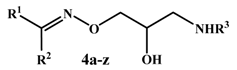
Synthesis, Docking Study and β-Adrenoceptor Activity of Some New Oxime Ether Derivatives
Abstract
:1. Introduction
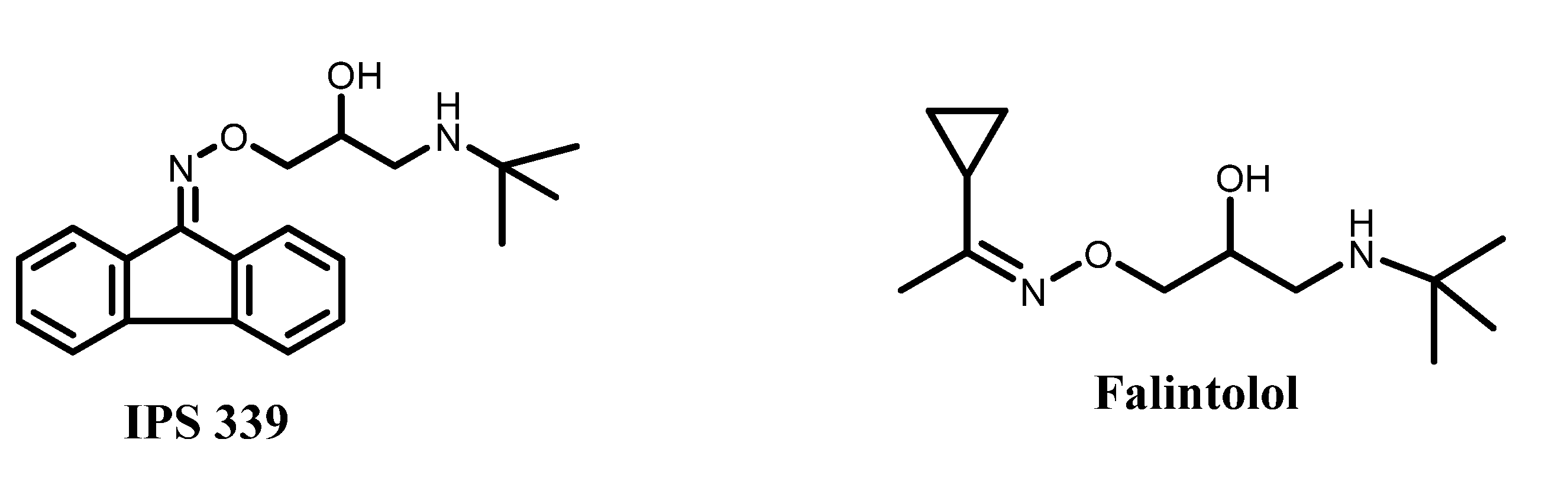
2. Results and Discussion
2.1. Chemistry

| Compound No. | R1 | R2 | Compound No. | R1 | R2 |
|---|---|---|---|---|---|
| a |  |  | f | CH3 |  |
| b |  | 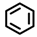 | g | 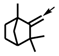 | |
| c |  |  | h | 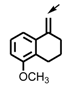 | |
| d | CH3 |  | i |  |  |
| e | CH3 | 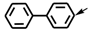 | j | CH3 |  |
2.2. Molecular Docking
| Compound No. | R1 | R2 | R3 | Compound No. | R1 | R2 | R3 |
|---|---|---|---|---|---|---|---|
| 4a |  |  |  | 4n |  | CH3 | 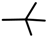 |
| 4b |  |  |  | 4o |  | CH3 |  |
| 4c |  |  |  | 4p | 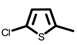 | CH3 |  |
| 4d |  |  |  | 4q |  | CH3 |  |
| 4e |  |  |  | 4r |  | CH3 |  |
| 4f |  |  |  | 4s |  |  | |
| 4g |  |  |  | 4t |  |  | |
| 4h |  |  |  | 4u |  |  | |
| 4i |  |  |  | 4v |  |  | |
| 4j |  | CH3 |  | 4w |  |  | |
| 4k |  | CH3 |  | 4x |  |  | |
| 4l |  | CH3 |  | 4y |  |  |  |
| 4m |  | CH3 |  | 4z |  | CH3 |  |
| Comp. | β1-Receptor | β2-Receptor | ||
|---|---|---|---|---|
| MolDock Score | H-Bond E. | MolDock Score | H-Bond E. | |
| 4a | −115.13 | −2.5 | −100.806 | −4.35757 |
| 4b | −122.697 | −4.58252 | −99.6442 | −2.42254 |
| 4c | −142.285 | −5.19707 | −105.6596 | −3.42955 |
| 4d | −121.41 | −3.59754 | −86.4381 | −1.16172 |
| 4e | −127.903 | −3.96668 | −88.3577 | −0.04374 |
| 4f | −153.964 | −5 | −95.1594 | 0 |
| 4g | −125.653 | −2.5 | −92.7053 | −3.4013 |
| 4h | −124.044 | −2.83065 | −85.3741 | −0.69094 |
| 4i | −145.546 | −5.2648 | −90.616 | −2.98878 |
| 4j | −114.594 | −2.62113 | −79.1286 | 0 |
| 4k | −120.406 | −2.5 | −90.5964 | −2.63215 |
