Antioxidant, Antimicrobial and Phytochemical Variations in Thirteen Moringa oleifera Lam. Cultivars
Abstract
:1. Introduction
2. Results and Discussion
2.1. DPPH Radical Scavenging Activity
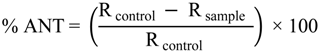
2.2. β-Carotene-Linoleic Acid Model System (CLAMS) Activity
| Cultivar | Cultivar Origin | Antioxidant Activity | ||
|---|---|---|---|---|
| DPPH Scavenging ActivityEC50 (µg/mL) | ANT (%) | ORR | ||
| TOT4880 | USA | 16.70 ± 0.00 a | 37.46 ± 2.35 a | 0.73 ± 0.02 e |
| TOT4977 | AVRDC | 20.91 ± 0.00 ab | 87.71 ± 1.81 efg | 0.02 ± 0.00 a |
| TOT5077 | AVRDC | 22.45 ± 1.70 ab | 44.40 ± 1.66 ab | 0.51 ± 0.03 cde |
| TOT5028 | AVRDC | 16.40 ± 3.04 a | 98.00 ± 0.16 g | 0.02 ± 0.00 a |
| TOT5169 | AVRDC | 26.88 ± 2.30 bc | 83.50 ± 0.33 de | 0.16 ± 0.13 ab |
| SH | Silver Hill (SA) | 23.96 ± 1.41 abc | 94.83 ± 1.77 fg | 0.05 ± 0.02 a |
| TOT4893 | AVRDC | 20.06 ± 4.29 ab | 87.71 ± 0.50 efg | 0.02 ± 0.01 a |
| CHM | Silver Hill (SA) | 32.56 ± 6.57 c | 94.04 ± 0.10 efg | 0.06 ± 0.00 ab |
| TOT5330 | AVRDC | 22.00 ± 2.58 ab | 96.49 ± 1.16 g | 0.72 ± 0.45 e |
| TOT7266 | AVRDC | 16.54 ± 2.72 a | 63.42 ± 1.83 bc | 0.27 ± 0.08 bc |
| TOT4100 | Taiwan | 16.78 ± 1.50 a | 55.83 ± 1.01 ab | 0.39 ± 0.06 cd |
| TOT4951 | AVRDC | 14.57 ± 0.65 a | 86.02 ± 0.12 ef | 0.24 ± 0.13 ab |
