Kinetics of Glycoxidation of Bovine Serum Albumin by Methylglyoxal and Glyoxal and its Prevention by Various Compounds
Abstract
:1. Introduction
2. Results
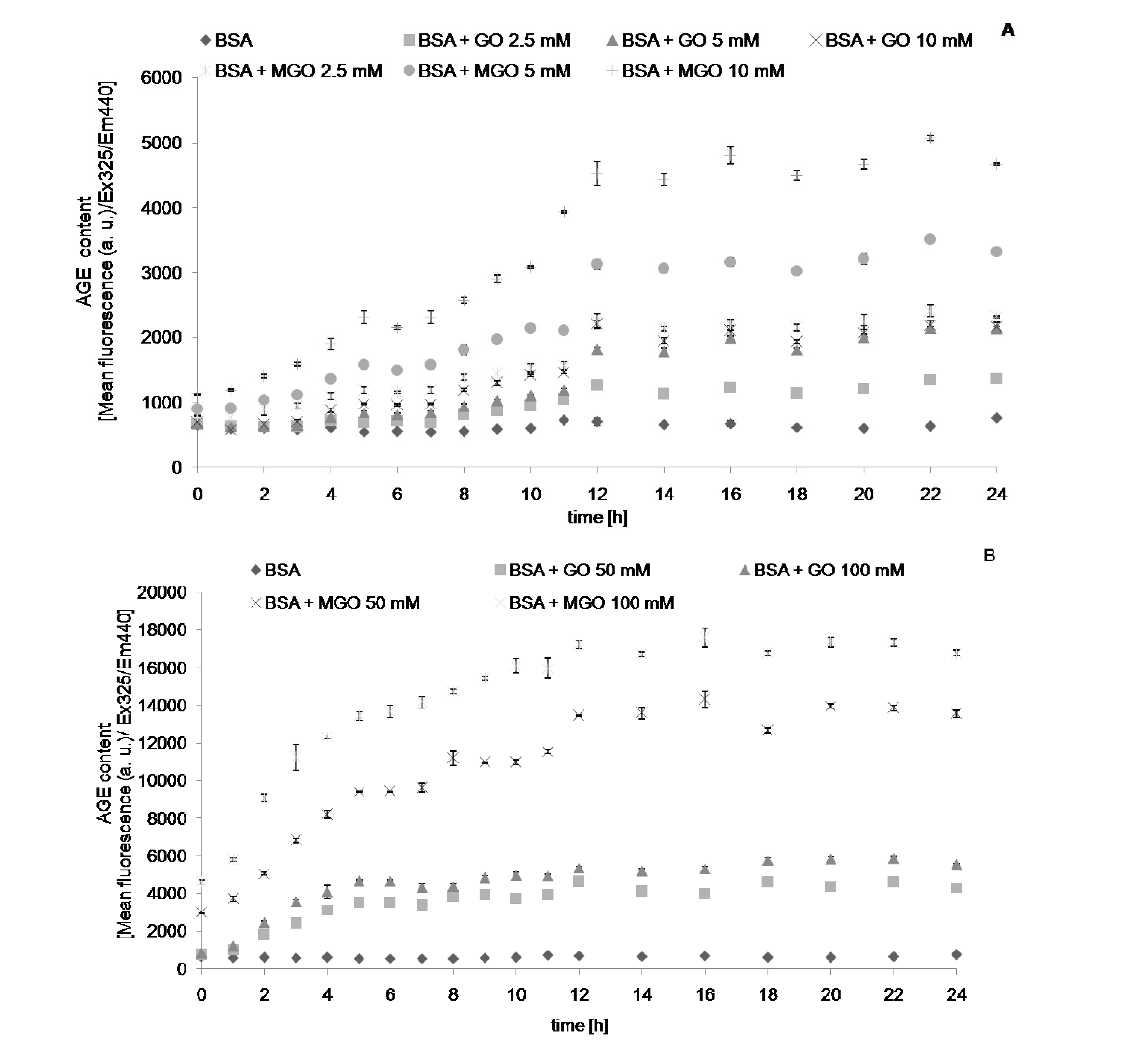
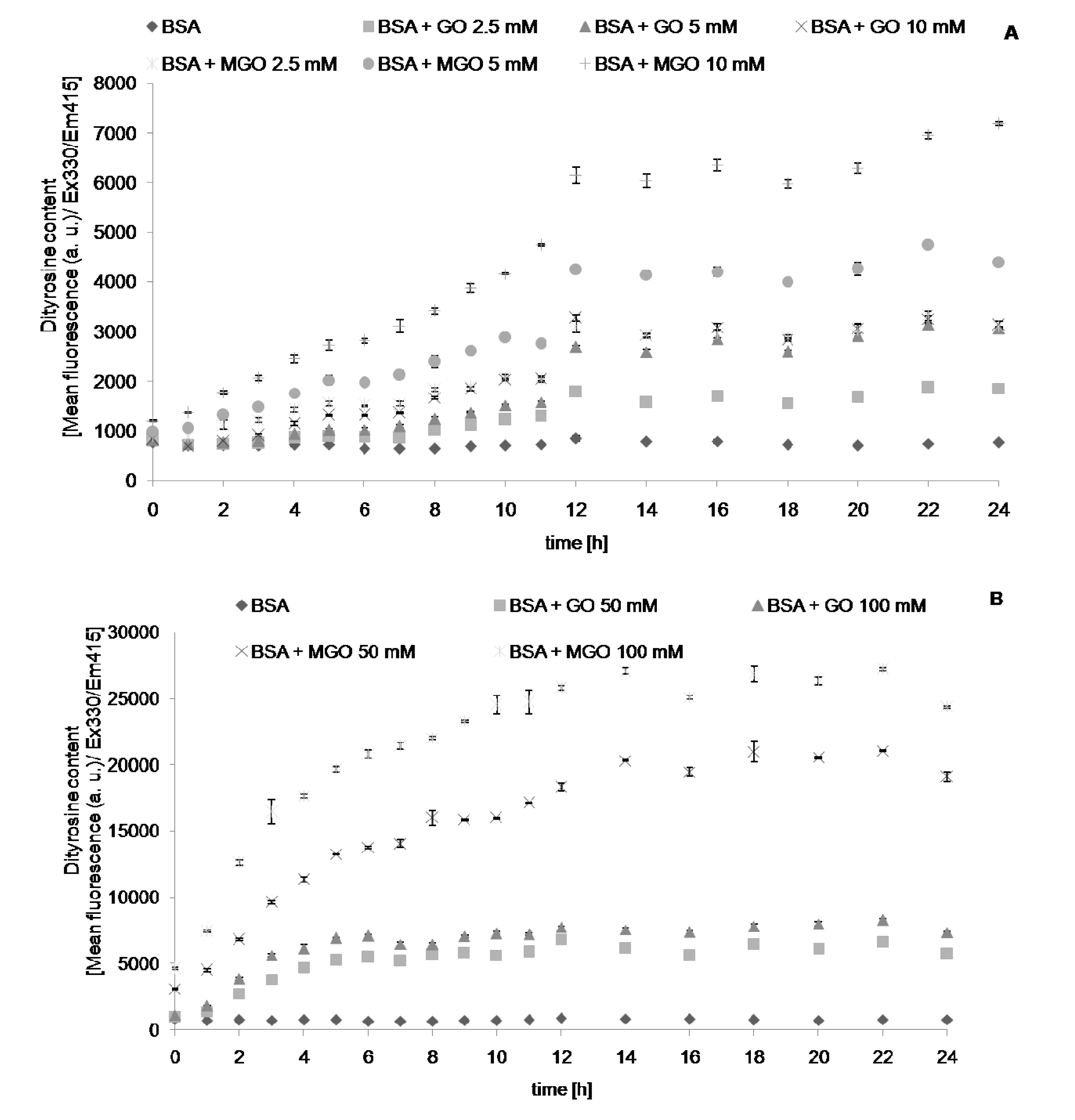



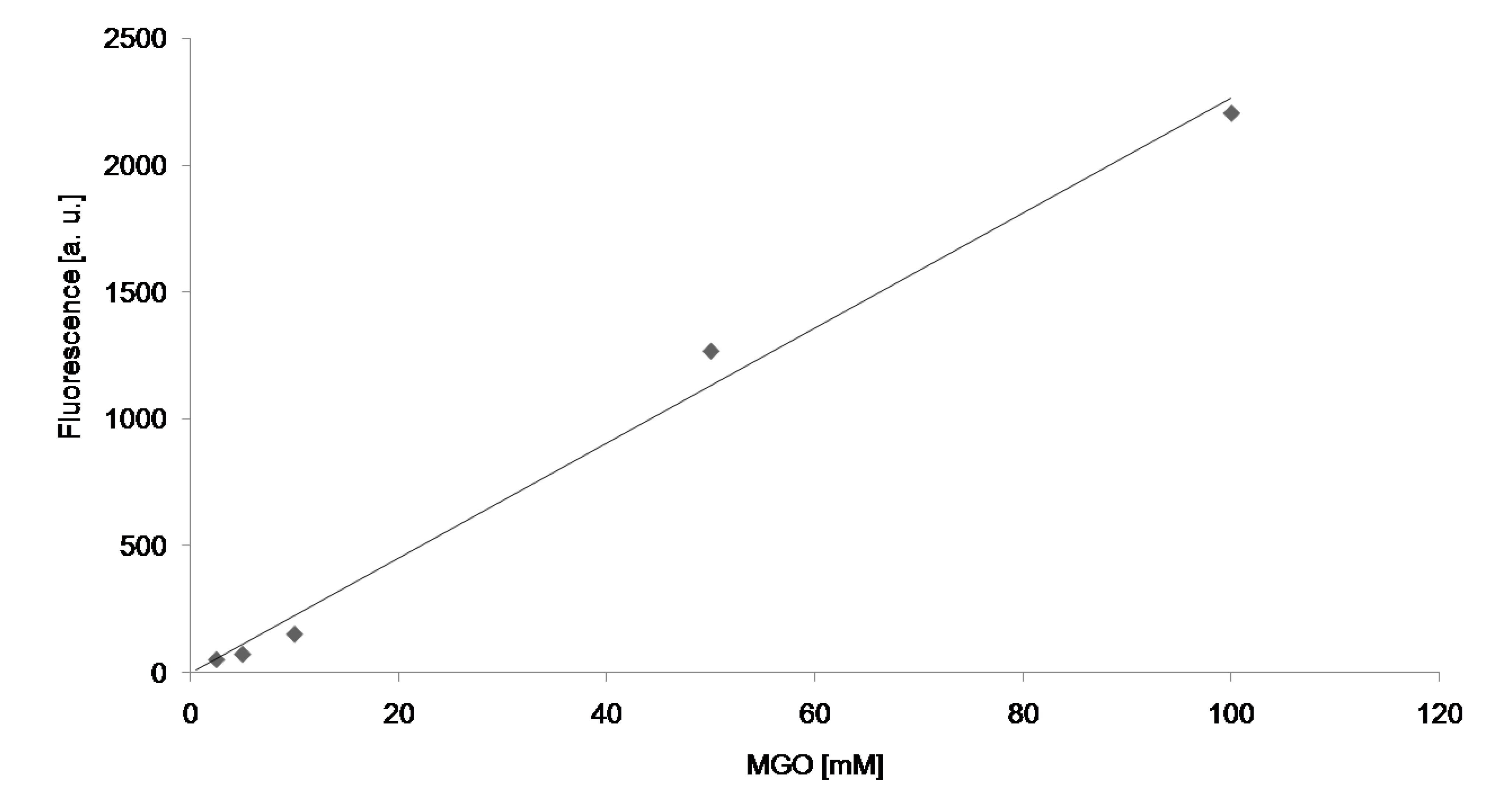
| Modification | Glyoxal | Methylglyoxal |
|---|---|---|
| AGE formation | 10.4 | 22.7 |
| Dityrosine formation | 48.9 | 121.3 |
| N-Formylkynurenine formation | 11.3 | 27.1 |
| Kynurenine formation | 12.5 | 19.0 |
| Compound | AGE | Dityrosine | N′-formylkynurenine | Kynurenine |
|---|---|---|---|---|
| 2,2,6,6-Tetramethyl-4-[(nonanoylamino)piperidin-1-yl]oxyl | 97.8 ± 4.78 | 96.25 ± 4.4 | 97.55 ± 4.41 | 326.09 ± 11.77 * |
| 3-Bromopyruvate | 216.01 ± 7.89 * | 208.88 ± 6.92 * | 221.49 ± 7.92 * | 120.5 ± 8.34 * |
| 4-Hydroxy-TEMPO | 80.54 ± 5.79 * | 75.62 ± 4.61 * | 78.33 ± 5.27 * | 162.6 ± 6.02 * |
| Ascorbic acid | 104.4 ± 2.09 * | 111.97 ± 2.85 * | 109.21 ± 2.49 * | 182.54 ± 5.77 * |
| Bisphenol A | 155.2 ± 5.22 * | 155.17 ± 3.91 * | 154.4 ± 3.68 * | 92.07 ± 4.51 * |
| Captopril | 88.84 ± 3.17 * | 93.46 ± 3.4 * | 93.56 ± 4.41 * | 13.78 ± 0.45 * |
| 1-Cyano-4-hydroxycinnamic acid | 3.5 ± 0.213 * | 3.94 ± 0.09 * | 3.69 ± 0.16 * | 81.72 ± 5.24 * |
| Cysteamine | 65.19 ± 2.69 * | 63.73 ± 2.84 * | 64.36 ± 2.97 * | 30.95 ± 0.47 * |
| Ellagic acid | 47.02± 0.51 * | 35.49 ± 0.22 * | 45.6 ± 0.33 * | 48.1 ± 1.32 * |
| Genistein | 27.17 ± 0.31 * | 25.98 ± 0.34 * | 27.92 ± 0.52 * | 85.90 ± 3.79 * |
| 4-Hydroxycinnamic acid | 91.78 ± 3.39 * | 92.35 ± 2.54 * | 89.67± 3.67 * | 89.4 ± 3.35 * |
| Lipoic acid | 79.19 ± 2.42 * | 80.86 ± 2.33 * | 81.69 ± 2.79 * | 123.25 ± 4.41 * |
| Metformin | 178.72 ± 5.34 * | 100.93 ± 2.6 | 163.44 ± 4.47 * | 146.51 ± 3.50 * |
| Na-pyruvate | 100.52 ± 1.9 | 95.09 ± 1.75 * | 101.61 ± 2.22 | 63.33 ± 1.84 |
| Naringin | 19.84 ± 0.68 | 16.87 ± 0.26 | 18.79 ± 0.49 | 69.32 ± 2.24 * |
| para-Aminobenzoic acid | 46.25 ± 1.28 * | 46.43 ± 1.35 * | 46.59 ± 1.21 * | 111.84 ± 6.47 * |
| Quinic acid | 57.29 ± 3.45 * | 57.27 ± 3.42 * | 61.35 ± 3.96 * | 128.99 ± 5.57 * |
