Authentication of Bulbus Fritillariae Cirrhosae by RAPD-Derived DNA Markers
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Identification of RAPD Marker for Bulbus Fritillariae Cirrhosae
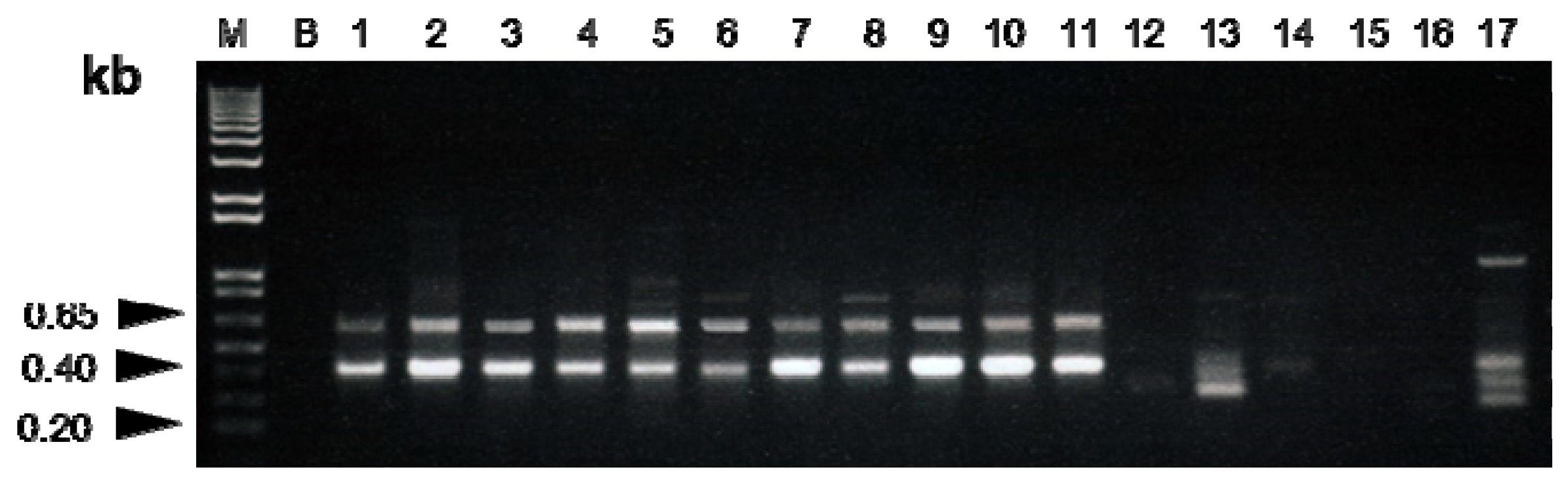
| Groups | Species | NO. of Indiv. | Origin |
|---|---|---|---|
| Chuanbeimu | Fritillaria cirrhosa | 2 | Kangdin County, Sichuan |
| F. unibracteata | 2 | Hongyuan County, Sichuan | |
| F. przewalskii | 2 | Min County, Gansu | |
| F. delavayi | 2 | Gongjue County, Tibet | |
| F. taipaiensis | 2 | Chongqing | |
| F. unibracteata Hsia et K. C. Hsia var. wabuensis | 1 | Mao County, Sichuan | |
| Yibeimu | F. pallidiflora | 1 | Yili County, Xinjiang |
| F. walujewii | 1 | Tacheng County, Xinjiang | |
| Zhebeimu | F. thunbergii | 1 | Ningbo, Zhejiang |
| Hubeibeimu | F. hupehensis | 1 | Yichang, Hubei |
| Pingbeimu | F. ussuriensis | 1 | Tonghua, Jilin |
| Puqibeimu | F. puqiensis | 1 | Puqi County, Hubei |
2.1.2. Cloning and Sequencing of the RAPD Marker

2.1.3. Amplification using SCAR Primers
| RADM
Primer | SCAR
Primer | Number of
base pairs (bp) | Sequence (5'-3') | G + C
Content (%) | Annealing
temperature |
|---|---|---|---|---|---|
| S158 | CBM-S | 20 | GAC CTG GTC TGT GGA TCG TT | 55 | 59 °C |
| CBM-AS | 20 | TGT CTC GCG CAA GTA CAA AC | 50 |

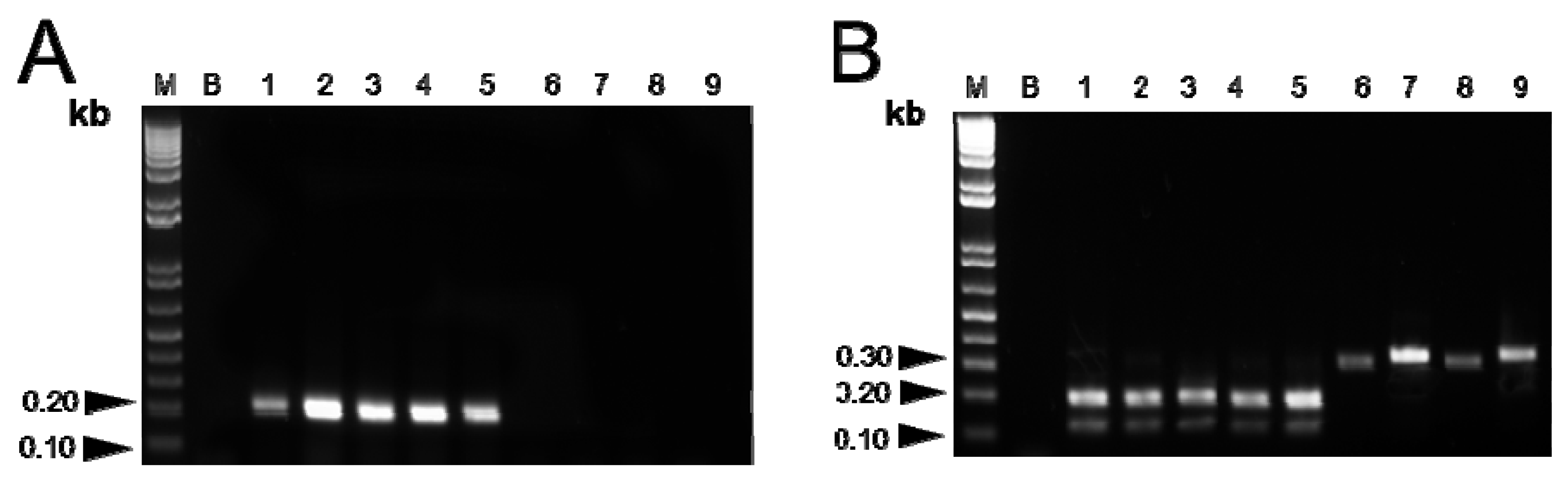
2.1.4. Validation of Results
| Column heading | PCR-RFLP | RAPD-SCAR |
|---|---|---|
| Specificity on
Fritillariae Cirrhosae Bulbus | Yes | Yes |
| Require high quality DNA | Yes | No |
| Require prior DNA sequence
Information | Yes | No |
| Experimental steps | 2 | 1 |
| Experimental duration | 6–8 h | 2–4 h |
2.2. Discussion
3. Experimental
3.1. Materials
3.2. DNA Extraction
3.3. RAPD Analysis
3.4. Screening Strategy and Identification of Specific RAPD
3.5. Cloning of the RAPD Amplicon
3.6. Sequencing, SCAR Primer Designing
3.7. Amplification of the Genomic Region
3.8. Validation of the Authentication Result
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Lin, B.Q.; Ji, H.; Li, P.; Jiang, Y.; Fang, W. Selective antagonism activity of alkaloids from bulbs Fritillariae at muscarinic receptors: Functional studies. Eur. J. Pharmacol. 2006, 551, 125–130. [Google Scholar] [CrossRef]
