A Novel DNA Nanosensor Based on CdSe/ZnS Quantum Dots and Synthesized Fe3O4 Magnetic Nanoparticles
Abstract
:1. Introduction
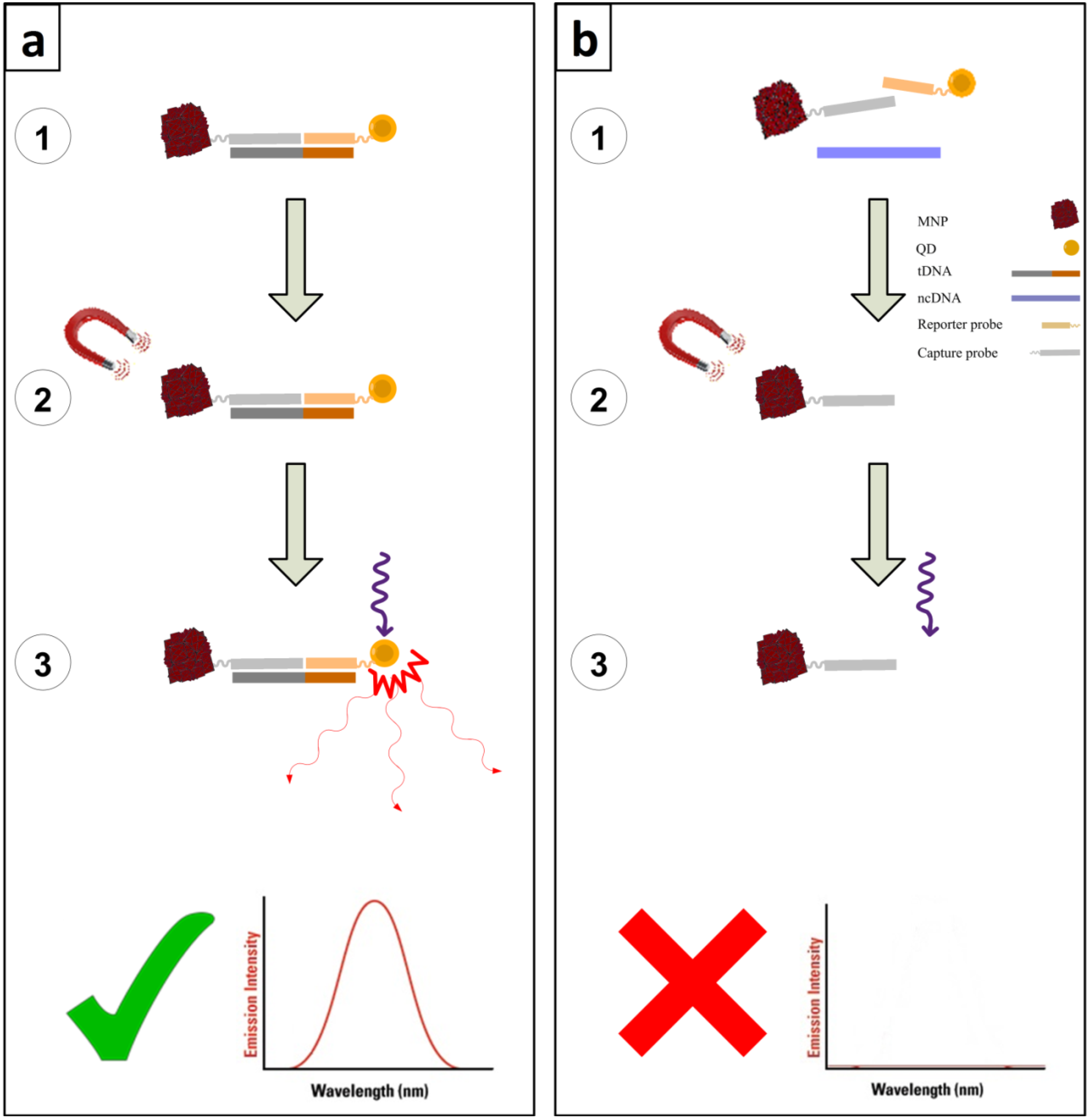
2. Results and Discussion

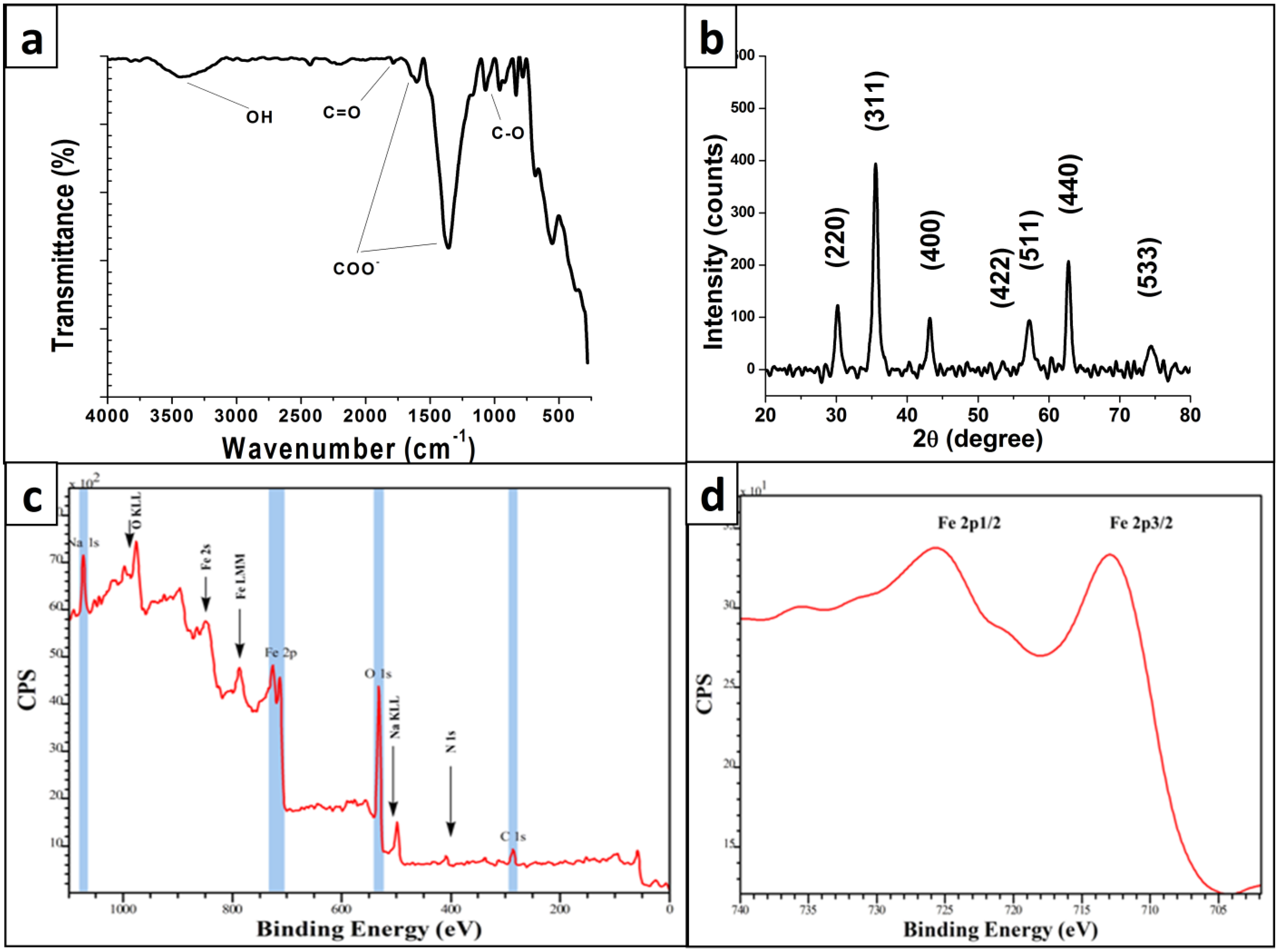

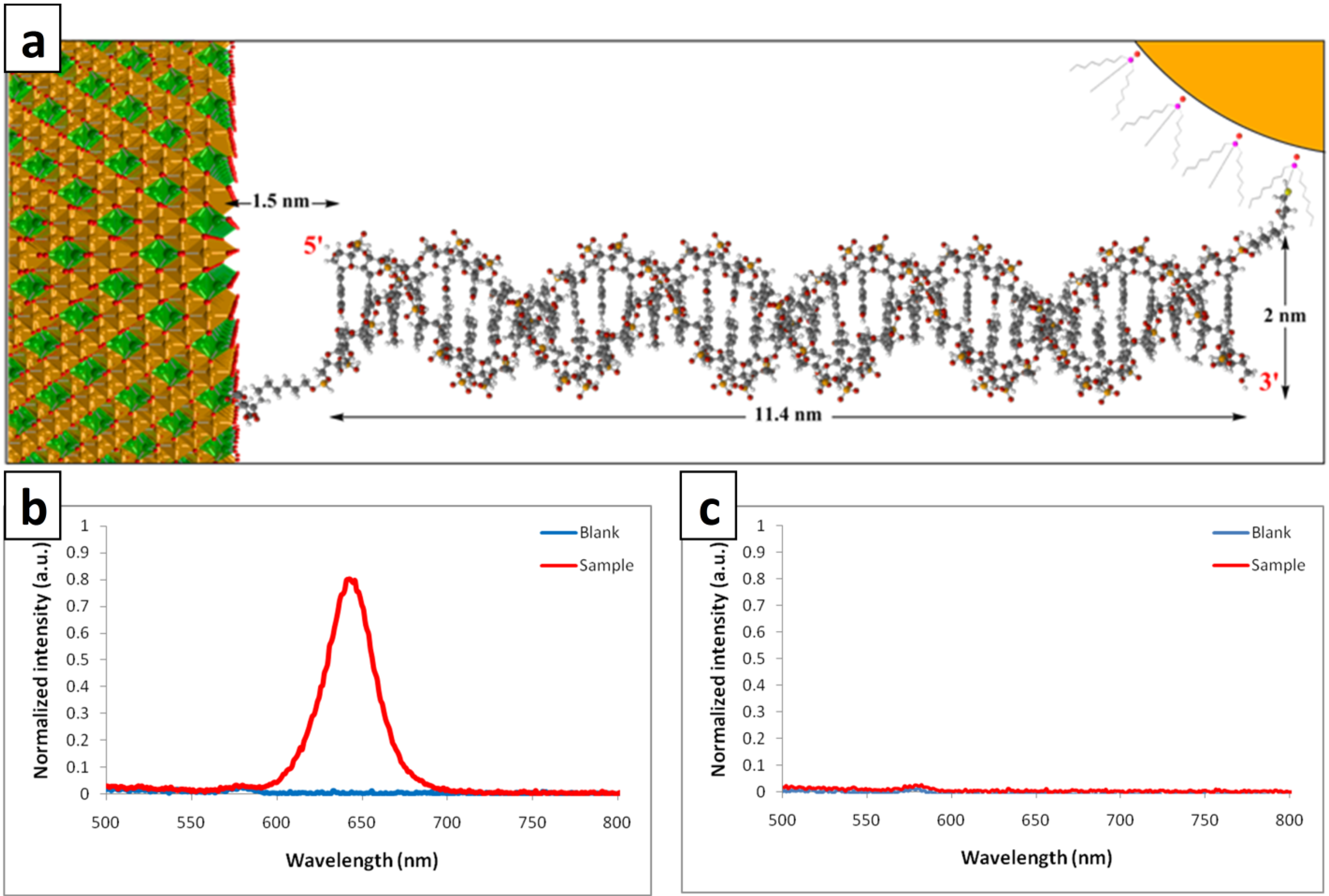
3. Experimental
3.1. Synthesis of Fe3O4 Magnetic Nanoparticles
3.2. Characterization of MNPs
3.3. Nucleic Acid Sequences
3.4. Surface Modification of Nanoparticles
3.5. Immobilization of DNA Sequences
3.6. DNA Hybridization
4. Conclusions
- Improve the biocompatibility and chemical stability, and tailor the dispersability and water solubility.
- Endow the iron oxide new physico-chemical properties, such as magnetic-optical properties, magnetic-electrical properties, magnetic-thermal properties, etc.
- Provide the iron oxide new functional end groups for the subsequent functionalized procedures or the subsequent applications, such as conjugation with the DNA, antibody, protein, etc.