| TEMPO | 78.11 ± 3.45 * | 75.09 ± 3.22 * | 80.35 ± 3.48 * | 106.35 ± 4.13 * |
| Tiron | 68.16 ± 4.16 * | 75.4 ± 4.15 * | 70.53 ± 4.54 * | 326.09 ± 11.77 * |
| Compound | AGE | Dityrosine | N′-formylkynurenine | Kynurenine |
|---|---|---|---|---|
| 2,2,6,6-Tetramethyl-4-[(nonanoylamino)piperidin-1-yl]oxyl | 80.94 ± 10.25 * | 82.6 ± 7.46 * | 82.03 ± 8.21 * | 74.68 ± 18.28 |
| 3-Bromopyruvate | 228.89 ± 11.14 * | 201.68 ± 8.82 * | 225.71 ± 9.33 * | 544.55 ± 28.26 * |
| 4-Hydroxy-TEMPO | 106.79 ± 11.96 | 104.33 ± 10.21 | 109.75 ± 11.15 | 116.48 ± 19.23 |
| Ascorbic acid | 217.49 ± 3.3 * | 226.31 ± 3.09 * | 214.49 ± 2.97 * | 1233.03 ± 5.13 * |
| Bisphenol A | 211.51 ± 4.41 * | 173.97 ± 5.28 * | 201.74 ± 3.91 * | 645.93 ± 17.76 * |
| Captopril | 118.21 ± 5.33 * | 114.82 ± 5.69 * | 120.55 ± 5.96 * | 160.94 ± 11.56 * |
| 1-Cyano-4-hydroxycinnamic acid | 2.48 ± 0.45 * | 3.60 ± 0.36 * | 3.00 ± 0.45 * | 0.78 ± 1.27 * |
| Cysteamine | 97.86 ± 14.26 | 90.96 ± 9.168 | 96.79 ± 10.56 | 221.00 ± 23.69 * |
| Ellagic acid | 50.67 ± 0.53 * | 31.76 ± 0.17 * | 46.59 ± 0.45 * | 137.45 ± 2.28 * |
| Genistein | 48.07 ± 1.11 * | 42.62 ± 1.01 * | 48.5± 0.99 * | 121.83 ± 7.36 * |
| 4-Hydroxycinnamic acid | 85.28 ± 6.09 * | 89.68 ± 8.33 * | 90.86 ± 11.44 | 117.76 ± 46.25 |
| Lipoic acid | 98.7 ± 4.86 | 97.7 ± 4.13 | 105.12 ± 4.19 * | 122.04 ± 8.49 * |
| Metformin | 112.98 ± 7.01 * | 118.51 ± 5.75 * | 118.95 ± 6.17 * | 140.2 ± 11.96 * |
| Na-pyruvate | 146.55 ± 6.16 * | 138.77 ± 11.67 * | 143.83 ± 11.52 * | 221.12 ± 22.40 * |
| Naringin | 37.86 ± 0.59 * | 28.18 ± 0.57 * | 35.88 ± 0.22 * | 206.21± 2.45 * |
| para-Aminobenzoic acid | 94.7 ± 4.86 | 96.59 ± 4.42 | 99.59 ± 4.22 | 129.67± 8.2 * |
| Quinic acid | 147.82 ± 10.8 * | 145.38 ± 8.21 * | 149.06 ± 8.75 * | 213.44 ± 14.32 * |
| TEMPO | 101.77 ± 6.94 | 98.71 ± 6.32 | 113.51± 10.75 * | 133.52 ± 17.71 * |
| Tiron | 95.96 ± 5.37 | 113.84 ± 9.77 * | 106.34 ± 10.88 | 170.80 ± 9.68 * |
3. Discussion
4. Experimental
 , where x- mean value of the sample with additive expressed in %, xo: value for control sample assumed as 100%, s: standard deviation, n: number of experiments (usually n = 3). The calculations were made in Excel.
