Fucoidan Inhibits the Proliferation of Human Urinary Bladder Cancer T24 Cells by Blocking Cell Cycle Progression and Inducing Apoptosis
Abstract
:1. Introduction
2. Results and Discussion
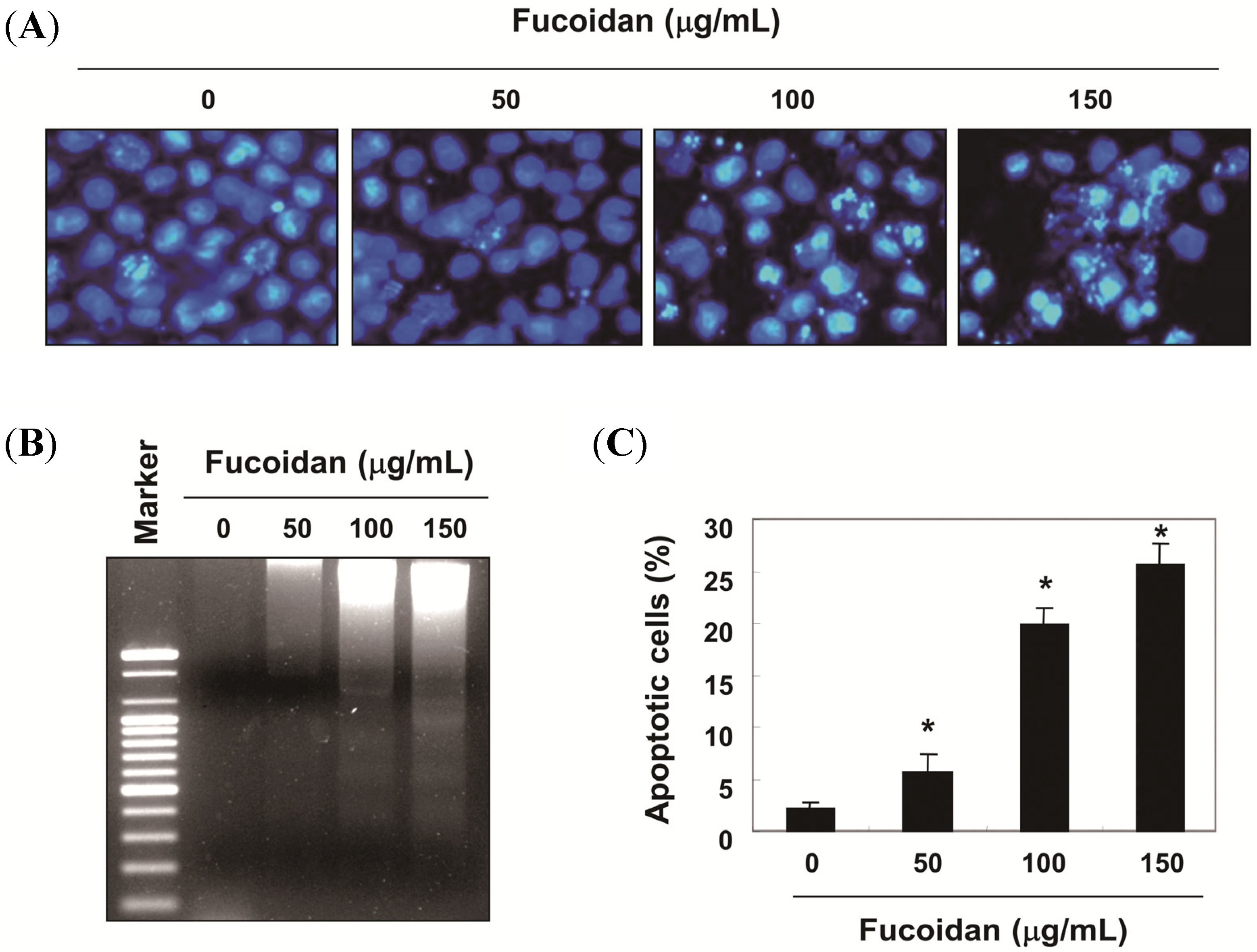
2.1. Fucoidan-Induced Growth Inhibition is Associated with the Induction of Apoptosis in T24 Cells

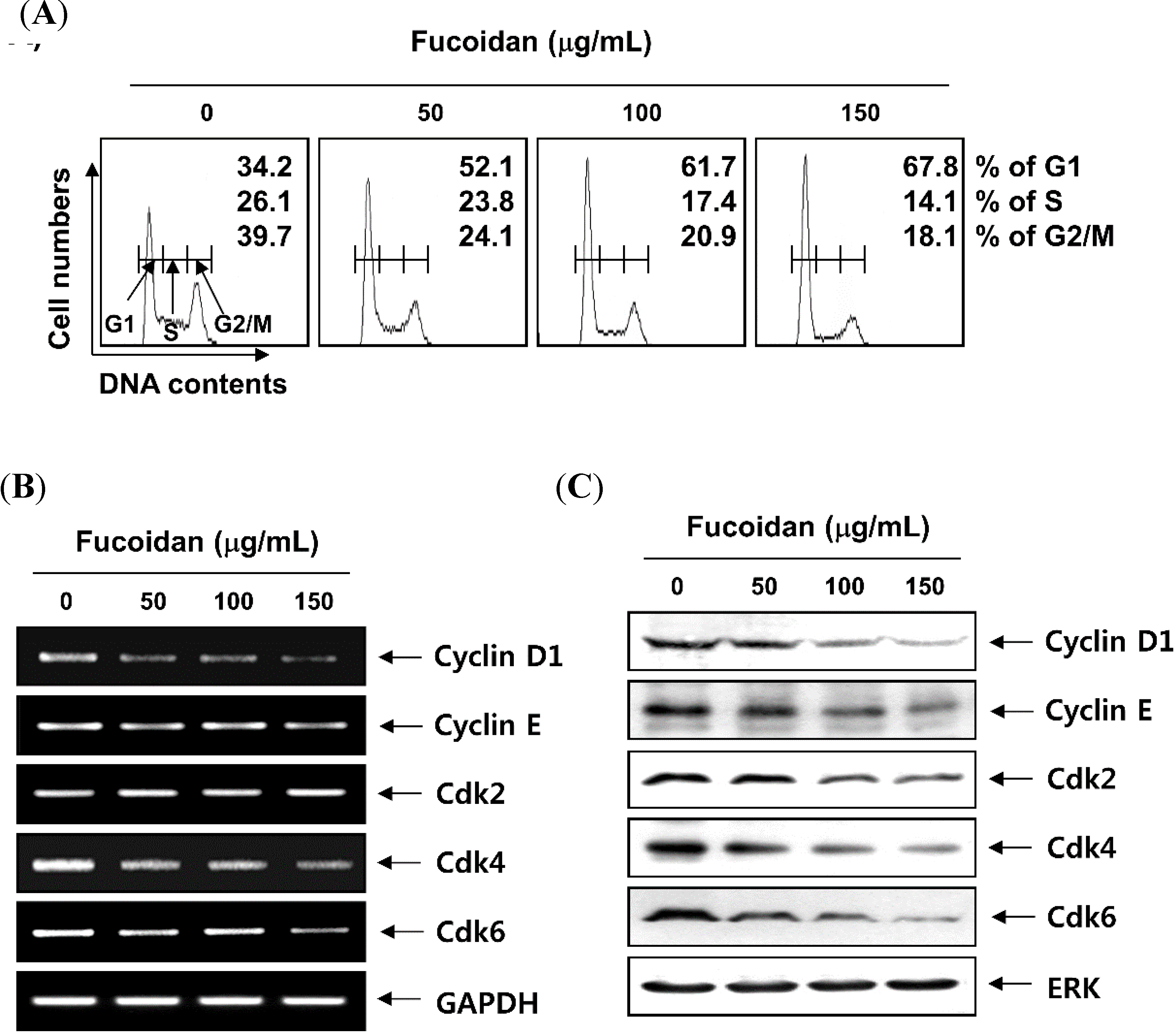
2.2. Fucoidan Induces Cell Cycle Arrest at G1 Phase in T24 Cells

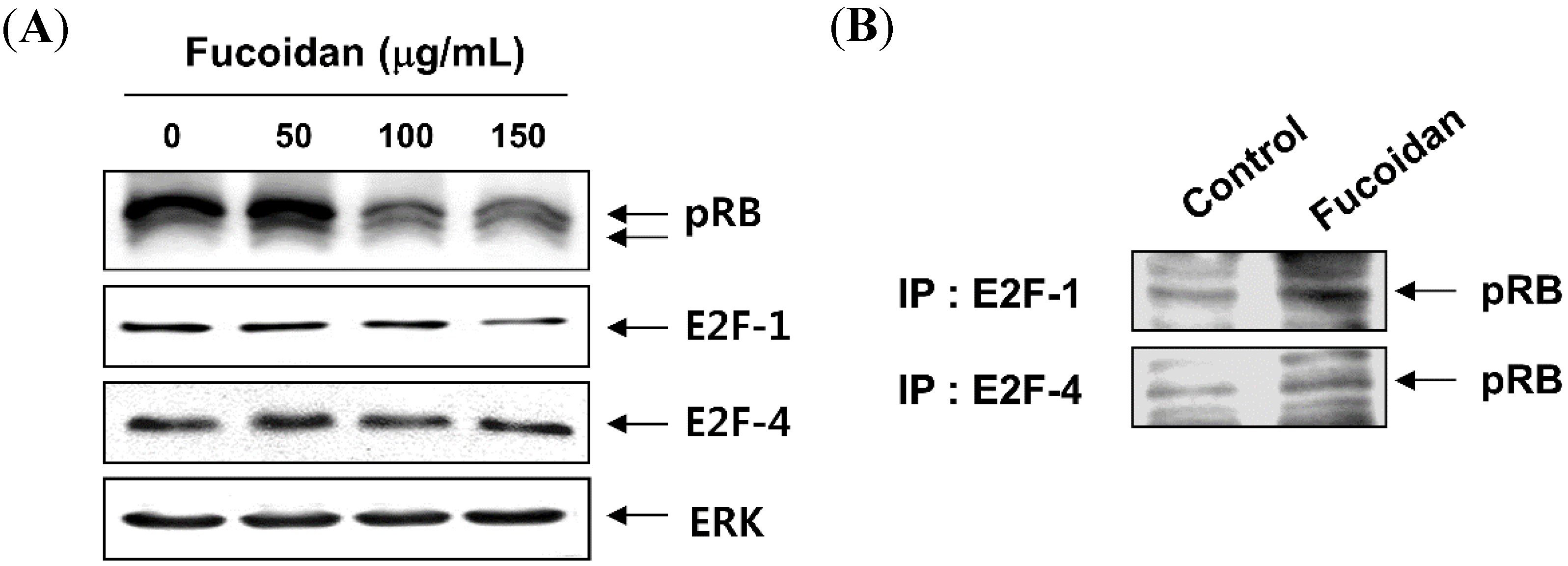
2.3. Fucoidan Inhibits pRB Phosphorylation and Increases the Binding of pRB and E2Fs in T24 Cells
2.4. Fucoidan Induces Apoptosis through a Caspase-Dependent Cascade in T24 Cells
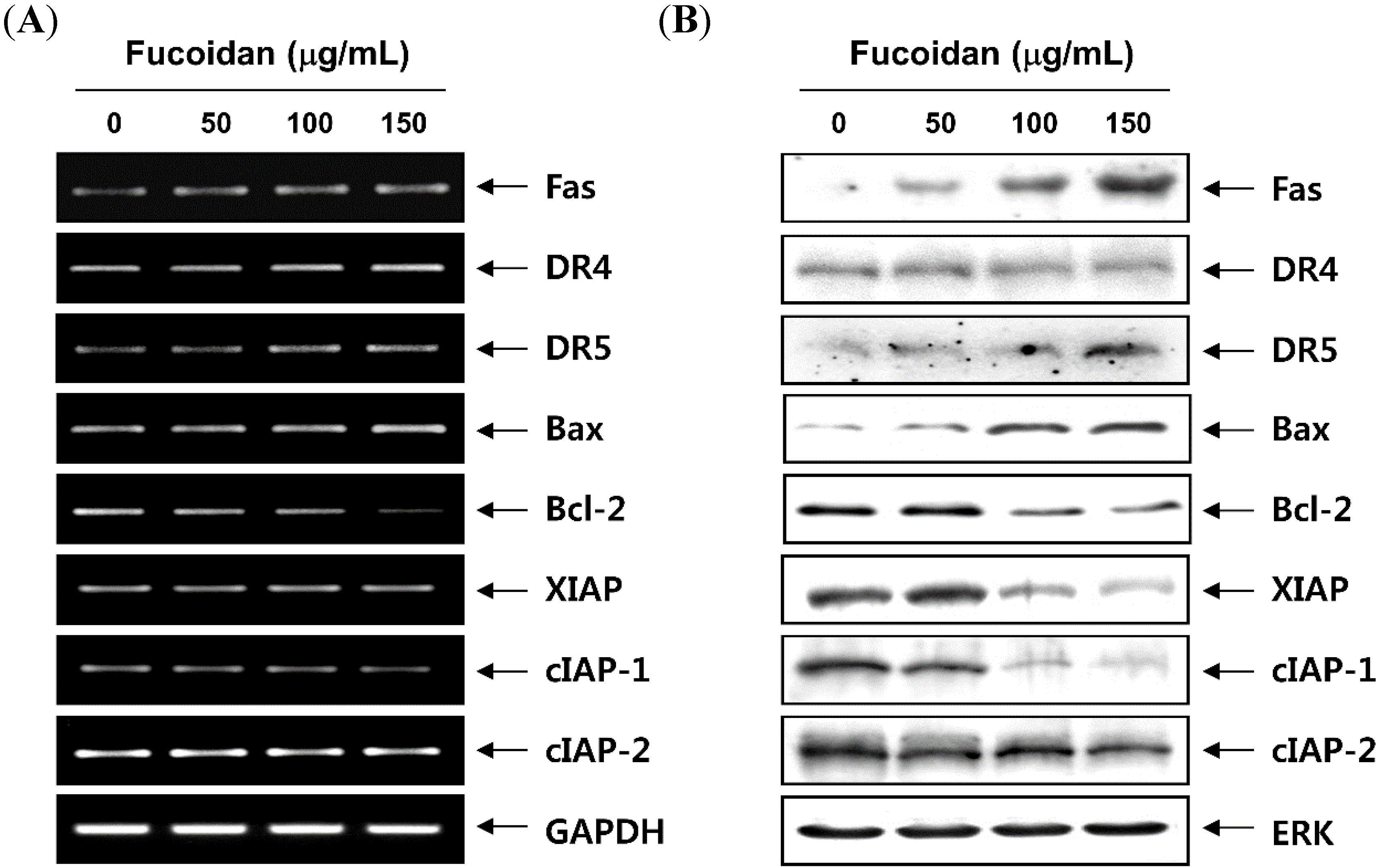
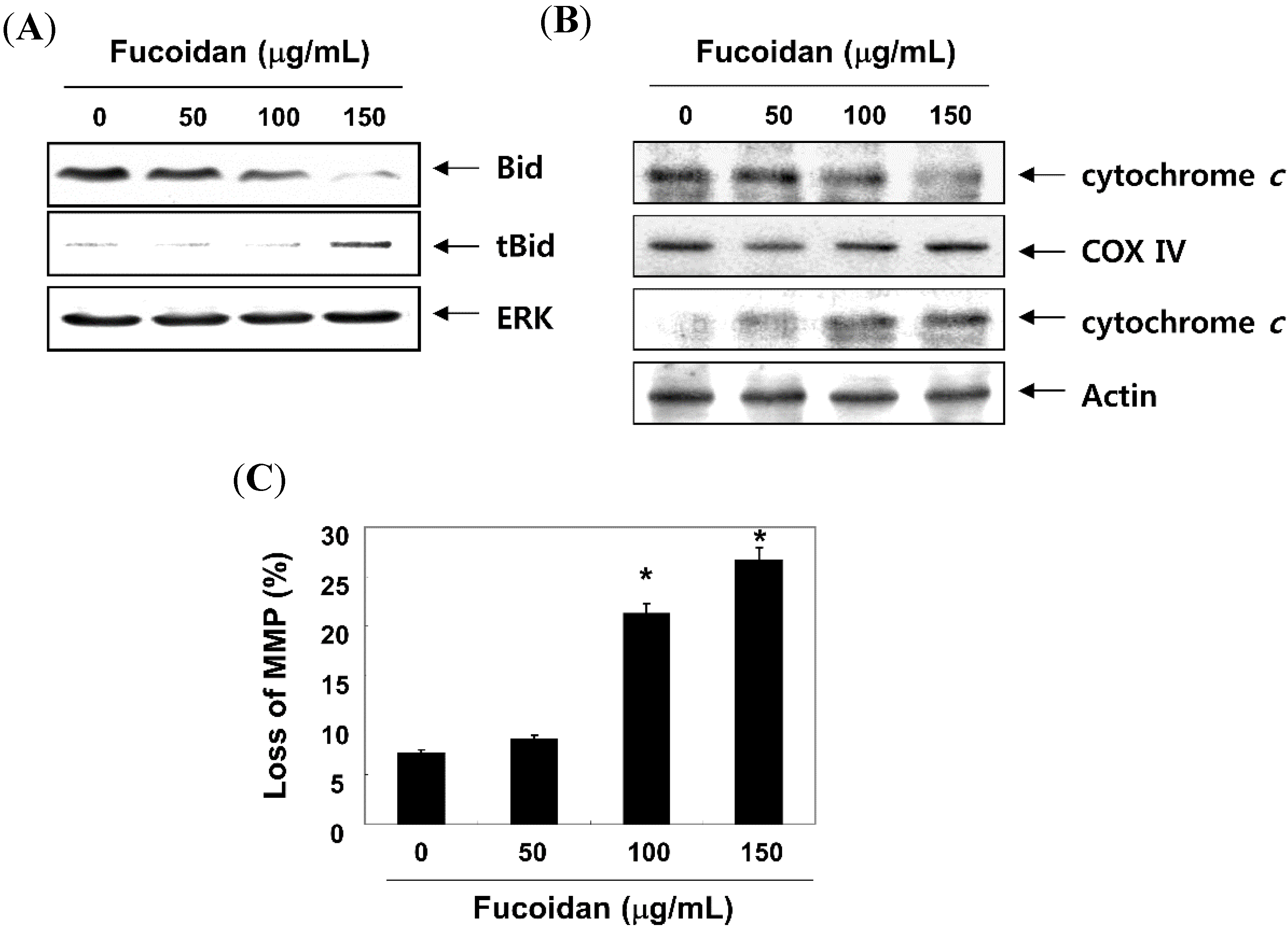
2.5. Fucoidan Modulates the Levels of Apoptosis-Related Factors and MMP Values in T24 Cells

2.6. Discussion
3. Experimental
3.1. Materials
3.2. Cell Culture, MTT Assay and Determinations of Cell Morphology
3.3. Nuclear Staining with DAPI
3.4. DNA Fragmentation Assay
3.5. Measurement of Apoptosis by Flow Cytometry
3.6. Cell Cycle Analysis
3.7. RNA Extraction and RT-PCR
3.8. Western Blot Analysis
3.9. Co-Immunoprecipitation Assay
3.10. In Vitro Caspase Activity Assay
3.11. Assay of MMP (ΔΨm)
3.12. Statistical Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sperka, T.; Wang, J.; Rudolph, K.L. DNA damage checkpoints in stem cells, ageing and cancer. Nat. Rev. Mol. Cell Biol. 2012, 13, 579–590. [Google Scholar]
