N-Substituted 2-Isonicotinoylhydrazinecarboxamides — New Antimycobacterial Active Molecules
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry

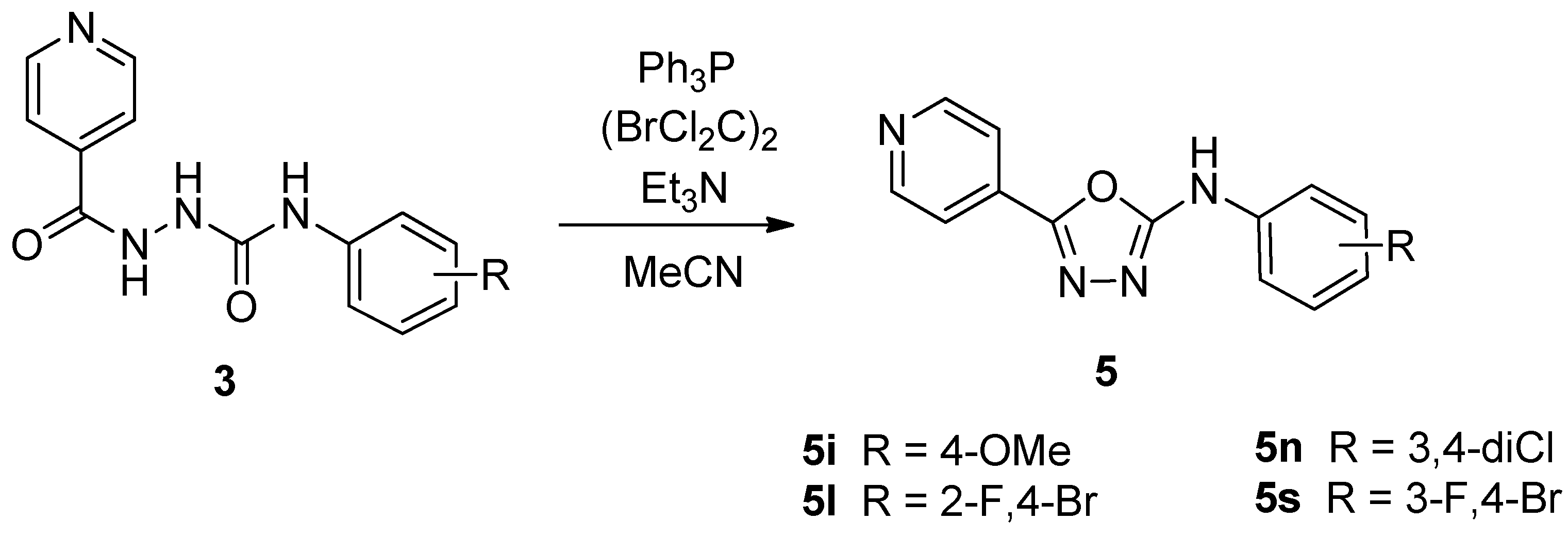
2.2. Antimycobacterial Activity
| Code | R | MIC [μM] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M. tuberculosis | M. avium | M. kansasii | M. kansasii | C logP | ||||||||
| 14 d | 21 d | 14 d | 21 d | 7 d | 14 d | 21 d | 7 d | 14 d | 21 d | |||
| 3a | 4-methyl | 62.5 | 62.5 | 16 | 32 | 62.5 | 125 | >125 | 62.5 | 62.5 | 125 | 1.13 |
| 3b | 4-isopropyl | 16 | 32 | 16 | 32 | 16 | 16 | 32 | 8 | 8 | 16 | 1.87 |
| 3c | 4-tert-butyl | 8 | 16 | 62.5 | 125 | 32 | 62.5 | 125 | 32 | 62.5 | 62.5 | 2.34 |
| 3d | 4-n-butyl | 4 | 8 | 62.5 | 125 | 62.5 | 125 | 125 | 32 | 62.5 | 62.5 | 2.38 |
| 3e | 4-pentyl | 32 | 62.5 | 62.5 | 125 | 32 | 62.5 | 125 | 32 | 62.5 | 125 | 2.79 |
| 3f | 4-hexyl | 32 | 62.5 | 62.5 | 62.5 | 32 | 125 | 125 | 62.5 | 125 | 125 | 3.21 |
| 3g | 4-heptyl | 16 | 32 | 32 | 62.5 | 32 | 125 | 125 | 62.5 | 62.5 | 125 | 3.63 |
| 3h | 4-octyl | 1 | 2 | 250 | 500 | 250 | 500 | >500 | 125 | 250 | 500 | 4.05 |