| 4l | −155.836 | −6.1153 | −103.123 | −0.16123 |
| 4m | −125.091 | −3.98536 | −74.7409 | −0.97756 |
| 4n | −120.574 | −2.18542 | −73.6041 | −3.78389 |
| 4o | −167.406 | −4.49787 | −91.127 | −2.5557 |
| 4p | −105.452 | −1.63375 | −113.5079 | −4.03237 |
| 4q | −114.132 | −2.62848 | −117.075 | −4.34578 |
| 4r | −143.244 | −2.15101 | −102.028 | −0.41247 |
| 4s | −108.994 | −1.50057 | −85.4672 | −3.26981 |
| 4t | −108.574 | −2.79887 | −84.921 | −0.78187 |
| 4u | −148.747 | −4.41468 | −100.887 | −0.03188 |
| 4v | −105.443 | −1.00129 | −84.9409 | −1.98422 |
| 4w | −105.034 | 0 | −85.1195 | −0.99963 |
| 4x | −139.094 | −4.07406 | −99.449 | −0.47872 |
| 4y | −152.107 | −5.54788 | −93.6354 | −2.4702 |
| 4z | −125.4 | −2.28939 | −84.4559 | −2.16834 |
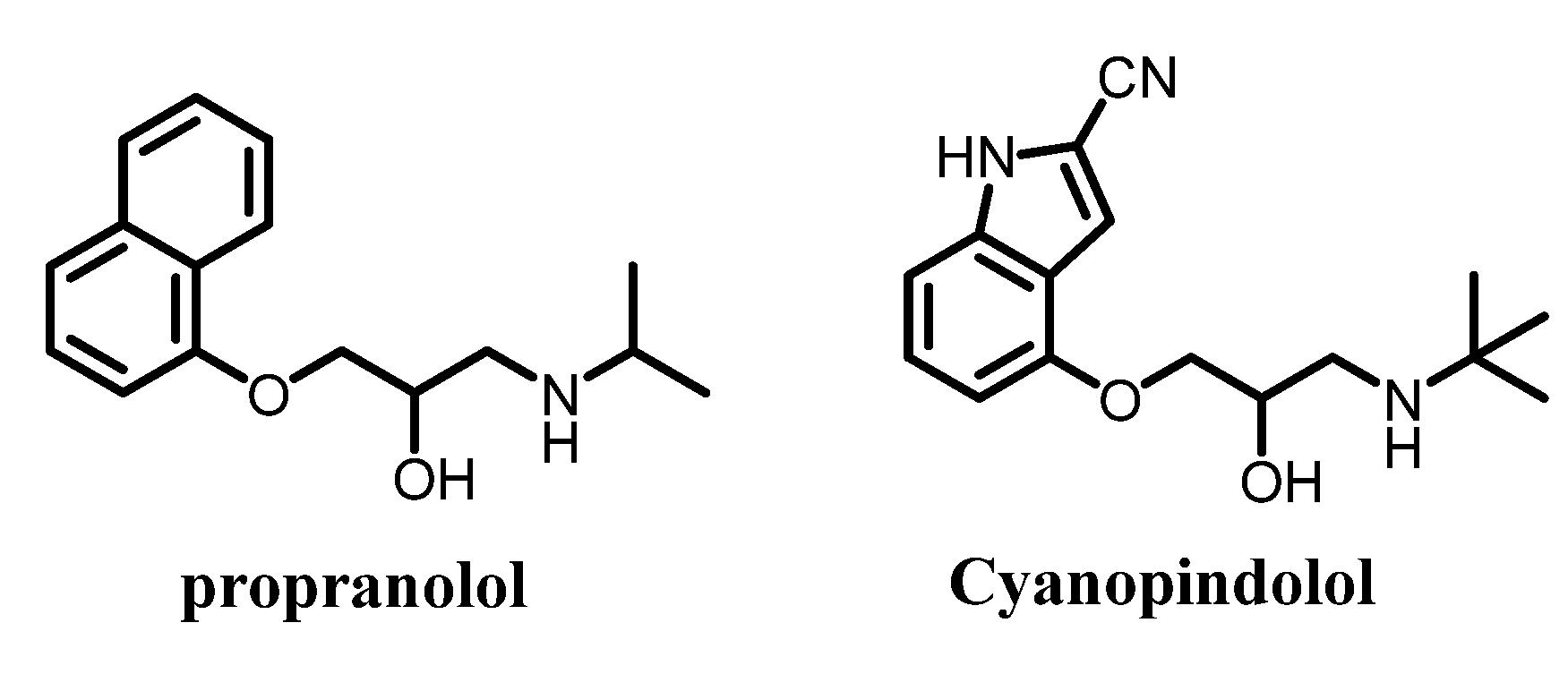
| Propranolol | −110.517 | −2.53142 | −88.1631 | −3.92249 |
| IPS 339 | −104.212 | −2.91744 | −95.141 | −2.87182 |
| Cyanopindolol | −129.204 | −10.7754 | −85.6768 | −2.98876 |
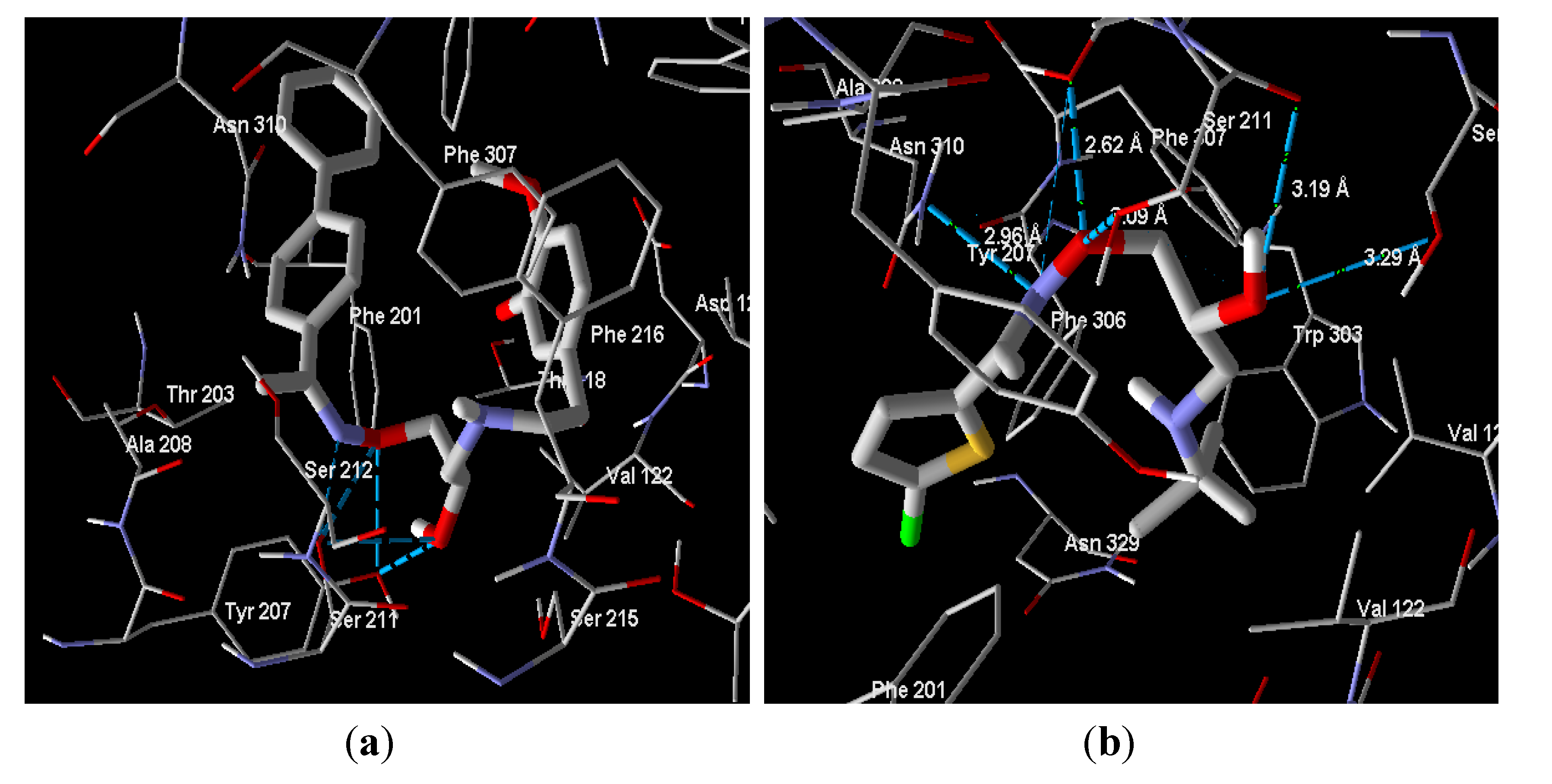
2.3. Biological Evaluation
2.3.1. In Vitro Screening for β1-Adrenoceptor Activity in Isolated Guinea Pig Atria
| Compound No. | β1-antagonist | Compd. No. | β1-antagonist | ||
|---|---|---|---|---|---|
| % inhibition in heart rate | % inhibition in contractility | % inhibition in heart rate | % Inhibition in contractility | ||
| 4a | 3% | 0% | 4o | 1% | 0% |
| 4b | 2% | 0% | 4p | 6% | 0% |
| 4c | 6% | 1% | 4q | 2% | 0% |
| 4e | 0% | 0% | 4r | 31% | 55% |
| 4f | 29% | 30% | 4u | 2% | 0% |
| 4g | 2% | 0% | 4x | 0% | 0% |
| 4i | 25% | 40% | 4y | 26% | 35% |
| 4k | 3% | 1% | 4z | 22% | 30% |
| 4l | 28% | 45% | Propranolol | 70% | 60% |
2.3.2. In Vitro Screening for β2-Adrenoceptor Activity in Isolated Guinea pig Trachea
| Compound No. | β2-agonist | β2-antagonist | Compound No. | β2-agonist | β2-antagonist |
|---|---|---|---|---|---|
| 4a | - | - | 4p | - | + |