| Limpopo | Limpopo (SA) | 21.44 ± 1.08 ab | 73.59 ± 0.69 cd | 0.46 ± 0.21 de |
| Ascorbic acid | 71.11 ± 0.01 d | 81.45 ± 1.72de | 0.19 ± 0.02 ab | |
2.3. Ferric-Reducing Power Assay Activity
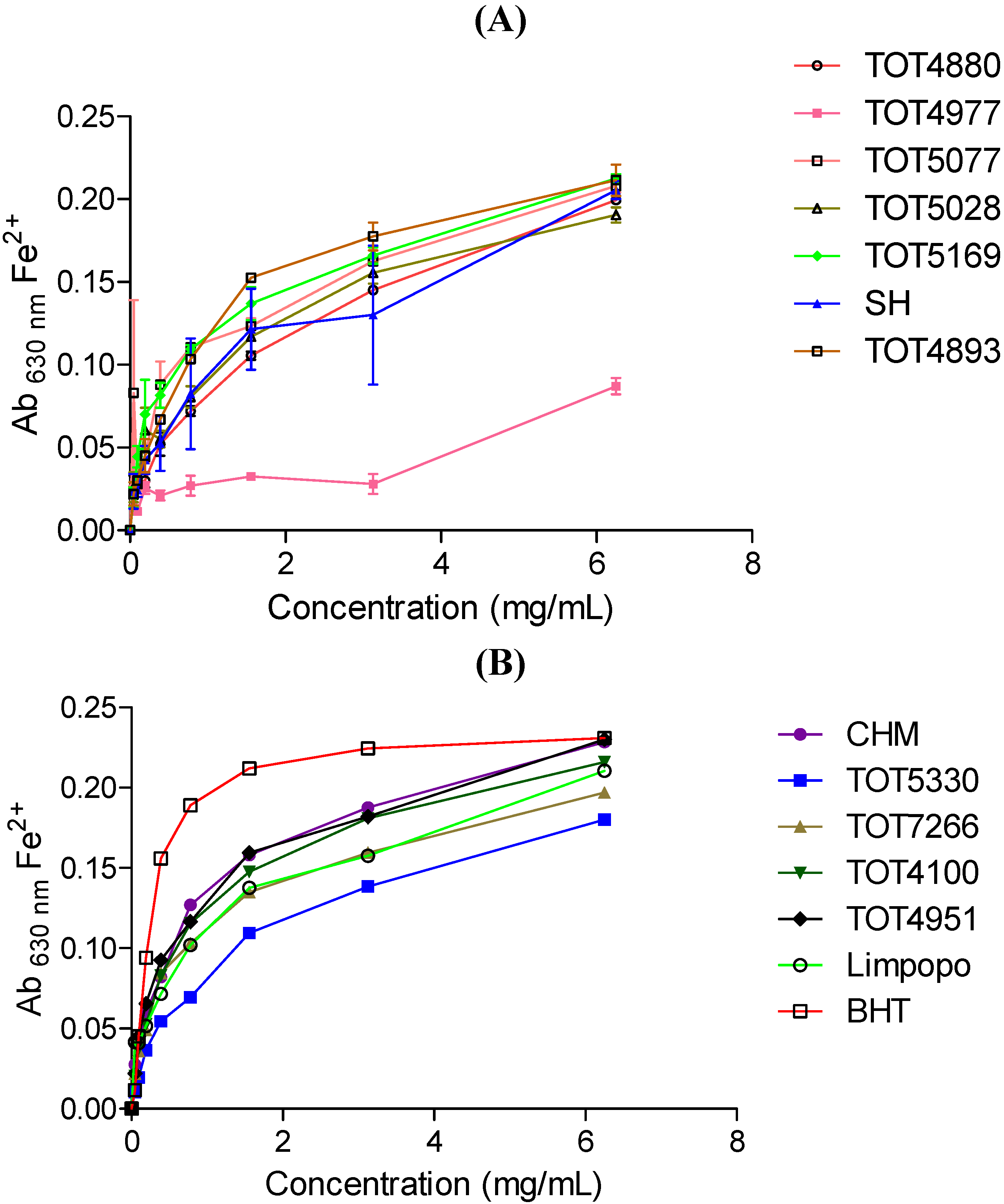
2.4. Antimicrobial Activity
| Solvent | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Acetone | EtOH | Water | |||||||
| Microorganism | |||||||||
| Variety | K.p. | S.a. | C.a. | K.p. | S.a. | C.a. | K.p. | S.a. | C.a. |
| TOT4880 | 0.78 | 3.125 | 3.125 | 0.78 | 1.56 | 3.125 | 1.56 | 6.25 | 6.25 |
| TOT4977 | 0.78 | 1.56 | 3.125 | 1.56 | 1.56 | 3.125 | 1.56 | 6.25 | 6.25 |
| TOT5077 | 0.78 | 3.125 | 3.125 | 0.78 | 0.78 | 3.125 | 1.56 | 6.25 | 6.25 |
| TOT5028 | 0.78 | 3.125 | 3.125 | 1.56 | 1.56 | 3.125 | 1.56 | 6.25 | >6.25 |
| TOT5169 | 0.78 | 3.125 | 3.125 | 1.56 | 1.56 | 3.125 | 1.56 | 6.25 | 6.25 |
| SH | 0.78 | 3.125 | 3.125 | 1.56 | 1.56 | 3.125 | 1.56 | 6.25 | 6.25 |
| TOT4893 | 0.78 | 1.56 | 3.125 | 1.56 | 1.56 | 6.25 | 1.56 | >6.25 | 3.125 |
| CHM | 0.78 | 3.125 | 3.125 | 0.78 | 1.56 | 6.25 | 1.56 | 6.25 | 6.25 |
| TOT5330 | 0.78 | 3.125 | 3.125 | 1.56 | 1.56 | 6.25 | 1.56 | 6.25 | 3.125 |
| TOT7266 | 0.78 | 3.125 | 3.125 | 1.56 | 1.56 | 6.25 | 1.56 | 6.25 | 3.125 |
| TOT4100 | 0.78 | 3.125 | 3.125 | 1.56 | 1.56 | 6.25 | 1.56 | 6.25 | 3.125 |