| 3i | 4-OMe | 62.5 | 62.5 | 32 | 62.5 | >125 | >125 | >125 | >125 | >125 | >125 | 0.51 |
| 3j | 4-CF3 | 62.5 | 62.5 | >125 | >125 | >125 | >125 | >125 | 125 | 125 | 125 | 1.56 |
| 3k | 2,4-diBr | 8 | 8 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 2.3 |
| 3l | 2-F,4-Br | 32 | 32 | >125 | >125 | >125 | >125 | >125 | 125 | >125 | >125 | 1.63 |
| 3m | 2-Br,4-F | 16 | 16 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 1.63 |
| 3n | 3,4-diCl | 125 | 125 | >125 | >125 | 125 | 125 | 125 | 125 | 125 | 125 | 1.75 |
| 3o | 3-Cl,4-F | 62.5 | 125 | >62.5 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 1.35 |
| 3p | 3-CF3,4-Cl | 32 | 32 | 125 | 125 | 62.5 | 125 | 125 | 62.5 | 125 | 125 | 2.12 |
| 3q | 3-CF3,4-Br | 8 | 8 | >125 | >125 | >125 | >125 | >125 | 125 | 125 | 125 | 2.39 |
| 3r | 3-CF3,4-F | 32 | 32 | >125 | >125 | 125 | 125 | 125 | 62.5 | 62.5 | 125 | 1.72 |
| 3s | 3-F,4-Br | 62.5 | 62.5 | >125 | >125 | 125 | >125 | >125 | 125 | 125 | >125 | 1.63 |
| 3t | 3,5-diCl | 32 | 62.5 | 125 | 125 | 125 | 125 | 125 | 125 | 125 | 125 | 1.75 |