, where x- mean value of the sample with additive expressed in %, xo: value for control sample assumed as 100%, s: standard deviation, n: number of experiments (usually n = 3). The calculations were made in Excel.5. Conclusions
Acknowledgments
Author Contributions
Conflicts of interest
References
- Seidler, N.W. Basic biology of GAPDH. Adv. Exp. Med. Biol. 2013, 985, 1–36. [Google Scholar] [CrossRef]
- Rondeau, P.; Bourdon, E. The glycation of albumin: Structural and functional impacts. Biochimie 2011, 93, 645–658. [Google Scholar] [CrossRef]
- Kalapos, M.P. Methylglyoxal in living organisms. Chemistry; biochemistry; toxicology and biological implications. Toxicol. Lett. 1999, 110, 145–175. [Google Scholar] [CrossRef]
- McLellan, A.C.; Thornalley, P.J.; Benn, J.; Sonksen, P.H. Glyoxalase system in clinical diabetes mellitus and correlation with diabetic complication. Clin. Sci. 1994, 87, 21–29. [Google Scholar]
- Thornalley, P.J. Protein and nucleotide damage by glyoxal and methylglyoxal in physiological systems—Role in ageing and disease. Drug Metabol. Drug Interact. 2008, 23, 125–150. [Google Scholar] [CrossRef]
- Turk, Z. Glycotoxines; carbonyl stress and relevance to diabetes and its complications. Physiol. Res. 2010, 59, 147–156. [Google Scholar]
- Tarwadi, K.V.; Agte, V.V. Effect of micronutrients on methylglyoxal-mediated in vitro glycation of albumin. Biol. Trace Elem. Res. 2011, 143, 717–725. [Google Scholar] [CrossRef]
- Lv, L.; Shao, X.; Chen, H.; Ho, C.T.; Sang, S. Genistein inhibits advanced glycation end product formation by trapping methylglyoxal. Chem. Res. Toxicol. 2011, 24, 579–586. [Google Scholar] [CrossRef]
- Ihnat, M.A.; Thorpe, J.E.; Ceriello, A. Hypothesis: The “metabolic memory”; the new challenge of diabetes. Diabet Med. 2007, 24, 582–586. [Google Scholar] [CrossRef]
- Bento, C.F.; Marques, F.; Fernandes, R.; Pereira, P. Methylglyoxal alters the function and stability of critical components of the protein quality control. PLoS One 2010, 5, e13007. [Google Scholar] [CrossRef]