| 4e | - | - | 4q | - | + |
| 4g | - | - | 4r | - | - |
| 4i | - | - | 4t | - | - |
| 4k | - | - | 4v | - | - |
| 4m | - | - | Propranolol | - | + |
3. Experimental
3.1. General Information
3.1.1. General Procedure for Synthesis of O-Oxiran-2-ylmethyl Oximes 3a–h
3.1.2. General Procedure for Synthesis of O-(3-Alkylamino-2-hydroxypropyl)oxime Derivatives 4a–z
3.2. Molecular Docking Methodology
3.3. Biological Evaluation
3.3.1. In Vitro Screening for β1-Adrenoceptor Activity in Isolated Guinea Pig Atria
3.3.2. In Vitro Screening for β2-Adrenoceptor Activity in Isolated Guinea Pig Trachea
4. Conclusions
Supplementary Materials
Acknowledgments
Conflictts of Interest
References
- Hoffman, B.B. Adrenoceptor antagonist drugs. In Basic and Clinical Pharmacology, 10th ed.; Katzung, B.G., Ed.; McGraw-Hill: New York, NY, USA, 2007. [Google Scholar]
- Imbs, J.L.; Miesch, F.; Schwartz, J.; Velly, J.; Leclerc, G.; Mann, A.; Wermuth, C.G. A potent new beta2-adrenoceptor blocking agent. Br. J. Pharmacol. 1977, 60, 357–362. [Google Scholar] [CrossRef]
- Leclerc, G.; Mann, A.; Wermuth, C.G.; Bieth, N.; Schwartz, J. Synthesis and beta-adrenergic blocking activity of a novel class of aromatic oxime ethers. J. Med. Chem. 1977, 20, 1657–1662. [Google Scholar] [CrossRef]
- Bouzoubaa, M.; Leclerc, G.; Decker, N.; Schwartz, J.; Andermann, G. Synthesis and beta-adrenergic blocking activity of new aliphatic and alicyclic oxime ethers. J. Med. Chem. 1984, 27, 1291–1294. [Google Scholar] [CrossRef]
- Hothersall, J.D.; Black, J.; Caddick, S.; Vinter, J.G.; Tinker, A.; Baker, J.R. The design, synthesis and pharmacological characterization of novel beta(2)-adrenoceptor antagonists. Br. J. Pharmacol. 2011, 164, 317–331. [Google Scholar] [CrossRef]