- Canavese, M.; Santo, L.; Raje, N. Cyclin dependent kinases in cancer: Potential for therapeutic intervention. Cancer Biol. Ther. 2012, 13, 451–457. [Google Scholar]
- Lee, M.H.; Yang, H.Y. Negative regulators of cyclin-dependent kinases and their roles in cancers. Cell Mol. Life Sci. 2001, 58, 1907–1922. [Google Scholar]
- Li, A.; Blow, J.J. The origin of CDK regulation. Nat. Cell Biol. 2001, 3, E182–E184. [Google Scholar]
- Paternot, S.; Bockstaele, L.; Bisteau, X.; Kooken, H.; Coulonval, K.; Roger, P.P. Rb inactivation in cell cycle and cancer: The puzzle of highly regulated activating phosphorylation of CDK4 versus constitutively active CDK-activating kinase. Cell Cycle 2010, 9, 689–699. [Google Scholar]
- Dobashi, Y.; Takehana, T.; Ooi, A. Perspectives on cancer therapy: Cell cycle blockers and perturbators. Curr. Med. Chem. 2003, 10, 2549–2558. [Google Scholar]
- Hensley, P.; Mishra, M.; Kyprianou, N. Targeting caspases in cancer therapeutics. Biol. Chem. 2013, 394, 831–843. [Google Scholar]
- MacKenzie, S.H.; Clark, A.C. Targeting cell death in tumors by activating caspases. Curr. Cancer Drug Targets 2008, 8, 98–109. [Google Scholar]
- Chung, K.M.; Yu, S.W. Interplay between autophagy and programmed cell death in mammalian neural stem cells. BMB Rep. 2013, 46, 383–390. [Google Scholar]
- Brenner, D.; Mak, T.W. Mitochondrial cell death effectors. Curr. Opin. Cell Biol. 2009, 21, 871–877. [Google Scholar]