- Vistoli, G.; de Maddis, D.; Cipak, A.; Zarkovic, N.; Carini, M.; Aldini, G. Advanced glycoxidation and lipoxidation end products (AGEs and ALEs): an overview of their mechanisms of formation. Free Radic. Res. 2013, 47 (Suppl. 1), 3–27. [Google Scholar]
- Muthenna, P.; Akileshwari, C.; Saraswat, M.; Bhanuprakash Reddy, G. Inhibition of advanced glycation end-product formation on eye lens protein by rutin. Br. J. Nutr. 2012, 107, 941–949. [Google Scholar] [CrossRef]
- Aldini, G.; Vistoli, G.; Stefek, M.; Chondrogianni, N.; Grune, T.; Sereikaite, J.; Sadowska-Bartosz, I.; Bartosz, G. Molecular strategies to prevent; inhibit; and degrade advanced glycoxidation and advanced lipoxidation end products. Free Radic. Res. 2013, 47 (Suppl. 1), 93–137. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Kiyota, N.; Tsurushima, K.; Yoshitomi, M.; Mera, K.; Sakashita, N.; Takeya, M.; Ikeda, T.; Araki, T.; Nohara, T.; et al. Natural compounds containing a catechol group enhance the formation of N′-(carboxymethyl)lysine of the Maillard reaction. Free Radic. Biol. Med. 2011, 50, 883–891. [Google Scholar] [CrossRef]
- Sadowska-Bartosz, I.; Adamczyk-Sowa, M.; Galiniak, S.; Mucha, S.; Pierzchala, K.; Bartosz, G. Oxidative modification of serum proteins in multiple sclerosis. Neurochem. Int. 2013, 6, 507–516. [Google Scholar]
- Kaufmann, E.; Boehm, B.O.; Süssmuth, S.D.; Kientsch-Engel, R.; Sperfeld, A.; Ludolph, A.C.; Tumani, H. The advanced glycation end-product N epsilon-(carboxymethyl)lysine level is elevated in cerebrospinal fluid of patients with amyotrophic lateral sclerosis. Neurosci. Lett. 2004, 371, 226–229. [Google Scholar] [CrossRef]
- Anguizola, J.; Matsuda, R.; Barnaby, O.S.; Hoy, K.S.; Wa, C.; DeBolt, E.; Koke, M.; Hage, D.S. Review: Glycation of human serum albumin. Clin. Chim. Acta 2013, 425, 64–76. [Google Scholar] [CrossRef]
- Bourdon, N.; Loreau, N.; Blache, D. Glucose and free radicals impair the antioxidant properties of serum albumin. FASEB J. 1999, 13, 233–244. [Google Scholar]
