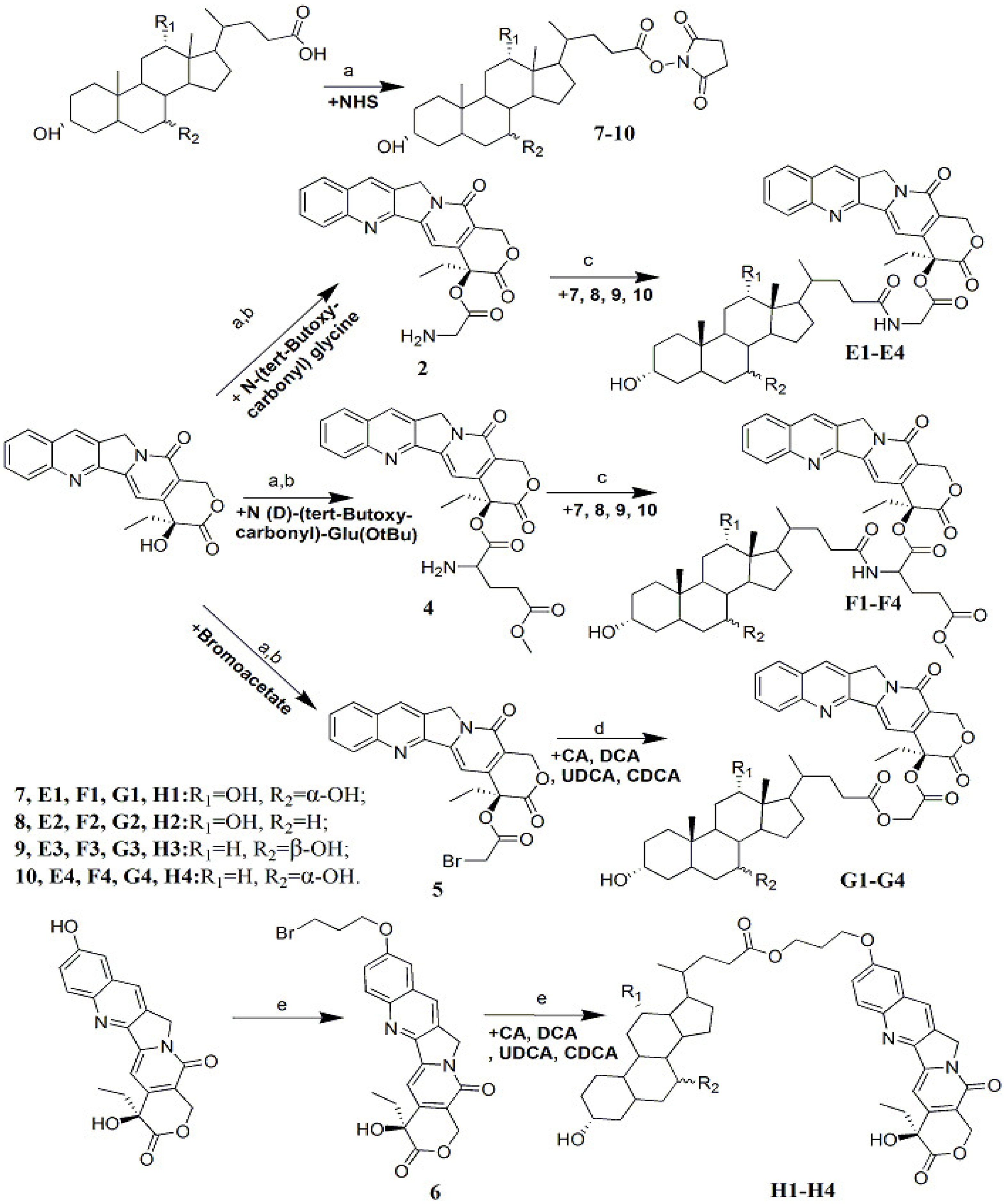
3.2. Compound Synthesis
Synthesis of Camptothecin-20 (S)-O-(N-(tert-butoxycarbonyl) glycine) Ester (1) and Compound Camptothecin-20 (S)-O-glycine ester (2)
EDCI (383.4 mg, 2 mmol) and DMAP (36 mg, 0.34 mmol) were added to a solution with CPT (139.2 mg, 0.4 mmol) and N-(tert-butoxycarbonyl) glycine (140.1 mg, 0.8 mmol) in dichloromethane (15 mL) and stirred at room temperature for 2 h. Chloroform (35 mL) was subsequently added, and the organic phase was washed with water (50 mL) and saturated sodium bicarbonate aqueous solution (50 mL), and then dried using Mg2SO4 before evaporating to dryness. Compound 1, a cream-coloured solid was obtained. Total yield 94.8%; M.p. 213.6–214.3 °C; IR (KBr) υ 3315, 3052, 2976, 2936, 2881, 2351, 2320, 1757, 1714, 1652, 1606, 1553, 1500, 1456, 1402, 1392, 1357, 1251, 1157, 1053, 1029, 947, 761, 723 cm−1; 1H-NMR (500 MHz, CDCl3-d6): δ 4.062–4.108 (1H, d, J = 25 Hz, -CH2-), 4.195–4.243 (1H, d, J = 24 Hz, -CH2-), 4.992 (1h, s, -NH-), 5.306 (2H, s, H-17), 5.391–5.426 (1H, d, J = 17.5 Hz, H-5), 5.681–5.715 (1H, d, J = 17 Hz, H-5), 7.399 (1H, s, H-14), 7.682–7.712 (1H, t, J = 15 Hz, H-10), 7.850–7.880 (1H, t, J = 15 Hz, H-11), 7.953–7.968 (1H, d, J = 7.5 Hz, H-12), 8.302–8.319 (1H, d, J = 8.5 Hz, H-9), 8.435 (1H, s, H-7); MS (ESI): m/z 506.1 (M+H)+. Compound 1 (191.5 mg) was added to a solution of hydrogen chloride/methanol (10 mL). The mixture was stirred at room temperature for 3 h and evaporated to dryness to yield compound 2 as a yellow-coloured solid. Total yield 95%; MS (ESI): m/z 441.5 (M+H)+.
Synthesis of 3α,7α,12α-Trihydroxy-5β-cholan-24-oic Acid 2,5-dioxopyrrolidin-1-yl Ester (7)
