Anti-Amoebic Properties of Carbonyl Thiourea Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Carbonyl Thiourea Derivatives
2.2. Anti-Amoebic Properties: IC50 Values

| Code | Chemical name | MW | Molecular structure |
|---|---|---|---|
| M1 | 2-(3-Benzoylthioureido)propanoic acid | 252.29 |  |
| M2 | 3-(3-Benzoylthioureido)propanoic acid | 252.29 |  |
| M3 | N-(2-Chlorophenyl)-N'-(4-chlorobutanoyl)thiourea | 291.20 |  |
| M4 | N-(3-Chlorophenyl)-N'-(4-chlorobutanoyl)thiourea | 291.20 |  |
| Compound | IC50 (µg·mL−1) | ||||
|---|---|---|---|---|---|
| A. castellanii (CCAP 1501/2A) | A. polyphaga (CCAP 1501/3A) | ||||
| M1 | 2.39 ± 0.24 | 3.74 ± 0.44 | |||
| M2 | 3.34 ± 0.41 | 3.76 ± 0.27 | |||
| M3 | 8.07 ± 0.65 | 8.52 ± 0.81 | |||
| M4 | 8.87 ± 0.27 | 9.30 ± 0.55 | |||
| IC50 (µM) | |||||
| A. castellanii (CCAP 1501/2A) | Percentage of strength (%) | A. polyphaga (CCAP 1501/3A) | Percentage of strength (%) | ||
| M1 | 9.47 | 73.5 | 14.84 | 52.4 | |
| M2 | 13.24 | 52.6 | 14.90 | 52.1 | |
| M3 | 27.70 | 25.1 | 29.25 | 26.6 | |
| M4 | 30.46 | 22.9 | 31.91 | 24.3 | |
| Chlorhexidine | 6.96 | 100.0 | 7.77 | 100.0 | |
2.3. Morphological Changes in Acanthamoeba
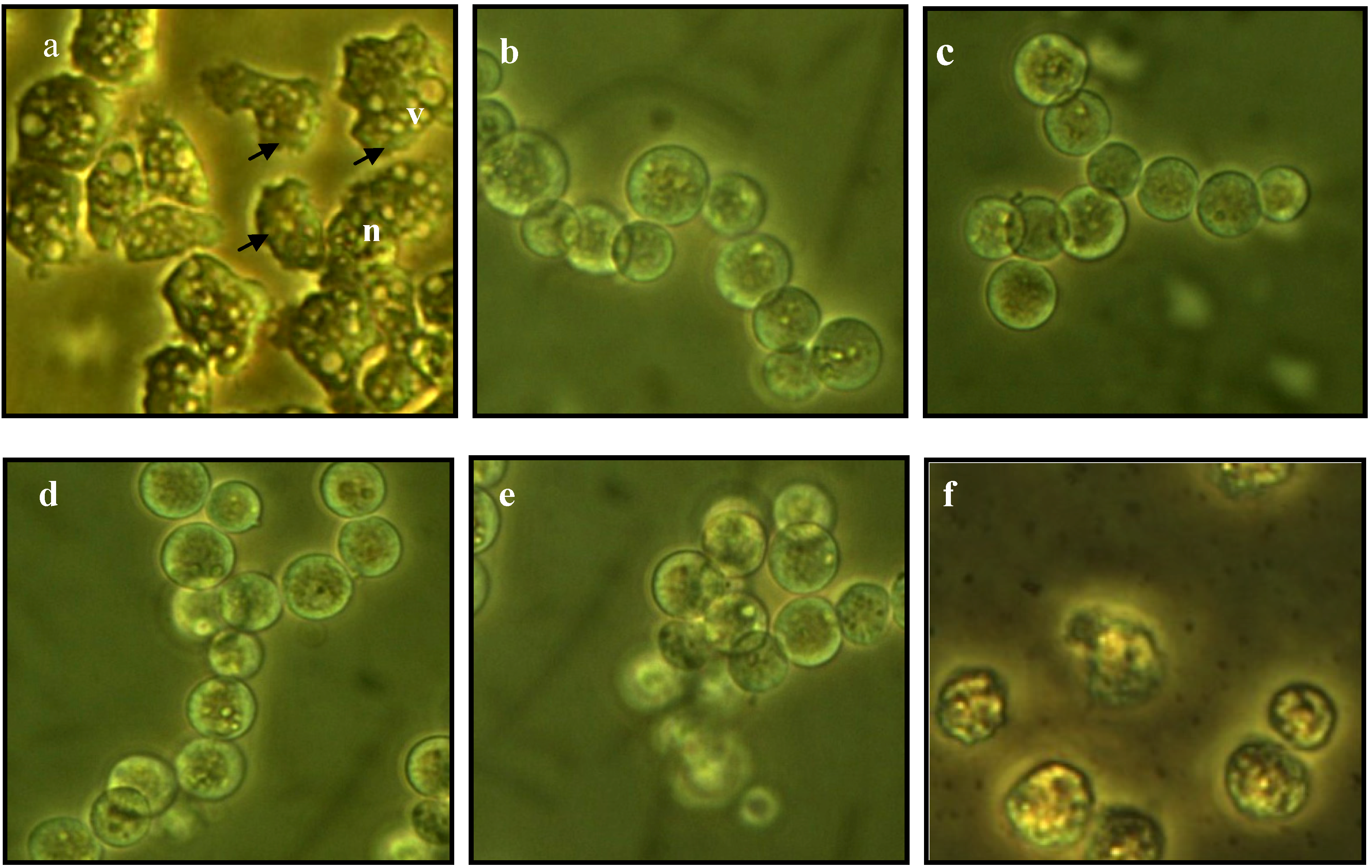
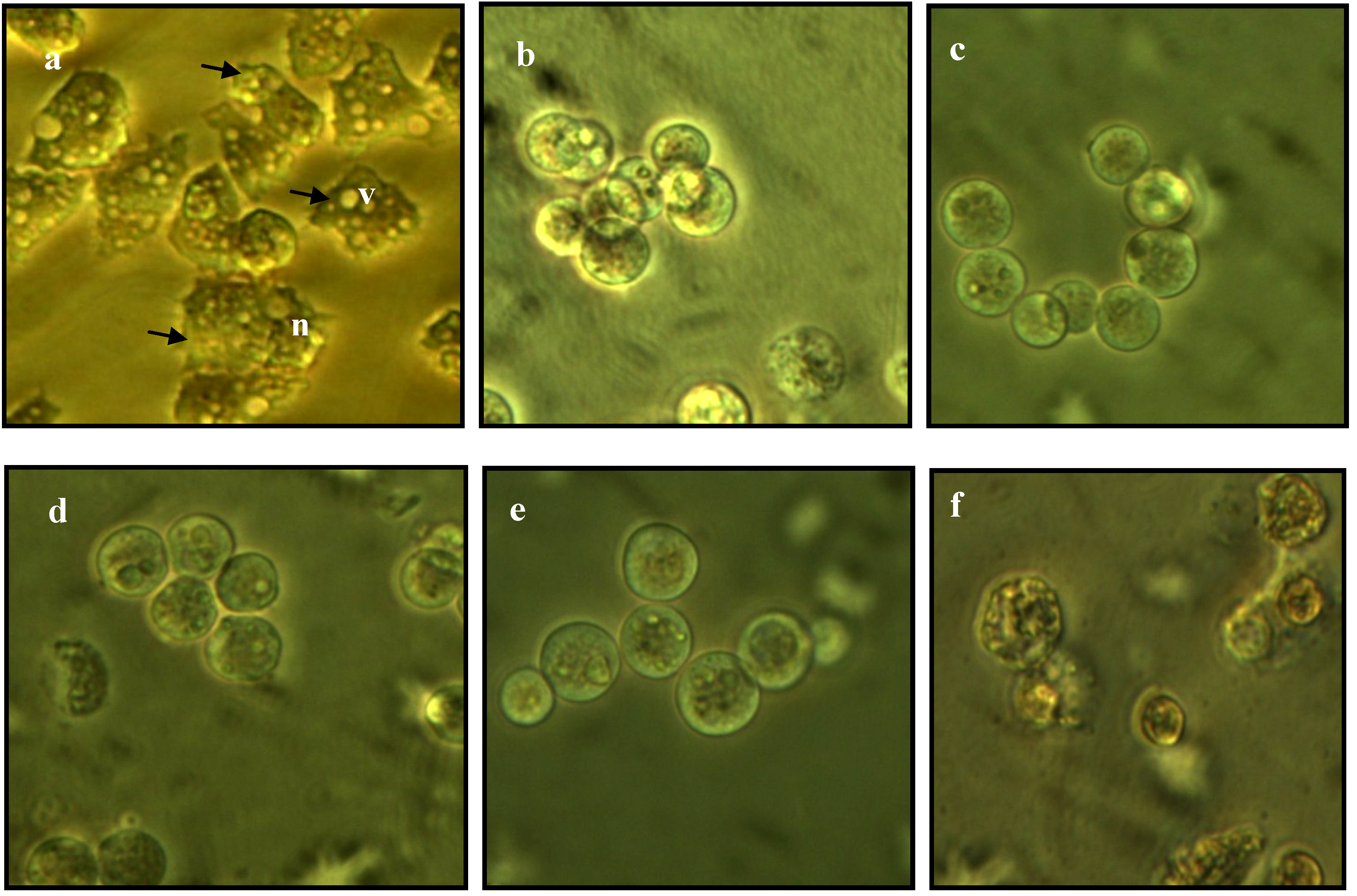
2.4. Integrity of Acanthamoeba Membrane
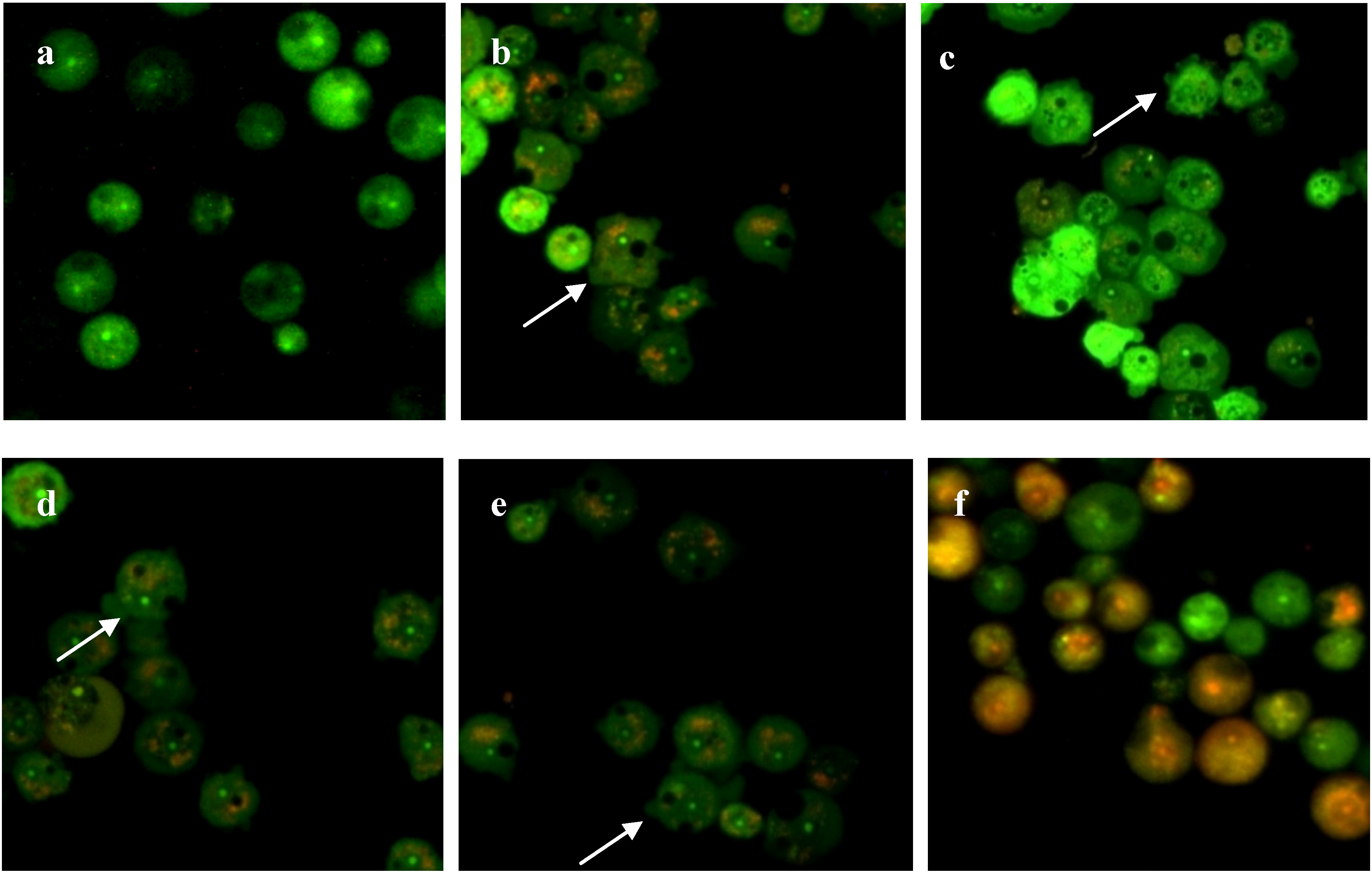

3. Experimental
3.1. General Information
3.2. Synthesis of Carbonyl Thiourea Derivatives
3.3. Characterization of the Newly-Synthesized Carbonyl Thiourea Derivatives
3.4. Determination of IC50 Values
3.5. Observation of Changes in Acanthamoeba Morphology
3.6. Evaluation of Acanthamoeba Membrane Integrity
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- De Jonckheere, J.F. Ecology of Acanthamoeba. Rev. Infect. Dis. 1991, 13, S385–S387. [Google Scholar] [CrossRef]
