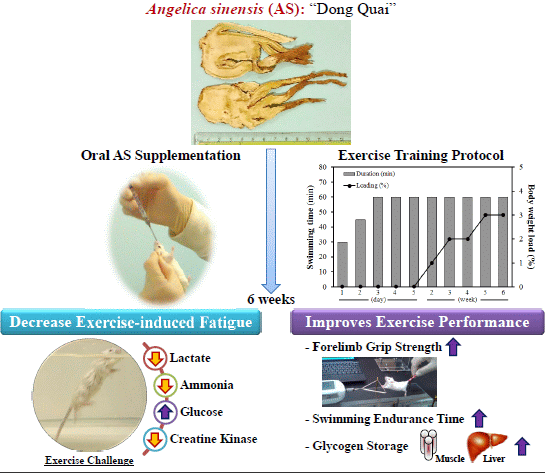
Angelica sinensis Improves Exercise Performance and Protects against Physical Fatigue in Trained Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Body Weight and Other Metabolism-Related Organ Weights

| Characteristic | Vehicle Control | Exercise Control | Ex-AS1 | Ex-AS5 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Weight (g) | ||||||||||||||||
| Liver | 2.16 | ± | 0.03 | 2.14 | ± | 0.03 | 2.11 | ± | 0.04 | 2.14 | ± | 0.04 | ||||
| Kidney | 0.61 | ± | 0.01 | 0.65 | ± | 0.02 | 0.64 | ± | 0.01 | 0.66 | ± | 0.01 | ||||
| EFP | 0.52 | ± | 0.03 | 0.47 | ± | 0.02 | 0.44 | ± | 0.04 | 0.46 | ± | 0.04 | ||||
| Muscle | 0.36 | ± | 0.01 | 0.36 | ± | 0.01 | 0.36 | ± | 0.01 | 0.38 | ± | 0.00 | ||||
| BAT | 0.13 | ± | 0.01 | 0.15 | ± | 0.01 | 0.15 | ± | 0.01 | 0.13 | ± | 0.01 | ||||
| Relative weight (%) | ||||||||||||||||
| Liver | 6.07 | ± | 0.07 | 5.97 | ± | 0.07 | 5.93 | ± | 0.08 | 5.84 | ± | 0.13 | ||||
| Kidney | 1.72 | ± | 0.03 | 1.81 | ± | 0.04 | 1.81 | ± | 0.03 | 1.81 | ± | 0.03 | ||||
| EFP | 1.45 | ± | 0.06 | 1.30 | ± | 0.07 | 1.25 | ± | 0.10 | 1.25 | ± | 0.10 | ||||
| Muscle | 1.01 | ± | 0.02 | 1.02 | ± | 0.02 | 1.02 | ± | 0.03 | 1.03 | ± | 0.02 | ||||
| BAT | 0.37 | ± | 0.01 | 0.42 | ± | 0.02 | 0.41 | ± | 0.02 | 0.37 | ± | 0.02 | ||||
2.2. Effect of AS on Forelimb Grip Strength
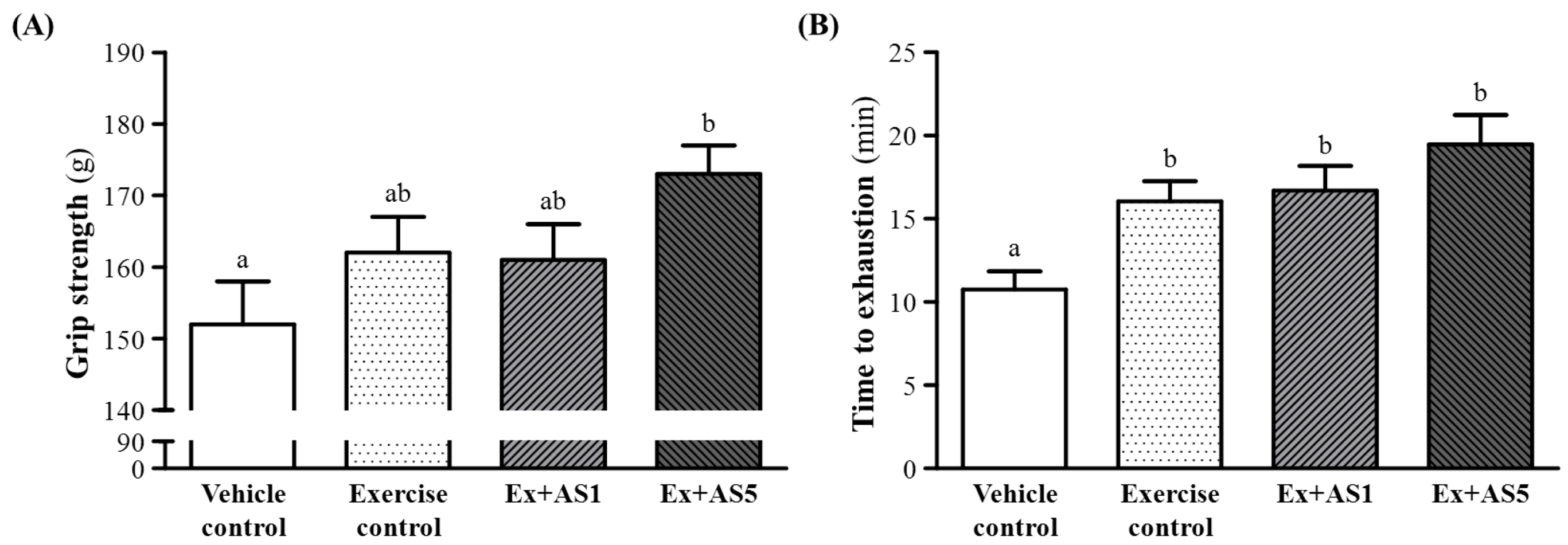
2.3. Effect of AS on Exercise Performance in a Weight-Loaded Swimming Test
2.4. Effect of Exercise Combined with AS Supplementation on Mouse Serum Lactate, Ammonia, and Glucose Levels and CK Activity after Acute Exercise Challenge
2.5. Effect of AS Supplementation on Hepatic and Muscle Glycogen Level

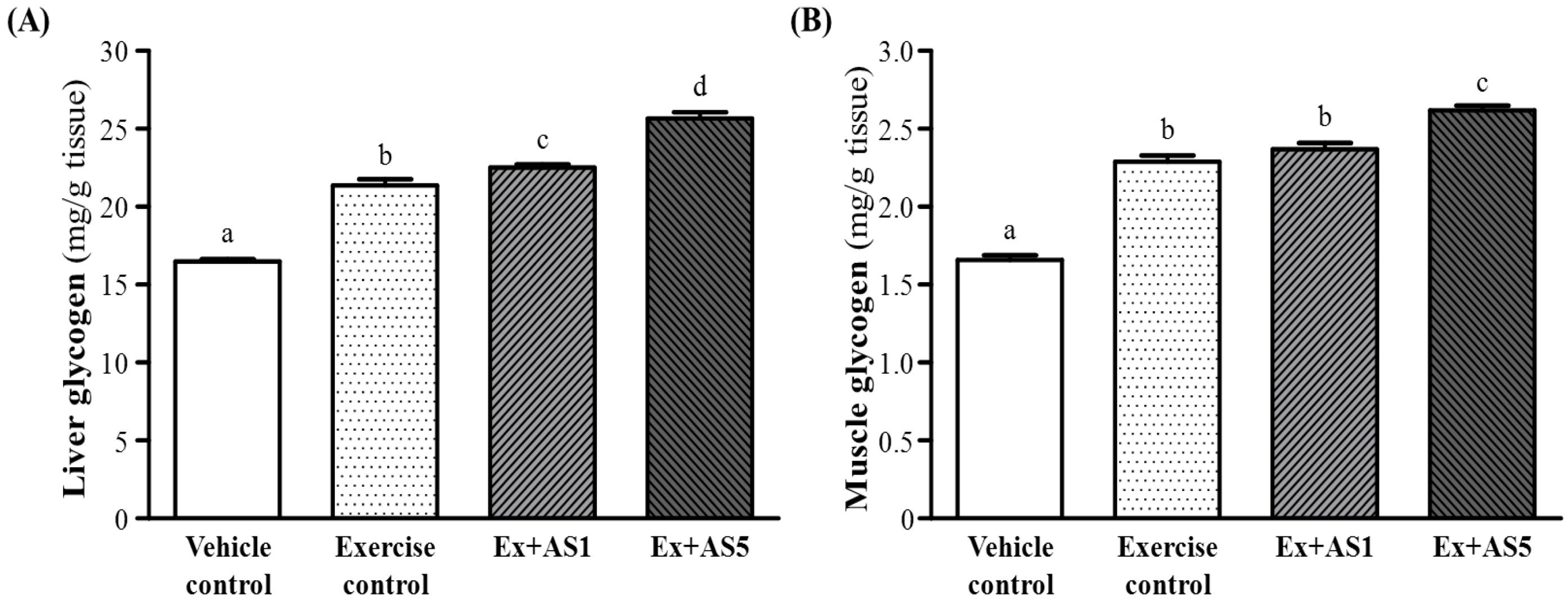
2.6. Effect of AS Supplementation on Biochemical Analyses at the End of the Experiment
| Parameter | Vehicle Control | Exercise Control | Ex-AS1 | Ex-AS5 |
|---|---|---|---|---|
| AST (U/L) | 63 ± 3 | 69 ± 4 | 66 ± 3 | 67 ± 3 |
| ALT (U/L) | 42 ± 3 a | 54 ± 2 b | 47 ± 2 ab | 48 ± 3 ab |