CA (40.8 mg, 0.1 mmol) was dissolved in DMSO (2 mL). N-Hydroxysulphosuccinimide (NHS, 172.5 mg, 1.5 mmol) and EDCI (286.5 mg, 1.5 mmol) were added and the mixture was stirred at room temperature for 2 h. Chloroform (100 mL) was subsequently added and the organic phase was washed with water (100 mL) and saturated sodium bicarbonate aqueous solution (100 mL). The organic solution was dried using Mg2SO4 and evaporated to dryness. The crude product was separated and purified by column chromatography to obtain a white solid. Yield: 60%; M.p. 103–106 °C; IR (KBr) υ 3377, 3361, 2937, 2968, 1815, 1753, 1658, 1465, 1446, 1435, 1411, 1375, 1296, 1249, 1209, 1112, 1074 1045 cm−1; 1H-NMR (500 MHz, DMSO-d6): δ 0.70 (3H, s, H-18), 0.88 (3H, s, H-19), 1.05 (3H, s, H-21), 2.83 (4H, d, J = 3.5 Hz, CH2), 3.53 (1H, m, 3α-OH), 3.87 (1H, s, 7α-OH), 4.02 (1H, s, 12α-OH); MS (ESI): m/z, 528.6 (M+Na)+
Synthesis of 3α,12α-Dihydroxy-5β-cholan-24-oic Acid 2,5-dioxopyrrolidin-1-yl Ester (8)
The synthesis was carried out as described for compound 7, and a white solid was obtained. Yield: 65%; M.p. 181–184 °C; IR (KBr) υ 3363, 2973, 2893, 2868, 1813, 1784, 1743, 1463, 1446, 1436, 1367, 1338, 1205, 1112, 1068, 1045, 1028 cm-1; 1H-NMR (500 MHz, DMSO-d6): δ 0.65 (3H, s, H-18), 0.90 (3H, s, H-19), 1.03 (3H, s, H-21), 2.83–2.85 (4H, d, CH2), 3.63 (1H, m, 3α-OH), 4.07 (1H, s, 12α-OH), MS (ESI): m/z, 530.6 (M+H2O+Na)+.
Synthesis of 3α,7β-Dihydroxy-5β-cholan-24-oic Acid 2,5-dioxo-pyrrolidin-1-yl Ester (9)
The synthesis was carried out as described for compound 7, and a white solid was obtained.
Synthesis of 3α, 7α-Dihydroxy-5β-cholan-24-oic Acid 2,5-dioxopyrrolidin-1-yl Ester (10)
The synthesis was carried out as described for compound 7, and a white solid was obtained. Yield: 63%; M.p. 241–243 °C; IR (KBr) υ 3412, 2931, 2868, 1815, 1784, 1743, 1732, 1448, 1435, 1413, 1367, 1330, 1289, 1207, 1168, 1128, 1111, 1072, 1047, 1001, 979 cm−1; 1H-NMR (500 MHz, DMSO-d6): δ 0.67 (3H, s, H-18), 0.90 (3H, s, H-19), 0.97 (3H, s, H-21), 2.83-2.85 (4H, d, CH2), 3.44 (1H, m, 3α-OH), 3.85 (1H, s, 7α-OH); MS (ESI): m/z 544.8 (M+Na+CH3OH)+.