- Jin, Z.; El-Deiry, W.S. Overview of cell death signaling pathways. Cancer Biol. Ther. 2005, 4, 139–163. [Google Scholar]
- Fadeel, B.; Orrenius, S. Apoptosis: A basic biological phenomenon with wide-ranging implications in human disease. J. Intern. Med. 2005, 258, 479–517. [Google Scholar]
- Mišurcová, L.; Škrovánková, S.; Samek, D.; Ambrožová, J.; Machů, L. Health benefits of algal polysaccharides in human nutrition. Adv. Food Nutr. Res. 2012, 66, 75–145. [Google Scholar]
- Kim, S.K.; Li, Y.X. Medicinal benefits of sulfated polysaccharides from sea vegetables. Adv. Food Nutr. Res. 2011, 64, 391–402. [Google Scholar]
- Gomez-Gutierrez, C.M.; Guerra-Rivas, G.; Soria-Mercado, I.E.; Ayala-Sánchez, N.E. Marine edible algae as disease preventers. Adv. Food Nutr. Res. 2011, 64, 29–39. [Google Scholar]
- Bilan, M.I.; Grachev, A.A.; Ustuzhanina, N.E.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. Structure of a fucoidan from the brown seaweed Fucus. evanescens C. Ag. Carbohydr. Res. 2002, 337, 719–730. [Google Scholar]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar]
- Wang, W.; Wang, S.X.; Guan, H.S. The antiviral activities and mechanisms of marine polysaccharides: An overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar]
- Senni, K.; Pereira, J.; Gueniche, F.; Delbarre-Ladrat, C.; Sinquin, C.; Ratiskol, J.; Godeau, G.; Fischer, A.M.; Helley, D.; Colliec-Jouault, S. Marine polysaccharides: A source of bioactive molecules for cell therapy and tissue engineering. Mar. Drugs 2011, 9, 1664–1681. [Google Scholar] [CrossRef]