- Ramkissoon, J.S.; Mahomoodally, M.F.; Ahmed, N.; Subratty, A.H. Antioxidant and anti-glycationactivities correlates with phenolic composition of tropical medicinal herbs. Asian Pac. J. Trop. Med. 2013, 6, 561–569. [Google Scholar] [CrossRef]
- Rezg, R.; El-Fazaa, S.; Gharbi, N.; Mornagui, B. Bisphenol A and human chronic diseases: Current evidences; possible mechanisms; and future perspectives. Environ. Int. 2013, 64C, 83–90. [Google Scholar]
- Griffiths, H.R.; Lunec, J.; Blake, D.R. Oxygen radical induced fluorescence in proteins; identification of the fluorescent tryptophan metabolite; N-formyl kynurenine; as a biological index of radical damage. Amino Acids 1992, 3, 183–194. [Google Scholar] [CrossRef]
- Royer, C.A. Probing protein folding and conformational transitions with fluorescence. Chem. Rev. 2006, 106, 1769–1784. [Google Scholar] [CrossRef]
- Le Guen, C.A.; Bain, S.; Barnett, A.H.; Lunec, J. Captopril inhibits the fluorescence development associated with glycation of proteins. Agents Actions 1992, 36, 264–270. [Google Scholar]
- Jakus, V.; Hrnciarová, M.; Cársky, J.; Krahulec, B.; Rietbrock, N. Inhibition of nonenzymatic protein glycation and lipid peroxidation by drugs with antioxidant activity. Life Sci. 1999, 65, 1991–1993. [Google Scholar] [CrossRef]
- Fan, X.; Xiaoqin, L.; Potts, B.; Strauch, C.M.; Nemet, I.; Monnier, V.M. Topical application of l-arginine blocks advanced glycation by ascorbic acid in the lens of hSVCT2 transgenic mice. Mol. Vis. 2011, 17, 2221–2227. [Google Scholar]
- Tupe, R.; Agte, V.V. Interaction of zinc; ascorbic acid; and folic acid in glycation with albumin as protein model. Biol. Trace Elem. Res. 2010, 138, 346–357. [Google Scholar] [CrossRef]
- Krone, C.A.; Ely, J.T. Ascorbic acid; glycation; glycohemoglobin and aging. Med. Hypotheses 2004, 62, 275–279. [Google Scholar] [CrossRef]
- Vallianou, N.; Evangelopoulos, A.; Koutalas, P. Alpha-lipoic acid and diabetic neuropathy. Rev. Diabet. Stud. 2009, 6, 230–236. [Google Scholar] [CrossRef]
- Das, UN. Pyruvate is an endogenous anti-inflammatory and anti-oxidant molecule. Med. Sci. Monit. 2006, 12, RA79–RA84. [Google Scholar]