Synthesis of Camptothecin-20 (S)-O-glycine Ester-[N-(3′α,7′α,12′α-trihydroxy-24′-carbonyl-5′β-cholane)] (E1)
Triethylamine (100 µL) was added to a solution of compound 7 (50.5 mg, 0.1 mmol) and compound 2 (44 mg, 0.1 mmol) in DMSO (2 mL), and the mixture was stirred at room temperature for 2 h (monitored by TLC). Water was then added to the solvent, and a pale yellow precipitate formed immediately. The mixture was filtered, and the solid was washed with water. The crude product was separated and purified by column chromatography to obtain a yellow solid. Yield: 75%; M.p. 200–203 °C; IR (KBr) υ 3419, 3062, 2924, 2858, 2603, 2495, 2308, 1755, 1716, 1660, 1604, 1558, 1504, 1456, 1402, 1377, 1352, 1232, 1076, 1049, 981, 950 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.492 (3H, s, H-18 (CA)), 3.190 (1H, s, H-3 (CA)), 3.550 (1H, s, H-7 (CA)), 3.722 (1H, s, H-12 (CA)), 3.950 (1H, s, 3α-OH(CA)), 3.950–3.985 (1H, d, J = 17.5 Hz, -CH2-), 4.038 (1H, s, 7α-OH(CA)), 4.045 (1H, d, -CH2-), 4.280 (1H, s, 12α-OH(CA)), 3.985-5.300 (2H, s, H-17), 5.494 (2H, s, H-5), 7.165 (1H, s, H-14), 7.723–7.738 (1H, t, H-10), 7.875 (1H, t, H-11), 8.127–8.143 (1H, d, J = 8 Hz, H-12), 8.173–8.190 (1H, d, J = 8.5 Hz, H-9), 8.330 (1H, s, -NH-), 8.702 (1H, s, H-7); MS (ESI): m/z 796.7 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-glycine Ester-[N-(3′α,12′α-dihydroxy-24′-carbonyl-5′β-cholane)] (E2)
The synthesis was carried out as described for compound E1, and a yellow solid was obtained. Yield: 74%; M.p. 203–204 °C; IR(KBr) υ 3421, 3064, 2927, 2864, 2370, 1751, 1662, 1616, 1560, 1500, 1458, 1402, 1375, 1325, 1232, 1166, 1051, 997, 891 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.567 (3H, s, H-19 (DCA)), 0.842–0.855 (3H, d, J = 6.5 Hz, H-21 (DCA)), 0.878 (3H, s, H-18 (DCA)), 3.60 (1H, s, H-3 (DCA)), 4.015 (1H, s, H-12 (DCA)), 4.120–4.166 (1H, t, J = 22.4 Hz, -CH2-), 4.437–4.487 (1H, t, J = 25 Hz, -CH2-), 5.298 (2H, s, H-17), 5.395–5.429 (1H, m, J = 17 Hz, H-5), 5.687–5.721 (1H, m, J = 17 Hz, H-5), 5.853 (1H, s, -NH-), 7.262 (1H, s, H-14), 7.681 (1H, t, H-10), 7.848 (1H, t, H-11), 7.936–7.952 (1H, d, J = 8 Hz, H-12), 8.252–8.269 (1H, d, J = 7 Hz, H-9), 8.401 (1H, s, H-7); MS (ESI): m/z 780.5 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-glycine Ester-[N-(3′α,7′β-dihydroxy-24′-carbonyl-5′β-cholane)] (E3)
The synthesis was carried out as described for compound E1, and a yellow solid was obtained. Yield: 75%; M.p. 209–211 °C; IR(KBr) υ 3419, 3062, 2927, 2861, 2349, 1755, 1662, 1606, 1558, 1506, 1456, 1402, 1375, 1296, 1232, 1182, 1134, 1051, 1014, 952, 902, 891 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.591 (3H, s, H-18 (UDCA)), 3.551 (1H, s, H-3 (UDCA)), 3.573 (1H, s, H-7 (UDCA)), 4.154-4.162 (1H, m, -CH2-), 4.460–4.472 (1H, m, -CH2-), 5.311 (2H, s, H-17), 5.390–5.424 (1H, m, J = 17 Hz, H-5), 5.683–5.717 (1H, m, J = 17.5 Hz, H-5), 5.90 (1H, s, -NH-), 7.403 (1H, s, H-14), 7.710–7.726 (1H, t, H-10), 7.882 (1H, t, H-11), 7.962-7.978 (1H, d, J = 8 Hz, H-12), 8.330–8.346 (1H, d, J = 8 Hz, H-9), 8.458 (1H, s, H-7); MS(ESI): m/z 781.1 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-glycine Ester-[N-(3′α,7′α-dihydroxy-24′-carbonyl-5′β-cholane)] (E4)