Acknowledgments
Conflicts of Interest
References
- Wan, J.; Cai, W.; Meng, X.; Liu, E. Monodisperse water-soluble magnetite nanoparticles prepared by polyol process for high-performance magnetic resonance imaging. Chem. Commun. 2007, 2007, 5004–5006. [Google Scholar]
- Iida, H.; Takayanagi, K.; Nakanishi, T.; Osaka, T. Synthesis of Fe3O4 nanoparticles with various sizes and magnetic properties by controlled hydrolysis. J. Colloid Interf. Sci. 2007, 314, 274–280. [Google Scholar] [CrossRef]
- Neuberger, T.; Schöpf, B.; Hofmann, H.; Hofmann, M.; von Rechenberg, B. Superparamagnetic nanoparticles for biomedical applications: Possibilities and limitations of a new drug delivery system. J. Magn. Magn. Mater. 2005, 293, 483–496. [Google Scholar] [CrossRef]
- Veiseh, O.; Gunn, J.W.; Zhang, M. Design and fabrication of magnetic nanoparticles for targeted drug delivery and imaging. Adv. Drug Deliv. Rev. 2010, 62, 284–304. [Google Scholar] [CrossRef]
- Zhu, J.; Wei, S.; Chen, M.; Gu, H.; Rapole, S.B.; Pallavkar, S.; Ho, T.C.; Hopper, J.; Guo, Z. Magnetic nanocomposites for environmental remediation. Adv. Powder Technol. 2013, 24, 459–467. [Google Scholar] [CrossRef]
- Wang, S.X.; Bae, S.Y.; Li, G.; Sun, S.; White, R.L.; Kemp, J.T.; Webb, C.D. Towards a magnetic microarray for sensitive diagnostics. J. Magn. Magn. Mater. 2005, 293, 731–736. [Google Scholar] [CrossRef]
- Paleček, E.; Fojta, M. Magnetic beads as versatile tools for electrochemical DNA and protein biosensing. Talanta 2007, 74, 276–290. [Google Scholar] [CrossRef]
- Denkbaş, E.B.; Kiliçay, E.; Birlikseven, C.; Öztürk, E. Magnetic chitosan microspheres: Preparation and characterization. React. Funct. Polym. 2002, 50, 225–232. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef]
- Boyer, C.; Whittaker, M.R.; Bulmus, V.; Liu, J.; Davis, T.P. The design and utility of polymer-stabilized iron-oxide nanoparticles for nanomedicine applications. NPG Asia Mater. 2010, 2, 23–30. [Google Scholar] [CrossRef]
- Corchero, J.L.; Villaverde, A. Biomedical applications of distally controlled magnetic nanoparticles. Trends Biotechnol. 2009, 27, 468–476. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Thanh, N.K.T.; Jones, S.K.; Dobson, J. Progress in applications of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2009, 42, 224001. [Google Scholar] [CrossRef]
- Sun, S.; Zeng, H. Size-Controlled Synthesis of Magnetite Nanoparticles. J. Am. Chem. Soc. 2002, 124, 8204–8205. [Google Scholar] [CrossRef]
- Hergt, R.; Dutz, S.; Müller, R.; Zeisberger, M. Magnetic particle hyperthermia: Nanoparticle magnetism and materials development for cancer therapy. J. Phys. Condens Mat. 2006, 18, S2919. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses; Wiley-Vch: Weinheim, Berlin, Germany, 2003. [Google Scholar]
- Teja, A.S.; Koh, P.-Y. Synthesis, properties, and applications of magnetic iron oxide nanoparticles. Prog. Cryst. Growth Charact. Mater. 2009, 55, 22–45. [Google Scholar] [CrossRef]
- Majewski, P.; Thierry, B. Functionalized magnetite nanoparticles-synthesis, properties, and bio-applications. Crit. Rev. Solid State Mater. Sci. 2007, 32, 203–215. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. Crystal Structure. In The Iron Oxides; Wiley-VCH Verlag GmbH & Co. KGaA: Zurich, Switzerland, 2004; pp. 9–38. [Google Scholar]
- Johnson, B.K.; Prud’homme, R.K. Mechanism for Rapid Self-Assembly of Block Copolymer Nanoparticles. Phys. Rev. Lett. 2003, 91, 118302. [Google Scholar] [CrossRef]
- Katz, E.; Willner, I. Integrated Nanoparticle-Biomolecule Hybrid Systems: Synthesis, Properties, and Applications. Angew. Chem. Int. Edit. 2004, 43, 6042–6108. [Google Scholar] [CrossRef]