- Fitton, J.H. Therapies from fucoidan; multifunctional marine polymers. Mar. Drugs 2011, 9, 1731–1760. [Google Scholar]
- Kyung, J.; Kim, D.; Park, D.; Yang, Y.H.; Choi, E.K.; Lee, S.P.; Kim, T.S.; Lee, Y.B.; Kim, Y.B. Synergistic anti-inflammatory effects of Laminaria. japonica fucoidan and Cistanche. tubulosa extract. Lab. Anim. Res. 2012, 28, 91–97. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Manivasagan, P.; Venkatesan, J.; Kim, S.K. Brown seaweed fucoidan: Biological activity and apoptosis, growth signaling mechanism in cancer. Int. J. Biol. Macromol. 2013, 60, 366–374. [Google Scholar] [CrossRef]
- Hsu, H.Y.; Lin, T.Y.; Hwang, P.A.; Tseng, L.M.; Chen, R.H.; Tsao, S.M.; Hsu, J. Fucoidan induces changes in the epithelial to mesenchymal transition and decreases metastasis by enhancing ubiquitin-dependent TGFβ receptor degradation in breast cancer. Carcinogenesis 2013, 34, 874–884. [Google Scholar] [CrossRef]
- Liu, F.; Wang, J.; Chang, A.K.; Liu, B.; Yang, L.; Li, Q.; Wang, P.; Zou, X. Fucoidan extract derived from Undaria. pinnatifida inhibits angiogenesis by human umbilical vein endothelial cells. Phytomedicine 2012, 19, 797–803. [Google Scholar] [CrossRef]
- Park, H.S.; Hwang, H.J.; Kim, G.Y.; Cha, H.J.; Kim, W.J.; Kim, N.D.; Yoo, Y.H.; Choi, Y.H. Induction of apoptosis by fucoidan in human leukemia U937 cells through activation of p38 MAPK and modulation of Bcl-2 family. Mar. Drugs 2013, 11, 2347–2364. [Google Scholar] [CrossRef]