This was carried out as described for compound E1, and a yellow solid was obtained.Yield: 76%; M.p. 202–203 °C; IR(KBr) υ 3435, 3062, 2929, 2866, 1755, 1662, 1616, 1558, 1521, 1506, 1456, 1402, 1375, 1296, 1255, 1182, 1058, 976, 959 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.594 (3H, s, H-19 (CDCA)), 3.402 (1H, s, H-3 (CDCA)), 3.802 (1H, s, H-7 (CDCA)), 4.167-4.180 (1H, m, -CH2-), 4.432–4.452 (1H, m, -CH2-), 5.294–5.302 (2H, d, J = 4 Hz, H-17), 5.396–5.430 (1H, m, J = 17 Hz, H-5), 5.684–5.718 (1H, m, J = 17.5 Hz, H-5), 5.811 (1H, s, -NH-), 7.285 (1H, s, H-14), 7.688 (1H, t, H-10), 7.858 (1H, t, H-11), 7.942–7.958 (1H, d, J = 8 Hz, H-12), 8.271–8.288 (1H, d, J = 8 Hz, H-9), 8.415 (1H, s, H-7); MS (ESI): m/z 780.8 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-(N -(tert-butoxycarbonyl)-(d)-Glu(OtBu)) Ester (3) and Camptothecin-20 (S)-O-(d-Glu(OMe)) Ester (4)
This was carried out as described for compound 1, and compound 3, and a yellow solid was obtained. Yield: 81%; M.p. 178–179 °C; IR (KBr) υ 3419, 3307, 3062, 2924, 2858, 2623, 2495, 2308, 1755, 1660, 1604, 1558, 1504, 1456, 1402, 1377, 1352, 1232, 1076, 1049, 981, 950, 914, 900 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 4.543 (1H, s, -CH-), 5.099 (1H, s,-NH-), 5.276 (2H, s, H-17), 5.368–5.402 (1H, d, J = 17 Hz, H-5), 5.687–5.721 (1H, d, J = 17 Hz, H-5), 7.408 (1H, s, H-14), 7.647–7.676 (1H, t, J = 14.5 Hz, H-10), 7.809–7.839 (1H, t, J = 15 Hz, H-11), 7.920–7.936 (1H, d, J = 8 Hz, H-12), 8.261–8.279 (1H, d, J = 9 Hz, H-9), 8.376 (1H, s, H-7); MS (ESI): m/z 634.4 (M+H)+. Compound 3(191.5 mg) was added to hydrogen chloride/methanol (10 mL) and the mixture was stirred at room temperature for 12 h (monitored by TLC). Chloroform (50 mL) was subsequently added, and the organic phase was washed with water (50 mL). The organic solution was dried (Mg2SO4) and evaporated to dryness, producing compound 4, a yellow solid. Total yield 90%; MS (ESI): m/z 527.5 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-(d-Glu (OMe)) Ester-[N-(3′α,7′α,12′α-trihydroxy-24′-carbonyl-5′β-cholane)] (F1)
This was carried out as described for compound E1, and a yellow solid was obtained. Yield: 70%; M.p. 195–196 °C; IR (KBr) υ 3431, 3057, 2926, 2856, 1747, 1662, 1604, 1456, 1402, 1375, 1352, 1232, 1166, 1195, 1049, 1016, 979 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.547 (3H, s, H-19 (CA)), 3.094–3.119 (2H, q, J = 12.5 Hz, H-3 (Glu)), 2.556–2.571 (2H, d, J = 7.5 Hz, H-4 (Glu)), 3.46 (1H, s, H-3 (CA)), 3.762 (3H, s, H-1 (CH3)), 3.762 (1H, s, H-7 (CA)), 3.849 (1H, s, H-3 (CA)), 4.96 (1H, s, H-2 (Glu)), 5.286 (2H, s,H-17), 5.374–5.408 (1H, d, J = 17 Hz,H-5), 5.681–5.715 (1H, d, J = 17 Hz, H-5), 6.56 (1H, s, H-1 (NH)), 7.419 (1H, s, H-14), 7.690 (1H, t, H-10), 7.856 (1H, t, H-11), 7.926–7.942 (1H, d, J = 8 Hz, H-12), 8.310–8.327 (1H, d, J = 8.5 Hz, H-9), 8.415 (1H, s, H-7); MS (ESI): m/z 882.5 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-(d-Glu(OMe))Ester-[N-(3′α,12′α-dihydroxy-24′-carbonyl-5′β-cholane)] (F2)