- Page, F.C. A New Key to Freshwater and Soil Gymnamoebae. In Freshwater Biological Association; Culture Collection of Algae and Protozoa: Ambleside, Cumbria, UK, 1988; p. 122. [Google Scholar]
- Narasimhan, S.; Madhavan, H.; Therese, L. Development and application of an in vitro susceptibility test for Acanthamoeba species isolated from keratitis to polyhexamethylene biguanide and chlorhexidine. Cornea 2002, 21, 203–205. [Google Scholar] [CrossRef]
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba spp. as agents of disease in humans. Clin. Microbiol. Rev. 2003, 16, 273–307. [Google Scholar] [CrossRef]
- Elder, M.J.; Dart, J.K.G. Chemotherapy for Acanthamoeba keratitis. Lancet 1995, 345, 791–792. [Google Scholar]
- Larkin, D.F.P.; Kilvington, S.; Dart, J.K.G. Treatment of Acanthamoeba keratitis with polyhexamethylene biguanide. Ophthalmology 1992, 99, 185–191. [Google Scholar] [CrossRef]
- Seal, D.V. Acanthamoeba keratitis update—Incidence, molecular epidemiology and new drugs for treatment. Eye 2003, 17, 893–905. [Google Scholar] [CrossRef]
- Murdoch, D.; Gray, T.B.; Cursons, R.; Parr, D. Acanthamoebakeratitis in New Zealand, including two cases with in vivo resistance to polyhexamethylene biguanide. Aust. New Zeal. J. Ophthalmol. 1998, 26, 231–236. [Google Scholar] [CrossRef]
- Turner, N.A.; Russell, A.D.; Furr, J.R.; Lloyd, D. Emergence of resistance to biocides during differentiation of Acanthamoeba castellanii. J. Antimicrob. Chemother. 2000, 46, 27–34. [Google Scholar]
- Ficker, L.; Seal, D.; Warhurst, D.; Wright, P. Acanthamoeba keratitis: Resistance to medical therapy. Eye 1990, 4, 835–838. [Google Scholar] [CrossRef]
- Abid, M.; Agarwal, S.M.; Azam, A. Synthesis and anti-amoebic activity of metronidazole thiosemicarbazone analogues. Eur. J. Med. Chem. 2008, 43, 2035–2039. [Google Scholar] [CrossRef]
