614–619. [Google Scholar] [CrossRef]
- Mbaze, L.M.; Poumale, H.M.P.; Wansi, J.D.; Lado, J.A.; Khan, S.N.; Iqbal, M.C.; Ngadjui, B.T.; Laatsch, H. α-Glucosidase inhibitory pentacyclic triterpenes from the stem bark of Fagara tessmannii (Rutaceae). Phytochemistry 2007, 68, 591–595. [Google Scholar] [CrossRef]
- González-Baró, A.C.; Parajón-Costa, B.S.; Franca, C.A.; Pis-Diez, R. Theoretical and spectroscopic study of vanillic acid. J. Mol. Struct. 2008, 889, 204–210. [Google Scholar] [CrossRef]
- Kalinowska, M.; Piekut, J.; Bruss, A.; Follet, C.; Sienkiewicz-Gromiuk, J.; Świsłocka, R.; Rzączyńska, Z.; Lewandowski, W. Spectroscopic (FT-IR, FT-Raman, 1H, 13C NMR, UV/VIS), thermogravimetric and antimicrobial studies of Ca(II), Mn(II), Cu(II), Zn(II) and Cd(II) complexes of ferulic acid. Spectrochim. ActaA 2014, 122, 631–638. [Google Scholar] [CrossRef]
- De-Eknamkul, W.; Potduang, B. Biosynthesis of β-sitosterol and stigmasterol in Croton sublyratus proceeds via a mixed origin of isoprene units. Phytochemistry 2003, 62, 389–398. [Google Scholar] [CrossRef]
- Kojima, H.; Sato, N.; Hatano, A.; Ogura, H. Sterol glucosides from Prunella vulgaris. Phytochemistry 1990, 29, 2351–2355. [Google Scholar] [CrossRef]
- Phan, M.A.T.; Wang, J.; Tang, J.; Lee, Y.Z.; Ng, K. Evaluation of α-glucosidase inhibition potential of some flavonoids from Epimedium brevicornum. LWT-Food Sci. Technol. 2013, 53, 492–498. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–4 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sheng, Z.; Dai, H.; Pan, S.; Wang, H.; Hu, Y.; Ma, W. Isolation and Characterization of an α-Glucosidase Inhibitor from Musa spp. (Baxijiao) Flowers. Molecules 2014, 19, 10563-10573. https://doi.org/10.3390/molecules190710563
Sheng Z, Dai H, Pan S, Wang H, Hu Y, Ma W. Isolation and Characterization of an α-Glucosidase Inhibitor from Musa spp. (Baxijiao) Flowers. Molecules. 2014; 19(7):10563-10573. https://doi.org/10.3390/molecules190710563
Chicago/Turabian StyleSheng, Zhanwu, Haofu Dai, Siyi Pan, Hui Wang, Yingying Hu, and Weihong Ma. 2014. "Isolation and Characterization of an α-Glucosidase Inhibitor from Musa spp. (Baxijiao) Flowers" Molecules 19, no. 7: 10563-10573. https://doi.org/10.3390/molecules190710563
APA StyleSheng, Z., Dai, H., Pan, S., Wang, H., Hu, Y., & Ma, W. (2014). Isolation and Characterization of an α-Glucosidase Inhibitor from Musa spp. (Baxijiao) Flowers. Molecules, 19(7), 10563-10573. https://doi.org/10.3390/molecules190710563