This was carried out as described for compound E1, and a yellow solid was obtained. Yield: 75%; M.p. 199–200 °C; IR (KBr) υ 3438, 3059, 2930, 2860, 1747, 1662, 1610, 1556, 1402, 1365, 1304, 1258, 1169, 1120,1086, 1049, 1014, 983 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.486 (3H, s, H-19 (DCA)), 0.780 (3H, s, H-21), 2.568–2.594 (2H, t, J = 13 Hz, H-4 (Glu)), 3.502 (1H, s, H-3 (DCA)), 3.576 (1H, s, H-12 (DCA)), 3.707 (3H, s, H-6 (CH3)), 4.882–4.894 (1H, d, J = 6 Hz, H-2 (Glu)), 5.283 (2H, s, H-17), 5.372–5.406 (1H, d, J = 17, H-5), 5.680–5.714 (1H, d, J = 17 Hz, H-5), 6.279–6.292 (1H, d, J = 6.5 Hz, H-6 (N-H)), 7.363 (1H, s, H-14), 7.671 (1H, t, H-10), 7.841 (1H, t, H-11), 7.926–7.942 (1H, d, J = 8 Hz, H-12), 8.289–8.306 (1H, d, J = 8.5 Hz, H-9), 8.393 (1H, s, H-7); MS (ESI): m/z 866.9 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-(d-Glu(OMe)) Ester-[N-(3′α,7′β-dihydroxy-24′-carbonyl-5′β-cholane)] (F3)
This was carried out as described for compound E1, and a yellow solid was obtained. Yield: 72%; M.p. 187–189 °C; IR (KBr) υ 3421, 3059, 2929, 2864, 2364, 1747, 1662, 1616, 1558, 1521, 1502,1452,1381,1360,1232,1166,1083,1051,1014,952 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.492 (3H, s, H-19 (UDCA)), 0.660 (3H, s, H-21 (UDCA)), 2.564–2.594 (2H, t, J = 15 Hz, H-4 (Glu)), 3.56 (1H, s, H-3 (CA)), 3.622 (1H, s, H-17 (UDCA)), 3.707 (3H, s, H-6 (CH3)), 4.860 (1H, s, H-2 (Glu)), 5.290 (2H, s, H-17), 5.376–5.411 (1H, d, J = 17.5 Hz, H-5), 5.684–5.718 (1H, d, J = 17 Hz, H-5), 6.25 (1H, d, H-6 (NH)), 7.369 (1H, s, H-14), 7.679 (1H, t, H-10), 7.864 (1H, t, H-11), 7.933–7.949 (1H, d, J = 8 Hz, H-12), 8.296–8.313 (1H, d, J = 7.5 Hz, H-9), 8.402 (1H, s, H-7); MS (ESI): m/z 866.9 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-(d-Glu(OMe)) Ester-[N-(3′α 7′α-dihydroxy-24′-carbonyl-5′β-cholane)] (F4)
This was carried out as described for compound E1, and a yellow solid was obtained. Yield: 74%; M.p. 182–185 °C; IR(KBr) υ 3566, 3057, 2921, 2851, 2306, 1717, 1662, 1604, 1558, 1506, 1156, 1375, 1325, 1296, 1259, 1232, 1166, 1078, 1051, 1001,989,800 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.513 (3H, s, H-19 (CDCA)), 0.788 (3H, s, H-21 (CDCA)), 2.563–2.592 (2H, t, J = 14.5 Hz, H-4 (Glu)), 3.454 (1H, s, H-3 (CDCA)), 3.705 (3H, s, H-6 (CH3)), 3.764 (1H, s, H-7 (CA)), 4.888–4.899 (1H, d, J = 5.5, H-2 (Glu)), 5.283 (2H, s, H-17), 5.375–5.410 (1H, d, J = 17.5 Hz, H-5), 5.683–5.717 (1H, d, J = 17 Hz, H-5), 6.26 (1H, d, H-6 (NH)), 7.362 (1H, s, H-14), 7.673–7.688 (1H, t, J = 7.5 Hz, H-10), 7.831–7.845 (1H, t, J = 7 Hz, H-11), 7.926–7.942 (1H, d, J = 8 Hz, H-12), 8.290–8.307 (1H, d, J = 8.5 Hz, H-9) 8.393 (1H, s, H-7); MS (ESI): m/z 866.9 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-bromoacetate (5)
This was carried out as described for compound 1, and a yellow solid was obtained. Yield: 97%; M.p. 234–236 °C; IR (KBr) υ 3449, 2970, 1754, 1661, 1558, 1454, 1232, 1151, 1052, 990, 787, 761 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 1.00 (3H, t, J = 7.2 Hz, H-18), 2.16-2.37 (2H, m, H-19), 4.28 (2H, q, CH2), 5.31 (2H, s, H-5), 5.43 (1H, d, J = 17.5 Hz, H-17), 5.71 (1H, d, J= 17.2 Hz, H-17), 7.34 (1H, s, H-14), 7.70 (1H, t, J = 7.2 Hz, H-10), 7.87 (1H, t, J = 7.2 Hz, H-11), 7.97 (1H, d, J = 8.4 Hz, H-12), 8.28 (1H, d, J = 8.5 Hz, H-9), 8.45 (1H, s, H-7); MS (ESI): m/z 469.4 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-acetate-[N-(3′α,7′α,12′α-trihydroxy-24′-carbonyl-5′β-cholane)] (G1)