- Park, H.S.; Kim, G.Y.; Nam, T.J.; Kim, N.D.; Choi, Y.H. Anti-proliferative activity of fucoidan was associated with the induction of apoptosis and autophagy in AGS human gastric cancer cells. J. Food Sci. 2011, 76, T77–T83. [Google Scholar] [CrossRef]
- Zhang, Z.; Teruya, K.; Eto, H.; Shirahata, S. Fucoidan extract induces apoptosis in MCF-7 cells via a mechanism involving the ROS-dependent JNK activation and mitochondria-mediated pathways. PLoS One 2011, 6, e27441. [Google Scholar]
- Zhang, Z.; Teruya, K.; Yoshida, T.; Eto, H.; Shirahata, S. Fucoidan extract enhances the anti-cancer activity of chemotherapeutic agents in MDA-MB-231 and MCF-7 breast cancer cells. Mar. Drugs 2013, 11, 81–98. [Google Scholar]
- Xue, M.; Ge, Y.; Zhang, J.; Liu, Y.; Wang, Q.; Hou, L.; Zheng, Z. Fucoidan inhibited 4T1 mouse breast cancer cell growth in vivo and in vitro via downregulation of Wnt/β-catenin signaling. Nutr. Cancer 2013, 65, 460–468. [Google Scholar] [CrossRef]
- Xue, M.; Ge, Y.; Zhang, J.; Wang, Q.; Hou, L.; Liu, Y.; Sun, L.; Li, Q. Anticancer properties and mechanisms of fucoidan on mouse breast cancer in vitro and in vivo. PLoS One 2012, 7, e43483. [Google Scholar]
- Jin, J.O.; Song, M.G.; Kim, Y.N.; Park, J.I.; Kwak, J.Y. The mechanism of fucoidan-induced apoptosis in leukemic cells: Involvement of ERK1/2, JNK, glutathione, and nitric oxide. Mol. Carcinog. 2010, 49, 771–782. [Google Scholar]