| 3u | 2,4,6-triCl | 4 | 4 | >125 | >125 | >125 | >125 | >125 | 125 | 125 | 125 | 2.31 |
 | ||||||||||||
| 3v | - | 16 | 32 | 32 | 62.5 | 32 | 125 | 125 | 32 | 62.5 | 125 | 1.47 |
 | ||||||||||||
| 5i | 4-OMe | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 2.2 |
| 5l | 2-F,4-Br | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 3.31 |
| 5n | 3,4-diCl | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 3.44 |
| 5s | 3-F,4-Br | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | >125 | 3.31 |
| INH | 0.5 | 0.5-1 | >250 | >250 | >250 | >250 | >250 | 4 | 8 | 8 | −0.6 | |
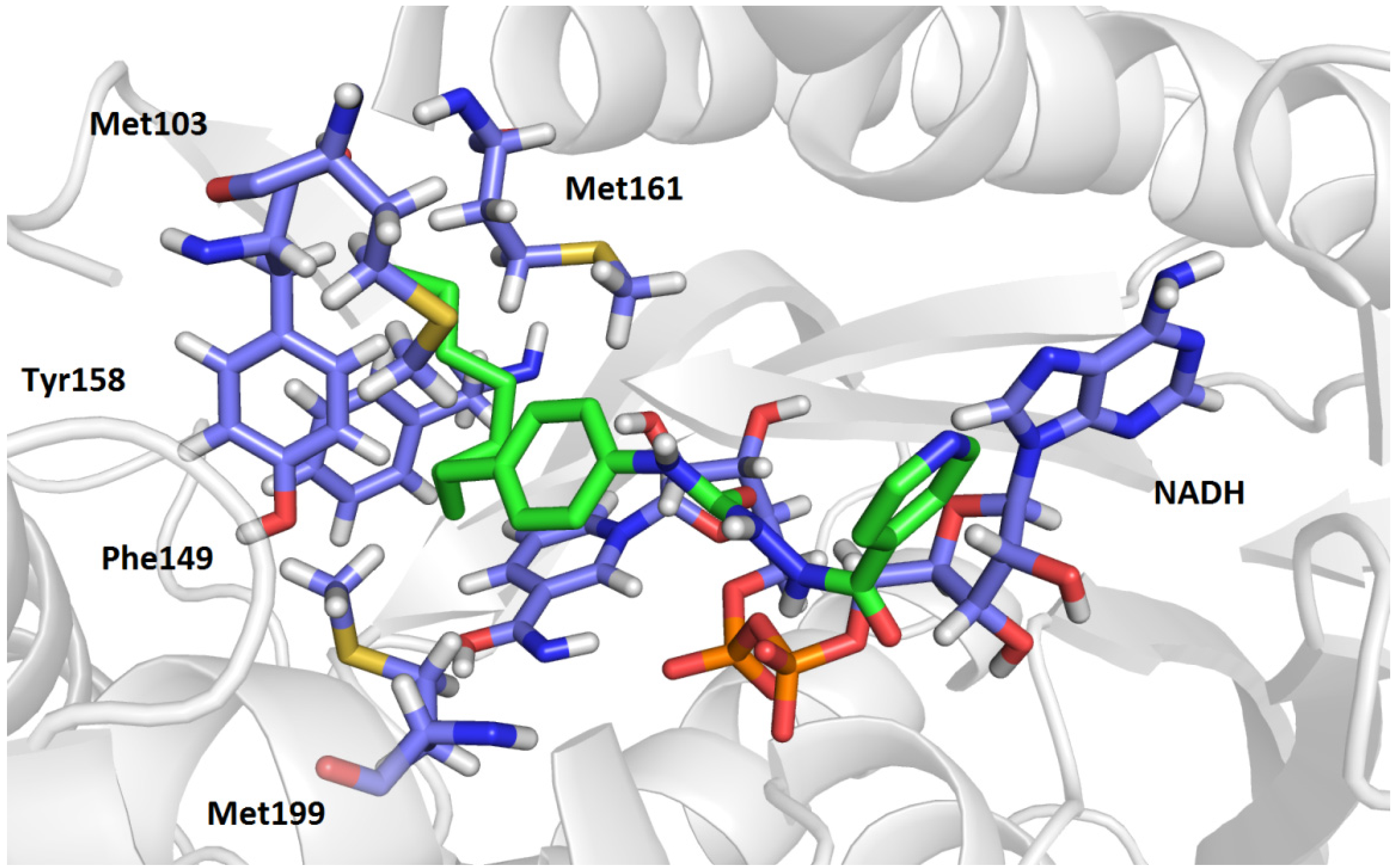
2.3. Molecular Modelling Studies
3. Experimental
3.1. General Information
3.2. Chemistry
3.2.1. Synthesis of Isonicotinoylhydrazinecarboxamides 3
3.2.2. General Procedure for the Synthesis of N-Substituted-1,3,4-oxadiazol-2-amines 5
3.3. In Vitro Antimycobacterial Evaluation
3.4. Molecular Docking Studies
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. Global Tuberculosis Report 2013. Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 26 February 2014).