Triethylamine (50 µL) was added to a solution of compound 5 (46.9 mg, 0.1 mmol) and CA (81.6 mg, 0.2 mmol) in N,N-dimethylformamide (DMF) (1 mL), and the mixture was stirred at room temperature for 12 h (monitored by TLC). Chloroform (50 mL) was subsequently added and the organic phase was washed with water (50 mL) before drying (Mg2SO4) and evaporation to dryness. The crude product was separated and purified by column chromatography, producing a yellow solid. Yield: 62%; M.p. 230–233 °C; IR(KBr) υ 3460, 3062, 2926, 2861, 2345, 2322, 1751, 1666, 1618, 1560, 1506, 1458, 1402, 1379, 1296, 1254, 1232, 1192, 1149, 1076, 1049, 1018, 999, 981, 912 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.624 (3H, s, H-19 (CA)), 3.421 (1H, s, H-3 (CA)), 3.801 (1H, s, H-7 (CA)), 3.917 (1H, s, H-12 (CA)), 4.767–4.881 (2H, q, J = 57 Hz, H-1 (CH2)), 5.301 (2H, s, H-17), 5.398–5.443 (1H, d, J = 17.5 Hz, H-5), 5.674–5.708 (1H, d, J = 17 Hz, H-5), 7.355 (1H, s, H-14), 7.689 (1H, t, H-10), 7.854 (1H, t, H-11), 7.946–7.963 (1H, d, J = 8.5 Hz, H-12), 8.279–8.296 (1H, d, J = 8.5 Hz, H-9) 8.425 (1H, s, H-7); MS (ESI): m/z 797.7 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-acetate-[N-(3′α,12′α-dihydroxy-24′-carbonyl-5′β-cholane)] (G2)
This was carried out as described for compound G1, and a yellow solid was obtained. Yield: 63%; M.p. 221–224 °C; IR(KBr) υ 3550, 3421, 3089, 2935, 2862, 2349, 1749, 1666, 1616, 1560, 1500, 1456, 1402, 1379, 1365, 1352, 1298, 1230, 1085, 1043, 945, 923 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.610 (3H, s, H-19 (DCA)), 3.600 (1H, s, H-3 (DCA)), 3.913 (1H, s, H-12 (DCA)), 4.769–4.882 (2H, q, J = 56.5 Hz, H-1 (CH2)), 5.304 (2H, s, H-17), 5.397–5.432 (1H, d, J = 17.5 Hz, H-5), 5.670–5.705 (1H, d, J = 17 Hz, H-5), 7.365 (1H, s, H-14), 7.699–7.715 (1H, t, J = 8 Hz, H-10), 7.862 (1H, t, H-11), 7.956–7.972 (1H, d, J = 8 Hz, H-12), 8.294 (1H, d, J = 8.5 Hz, H-9), 8.440 (1H, s, H-7); MS (ESI): m/z 781.8 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-acetate-[N-(3′α,7′β-dihydroxy-24′-carbonyl-5′β-cholane)] (G3)
This was carried out as described for compound G1, and a yellow solid was obtained. Yield: 62%; M.p. 229–230 °C; IR(KBr) υ 3504, 3446, 3064, 2929, 2861, 2315, 1755, 1670, 1618, 1560, 1500, 1456, 1404, 1298, 1232, 1151, 1101, 1051, 1014, 948, 927 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.592 (3H, s, H-19 (UDCA)), 3.568 (2H, s, H-3, H-7 (UDCA)), 4.738–4.771 (1H, d, J = 17.5, H-1 (CH2)), 4.851–4.884 (1H, d, J = 16.5 Hz, H-1 (CH2)), 5.293 (2H, s, H-17), 5.395–5.430 (1H, d,J = 17.5 Hz, H-5), 5.670–5.705 (1H, d, J = 17.5 Hz, H-5) 7.281 (1H, s, H-14), 7.678–7.693 (1H, t, J = 7.5 Hz, H-10), 7.842–7.845 (1H, t, J = 1.5 Hz, H-11), 7.940–7.956 (1H, d, J = 8 Hz,H-12), 8.242–8.259 (1H, d, J = 8.5 Hz,H-9), 8.400 (1H, s, H-7); MS (ESI): m/z 781.8 (M+H)+.