| ALP (U/L) | 49 ± 3 | 63 ± 6 | 59 ± 3 | 54 ± 2 |
| LDH (U/L) | 301 ± 19 | 273 ± 23 | 304 ± 15 | 297 ± 20 |
| Albumin (g/dL) | 3.6 ± 0.1 | 3.8 ± 0.1 | 3.7 ± 0.1 | 3.7 ± 0.0 |
| TBIL (μg/dL) | 0.19 ± 0.03 | 0.22 ± 0.03 | 0.23 ± 0.03 | 0.20 ± 0.02 |
| TP (g/dL) | 4.7 ± 0.1 | 4.6 ± 0.1 | 4.7 ± 0.1 | 4.8 ± 0.1 |
| BUN (mg/dL) | 23.4 ± 0.8 | 23.1 ± 1.0 | 23.8 ± 1.2 | 21.9 ± 0.8 |
| Creatinine (mg/dL) | 0.13 ± 0.01 | 0.12 ± 0.01 | 0.14 ± 0.01 | 0.15 ± 0.01 |
| UA (mg/dL) | 1.43 ± 0.11 b | 0.83 ± 0.05 a | 1.45 ± 0.07 b | 1.10 ± 0.07 ab |
| TG (mg/dL) | 228 ± 21 b | 184 ± 20 ab | 147 ± 8 a | 164 ± 11 ab |
| TC (mg/dL) | 111 ± 4 | 104 ± 4 | 122 ± 5 | 122 ± 5 |
| Glucose (mg/dL) | 180 ± 6 | 182 ± 7 | 185 ± 6 | 175 ± 5 |
2.7. Effect of AS Supplementation on Histological Examinations at the End of the Experiment
2.8. Discussion

3. Experimental
3.1. Experimental Design
3.2. Swimming Exercise Training
3.3. Swim to Exhaustion Exercise Test
3.4. Forelimb Grip Strength
3.5. Determination of Blood Biochemical Variables
3.6. Tissue Glycogen Determination
3.7. Histology Staining of Tissues
3.8. Analysis of Ferulic Acid of AS by HPLC/CAD
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mehta, R.K.; Agnew, M.J. Influence of mental workload on muscle endurance, fatigue, and recovery during intermittent static work. Eur. J. Appl. Physiol. 2012, 112, 2891–2902. [Google Scholar] [CrossRef]
- Endoh, T.; Nakajima, T.; Sakamoto, M.; Komiyama, T. Effects of muscle damage induced by eccentric exercise on muscle fatigue. Med. Sci. Sports Exerc. 2005, 37, 1151–1156. [Google Scholar]
- Fitts, R.H. Cellular mechanisms of muscle fatigue. Physiol. Rev. 1994, 74, 49–94. [Google Scholar]
- Davis, J.M.; Bailey, S.P. Possible mechanisms of central nervous system fatigue during exercise. Med. Sci. Sports Exerc. 1997, 29, 45–57. [Google Scholar] [CrossRef]
- Coombes, J.S.; Rowell, B.; Dodd, S.L.; Demirel, H.A.; Naito, H.; Shanely, R.A.; Powers, S.K. Effects of vitamin E deficiency on fatigue and muscle contractile properties. Eur. J. Appl. Physiol. 2002, 87, 272–277. [Google Scholar] [CrossRef]