- Zhao, W.; Devamanoharan, P.S.; Varma, S.D. Fructose induced deactivation of antioxidant enzymes: Preventive effect of pyruvate. Free Radic. Res. 2000, 33, 23–30. [Google Scholar] [CrossRef]
- Hegde, K.R.; Varma, S.D. Prevention of cataract by pyruvate in experimentally diabetic mice. Mol. Cell. Biochem. 2005, 269, 115–120. [Google Scholar] [CrossRef]
- Akberova, S.I.; Musaev, P.I.; Magomedov, N.M.; Babaev, K.F.; Gakhramanov, K.M.; Stroeva, O.G. Para-aminobenzoic acid as an antioxidant. Dokl. Akad. Nauk 1998, 361, 419–421. [Google Scholar]
- Greenstock, C.L.; Miller, R.W. The oxidation of tiron by superoxide anion. Kinetics of the reaction in aqueous solution in chloroplasts. Biochim. Biophys. Acta 1975, 396, 11–16. [Google Scholar] [CrossRef]
- Krishna, C.M.; Liebmann, J.E.; Kaufman, D.; DeGraff, W.; Hahn, S.M.; McMurry, T.; Mitchell, J.B.; Russo, A. The catecholic metal sequestering agent 1;2-dihydroxybenzene-3;5-disulfonate confers protection against oxidative cell damage. Arch. Biochem. Biophys. 1992, 294, 98–106. [Google Scholar] [CrossRef]
- Soule, B.P.; Hyodo, F.; Matsumoto, K.; Simone, N.L.; Cook, J.A.; Krishna, M.C.; Mitchell, J.B. The chemistry and biology of nitroxide compounds. Free Radic. Biol. Med. 2007, 42, 1632–1650. [Google Scholar] [CrossRef]
- Bournival, J.; Francoeur, M.A.; Renaud, J.; Martinoli, M.G. Quercetin and sesamin protect neuronal PC12 cells from high-glucose-induced oxidation; nitrosative stress; and apoptosis. Rejuvenation Res. 2012, 15, 322–333. [Google Scholar] [CrossRef]
- Xie, Y.; Chen, X. Structures required of polyphenols for inhibiting advanced glycation end products formation. Curr. Drug Metab. 2013, 14, 414–431. [Google Scholar] [CrossRef]
- Obrenovich, M.E.; Nair, N.G.; Beyaz, A.; Aliev, G.; Reddy, V.P. The role of polyphenolic antioxidants in health; disease; and aging. Rejuvenation Res. 2010, 13, 631–643. [Google Scholar] [CrossRef]
- Razzaghi-Asl, N.; Garrido, J.; Khazraei, H.; Borges, F.; Firuzi, O. Antioxidant properties of hydroxycinnamic acids: A review of structure-activity relationships. Curr. Med. Chem. 2013, 20, 4436–4450. [Google Scholar] [CrossRef]