Synthesis of Camptothecin-20 (S)-O-acetate-[N-(3′α,7′α-dihydroxy-24′-carbonyl-5′β-cholane)] (G4)
This was carried out as described for compound G1, and a yellow solid was obtained. Yield: 63%; M.p. 228–229 °C; IR (KBr) υ 3564, 3481, 3064, 2926, 2854, 2347, 1755, 1670, 1620, 1560, 1500, 1458, 1404, 1381, 1354, 1232, 1203, 1147, 1011, 979 cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.592 (3H, s, H-19 (CDCA)), 3.454 (1H, s, H-3 (CDCA)), 3.800 (1H, s, H-7 (CDCA)), 4.746-4.779 (1H, d, J = 16.5 Hz, H-1 (CH2)), 4.845–4.878 (1H, d, J = 16.5 Hz, H-1 (CH2)), 5.288 (2H, s, H-17), 5.397–5.431 (1H, d, J = 17 Hz, H-5), 5.668–5.702 (1H, d, J = 17 Hz, H-5), 7.290 (1H, s, H-14), 7.680 (1H, t, H-10), 7.843 (1H, t, H-11), 7.940–7.956 (1H, d, J = 2 Hz, H-12), 8.248–8.265 (1H, d, J = 8.5 Hz, H-9), 8.404 (1H, s, H-7); MS (ESI): m/z 781.9 (M+H)+.
Synthesis of 10-O-(3-Bromopropyl)-20(S)-camptothecin (6)
To a solution of 10-hydroxy CPT (182 mg, 0.5 mmol) and potassium carbonate (345.94 mg, 2.5 mmol) in DMF (1.5 mL) 1,3-dibromopropane (100 µL) was added, and the mixture was stirred at 60 °C for 10 h under nitrogen conditions (monitored by TLC). Water was then added to the solvent, and a pale yellow precipitate appeared immediately. The mixture was filtered and the solid was washed with water. The crude product was separated and purified by column chromatography to obtain a yellow solid. Yield: 85%; M.p. 235–237 °C; IR (KBr) υ 3398, 2955, 2925, 1747,1660, 1595, 1556, 1508, 1429, 1370, 1237, 1147, 1105, 1049, 1015, 532 cm−1; 1H-NMR (500 MHz, DMSO-d6) δ: 0.88 (3H, t, J = 7.4 Hz, H-18), 1.86 (2H, m, H-19), 2.36 (2H, t, J =5.6 Hz, CH2), 3.74 (2H, t, J = 5.6 Hz, CH2), 4.26 (2H, t, J = 5.6 Hz, CH2), 5.25 (2H, s, H-5), 5.41 (2H, s, H-17), 6.53 (1H, s, 20-OH), 7.27 (1H, s, H-14), 7.50 (1H, d, J = 10 Hz, H-11), 7.55 (1H, s, H-9), 8.05 (1H, d, J = 10 Hz, h-12), 8.53 (1H, s, H-7); MS (ESI): m/z 485.0 (M+H)+.
Synthesis of 10-O-(3-O-(3′α,7′α,12′α-Trihydroxy-24′-carbonyl-5′β-cholan)-propyl)-20(S)-camptothecin (H1)
CA (81.6 mg, 0.2 mmol) was added to a solution of compound 6 (47 mg, 0.1 mmol) and potassium carbonate (34.5 mg, 0.25 mmol) in 1 mL DMF, and the mixture was stirred at 60 °C for 10 h under nitrogen conditions (monitored by TLC). Water was then added to the solvent, and a pale yellow precipitate appeared immediately. The mixture was filtered and the solid was washed with water. The crude product was separated and purified by column chromatography to obtain a yellow solid. Yield: 73%; M.p. 195–200 °C; IR (KBr) υ 3419, 3095, 2926, 2866, 2349, 1739, 1658, 1622, 1597, 1558, 1504, 1463, 1371, 1296, 1238, 1157, 1047, 912cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.598 (3H, s, H-19), 3.399 (1H, s, H-3 (CA)), 3.720 (1H, s, H-7 (CA)), 3.86 (1H, s, H-20), 3.926 (1H, s, H-12 (CA)), 4.26 (2H, t, H-1 (CH2)), 4.329–4.353 (2H, t, J = 12 Hz, H-2 (CH2)), 5.324–5.348 (1H, d, J = 12 Hz, H-5), 5.348 (2H, s, H-17), 5.712–5.745 (1H, d, J = 16.5 Hz, H-5), 7.176 (1H, s, H-14), 7.460–7.484 (1H, d, J = 12 Hz, H-11), 7.656 (1H, s, H-9), 8.132–8.150 (1H, d, J = 9 Hz, H-12), 8.274 (1H, s, H-7); MS (ESI): m/z 813.9 (M+H)+.