- Wenzel, D.; Knies, R.; Matthey, M.; Klein, A.M.; Welschoff, J.; Stolle, V.; Sasse, P.; Roll, W.; Breuer, J.; Fleischmann, B.K. Beta(2)-adrenoceptor antagonist ici 118,551 decreases pulmonary vascular tone in mice via a G(i/o) protein/nitric oxide-coupled pathway. Hypertension 2009, 54, 157–163. [Google Scholar]
- Citterio, A.; Filippini, L. Homolytic c-alkylation of aldoximes. Synthesis 1986, 1986, 473–474. [Google Scholar] [CrossRef]
- Zaitsev, A.B.; Vasil'tsov, A.M.; Schmidt, E.Y.; Mikhaleva, A.I.; Morozova, L.V.; Afonin, A.V.; Ushakov, I.A.; Trofimov, B.A. O-vinyldiaryl-and O-vinylaryl (hetaryl) ketoximes: A breakthrough in O-vinyloxime chemistry. Tetrahedron 2002, 58, 10043–10046. [Google Scholar] [CrossRef]
- Maheswara, M.; Siddaiah, V.; Gopalaiah, K.; Rao, V.M.; Rao, C.V. A simple and effective glycine-catalysed procedure for the preparation of oximes. J. Chem. Res. 2006, 2006, 362–363. [Google Scholar] [CrossRef]
- Campbell, K.N.; Campbell, B.K.; Chaput, E.P. The reaction of grignard reagents with oximes. Ii. The action of aryl grignard reagents with mixed ketoximes. J. Org. Chem. 1943, 8, 99–102. [Google Scholar] [CrossRef]
- Crovetti, A.J.; Stein, R.G. Certain o-substituted thiophene oxime carbamates used as antibacterial and antifungal agents. US Patent 4,061,764, December 1977. [Google Scholar]
- Cottingham, R. The beckmann rearrangement of fenchone oxime. J. Org. Chem. 1960, 25, 1473–1476. [Google Scholar] [CrossRef]
- Hardegger, E.; Redlich, D.; Gal, A. Über steroide und sexualhormone. (114. Mitteilung). Versuche zur herstellung von 4, 13-dioxychrysen-derivaten. Helv. Chim. Acta 1945, 28, 628–637. [Google Scholar] [CrossRef]
- Delgado, A.; Garcia, J.M.; Mauleon, D.; Minguillon, C.; Subirats, J.R.; Feliz, M.; Lopez, F.; Velasco, D. Synthesis and conformational analysis of 2-amino-1, 2, 3, 4-tetrahydro-1-naphthalenols. Can. J. Chem. 1988, 66, 517–527. [Google Scholar] [CrossRef]