- Hui, C.; Shen, C.; Yang, T.; Bao, L.; Tian, J.; Ding, H.; Li, C.; Gao, H.J. Large-Scale Fe3O4 Nanoparticles Soluble in Water Synthesized by a Facile Method. J. Phys. Chem. C 2008, 112, 11336–11339. [Google Scholar]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef]
- Qiao, R.; Yang, C.; Gao, M. Superparamagnetic iron oxide nanoparticles: From preparations to in vivo MRI applications. J. Mater. Chem. 2009, 19, 6274–6293. [Google Scholar] [CrossRef]
- Hyeon, T. Chemical synthesis of magnetic nanoparticles. Chem. Commun. 2003, 2003, 927–934. [Google Scholar] [CrossRef]
- Dunin-Borkowski, R.E.; McCartney, M.R.; Frankel, R.B.; Bazylinski, D.A.; Pósfai, M.; Buseck, P.R. Magnetic microstructure of magnetotactic bacteria by electron holography. Science 1998, 282, 1868–1870. [Google Scholar] [CrossRef]
- Pan, Y.; Lin, W.; Li, J.; Wu, W.; Tian, L.; Deng, C.; Liu, Q.; Zhu, R.; Winklhofer, M.; Petersen, N. Reduced efficiency of magnetotaxis in magnetotactic coccoid bacteria in higher than geomagnetic fields. Biophys. J. 2009, 97, 986–991. [Google Scholar] [CrossRef]
- Cao, S.W.; Zhu, Y.J. Iron oxide hollow spheres: Microwave-hydrothermal ionic liquid preparation, formation mechanism, crystal phase and morphology control and properties. Acta Mater. 2009, 57, 2154–2165. [Google Scholar] [CrossRef]
- Wang, Y.; Maksimuk, S.; Shen, R.; Yang, H. Synthesis of iron oxide nanoparticles using a freshly-made or recycled imidazolium-based ionic liquid. Green Chem. 2007, 9, 1051–1056. [Google Scholar] [CrossRef]
- Bharde, A.; Wani, A.; Shouche, Y.; Joy, P.A.; Prasad, B.L.V.; Sastry, M. Bacterial Aerobic Synthesis of Nanocrystalline Magnetite. J. Am. Chem. Soc. 2005, 127, 9326–9327. [Google Scholar]
- Robinson, D.B.; Persson, H.H.J.; Zeng, H.; Li, G.; Pourmand, N.; Sun, S.; Wang, S.X. DNA-Functionalized MFe2O4 (M = Fe, Co, or Mn) Nanoparticles and Their Hybridization to DNA-Functionalized Surfaces. Langmuir 2005, 21, 3096–3103. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Fang, C.; Zhang, M. Multifunctional magnetic nanoparticles for medical imaging applications. J. Mater. Chem. 2009, 19, 6258–6266. [Google Scholar] [CrossRef]
- Dias, A.M.G.C.; Hussain, A.; Marcos, A.S.; Roque, A.C.A. A biotechnological perspective on the application of iron oxide magnetic colloids modified with polysaccharides. Biotechnol. Adv. 2011, 29, 142–155. [Google Scholar] [CrossRef]
- Tomitaka, A.; Ueda, K.; Yamada, T.; Takemura, Y. Heat dissipation and magnetic properties of surface-coated Fe3O4 nanoparticles for biomedical applications. J. Magn. Magn. Mater. 2012, 324, 3437–3442. [Google Scholar] [CrossRef]
- Tran, P.H.-L.; Tran, T.T.-D.; van Vo, T.; Lee, B.-J. Promising iron oxide-based magnetic nanoparticles in biomedical engineering. Arch. Pharm. Res. 2012, 35, 2045–2061. [Google Scholar] [CrossRef]
- Jolivet, J.-P.; Chanéac, C.; Tronc, E. Iron oxide chemistry. From molecular clusters to extended solid networks. Chem. Commun. 2004, 2004, 481–483. [Google Scholar] [CrossRef]
- Xie, J.; Sun, S. Monodisperse Magnetic Nanoparticles: Chemical Synthesis and Surface Modification. Nanomater. Inorg. Bioinorg. Perspect. 2013, 475. [Google Scholar] [CrossRef]
- Martínez-Mera, I.; Espinosa-Pesqueira, M.E.; Pérez-Hernández, R.; Arenas-Alatorre, J. Synthesis of magnetite (Fe3O4) nanoparticles without surfactants at room temperature. Mater. Lett. 2007, 61, 4447–4451. [Google Scholar] [CrossRef]