| TOT4951 | 0.78 | 3.125 | 3.125 | 1.56 | 0.78 | 3.125 | 6.25 | 6.25 | 3.125 |
| Limpopo | 0.78 | 1.56 | 1.56 | 1.56 | 1.56 | 3.125 | 6.25 | 3.125 | 3.125 |
2.5. Total Phenolics and Flavonoid Content

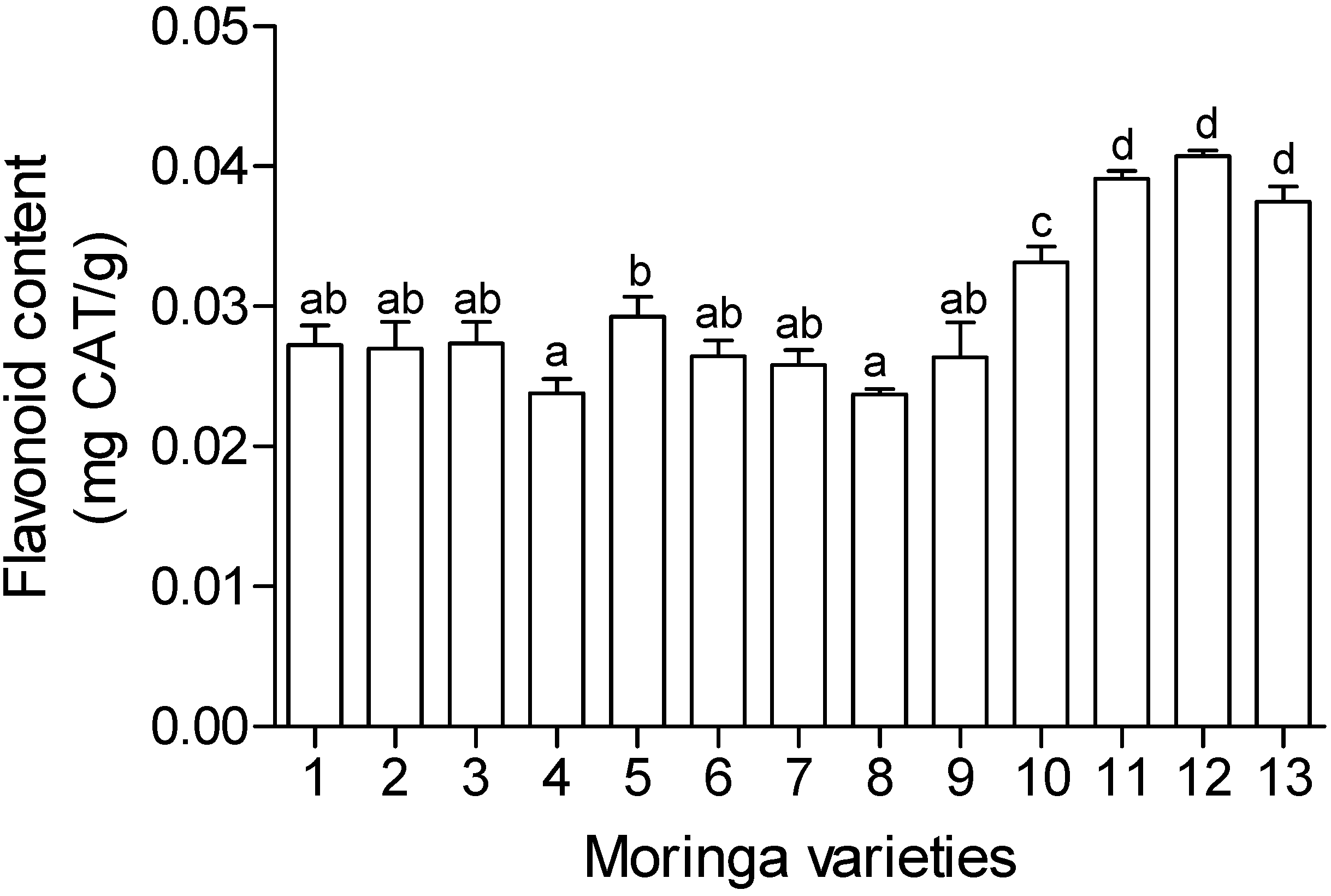
3. Experimental
3.1. General
3.2. Sample Preparation
3.3. Bioassays
3.3.1. DPPH Radical Scavenging Activity
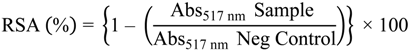
3.3.2. Ferric-Reducing Power Assay
3.3.3. β-Carotene-Linoleic Acid Model System (CLAMS)
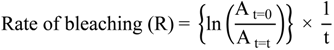
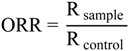
3.3.4. Antibacterial Microdilution Assay
3.3.5. Antifungal Microdilution Bioassay
3.3.6. Determination of Total Phenolics and Flavonoids
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nafiu, M.O.; Salawu, M.O.; Kazeem, M.I. Antioxidant Activity of African Medicinal Plants. In Medicinal Plant Research in Africa; Kuete, V., Ed.; Elservier: London, UK, 2013. [Google Scholar]
- Révész, D.; Verhoeven, J.E.; Milaneschi, Y.; de Geus, E.J.C.N.; Wolkowitz, O.M.; Penninx, B.W.J.H. Dysregulated physiological stress systems and accelerated cellular ageing. Neurobiol. Ageing 2014, 35, 1422–1430. [Google Scholar] [CrossRef]