- Wong, E.B.; Cohen, K.A.; Bishai, W.R. Rising to the challenge: New therapies for tuberculosis. Trends Microbiol. 2013, 21, 493–501. [Google Scholar]
- Wang, J.Y.; Burger, R.M.; Drlica, K. Role of superoxide in catalase-peroxidase-mediated isoniazid action against mycobacteria. Antimicrob. Agents Chemother. 1998, 42, 709–711. [Google Scholar]
- Vinšová, J.; Imramovský, A.; Jampílek, J.; Monreal, J.F.; Doležal, M. Recent Advances on Isoniazide Derivatives. Anti-Infect. Agents Med. Chem. 2008, 7, 1–20. [Google Scholar] [CrossRef]
- Schroeder, E.K.; de Souza, O.N.; Santos, D.S.; Blanchard, J.S.; Basso, L.A. Drugs that inhibit mycolic acid biosynthesis in Mycobacterium tuberculosis. Curr. Pharm. Biotechnol. 2002, 3, 197–225. [Google Scholar] [CrossRef]
- Scior, T.; Garcés-Eisele, S.J. Isoniazid is not a lead compound for its pyridyl ring derivatives, Isonicotinoyl amides, hydrazides, and hydrazones: A Critical Review. Curr. Med. Chem. 2006, 13, 2205–2219. [Google Scholar] [CrossRef]
- Bernardes-Génisson, V.; Deraeve, C.; Chollet, A.; Bernadou, J.; Pratviel, G. Isoniazid: An uptake on the multiple mechanisms for a singular action. Curr. Med. Chem. 2013, 20, 4370–4385. [Google Scholar] [CrossRef]
- Judge, V.; Narasimhan, B.; Ahuja, M. Isoniazid: The magic molecule. Med. Chem. Res. 2012, 21, 3940–3957. [Google Scholar] [CrossRef]
- Imramovský, A.; Polanc, S.; Vinšová, J.; Kočevar, M.; Jampílek, J.; Rečková, Z.; Kaustová, J. A new modification of anti-tubercular active molecules. Bioorg. Med. Chem. 2007, 15, 2551–2559. [Google Scholar] [CrossRef]
- Vavříková, E.; Polanc, S.; Kočevar, M.; Košmrlj, J.; Horváti, K.; Bősze, S.; Stolaříková, J.; Imramovský, A.; Vinšová, J. New series of isoniazid hydrazones linked with electron-withdrawing substituents. Eur. J. Med. Chem. 2011, 46, 5902–5909. [Google Scholar] [CrossRef]
- Vavříková, E.; Polanc, S.; Kočevar, M.; Horváti, K.; Bősze, S.; Stolaříková, J.; Vávrová, K.; Vinšová, J. New fluorine-containing hydrazones active against MDR-tuberculosis. Eur. J. Med. Chem. 2011, 46, 4937–4945. [Google Scholar] [CrossRef]
- Sriram, D.; Yogeeswari, P.; Priya, D.Y. Antimycobacterial activity of novel N-(substituted)-2-isonicotinoylhydrazinocarbothioamide endowed with high activity towards isoniazid resistant tuberculosis. Biomed. Pharmacother. 2009, 63, 36–39. [Google Scholar] [CrossRef]
- Navarrete-Vázquez, G.; Molina-Salinas, G.M.; Duarte-Fajardo, Z.V.; Vargas-Villarreal, J.; Estrada-Soto, S.; Gonzalez-Salazar, F.; Hernandez-Nunez, E.; Said-Fernandes, S. Synthesis and antimycobacterial activity of 4-(5-substituted-1,3,4-oxadiazol-2-yl)pyridines. Bioorg. Med. Chem. 2007, 15, 5502–5508. [Google Scholar] [CrossRef]
- Košmrlj, J.; Kočevar, M.; Polanc, S. A Mild Approach to 1,3,4-Oxadiazoles and Fused 1,2,4-Triazoles. Diazenes as Intermediates? Synlett 1996, 652–654. [Google Scholar]
- Ventura, C.; Martins, F. Application of Quantitative Structure-Activity Relationships to the Modeling of Antitubercular Compounds. 1. The Hydrazide Family. J. Med. Chem. 2008, 51, 612–624. [Google Scholar] [CrossRef]
- Ahsan, M.J.; Samy, J.G.; Khalilullah, H.; Nomani, M.S.; Saraswat, P.; Gaur, R.; Singh, A. Molecular properties prediction and synthesis of novel 1,3,4-oxadiazole analogues as potent antimicrobial and antitubercular agents. Bioorg. Med. Chem. Lett. 2011, 21, 7246–7250. [Google Scholar] [CrossRef]