- Balsamo, A.; Macchia, M.; Martinelli, A.; Rossello, A. The [(methyloxy) imino] methyl moiety (moimm) in the design of a new type of β-adrenergic blocking agent. Eur. J. Med. Chem. 1999, 34, 283–291. [Google Scholar] [CrossRef]
- Barril, X.; Morley, S.D. Unveiling the full potential of flexible receptor docking using multiple crystallographic structures. J. Med. Chem. 2005, 48, 4432–4443. [Google Scholar] [CrossRef]
- Rasmussen, S.G.; Choi, H.-J.; Rosenbaum, D.M.; Kobilka, T.S.; Thian, F.S.; Edwards, P.C.; Burghammer, M.; Ratnala, V.R.; Sanishvili, R.; Fischetti, R.F. Crystal structure of the human β2 adrenergic G-protein-coupled receptor. Nature 2007, 450, 383–387. [Google Scholar] [CrossRef]
- Cherezov, V.; Rosenbaum, D.M.; Hanson, M.A.; Rasmussen, S.G.; Thian, F.S.; Kobilka, T.S.; Choi, H.-J.; Kuhn, P.; Weis, W.I.; Kobilka, B.K. High-resolution crystal structure of an engineered human β2-adrenergic g protein–coupled receptor. Science 2007, 318, 1258–1265. [Google Scholar] [CrossRef]
- Warne, T.; Serrano-Vega, M.J.; Baker, J.G.; Moukhametzianov, R.; Edwards, P.C.; Henderson, R.; Leslie, A.G.; Tate, C.G.; Schertler, G.F. Structure of a b 1-adrenergic g-protein-coupled receptor. Nature 2008, 454, 486–491. [Google Scholar] [CrossRef]
- Abdel-Aziz, H.A.; Al-Rashood, K.A.; Ghabbour, H.A.; Fun, H.K.; Chia, T.S. 1-(4-Methyl-phen-yl)-2-(phenyl-sulfon-yl)ethanone. Acta Crystallogr. E 2012, 68, o1033. [Google Scholar]
- Kitchen, D.B.; Decornez, H.; Furr, J.R.; Bajorath, J. Docking and scoring in virtual screening for drug discovery: Methods and applications. Nat. Rev. Drug Discov. 2004, 3, 935–949. [Google Scholar] [CrossRef]
- Tesfamariam, B.; Allen, G.T. B1-and β2-adrenoceptor antagonist activities of ici-215001, a putative β3-adrenoceptor agonist. Br. J. Pharmacol. 1994, 112, 55–58. [Google Scholar] [CrossRef]