- Katalinic, V.; Modun, D.; Music, I.; Boban, M. Gender differences in antioxidant capacity of rat tissues determined by 2,2-azinobis (3-ethylbenzothiazoline 6-sulfonate; ABTS) and ferric reducing antioxidant power (FRAP) assays. Comp. Biochem. Physiol. 2005, 140, 47–52. [Google Scholar]
- Orhan, I.; Kartal, M.; Sezer, M.; Şenol, F.S.; Yilmaz, G.; Şener, B.S. Free radical scavenging properties and phenolic characterization of some edible plants. Food Chem. 2009, 114, 276–281. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. The antioxidant and free radical scavenging activities of processed cowpea (Vigna unguiculata (L.) Walp.) seed extracts. Food Chem. 2007, 101, 10–19. [Google Scholar] [CrossRef]
- Lorencini, M.; Brohem, C.A.; Dieamant, G.C.; Zanchin, N.I.T.; Maibach, H.I. Active ingredients against human epidermal ageing. Ageing Res. Rev. 2014, 15, 100–115. [Google Scholar]
- Singh, R.S.G.; Negi, P.S.; Radha, C. Phenolic composition, antioxidant and antimicrobial activities of free and bound phenolic extracts of Moringa oleifera seed flour. J. Funct. Foods 2013, 5, 1883–1891. [Google Scholar] [CrossRef]
- Radovich, T. Farm and Forestry Production and Marketing Profile for Moringa (Moringa oleifera). Specialty Crops for Pacific Island Agroforestry; Elevitch, C.R., Ed.; Permanent Agriculture Resources (PAR): Holualoa, HI, USA, 2011. Available online: http://agroforestry.net/scps (accessed on 14 April 2014).