Synthesis of 10-O-(3-O-(3′α,12′α-Dihydroxy-24′-carbonyl-5′β-cholan)-propyl)-20(S)-camptothecin (H2)
This was carried out as described for compound H1, and a yellow solid was obtained. Yield: 75%; M.p. 193–197 °C; IR (KBr) υ 3400, 3097, 2926, 2858, 2349, 1739, 1658, 1622, 1595, 1556, 1504, 1446, 1371, 1296, 1238, 1155, 1095, 1045, 968, 912 cm−1; 1H-NMR (500 MHz, DMSO-d6) δ: 0.316 (3H, s, H-19), 3.611 (1H, s, H-3 (DCA)), 4.120–4.127 (1H, s, H-7 (DCA)), 4.227 (4H, s, H-1, H-2 (CH2)), 4.227 (1H, s, OH-3 (DCA)), 4.417–4.426 (1H, s, OH-7 (DCA)), 5.275 (2H, s, H-17), 5.417 (2H, s, H-5), 6.498 (1H, s, OH-20), 7.295 (1H, d, H-14), 7.532 (2H, d, H-11, H-9), 8.061–8.079 (1H, d, J = 9 Hz, H-12), 8.542 (1H, s, H-7); MS (ESI): m/z 797.7 (M+H)+.
Synthesis of 10-O-(3-O-(3′α,7′β-Dihydroxy-24′-carbonyl-5′β-cholan)-propyl)-20(S)-camptothecin (H3)
This was carried out as described for compound H1, and a yellow solid was obtained. Yield: 73%; M.p. 201–205 °C; IR (KBr) υ 3350, 3095, 2926, 2858, 2357, 1739, 1660, 1600, 1556, 1504, 1454, 1371, 1238, 1157, 1103, 1047, 1012, 964, 900, 827 cm-1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.595 (3H, s, H-19 (UDCA)), 3.456–3.461 (1H, s, H-3 (UDCA)), 3.578 (1H, s, H-12 (UDCA)), 3.857 (1H, s, OH-20), 4.227 (2H, t, H-1 (CH2)), 4.338 (2H, t, H-2 (CH2)), 5.293 (2H, s, H-17), 5.293–5.325 (1H, d, J = 16 Hz, H-5), 5.729–5.761 (1H, d, J = 16 Hz, H-5), 7.164 (1H, s, H-14), 7.462–7.483 (1H, d, J = 11 Hz, H-11), 7.648 (1H, s, H-9), 8.129–8.147 (1H, d, J = 8 Hz, H-12), 8.251 (1H, s, H-7); MS (ESI): m/z 797.9 (M+H)+.
Synthesis of 10-O-(3-O-(3′α,7′α-Dihydroxy-24′-carbonyl-5′β-cholan)-propyl)-20(S)-camptothecin (H4)
This was carried out as described for compound H1, and a yellow solid was obtained. Yield: 71%; M.p. 202–205 °C; IR (KBr) υ 3093, 2924, 2854, 2351, 1732, 1660, 1624, 1558, 1506, 1456, 1373, 1240, 1157, 1103, 1047, 1001, 977, 945, 825cm−1; 1H-NMR (500 MHz, CDCl3-d6) δ: 0.583 (3H, s, H-19 (CDCA)), 3.434 (1H, s, H-3 (CDCA)), 3.759 (1H, s, H-12 (CDCA)), 3.764 (1H, s, OH-20), 4.231-4.238 (2H, d, J = 3.5 Hz, H-1 (CH2)), 4.337–4.340 (2H, d, J = 1.5 Hz, H-2 (CH2)), 5.301 (2H, s, H-17), 5.301–5.339 (1H, d, J = 19 Hz, H-5), 5.724–5.757 (1H, d, J = 16.5 Hz, H-5), 7.168 (1H, s, H-14), 7.470–7.489 (1H, d, J = 9.5 Hz, H-11), 7.667 (1H, s, H-9), 8.144–8.172 (1H, d, J = 13 Hz, H-12), 8.256 (1H, s, H-7); MS (ESI): m/z 797.6 (M+H)+.