- You, L.; Zhao, M.; Regenstein, J.M.; Ren, J. In vitro antioxidant activity and in vivo anti-fatigue effect of loach (Misgurnus anguillicaudatus) peptides prepared by papain digestion. Food Chem. 2011, 124, 188–194. [Google Scholar] [CrossRef]
- Hsu, C.C.; Ho, M.C.; Lin, L.C.; Su, B.; Hsu, M.C. American ginseng supplementation attenuate creatine kinase level induces by submaximal exercise in humans. World J. Gastroenterol. 2005, 11, 5327–5331. [Google Scholar]
- Yeh, T.S.; Chuang, H.L.; Huang, W.C.; Chen, Y.M.; Huang, C.C.; Hsu, M.C. Astragalus. membranaceus improves exercise performance and ameliorates exercise-induced fatigue in trained mice. Molecules 2014, 19, 2793–2807. [Google Scholar] [CrossRef]
- Yeh, T.S.; Chan, K.H.; Hsu, M.C.; Liu, J.F. Supplementation with soybean peptides, taurine, Pueraria isoflavone, and ginseng saponin complex improves endurance exercise capacity in humans. J. Med. Food 2011, 14, 219–225. [Google Scholar] [CrossRef]
- Huang, C.C.; Hsu, M.C.; Huang, W.C.; Yang, H.R.; Hou, C.C. Triterpenoid-rich extract from Antrodia camphorata improves physical fatigue and exercise performance in mice. Evid. Based Complement. Alternat. Med. 2012, 2012, 364741. [Google Scholar]
- Li, W.; Wu, Y.; Liu, X.; Yan, C.; Liu, D.; Pan, Y.; Yang, G.; Yin, F.; Weng, Z.; Zhao, D.; et al. Antioxidant Properties of cis-Z,Z'-3a.7a',7a.3a'-Dihydroxy-ligustilide on Human Umbilical Vein Endothelial Cells in Vitro. Molecules 2013, 18, 520–534. [Google Scholar] [CrossRef]
- Zhang, W.L.; Zheng, K.Y.; Zhu, K.Y.; Zhan, J.Y.; Bi, C.W.; Chen, J.P.; Dong, T.T.; Choi, R.C.; Lau, D.T.; Tsim, K.W. Chemical and biological assessment of angelica roots from different cultivated regions in a chinese herbal decoction danggui buxue tang. Evid. Based Complement. Alternat. Med. 2013, 2013, 483286. [Google Scholar]
- Zhao, K.J.; Dong, T.T.; Tu, P.F.; Song, Z.H.; Lo, C.K.; Tsim, K.W. Molecular Genetic and Chemical Assessment of Radix Angelica (Danggui) in China. J. Agric. Food Chem. 2003, 51, 2576–2583. [Google Scholar] [CrossRef]