- Rane, R.A.; Gutte, S.D.; Sahu, N.U. Synthesis and evaluation of novel 1,3,4-oxadiazole derivatives of marine bromopyrrole alkaloids as antimicrobial agent. Bioorg. Med. Chem. Lett. 2012, 22, 6429–6432. [Google Scholar] [CrossRef]
- Foks, H.; Mieczkowska, J.; Janowiec, M.; Zwolska, Z.; Andrzejczyk, Z. Synthesis and tuberculostatic activity of methyl 3-isonicotinoyldithiocarbazate and S,S'-dimethyldithiocarbonate isonicotinoylhydrazone and their reactions with amines and hydrazines. Chem. Heterocycl. Comp. 2002, 38, 810–816. [Google Scholar] [CrossRef]
- Patel, M.B.; Modi, N.R.; Raval, J.P.; Menon, S.K. Calix[4]arene based 1,3,4-oxadiazole and thiadiazole derivatives: Design, synthesis, and biological evaluation. Org. Biomol. Chem. 2012, 10, 1785–1794. [Google Scholar] [CrossRef]
- Dessen, A.; Quemard, A.; Blanchard, J.S.; Jacobs, W.R.; Sacchettini, J.C. Crystal structure and function of the isoniazid target of Mycobacterium tuberculosis. Science 1995, 267, 1638–1641. [Google Scholar] [CrossRef]
- Quemard, A.; Sacchettini, J.C.; Dessen, A.; Vilcheze, C.; Bittman, R.; Jacobs, W.R.; Blanchard, J.S. Enzymatic Characterization of the Target for Isoniazid in Mycobacterium tuberculosis. Biochemistry 1995, 34, 8235–8241. [Google Scholar] [CrossRef]
- Rozwarski, D.A.; Vilcheze, C.; Sugantino, M.; Bittman, R.; Sacchettini, J.C. Crystal Structure of the Mycobacterium tuberculosis Enoyl-ACP Reductase, InhA, in Complex with NAD1 and a C16 Fatty Acyl Substrate. J. Biol. Chem. 1999, 274, 15582–15589. [Google Scholar]
- Xie, Y.Y.; Liu, J.L.; Yang, P.; Shi, X.J.; Li, J.J. Synthesis of 2-amino-1,3,4-oxadiazoles from isoselenocyanates via cyclodeselenization. Tetrahedron 2011, 67, 5369–5374. [Google Scholar] [CrossRef]
- Kaustová, J. Quantitative micromethod for drug susceptibility testing of mycobacteria in Sula’s medium. Klin. Microbiol. Inf. Lek. 1997, 3, 115–124. [Google Scholar]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera--a visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Morris, G.M.; Goodsell, D.S.; Halliday, R.S.; Huey, R.; Hart, W.E.; Belew, R.K.; Olson, A.J. Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J. Comput. Chem. 1998, 19, 1639–1662. [Google Scholar] [CrossRef]
- DeLano, W.L. The PyMol Molecular Graphics System, Version 1.1r1; Schrodinger, LLC. Available online: http://www.pymol.org (accessed on 23 March 2014).
- Sample Availability: Samples of the compounds 3a–v and 5i, 5l, 5n and 5s are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rychtarčíková, Z.; Krátký, M.; Gazvoda, M.; Komlóová, M.; Polanc, S.; Kočevar, M.; Stolaříková, J.; Vinšová, J. N-Substituted 2-Isonicotinoylhydrazinecarboxamides — New Antimycobacterial Active Molecules. Molecules 2014, 19, 3851-3868. https://doi.org/10.3390/molecules19043851
Rychtarčíková Z, Krátký M, Gazvoda M, Komlóová M, Polanc S, Kočevar M, Stolaříková J, Vinšová J. N-Substituted 2-Isonicotinoylhydrazinecarboxamides — New Antimycobacterial Active Molecules. Molecules. 2014; 19(4):3851-3868. https://doi.org/10.3390/molecules19043851
Chicago/Turabian StyleRychtarčíková, Zuzana, Martin Krátký, Martin Gazvoda, Markéta Komlóová, Slovenko Polanc, Marijan Kočevar, Jiřina Stolaříková, and Jarmila Vinšová. 2014. "N-Substituted 2-Isonicotinoylhydrazinecarboxamides — New Antimycobacterial Active Molecules" Molecules 19, no. 4: 3851-3868. https://doi.org/10.3390/molecules19043851