- Akamine, E.H.; Hohman, T.C.; Nigro, D.; Carvalho, M.H.C.; de Cássia Tostes, R.; Fortes, Z.B. Minalrestat, an aldose reductase inhibitor, corrects the impaired microvascular reactivity in diabetes. J. Pharmacol. Exp. Ther. 2003, 304, 1236–1242. [Google Scholar]
- Poli, E.; Coruzzi, G.; Bertaccini, G. Changes in the ionic environment may alter the kind of antagonism of some histamine H2-receptor blockers in the guinea pig papillary muscle. J. Pharmacol. Method. 1990, 23, 265–274. [Google Scholar] [CrossRef]
- Gan, L.-L.; Wang, M.-W.; Cheng, M.-S.; Pan, L. Trachea relaxing effects and beta2-selectivity of spff, a newly developed bronchodilating agent, in guinea pigs and rabbits. Biol. Pharm. Bull. 2003, 26, 323–328. [Google Scholar] [CrossRef]
- Hoefle, M.L.; Hastings, S.G.; Meyer, R.F.; Corey, R.M.; Holmes, A.; Stratton, C.D. Cardioselective. Beta.-adrenergic blocking agents. 1. 1-[(3, 4-dimethoxyphenethyl) amino]-3-aryloxy-2-propanols. J. Med. Chem. 1975, 18, 148–152. [Google Scholar] [CrossRef]
- El‐Ashmawy, M.; Lissavetzky, J.; Darias, V.; Martín‐Herrera, D. Synthesis and pharmacological study of a thiophene analogue of moprolol and related compounds. Arch. Pharm. 1991, 324, 45–48. [Google Scholar] [CrossRef]
- Abdel-Aziz, H.A.; Ghabbour, H.A.; Chantrapromma, S.; Fun, H.K. 3-Acetyl-1,5-diphenyl-1h-pyrazole-4-carbonitrile. Acta Crystallogr. E 2012, 68, o1095–o1096. [Google Scholar]
- Kassem, M.G.; Ghabbour, H.A.; Abdel-Aziz, H.A.; Fun, H.K.; Ooi, C.W. 3-Chloro-4-methyl-quinolin-2(1h)-one. Acta Crystallogr. E 2012, 68, o1043. [Google Scholar]
- Pinheiro, J.R.; Bitencourt, M.; da Cunha, E.F.; Ramalho, T.C.; Freitas, M.P. Novel anti-hiv cyclotriazadisulfonamide derivatives as modeled by ligand-and receptor-based approaches. Bioorg. Med. Chem. 2008, 16, 1683–1690. [Google Scholar] [CrossRef]
- Sapre, N.S.; Gupta, S.; Pancholi, N.; Sapre, N. Molecular docking studies on tetrahydroimidazo-[4,5,1-jk][1,4]-benzodiazepinone (TIBO) derivatives as HIV-1 NNRT inhibitors. J. Comput. Aid. Mol. Des. 2008, 22, 69–80. [Google Scholar] [CrossRef]