- Liu, J.Y.; Zhang, Y.; You, R.X.; Zeng, F.; Guo, D.; Wang, K.P. Polysaccharide isolated from Angelica sinensis inhibits hepcidin expression in rats with iron deficiency anemia. J. Med. Food 2012, 15, 923–929. [Google Scholar] [CrossRef]
- Hou, Y.Z.; Zhao, G.R.; Yang, J.; Yuan, Y.J.; Zhu, G.G.; Hiltunen, R. Protective effect of Ligusticum chuanxiong and Angelica sinensis on endothelial cell damage induced by hydrogen peroxide. Life Sci. 2004, 75, 1775–1786. [Google Scholar] [CrossRef]
- Zhuang, S.R.; Chiu, H.F.; Chen, S.L.; Tsai, J.H.; Lee, M.Y.; Lee, H.S.; Shen, Y.C.; Yan, Y.Y.; Shane, G.T.; Wang, C.K. Effects of a Chinese medical herbs complex on cellular immunity and toxicity-related conditions of breast cancer patients. Br. J. Nutr. 2012, 107, 712–718. [Google Scholar] [CrossRef]
- Yang, T.; Jia, M.; Zhou, S.; Pan, F.; Mei, Q. Antivirus and immune enhancement activities of sulfated polysaccharide from Angelica sinensis. Int. J. Biol. Macromol. 2012, 50, 768–772. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, H.G.; Li, X.H. A Chinese herbal decoction, Danggui Buxue Tang, improves chronic fatigue syndrome induced by food restriction and forced swimming in rats. Phytother. Res. 2011, 25, 1825–1832. [Google Scholar] [CrossRef]
- Bucci, L.R. Selected herbals and human exercise performance. Am. J. Clin. Nutr. 2000, 72, 624S–636S. [Google Scholar]
- Farnsworth, N.R.; Krause, E.C.; Bolton, J.L.; Pauli, G.F.; van Breemen, R.B.; Graham, J.G. The University of Illinois at Chicago/National Institutes of Health Center for Botanical Dietary Supplements Research for Women’s Health: From plant to clinical use. Am. J. Clin. Nutr. 2008, 87, 504S–508S. [Google Scholar]
- Gibson, H.; Edwards, R.H. Muscular exercise and fatigue. Sports Med. 1985, 2, 120–132. [Google Scholar] [CrossRef]
- Sahlin, K.; Tonkonogi, M.; Söderlund, K. Energy supply and muscle fatigue in humans. Acta Physiol. Scand. 1998, 162, 261–266. [Google Scholar] [CrossRef]