- Hyun, J.H.; Kim, S.C.; Kang, J.I.; Kim, M.K.; Boo, H.J.; Kwon, J.M.; Koh, Y.S.; Hyun, J.W.; Park, D.B.; Yoo, E.S.; et al. Apoptosis inducing activity of fucoidan in HCT-15 colon carcinoma cells. Biol. Pharm. Bull. 2009, 32, 1760–1764. [Google Scholar] [CrossRef]
- Yamasaki-Miyamoto, Y.; Yamasaki, M.; Tachibana, H.; Yamada, K. Fucoidan induces apoptosis through activation of caspase-8 on human breast cancer MCF-7 cells. J. Agric. Food Chem. 2009, 57, 8677–8682. [Google Scholar] [CrossRef]
- Cooper, M.J.; Haluschak, J.J.; Johnson, D.; Schwartz, S.; Morrison, L.J.; Lippa, M.; Hatzivassiliou, G.; Tan, J. p53 mutations in bladder carcinoma cell lines. Oncol. Res. 1994, 6, 569–579. [Google Scholar]
- Pei, X.H.; Xiong, Y. Biochemical and cellular mechanisms of mammalian CDK inhibitors: A few unresolved issues. Oncogene 2005, 24, 2787–2795. [Google Scholar] [CrossRef]
- Riou, D.; Colliec-Jouault, S.; Pinczon du Sel, D.; Bosch, S.; Siavoshian, S.; le Bert, V.; Tomasoni, C.; Sinquin, C.; Durand, P.; Roussakis, C. Antitumor and antiproliferative effects of a fucan extracted from ascophyllum nodosum against a non-small-cell bronchopulmonary carcinoma line. Anticancer Res. 1996, 16, 1213–1218. [Google Scholar]
- Aisa, Y.; Miyakawa, Y.; Nakazato, T.; Shibata, H.; Saito, K.; Ikeda, Y.; Kizaki, M. Fucoidan induces apoptosis of human HS-sultan cells accompanied by activation of caspase-3 and down-regulation of ERK pathways. Am. J. Hematol. 2005, 78, 7–14. [Google Scholar] [CrossRef]