- Jang, D.S.; Yoo, N.H.; Kim, N.H.; Lee, Y.M.; Kim, C.S.; Kim, J.; Kim, J.H.; Kim, J.S. 3;5-Di-O-caffeoyl-epi-quinic acid from the leaves and stems of Erigeron annuus inhibits protein glycation; aldose reductase; and cataractogenesis. Biol. Pharm. Bull. 2010, 33, 329–333. [Google Scholar]
- Ishibashi, Y.; Matsui, T.; Takeuchi, M.; Yamagishi, S. Metformin inhibits advanced glycation end products (AGEs)-induced renal tubular cell injury by suppressing reactive oxygen species generation via reducing receptor for AGEs (RAGE) expression. Horm. Metab. Res. 2012, 44, 891–895. [Google Scholar] [CrossRef]
- Ahmad, S.; Shahab, U.; Baig, M.H.; Khan, M.S.; Khan, M.S.; Srivastava, A.K.; Saeed, M.; Moinuddin. Inhibitory effect of metformin and pyridoxamine in the formation of early; intermediate and advanced glycation end-products. PLoS One 2013, 8, e72128. [Google Scholar]
- Diamanti-Kandarakis, E.; Alexandraki, K.; Piper, C.; Aessopos, A.; Paterakis, T.; Katsikis, I.; Panidis, D. Effect of metformin administration on plasma advanced glycation end product levels in women with polycystic ovary syndrome. Metabolism 2007, 56, 29–134. [Google Scholar]
- Ganapathy-Kanniappan, S.; Kunjithapatham, R.; Geschwind, J.F. Anticancer efficacy of the metabolic blocker 3-bromopyruvate: Specific molecular targeting. Anticancer Res. 2013, 33, 13–20. [Google Scholar]
- Briski, K.P.; Patil, G.D. Induction of Fos immunoreactivity labeling in rat forebrain metabolic loci by caudal fourth ventricular infusion of the monocarboxylate transporter inhibitor; alpha-cyano-4-hydroxycinnamic acid. Neuroendocrinology 2005, 82, 49–57. [Google Scholar] [CrossRef]
- Thornalley, P.J.; Yurek-George, A.; Argirov, O.K. Kinetics and mechanism of the reaction of aminoguanidine with the alpha-oxoaldehydes glyoxal; methylglyoxal; and 3-deoxyglucosone under physiological conditions. Biochem. Pharmacol. 2000, 60, 55–65. [Google Scholar] [CrossRef]
- Baynes, J.W.; Murray, D.B. The metal chelators; trientine and citrate; inhibit the development of cardiac pathology in the Zucker diabetic rat. Exp. Diabetes Res. 2009, 2009, 696378. [Google Scholar]