- Faizi, S.; Siddiqui, B.S.; Saleem, R.; Siddiqui, S.; Aftab, K. Isolation and structure elucidation of new nitrile and mustard oil glycosides from Moringa oleifera and their effect on blood pressure. J. Nat. Prod. 1994, 57, 1256–1261. [Google Scholar] [CrossRef]
- Faizi, S.; Siddiqui, B.S.; Saleem, R.; Siddiqui, S.; Aftab, K.; Gilani, A.H. Fully acetylated carbamate and hypotensive thiocarbamate glycosides from Moringa oleifera. Phytochemistry 1995, 38, 957–963. [Google Scholar]
- Parrotta, J.A. Healing Plants of Peninsular India; CAB International: Wallingford, NY, USA, 2001. [Google Scholar]
- Mekonnen, Y. The multi-purpose Moringa tree: Ethiopia. In Examples of the Development of Pharmaceutical Products from Medicinal Plants; Mekonnen, Y., Ed.; Research and Publication Office, Addis Ababa University: Addis Ababa, Ethiopia, 2002; Volume 10, pp. 111–118. [Google Scholar]
- Atoui, A.K.; Mansouri, A.; Boskou, G.; Kefalas, P. Tea and herbal infusions: Their antioxidant activity and phenolic profile. Food Chem. 2005, 89, 27–36. [Google Scholar] [CrossRef]
- Steenkamp, P.A.; Harding, N.M.; van Heerden, F.R.; van Wyk, B.-E. Identification of atractyloside by LC–ESI–MS in alleged herbal poisonings. Forensic. Sci. Int. 2006, 163, 81–92. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in food and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Fabry, W.; Okemo, P.O.; Ansorg, R. Antibacterial activity of East African medicinal plants. J. Ethnopharmacol. 1998, 60, 79–84. [Google Scholar] [CrossRef]
- Arnold, R.S.; Thom, K.A.; Sharma, S.; Phillips, M.; Johnson, J.K.; Morgan, D.J. Emergence of Klebsiella pneumoniae Carbapenemase (KPC)-Producing Bacteria. South Med. J. 2011, 104, 40–45. [Google Scholar] [CrossRef]
- Yigit, H.; Queenan, A.M.; Anderson, G.J.; Domenech-Sanchez, J.W.; Biddle, C.D.; Steward, S.; Alberti, K.; Bush, F.C. Novel carbapenem-hydrolyzing beta-lactamase KPC-1 from a carbapenem-resistant strain of Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2001, 45, 1151–1161. [Google Scholar] [CrossRef]
- Bennett, R.W.; Yeterian, M.; Smith, W.; Coles, C.M.; Sassaman, M.; McClure, F.D. Staphylococcus aureus identification characteristics and enterotoxigenicity. J. Food Sci. 1986, 51, 1337–1339. [Google Scholar] [CrossRef]
- Singer, A.J.; Talan, D.A. Management of skin abscesses in the era of Methicillin-Resistant Staphylococcus aureus. N. Engl. J. Med. 2014, 370, 1039–1047. [Google Scholar]
- Liu, C.; Bayer, A.; Cosgrove, S.E.; Daum, R.S.; Fridkin, S.K.; Gorwitz, R.J.; Kaplan, S.L.; Karchmer, A.W.; Levine, D.P.; Murray, B.E.; et al. Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-Resistant Staphylococcus aureus Infections in Adults and Children: Executive Summary. Clin. Infect. Dis. 2011, 52, 285–292. [Google Scholar] [CrossRef]
- Bourgaud, F.; Gravot, A.; Milesi, S.; Gontier, E. Production of plant secondary metabolites: A historical perspective. Plant Sci. 2001, 161, 839–851. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Siddhuraju, P.; Becker, K. Plant Secondary Metabolites; Humana Press: Totowa, NJ, USA, 2007. [Google Scholar]
- Bruneton, J. Pharmacognosy, Phytochemistry, Medicinal Plants; Intercept Ltd.: Hampshire, UK, 1995. [Google Scholar]
- Luseba, D.; Elgorashi, E.E.; Ntloedibe, D.T.; van Staden, J. Antibacterial, anti-inflammatory and mutagenic effects of some medicinal plants used in South Africa for treatment of wounds and retained placenta in livestock. S. Afr. J. Bot. 2007, 73, 378–383. [Google Scholar] [CrossRef]