- Li, D.; Tian, Y.; Ma, Y.; Benjamin, T. p150(Sal2) is a p53-independent regulator of p21(WAF1/CIP). Mol. Cell Biol. 2004, 24, 3885–3893. [Google Scholar] [CrossRef]
- Aguero, M.F.; Facchinetti, M.M.; Sheleg, Z.; Senderowicz, A.M. Phenoxodiol, a novel isoflavone, induces G1 arrest by specific loss in cyclin-dependent kinase 2 activity by p53-independent induction of p21WAF1/CIP1. Cancer Res. 2005, 65, 3364–3373. [Google Scholar]
- Khosravi-Far, R.; Esposti, M.D. Death receptor signals to mitochondria. Cancer Biol. Ther. 2004, 3, 1051–1057. [Google Scholar] [CrossRef]
- Ola, M.S.; Nawaz, M.; Ahsan, H. Role of Bcl-2 family proteins and caspases in the regulation of apoptosis. Mol. Cell Biochem. 2011, 351, 41–58. [Google Scholar] [CrossRef]
- Danson, S.; Dean, E.; Dive, C.; Ranson, M. IAPs as a target for anticancer therapy. Curr. Cancer Drug Targets 2007, 7, 785–794. [Google Scholar] [CrossRef]
- De Graaf, A.O.; de Witte, T.; Jansen, J.H. Inhibitor of apoptosis proteins: New therapeutic targets in hematological cancer? Leukemia 2004, 18, 1751–1759. [Google Scholar] [CrossRef]
- Bubeník, J.; Baresová, M.; Viklický, V.; Jakoubková, J.; Sainerová, H.; Donner, J. Established cell line of urinary bladder carcinoma (T24) containing tumour-specific antigen. Int. J. Cancer 1973, 11, 765–773. [Google Scholar] [CrossRef]