- Budakoti, A.; Bhat, A.R.; Athar, F.; Azam, A. Syntheses and evaluation of 3-(3-bromophenyl)-5-phenyl-1-(thiazolo[4,5-b]quinoxaline-2-yl)-2pyrazoline derivatives. Eur. J. Med. Chem. 2008, 43, 1749–1757. [Google Scholar] [CrossRef]
- Ziegler-Skylakakis, K.; Nill, S.; Pan, J.F.; Andrae, U. S-Oxygenation of thiourea results in the formation of genotoxic products. Environ. Mol. Mutagen. 1998, 31, 362–373. [Google Scholar] [CrossRef]
- Khan, S.A.; Singh, N.; Saleem, K. Synthesis, characterization and in vitro antibacterial activity of thiourea and urea derivatives of steroids. Eur. J. Med. Chem. 2008, 43, 2272–2277. [Google Scholar] [CrossRef]
- Zhong, Z.; Xing, R.; Liu, S.; Wang, L.; Chai, S.; Li, P. Synthesis of acyl thiourea derivatives of chitosan and their anti-microbial activities in vitro. Carbohydr. Res. 2008, 343, 566–570. [Google Scholar] [CrossRef]
- Eweis, M.; Elkholy, S.S.; Elsabee, M.Z. Antifungal efficacy of chitosan and its thiourea derivatives upon the growth of some sugar-beet pathogens. Int. J. Biol. Macromol. 2006, 38, 1–8. [Google Scholar] [CrossRef]
- Chen, S.; Wu, G.; Zeng, H. Preparation of high anti-microbial activity chitosan-Ag+ complex. Carbohydr. Polym. 2005, 60, 33–38. [Google Scholar] [CrossRef]
- Turan-Zitouni, G.; Sıvacı, D.M.; Kaplancıklı, Z.A.; Özdemir, A. Synthesis and anti-microbial activity of some pyridinyliminothiazoline derivatives. Il Farmaco 2002, 57, 569–572. [Google Scholar] [CrossRef]
- Phetsuksiri, B.; Jackson, M.; Scherman, H.; McNeil, M.; Besra, G.S.; Baulard, A.R.; Slayden, R.A.; DeBarber, A.E.; Barry, C.E., III; Baird, M.S.; et al. Unique mechanism of action of the thiourea drug isoxyl on Mycobacterium tuberculosis. J. Biol. Chem. 2003, 278, 53123–53130. [Google Scholar] [CrossRef]