- Mera, K.; Takeo, K.; Izumi, M.; Mruyama, T.; Nagai, R.; Otagiri, M. Effect of reactive-aldehydeson the modification and dysfunction of human serum albumin. J. Pharm. Sci. 2010, 99, 1614–1625. [Google Scholar] [CrossRef]
- Grimm, S.; Horlacher, M.; Catalgol, B.; Hoehn, A.; Reinheckel, T.; Grune, T. Cathepsins D and L reduce the toxicity of advanced glycation end products. Free Radic. Biol. Med. 2012, 52, 1011–1023. [Google Scholar] [CrossRef]
- Seneviratne, C.; Dombi, G.W.; Liu, W.; Dain, J.A. The in vitro glycation of human serum albumin in the presence of Zn(II). J. Inorg. Biochem. 2011, 105, 1548–1554. [Google Scholar] [CrossRef]
- Kang, Y.; Edwards, L.G.; Thornalley, P.J. Effect of methylglyoxal on human leukaemia 60 cell growth: modification of DNA G1 growth arrest and induction of apoptosis. Leuk. Res. 1996, 20, 397–405. [Google Scholar] [CrossRef]
- Meeprom, A.; Sompong, W.; Chan, C.B.; Adisakwattana, S. Isoferulic acid; a new anti-glycation agent; inhibits fructose- and glucose-mediated protein glycation in vitro. Molecules 2013, 18, 6439–6454. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Friedlander, M.; Capeillere-Blandin, C.; Nguyen-Khoa, T.; Nguyen, A.T.; Zingraff, J.; Jungers, P.; Descamps-Latscha, B. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996, 49, 1304–1313. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sadowska-Bartosz, I.; Galiniak, S.; Bartosz, G. Kinetics of Glycoxidation of Bovine Serum Albumin by Methylglyoxal and Glyoxal and its Prevention by Various Compounds. Molecules 2014, 19, 4880-4896. https://doi.org/10.3390/molecules19044880
Sadowska-Bartosz I, Galiniak S, Bartosz G. Kinetics of Glycoxidation of Bovine Serum Albumin by Methylglyoxal and Glyoxal and its Prevention by Various Compounds. Molecules. 2014; 19(4):4880-4896. https://doi.org/10.3390/molecules19044880
Chicago/Turabian StyleSadowska-Bartosz, Izabela, Sabina Galiniak, and Grzegorz Bartosz. 2014. "Kinetics of Glycoxidation of Bovine Serum Albumin by Methylglyoxal and Glyoxal and its Prevention by Various Compounds" Molecules 19, no. 4: 4880-4896. https://doi.org/10.3390/molecules19044880