- Thiessen, W.; Dubavik, A.; Lesnyak, V.; Gaponik, N.; Eychmuller, A.; Wolff, T. Amphiphilic and magnetic behavior of Fe3O4 nanocrystals. Phys. Chem. Chem. Phys. 2010, 12, 2063–2066. [Google Scholar] [CrossRef]
- Ji, L.; Zhou, L.; Bai, X.; Shao, Y.; Zhao, G.; Qu, Y.; Wang, C.; Li, Y. Facile synthesis of multiwall carbon nanotubes/iron oxides for removal of tetrabromobisphenol A and Pb(ii). J. Mater. Chem. 2012, 22, 15853–15862. [Google Scholar]
- Lai, Y.; Yin, W.; Liu, J.; Xi, R.; Zhan, J. One-Pot Green Synthesis and Bioapplication of l-Arginine-Capped Superparamagnetic Fe3O4 Nanoparticles. Nanoscale Res. Lett. 2010, 5, 302–307. [Google Scholar] [CrossRef]
- Yongxiong, L.; Claydon, J.S.; Ahmad, E.; Yongbing, X.; Thompson, S.M.; Wilson, K.; van der Laan, G. XPS and XMCD study of Fe3O4/GaAs interface. IEEE Trans. Magn. 2005, 41, 2808–2810. [Google Scholar] [CrossRef]
- Mills, P.; Sullivan, J.L. A study of the core level electrons in iron and its three oxides by means of X-ray photoelectron spectroscopy. J. Phys. D Appl. Phys. 1983, 16, 723. [Google Scholar] [CrossRef]
- Abbas, M.; Parvatheeswara Rao, B.; Naga, S.M.; Takahashi, M.; Kim, C. Synthesis of high magnetization hydrophilic magnetite (Fe3O4) nanoparticles in single reaction—Surfactantless polyol process. Ceram. Int. 2013, 39, 7605–7611. [Google Scholar] [CrossRef]
- Parkinson, G.S.; Novotny, Z.; Argentero, G.; Schmid, M.; Pavelec, J.; Kosak, R.; Blaha, P.; Diebold, U. Carbon monoxide-induced adatom sintering in a Pd-Fe3O4 model catalyst. Nat. Mater. 2013, 12, 724–728. [Google Scholar] [CrossRef]
- Hushiarian, R.; Yusof, N.A.; Dutse, S.W. Detection and control of Ganoderma boninense: Strategies and perspectives. SpringerPlus 2013, 2, 555. [Google Scholar] [CrossRef]
- Patolsky, F.; Gill, R.; Weizmann, Y.; Mokari, T.; Banin, U.; Willner, I. Lighting-up the dynamics of telomerization and DNA replication by CdSe-ZnS quantum dots. J. Am. Chem. Soc. 2003, 125, 13918–13919. [Google Scholar] [CrossRef]
- Wang, X.; Lou, X.; Wang, Y.; Guo, Q.; Fang, Z.; Zhong, X.; Mao, H.; Jin, Q.; Wu, L.; Zhao, H.; et al. QDs-DNA nanosensor for the detection of hepatitis B virus DNA and the single-base mutants. Biosens. Bioelectron. 2010, 25, 1934–1940. [Google Scholar] [CrossRef]
- Zhang, C.-Y.; Yeh, H.-C.; Kuroki, M.T.; Wang, T.-H. Single-quantum-dot-based DNA nanosensor. Nat. Mater. 2005, 4, 826–831. [Google Scholar] [CrossRef]
- Dong, X.-Y.; Mi, X.-N.; Wang, B.; Xu, J.-J.; Chen, H.-Y. Signal amplification for DNA detection based on the HRP-functionalized Fe3O4 nanoparticles. Talanta 2011, 84, 531–537. [Google Scholar] [CrossRef]
- Sharon, E.; Freeman, R.; Willner, I. CdSe/ZnS quantum dots-G-quadruplex/hemin hybrids as optical DNA sensors and aptasensors. Anal. Chem. 2010, 82, 7073–7077. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compound Fe3O4 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hushiarian, R.; Yusof, N.A.; Abdullah, A.H.; Ahmad, S.A.A.; Dutse, S.W. A Novel DNA Nanosensor Based on CdSe/ZnS Quantum Dots and Synthesized Fe3O4 Magnetic Nanoparticles. Molecules 2014, 19, 4355-4368. https://doi.org/10.3390/molecules19044355
Hushiarian R, Yusof NA, Abdullah AH, Ahmad SAA, Dutse SW. A Novel DNA Nanosensor Based on CdSe/ZnS Quantum Dots and Synthesized Fe3O4 Magnetic Nanoparticles. Molecules. 2014; 19(4):4355-4368. https://doi.org/10.3390/molecules19044355
Chicago/Turabian StyleHushiarian, Roozbeh, Nor Azah Yusof, Abdul Halim Abdullah, Shahrul Ainliah Alang Ahmad, and Sabo Wada Dutse. 2014. "A Novel DNA Nanosensor Based on CdSe/ZnS Quantum Dots and Synthesized Fe3O4 Magnetic Nanoparticles" Molecules 19, no. 4: 4355-4368. https://doi.org/10.3390/molecules19044355