- Paynter, O.E.; Burin, G.J.; Jaeger, R.B.; Gregorio, C.A. Goitrogens and thyroid follicular cell neoplasia. Evidence for a threshold process. Regul. Toxicol. Pharmacol. 1988, 8, 102–119. [Google Scholar] [CrossRef]
- Yusof, M.S.M.; Yamin, B.M. 3-(3-Benzoylthioureido) propionic acid. Acta Crystallogr. 2003, E59, o828–o829. [Google Scholar]
- Yusof, M.S.M.; Embong, N.F.; Yamin, B.M.; Ngah, N. 1-(4-Chlorobutanoyl)-3-(2-chloro phenyl)thiourea. Acta Crystallogr. 2012, E68, o1536. [Google Scholar]
- Deharo, E.; Bourdy, G.; Quenevo, C.; Munoz, V.; Ruiz, G.; Sauvain, M. A search for natural bioactive compounds in Bolivia through a multi disciplinary sciences approach. Part V. Evaluation of the antimalarial activity of plants used by the Tacana Indians. J. Ethnopharmacol. 2001, 77, 91–98. [Google Scholar] [CrossRef]
- McBride, J.; Ingram, R.P.; Henriquez, F.L.; Roberts, C.W. Development of colorimetric microtiter plate assay for assessment of anti-microbials against Acanthamoeba. J. Clin. Microbiol. 2005, 43, 629–634. [Google Scholar] [CrossRef]
- Patnaik, P. A Comprehensive Guide to the Hazardous Properties of Chemical Substances; Wiley-Interscience: Hoboken, NJ, USA, 2007; p. 904. [Google Scholar]
- Fustero, S.; Salavert, E.; Pina, B.; de Arellano, C.R.; Asensio, R. Novel strategy for the synthesis of fluorinated β-amino acid derivatives from Δ2-oxazolines. Tetrahedron 2001, 57, 6475–6486. [Google Scholar] [CrossRef]
- Ye, Y.H.; Huang, Y.S.; Wang, Z.Q.; Chen, S.M.; Tian, Y. Synthesis of new amino acid and peptide derivatives of estradiol and their binding affinities for the estrogen receptor. Steroids 1993, 58, 35–39. [Google Scholar] [CrossRef]
- Patel, N.B.; Shaikh, F.M. Synthesis and anti-microbial activity of new 4-thiazolidinone derivatives containing 2-amino-6-methoxybenzothiazole. Saudi Pharm. J. 2010, 18, 129–136. [Google Scholar] [CrossRef]