- Lee, S.J.; Hwang, S.O.; Noh, E.J.; Kim, D.U.; Nam, M.; Kim, J.H.; Nam, J.H.; Hoe, K.L. Transactivation of bad by vorinostat-induced acetylated p53 enhances doxorubicin-induced cytotoxicity in cervical cancer cells. Exp. Mol. Med. 2014, 46, e76. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Park, H.Y.; Kim, G.-Y.; Moon, S.-K.; Kim, W.J.; Yoo, Y.H.; Choi, Y.H. Fucoidan Inhibits the Proliferation of Human Urinary Bladder Cancer T24 Cells by Blocking Cell Cycle Progression and Inducing Apoptosis. Molecules 2014, 19, 5981-5998. https://doi.org/10.3390/molecules19055981
Park HY, Kim G-Y, Moon S-K, Kim WJ, Yoo YH, Choi YH. Fucoidan Inhibits the Proliferation of Human Urinary Bladder Cancer T24 Cells by Blocking Cell Cycle Progression and Inducing Apoptosis. Molecules. 2014; 19(5):5981-5998. https://doi.org/10.3390/molecules19055981
Chicago/Turabian StylePark, Hye Young, Gi-Young Kim, Sung-Kwon Moon, Wun Jae Kim, Young Hyun Yoo, and Yung Hyun Choi. 2014. "Fucoidan Inhibits the Proliferation of Human Urinary Bladder Cancer T24 Cells by Blocking Cell Cycle Progression and Inducing Apoptosis" Molecules 19, no. 5: 5981-5998. https://doi.org/10.3390/molecules19055981
APA StylePark, H. Y., Kim, G.-Y., Moon, S.-K., Kim, W. J., Yoo, Y. H., & Choi, Y. H. (2014). Fucoidan Inhibits the Proliferation of Human Urinary Bladder Cancer T24 Cells by Blocking Cell Cycle Progression and Inducing Apoptosis. Molecules, 19(5), 5981-5998. https://doi.org/10.3390/molecules19055981