- Young, A.J.; Castellani, J.W. Exertion-induced fatigue and thermoregulation in the cold. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 128, 769–776. [Google Scholar] [CrossRef]
- Yang, T.; Jia, M.; Meng, J.; Wu, H.; Mei, Q. Immunomodulatory activity of polysaccharide isolated from Angelica sinensis. Int. J. Biol. Macromol. 2006, 39, 179–184. [Google Scholar] [CrossRef]
- Nieman, D.C.; Pedersen, B.K. Review exercise and immune function. Recent developments. Sports Med. 1999, 27, 73–80. [Google Scholar] [CrossRef]
- Gunzer, W.; Konrad, M.; Pail, E. Exercise-induced immunodepression in endurance athletes and nutritional intervention with carbohydrate, protein and fat-what is possible, what is not? Nutrients 2012, 4, 1187–1212. [Google Scholar] [CrossRef]
- Xie, J.J.; Lu, J.; Qian, Z.M.; Yu, Y.; Duan, J.A.; Li, S.P. Optimization and comparison of five methods for extraction of coniferyl ferulate from Angelica sinensis. Molecules 2009, 14, 555–565. [Google Scholar] [CrossRef]
- You, Y.; Park, J.; Yoon, H.G.; Lee, Y.H.; Hwang, K.; Lee, J.; Kim, K.; Lee, K.W.; Shim, S.; Jun, W. Stimulatory effects of ferulic acid on endurance exercise capacity in mice. Biosci. Biotechnol. Biochem. 2009, 73, 1392–1397. [Google Scholar] [CrossRef]
- Warren, G.L.; Ingalls, C.P.; Lowe, D.A.; Armstrong, R.B. Excitation-contraction uncoupling: Major role in contraction-induced muscle injury. Exerc. Sport Sci. Rev. 2001, 29, 82–87. [Google Scholar] [CrossRef]
- Murase, T.; Haramizu, S.; Shimotoyodome, A.; Tokimitsu, I.; Hase, T. Green tea extract improves running endurance in mice by stimulating lipid utilization during exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R1550–R1556. [Google Scholar] [CrossRef]
- Calders, P.; Matthys, D.; Derave, W.; Pannier, J.L. Effect of branched-chain amino acids (BCAA), glucose, and glucose plus BCAA on endurance performance in rats. Med. Sci. Sports Exerc. 1999, 31, 583–587. [Google Scholar] [CrossRef]
- Cairns, S.P. Lactic acid and exercise performance: Culprit or friend? Sports Med. 2006, 36, 279–291. [Google Scholar] [CrossRef]