- Bowers, B.; Korn, E.D. The fine structure of Acanthamoeba castellanii, kinetics and morphology. I. The Trophozoite. J. Cell Biol. 1968, 39, 95–111. [Google Scholar] [CrossRef]
- Khan, N.A. Pathogenicity, morphology, and differentiation of Acanthamoeba. Curr. Microbiol. 2001, 43, 391–395. [Google Scholar] [CrossRef]
- Bowers, B.; Korn, E.D. Localization of lipophosphonoglycan on both sides of Acanthamoeba plasma membrane. J. Cell Biol. 1974, 62, 533–540. [Google Scholar] [CrossRef]
- Khan, N.A. Emerging Protozoan Pathogens; Taylor & Francis Group: Oxford, UK, 2008; pp. 5–24. [Google Scholar]
- Weisman, R.A. Differentiation in Acanthamoeba castellanii. Annu. Rev. Microbiol. 1976, 30, 189–219. [Google Scholar] [CrossRef]
- Coder, D.M. Assessment of cell viability. In Current Protocols in Cytometry, 2nd ed.; Wiley: New York, NY, USA, 1997; pp. 8–11. [Google Scholar]
- Darzynkiewicz, Z.; Juan, G.; Li, X.; Gorczyka, W.; Murakami, T.; Traganos, F. Cytometry in cell necrobiology: Analysis of apoptosis and accidental cell death (necrosis). Cytometry 1997, 27, 1–20. [Google Scholar] [CrossRef]
- Riss, T.L.; Moravec, R.A. Use of multiple assay endpoints to investigate the effects of incubation time, dose of toxin, and plating density in cell-based cytotoxicity assays. Assay Drug Dev. Technol. 2004, 2, 51–62. [Google Scholar] [CrossRef]
- Arnkt-Jovin, D.J.; Jovin, T.M. Fluorescence labeling and microscopy of DNA. Methods Cell Biol. 1989, 30, 417–448. [Google Scholar] [CrossRef]
- Puranam, K.L.; Boustany, R.M. Assessment of cell viability and histochemical methods in apoptosis. In Apoptosis in Neurobiology; Hannun, Y.A., Boustany, R.M., Eds.; CRC Press: Washington, DC, USA, 1999; p. 78. [Google Scholar]