- Mulaudzi, R.B.; Ndhlala, A.R.; Kulkarni, M.G.; van Staden, J. Pharmacological properties and protein binding capacity of phenolic extracts of some Venda medicinal plants used against cough and fever. J. Ethnopharmacol. 2012, 143, 185–193. [Google Scholar] [CrossRef]
- Karioti, A.; Hadjipavlou-Litina, D.; Mensah, M.L.K.; Fleischer, T.C.; Saltsa, H. Composition and antioxidant activity of the essential oils of Xylopia aethiopica (Dun) A. Rich. (Annonaceae) leaves, stem bark, root bark, and fresh and dried fruits, growing in Ghana. J. Agric. Food Chem. 2004, 52, 8094–8098. [Google Scholar]
- Lim, T.Y.; Lim, Y.Y.; Yule, C.M. Evaluation of antioxidant, antibacterial and anti-tyrosinase activities of four Macaranga species. Food Chem. 2009, 114, 594–599. [Google Scholar] [CrossRef]
- Amarowicz, R.; Pegg, R.B.; Rahimi-Moghaddam, P.; Barl, B.; Weil, J.A. Free radical scavenging capacity and antioxidant activity of selected plant species from the Canadian prairies. Food Chem. 2004, 84, 551–562. [Google Scholar] [CrossRef]
- Eloff, J.N. A sensitive and quick microplate method to determine the minimal inhibitory concentration of plant extracts for bacteria. Planta Med. 1998, 64, 711–713. [Google Scholar] [CrossRef]
- Masoko, P.; Picard, J.; Eloff, J.N. The antifungal activity of twenty-four southern Africa Combretum species (Combretaceae). S. Afr. J. Bot. 2007, 73, 173–183. [Google Scholar] [CrossRef]
- Makkar, H.P.S. Quantification of Tannins in Tree Foliage: A Laboratory Manual for the FAO/IAEA Co-Ordinated Research Project on Use of Nuclear and Related Techniques to Develop Simple Tannin Assay for Predicting and Improving the Safety and Efficiency of Feeding Ruminants on the Tanniniferous Tree Foliage; Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture: Vienna, Austria, 1999. [Google Scholar]
- Ndhlala, A.R.; Kasiyamhuru, A.; Mupure, C.; Chitindingu, K.; Benhura, M.A.; Muchuweti, M. Phenolic composition of Flacourtia indica, Opuntia megacantha and Sclerocarya birrea. Food Chem. 2007, 103, 82–87. [Google Scholar] [CrossRef]
- Hagerman, A.E. Tannin Chemistry; Miami University Press: Miami, FL, USA, 2002. [Google Scholar]
- Sample availability: Samples of the thirteen Moringa oleifera Lam., cultivars are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ndhlala, A.R.; Mulaudzi, R.; Ncube, B.; Abdelgadir, H.A.; Du Plooy, C.P.; Van Staden, J. Antioxidant, Antimicrobial and Phytochemical Variations in Thirteen Moringa oleifera Lam. Cultivars. Molecules 2014, 19, 10480-10494. https://doi.org/10.3390/molecules190710480
Ndhlala AR, Mulaudzi R, Ncube B, Abdelgadir HA, Du Plooy CP, Van Staden J. Antioxidant, Antimicrobial and Phytochemical Variations in Thirteen Moringa oleifera Lam. Cultivars. Molecules. 2014; 19(7):10480-10494. https://doi.org/10.3390/molecules190710480
Chicago/Turabian StyleNdhlala, Ashwell R., Rofhiwa Mulaudzi, Bhekumthetho Ncube, Hafiz A. Abdelgadir, Christian P. Du Plooy, and Johannes Van Staden. 2014. "Antioxidant, Antimicrobial and Phytochemical Variations in Thirteen Moringa oleifera Lam. Cultivars" Molecules 19, no. 7: 10480-10494. https://doi.org/10.3390/molecules190710480
APA StyleNdhlala, A. R., Mulaudzi, R., Ncube, B., Abdelgadir, H. A., Du Plooy, C. P., & Van Staden, J. (2014). Antioxidant, Antimicrobial and Phytochemical Variations in Thirteen Moringa oleifera Lam. Cultivars. Molecules, 19(7), 10480-10494. https://doi.org/10.3390/molecules190710480