- Lin, G.; Li, P.; Li, S.L.; Chan, S.W. Chromatographic analysis of Fritillaria isosteroidal alkaloids, the active ingredients of Beimu, the antitussive traditional Chinese medicinal herb. J. Chromatogr. A 2001, 935, 321–338. [Google Scholar] [CrossRef]
- The State Pharmacopoeia Commission of the People’s Republic of China. Pharmacopoeia of the People’s Republic of China, 2010 ed.; Chemical Industry Press: Beijing, China, 2010; Volume 1, p. 191. [Google Scholar]
- Chan, S.W.; Li, S.L.; Lin, G.; Li, P. Pharmacokinetic study and determination of imperialine, the major bioactive component in antitussive Fritillaria cirrhosa, in rat by high-performance liquid chromatography coupled with evaporative light-scattering detector. Anal. Biochem. 2000, 285, 172–175. [Google Scholar] [CrossRef]
- Yu, X.L.; Ji, H.; Wang, C.L.; Li, P. Survey of pharmacology researches of Bulbus Fritillariae. Chin. Tradit. Herb. Drugs 2000, 31, 313–315. [Google Scholar]
- Li, P.; Xu, G.J. Studies on resources of Chinese drugs Beimu. J. Plant Resour. Environ. 1993, 2, 12–17. [Google Scholar]
- Li, P.; Xu, G.J.; Xu, L.S. Studies on the Chinese drug beimu VIII. Identification of the commercial drugs on Fritillaria. Chin. Tradit. Herb. Drugs 1990, 21, 26–29. [Google Scholar]
- Li, H.J.; Jiang, Y.; Li, P. Characterizing distribution of steroidal alkaloids in Fritillaria spp. and related compound formulas by liquid chromatography–mass spectrometry combined with hierarchial cluster analysis. J. Chromatogr. A 2009, 1216, 2142–2149. [Google Scholar]
- Cai, Z.H.; Li, P.; Dong, T.T.X.; Tsim, K.W.K. Molecular diversity of 5S-rRNA spacer domain in Fritillaria species revealed by PCR analysis. Planta Med. 1999, 65, 360–364. [Google Scholar] [CrossRef]
- Li, Y.F.; Li, Y.X.; Lin, J.; Xu, Y.; Yan, F.; Tang, L.; Chen, F. Identification of bulb from Fritillaria cirrhosa by PCR with specific primers. Planta Med. 2003, 69, 186–188. [Google Scholar] [CrossRef]
- Wang, C.Z.; Li, P.; Ding, J.Y.; Peng, X.; Yuan, C.S. Simultaneous identification of Bulbus Fritillariae cirrhosae using PCR-RFLP analysis. Phytomedicine 2007, 14, 628–632. [Google Scholar] [CrossRef]