- Gehlhaar, D.K.; Bouzida, D.; Rejto, P.A. Fully Automated and Rapid Flexible Docking of Inhibitors Covalently Bound to Serine Proteases. In Evolutionary Programming VII; Porto, V.W., Saravanan, N., Waagen, D., Eiben, A.E., Eds.; Springer: Berlin, Heidelberg, Germany, 1998; pp. 449–461. [Google Scholar]
- Yang, J.M.; Chen, C.C. Gemdock: A generic evolutionary method for molecular docking. Proteins 2004, 55, 288–304. [Google Scholar] [CrossRef]
- Thomsen, R.; Christensen, M.H. Moldock: A new technique for high-accuracy molecular docking. J. Med. Chem. 2006, 49, 3315–3321. [Google Scholar] [CrossRef]
- Emmerson, J.; Mackay, D. The zig-zag tracheal strip. J. Pharm. Pharmacol. 1979, 31, 798–798. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 4y–z are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ghabbour, H.A.; El-Bendary, E.R.; El-Ashmawy, M.B.; El-Kerdawy, M.M. Synthesis, Docking Study and β-Adrenoceptor Activity of Some New Oxime Ether Derivatives. Molecules 2014, 19, 3417-3435. https://doi.org/10.3390/molecules19033417
Ghabbour HA, El-Bendary ER, El-Ashmawy MB, El-Kerdawy MM. Synthesis, Docking Study and β-Adrenoceptor Activity of Some New Oxime Ether Derivatives. Molecules. 2014; 19(3):3417-3435. https://doi.org/10.3390/molecules19033417
Chicago/Turabian StyleGhabbour, Hazem A., Eman R. El-Bendary, Mahmoud B. El-Ashmawy, and Mohamed M. El-Kerdawy. 2014. "Synthesis, Docking Study and β-Adrenoceptor Activity of Some New Oxime Ether Derivatives" Molecules 19, no. 3: 3417-3435. https://doi.org/10.3390/molecules19033417
APA StyleGhabbour, H. A., El-Bendary, E. R., El-Ashmawy, M. B., & El-Kerdawy, M. M. (2014). Synthesis, Docking Study and β-Adrenoceptor Activity of Some New Oxime Ether Derivatives. Molecules, 19(3), 3417-3435. https://doi.org/10.3390/molecules19033417