- Perrine, D.; Chenu, J.P.; Georges, P.; Lancelot, J.C.; Saturnino, C.; Robba, M. Amoebicidal efficiencies of various diamidines against two strains of Acanthamoeba polyphaga. Antimicrob. Agents Chemother. 1995, 39, 339–342. [Google Scholar] [CrossRef]
- Nakisah, M.A.; Ida Muryany, M.Y.; Fatimah, H.; Nor Fadilah, R.; Zalilawati, M.R.; Khamsah, S.; Habsah, M. Anti-amoebic properties of a Malaysian marine sponge Aaptos sp. on Acanthamoeba castellanii. World J. Microbiol. Biotechnol. 2012, 28, 1237–1244. [Google Scholar] [CrossRef]
- Asiri, S.; Ogbunade, P.O.J.; Warhust, D.C. In vitro assessment of susceptibility of Acanthamoeba polyphaga to drugs using combined methods of dye-binding assay and uptake of radiolabeled adenosine. Int. J. Parasitol. 1994, 24, 975–980. [Google Scholar] [CrossRef]
- Wright, C.W.; O’Neill, M.J.; Phillipson, J.D.; Warhurst, D.C. Use of microdilution to assess in vitro anti-amoebic activities of Bruceajavanica fruits, Simaroubaamara Stem, and a number of Quassinoids. Antimicrob. Agents Chemother. 1988, 32, 1725–1729. [Google Scholar] [CrossRef]
- Mascotti, K.; McCullough, J.; Burger, S.R. HPC viability measurement: Trypan blue versus acridine orange and propidium iodide. Transfusion 2000, 40, 693–696. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compoundsare available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ibrahim, M.A.; Mohd Yusof, M.S.; Amin, N.M. Anti-Amoebic Properties of Carbonyl Thiourea Derivatives. Molecules 2014, 19, 5191-5204. https://doi.org/10.3390/molecules19045191
Ibrahim MA, Mohd Yusof MS, Amin NM. Anti-Amoebic Properties of Carbonyl Thiourea Derivatives. Molecules. 2014; 19(4):5191-5204. https://doi.org/10.3390/molecules19045191
Chicago/Turabian StyleIbrahim, Maizatul Akma, Mohd Sukeri Mohd Yusof, and Nakisah Mat Amin. 2014. "Anti-Amoebic Properties of Carbonyl Thiourea Derivatives" Molecules 19, no. 4: 5191-5204. https://doi.org/10.3390/molecules19045191