- Xu, C.L.; Li, H.J.; Li, P.; Zhang, Y.; Wang, S. Molecular method for the identification of Bulbus Fritillariae Cirrhosae. J. Chin. Pharm. Univ. 2010, 41, 226–230. [Google Scholar]
- Um, J.Y.; Chung, H.S.; Kim, M.S.; Na, H.J.; Kwon, H.J.; Kim, J.J.; Lee, K.M.; Lee, S.J.; Lim, J.P.; Do, K.R.; et al. Molecular authentication of Panax ginseng species by RAPD analysis and PCR-RFLP. Biol. Pharm. Bull. 2001, 24, 872–875. [Google Scholar]
- Feng, K.; Wang, S.; Hu, D.J.; Yang, F.Q.; Wang, H.X.; Li, S.P. Random amplified polymorphic DNA (RAPD) analysis and the nucleosides assessment of fungal strains isolated from natural Cordyceps sinensis. J. Pharm. Biomed. Anal. 2009, 50, 522–526. [Google Scholar] [CrossRef]
- Chen, K.T.; Su, Y.C.; Lin, J.G.; Hsin, L.H.; Su, Y.P.; Su, C.H. Identification of Atractylodes plants in Chinese herbs and formulations by random amplified polymorphic DNA. Acta Pharmacol. Sin. 2001, 22, 493–497. [Google Scholar]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- BLAST Algorithm, 2nd ed.; an algorithm for comparing Primary biological sequence information. National Center of Biotechnology Information: Bethesda, MD, USA, 2002. Available online: http://www.ncbi.nlm.nih.gov/BLAST/ (accessed on 18 March 2014).
- Sample Availability: Samples of the Bulbus Fritillariae are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xin, G.-Z.; Lam, Y.-C.; Maiwulanjiang, M.; Chan, G.K.L.; Zhu, K.Y.; Tang, W.-L.; Dong, T.T.-X.; Shi, Z.-Q.; Li, P.; Tsim, K.W.K. Authentication of Bulbus Fritillariae Cirrhosae by RAPD-Derived DNA Markers. Molecules 2014, 19, 3450-3459. https://doi.org/10.3390/molecules19033450
Xin G-Z, Lam Y-C, Maiwulanjiang M, Chan GKL, Zhu KY, Tang W-L, Dong TT-X, Shi Z-Q, Li P, Tsim KWK. Authentication of Bulbus Fritillariae Cirrhosae by RAPD-Derived DNA Markers. Molecules. 2014; 19(3):3450-3459. https://doi.org/10.3390/molecules19033450
Chicago/Turabian StyleXin, Gui-Zhong, Yin-Ching Lam, Maitinuer Maiwulanjiang, Gallant K. L. Chan, Kevin Yue Zhu, Wai-Lun Tang, Tina Ting-Xia Dong, Zi-Qi Shi, Ping Li, and Karl W. K. Tsim. 2014. "Authentication of Bulbus Fritillariae Cirrhosae by RAPD-Derived DNA Markers" Molecules 19, no. 3: 3450-3459. https://doi.org/10.3390/molecules19033450