- Sánchez-Medina, L.; González-Badillo, J.J. Velocity loss as an indicator of neuromuscular fatigue during resistance training. Med. Sci. Sports Exerc. 2011, 43, 1725–1734. [Google Scholar] [CrossRef]
- Rico-Sanz, J.; Mendez Marco, M.T. Creatine enhances oxygen uptake and performance during alternating intensity exercise. Med. Sci. Sports Exerc. 2000, 32, 379–385. [Google Scholar] [CrossRef]
- Chen, W.C.; Huang, W.C.; Chiu, C.C.; Chang, Y.K.; Huang, C.C. Whey Protein improves exercise performance and biochemical profiles in trained mice. Med. Sci. Sports Exerc. 2014. [Google Scholar] [CrossRef]
- Matsumoto, K.; Ishihara, K.; Tanaka, K.; Inoue, K.; Fushiki, T. An adjustable-current swimming pool for the evaluation of endurance capacity of mice. J. Appl. Physiol. 1985, 81, 1843–1849. [Google Scholar]
- Wang, S.Y.; Huang, W.C.; Liu, C.C.; Wang, M.F.; Ho, C.S.; Huang, W.P.; Hou, C.C.; Chuang, H.L.; Huang, C.C. Pumpkin (Cucurbita moschata) fruit extract improves physical fatigue and exercise performance in mice. Molecules 2012, 17, 11864–11876. [Google Scholar] [CrossRef]
- Wu, R.E.; Huang, W.C.; Liao, C.C.; Chang, Y.K.; Kan, N.W.; Huang, C.C. Resveratrol protects against physical fatigue and improves exercise performance in mice. Molecules 2013, 18, 4689–4702. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yeh, T.-S.; Huang, C.-C.; Chuang, H.-L.; Hsu, M.-C. Angelica sinensis Improves Exercise Performance and Protects against Physical Fatigue in Trained Mice. Molecules 2014, 19, 3926-3939. https://doi.org/10.3390/molecules19043926
Yeh T-S, Huang C-C, Chuang H-L, Hsu M-C. Angelica sinensis Improves Exercise Performance and Protects against Physical Fatigue in Trained Mice. Molecules. 2014; 19(4):3926-3939. https://doi.org/10.3390/molecules19043926
Chicago/Turabian StyleYeh, Tzu-Shao, Chi-Chang Huang, Hsiao-Li Chuang, and Mei-Chich Hsu. 2014. "Angelica sinensis Improves Exercise Performance and Protects against Physical Fatigue in Trained Mice" Molecules 19, no. 4: 3926-3939. https://doi.org/10.3390/molecules19043926